Abstract
By way of introduction to the special issue on diisocyanates and their corresponding diamines, this brief overview presents, for the most commonly used diisocyanate monomers, a selection of physical-chemical properties that are relevant to exposure in the workplace and in the general environment, as well as a concise overview of diisocyanate reactions and some of their toxicological implications.
Keywords
Introduction
For any substance, the exposure potential depends, in large part, on basic physical-chemical properties such as density, hydrophobicity, physical state, and vapor pressure. The first part of this overview provides a brief summary of these properties for the most commercially important diisocyanates.
The potential effects of exposure to a substance, in contrast, are a direct consequence of the chemical reactivity of that substance. In the case of diisocyanates, the reactivity of the isocyanate (−N=C=O, or NCO) functional group toward water and biological (macro)molecules and the resulting reaction products are key. Therefore, the second part of this overview summarizes the most relevant diisocyanate reactions and provides some insight into their toxicological implications.
In addition, these brief summaries allow for the introduction of some terminology that is frequently used in the manuscripts that are included in the special issue.
Nomenclature and physical-chemical properties
Diisocyanates are monomers used to make polyurethane (PU) polymers and are characterized by having two NCO groups as their key reactive sites. They are distinguished from mono-isocyanates, like methyl isocyanate,
Structures and selected physical-chemical properties of the most commonly used diisocyanate monomers that are relevant to human and aquatic exposure. Structures drawn with PubChem Sketcher V2.4.
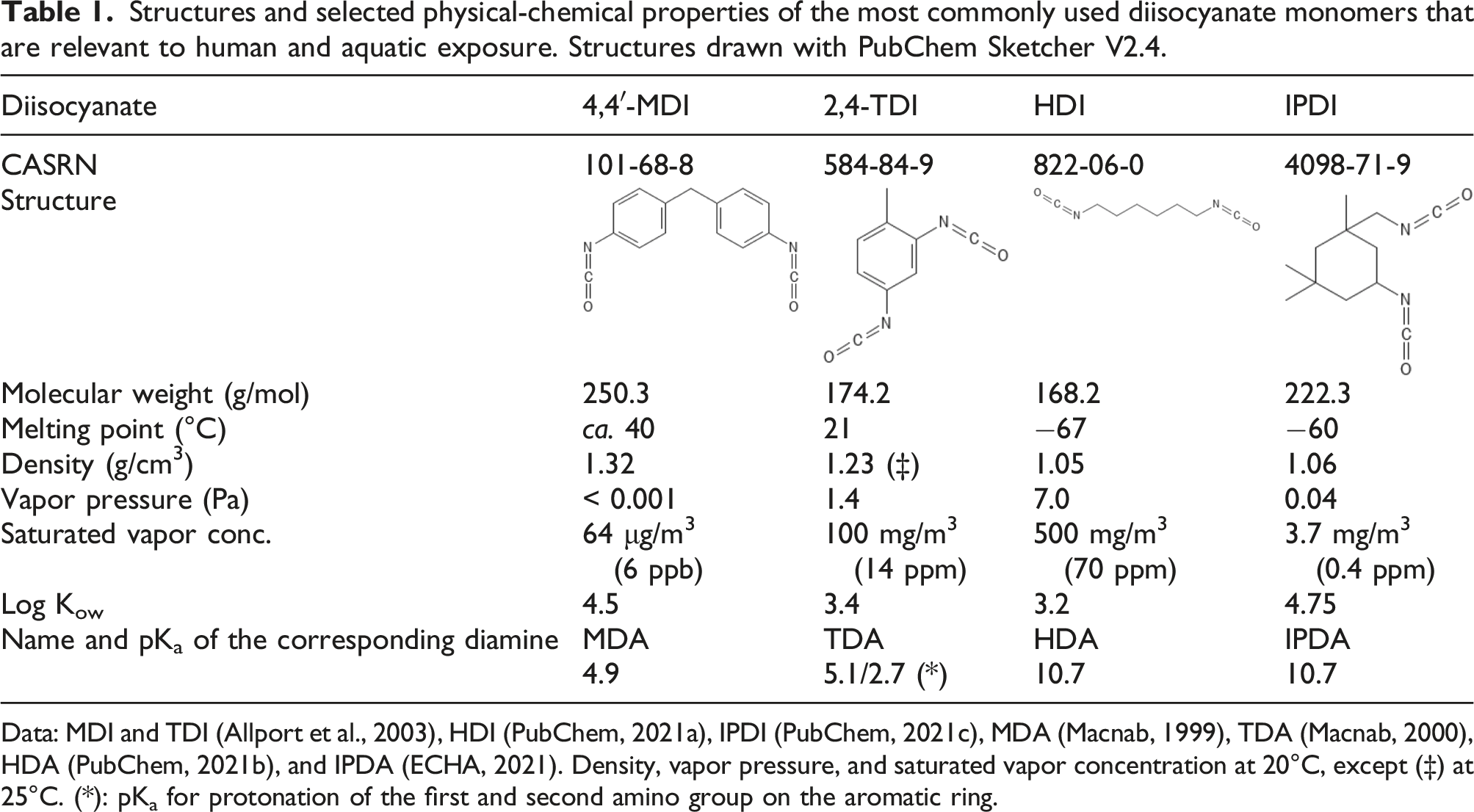
Data: MDI and TDI (Allport et al., 2003), HDI (PubChem, 2021a), IPDI (PubChem, 2021c), MDA (Macnab, 1999), TDA (Macnab, 2000), HDA (PubChem, 2021b), and IPDA (ECHA, 2021). Density, vapor pressure, and saturated vapor concentration at 20°C, except (‡) at 25°C. (*): pKa for protonation of the first and second amino group on the aromatic ring.
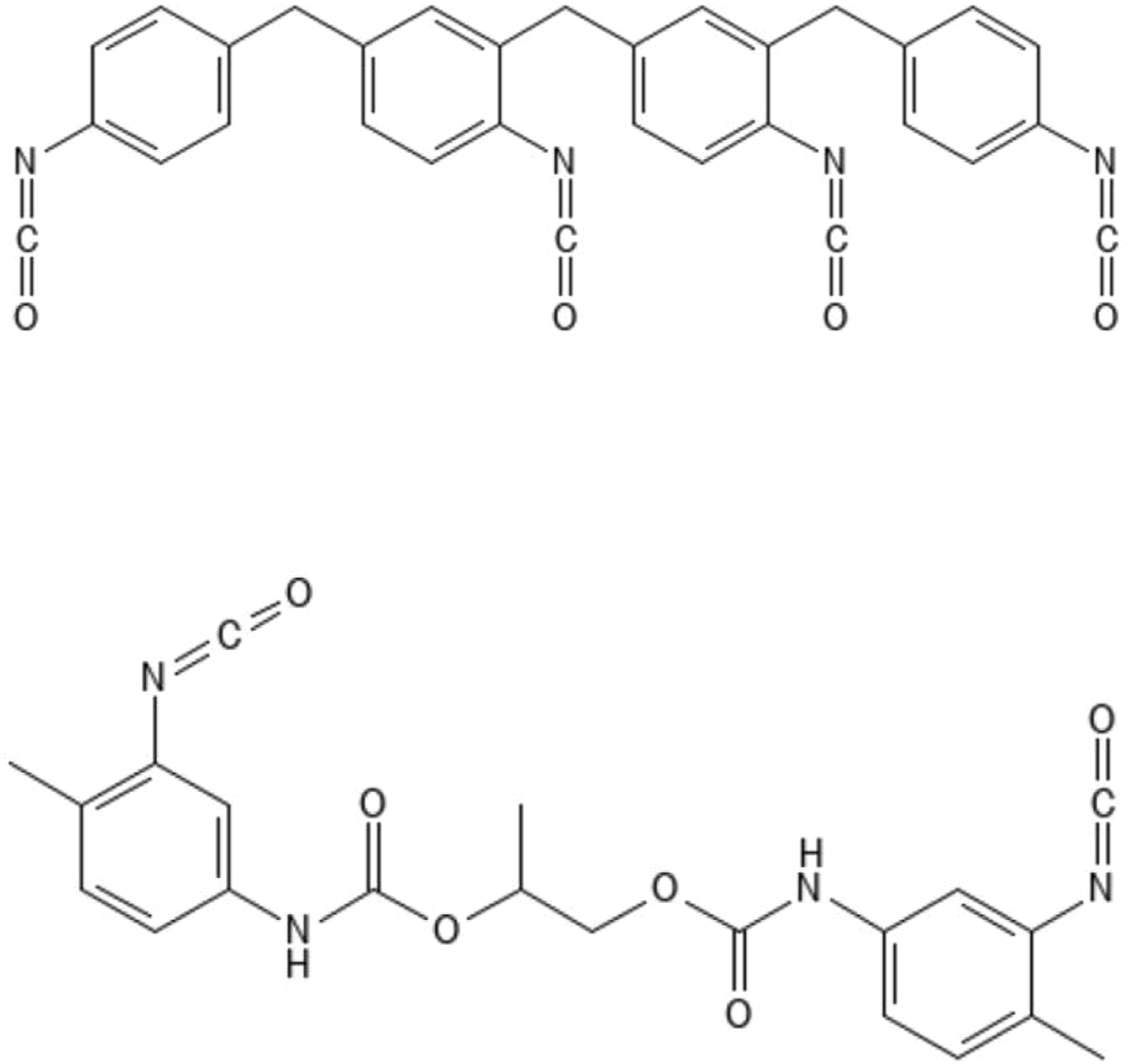
Structures of a 4-functional MDI homologue (top) and an oligomer constituent of a prepolymer of 2,4-TDI with propylene glycol (bottom). Structures drawn with PubChem Sketcher V2.4.
ADI and DII are denser than water and are hydrophobic (i.e., to the extent the term is meaningful for water-reactive substances, they have a very low “solubility” in water). ADI and DII differ, however, in physical appearance: while the ADI monomers are liquids under standard conditions, the DII monomers and mixtures typically have melting points in a temperature range between 5 and 40°C. HDI and TDI have significant vapor pressures, which is important regarding potential workplace exposure. The vapor pressure of IPDI is a factor 100 less than both HDI and TDI. In contrast, MDI has a vapor pressure at ambient conditions that results in a saturated vapor concentration (ca. 6 ppb) close to typical exposure limits. At higher concentrations, MDI would be present as an aerosol in the workplace atmosphere.
The corresponding diamines of ADI, hexamethylene diamine (HDA) and isophorone diamine (IPDA), are strong bases. At physiological pH, the aliphatic amino groups are completely protonated (R-NH3+), and hence, both HDA and IPDA are very soluble in water. The aromatic amino group is much less basic because of the resonance of the N-atom’s free electron pair with the π-electron system of the aromatic ring. At physiological pH, the corresponding diamines of DII, methylene dianiline (MDA) and toluene diamine (TDA), are only moderately soluble in water, since they are mostly present in the unprotonated, but reactive, R-NH2 form. The reactivity of the unprotonated aromatic amino group makes it subject to metabolic activation in the liver; whence, TDA and MDA are presumed to be human carcinogens (IARC, 1978, 1986).
General reactivity, reactions, and implications
The NCO group is electrophilic. Its reactivity is a function of the partial positive charge on its central C-atom induced by the more electronegative N- and O-atoms (Allport et al., 2003). Alkyl chains are electron donors and decrease the positive charge on the C-atom, whereas the aromatic ring withdraws electrons and increases the charge in the C-atom. Therefore, the reactivity of NCO groups increases in the sequence secondary aliphatic < primary aliphatic << ortho aromatic < para aromatic, whereby ortho (often called 2-NCO) and para (often called 4-NCO) refer to the position of the NCO group with respect to that of the methyl(ene) substituent.
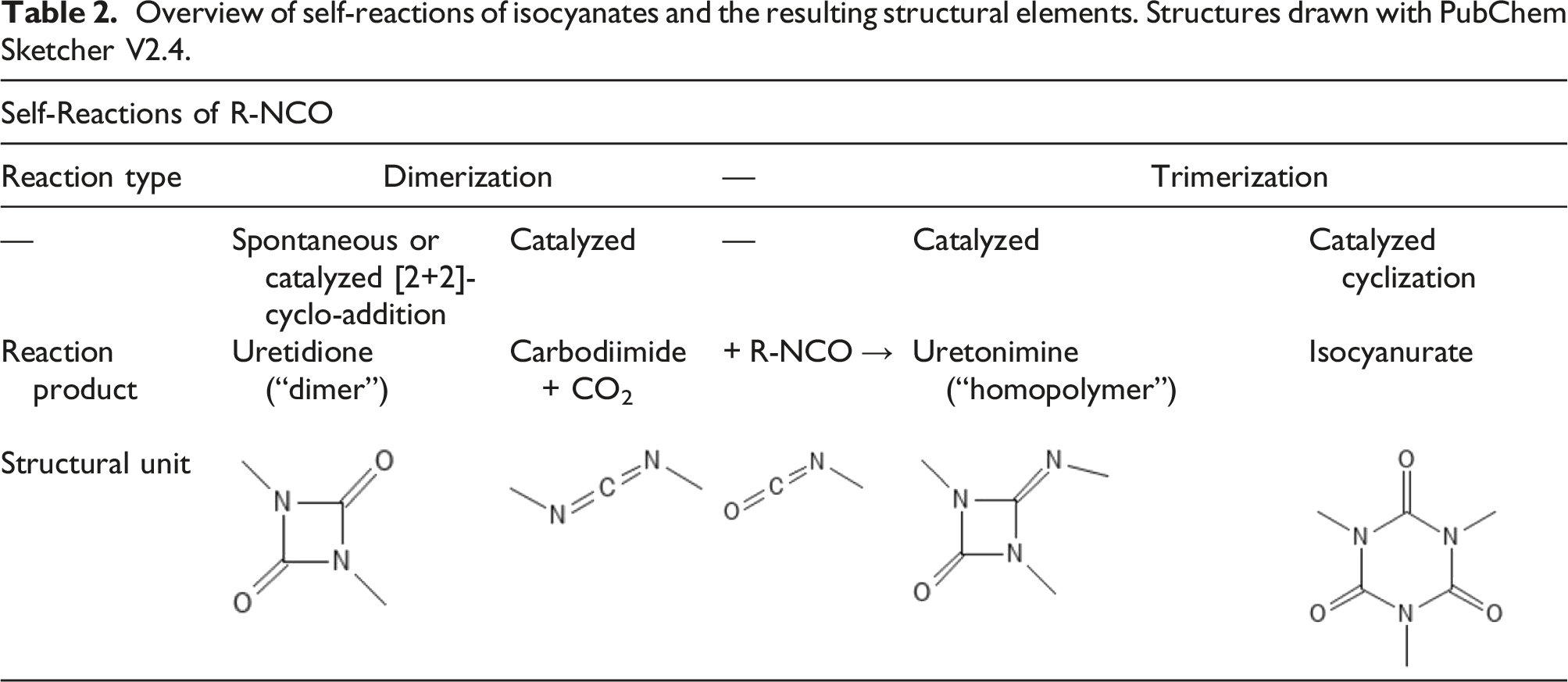
Overview of self-reactions of isocyanates and the resulting structural elements. Structures drawn with PubChem Sketcher V2.4.
Overview of the most important nucleophilic addition reactions to the isocyanate (NCO) group.
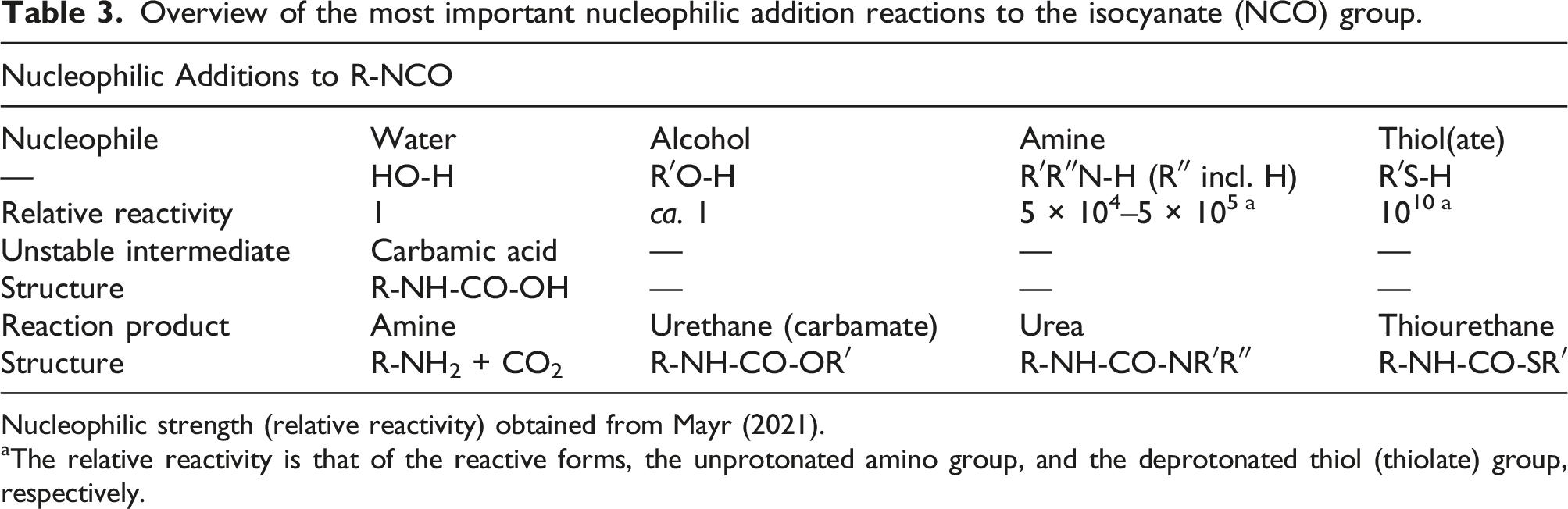
Nucleophilic strength (relative reactivity) obtained from Mayr (2021).
aThe relative reactivity is that of the reactive forms, the unprotonated amino group, and the deprotonated thiol (thiolate) group, respectively.
Whether in the human body or in the broader living environment, water is always present and the potential hydrolysis of the diisocyanate must be considered. However, the reaction of a diisocyanate with water rarely results in significant formation of the “textbook reaction product,” the corresponding diamine. Amino groups (e.g., present in the hydrolysis product itself or in proteins) and particularly thiolates (e.g., in glutathione (GSH)) are much stronger nucleophiles than water (see Table 3) and are, therefore, the dominant reaction partners for the NCO group, even if their concentration is typically limited to millimolar levels. GSH can function as a carrier to transfer the NCO moiety to proteins in a process called transcarbamoylation. The reaction between an isocyanate and an amine or a protein amino-group results in the formation of a substituted urea or a protein adduct. The resulting urea bonds are stable across relevant pH and temperature ranges (Sendijarevic et al., 2004). The formation of ureas and protein adducts constitutes an effective detoxification pathway for diisocyanates by limiting the availability of the reactive NCO group (Loddenkemper et al., 2017, 2019; Neuhahn et al., 2020; Schupp and Plehiers, 2022). At the same time, the formation of the same protein adducts can be the molecular trigger for the immunological processes that may lead to sensitization via dermal or respiratory route in susceptible individuals (Kimber et al., 2018).
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are employed by the International Isocyanate Institute, Inc. The Institute is funded by producers of MDI and TDI. Any views expressed in this paper and any conclusions are those of the authors, not necessarily of the International Isocyanate Institute, Inc. or its member companies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded with regular membership fees of the International Isocyanate Institute, Inc.
Ethical approval
This work did not involve interaction with human study subjects and no ethical approval was required.
