Abstract
The aim of this study was to provide realistic isocyanate and amine emission data when using different methylene diphenyl diisocyanate (MDI)-based polyurethane consumer products. Emission testing (air sampling) of diisocyanates and corresponding diamines was performed in a full-scale controlled-environment chamber during different work operations, such as gluing, mixing and foaming. The polyurethane products used were construction glue, one-component foam and two different two-component adhesives used in parquet flooring. Air sampling for isocyanates and amines was performed in the breathing zone of the worker and at different positions inside the controlled-environment chamber while the work operations were performed. Air sampling was also performed after the application, at different positions inside the chamber, to cover the post curing phase. Low air concentrations (0.1–0.7 μg MDI/m3, 0.03–0.2 μg isocyanate group (NCO)/m3) were found in the breathing zone and close to the work operation for some of the gluing applications. No methylene diphenyl diamine (MDA) concentrations above the limit of quantification were found for any of the applications in the breathing zone air. These results indicated that inhalation exposure to MDA or MDI would be expected to be minimal during application of do-it-yourself consumer products containing MDI.
Keywords
Introduction
Diisocyanates are mainly used to produce polyurethane (PU). PU are valuable polymers that are used in a wide range of applications and can be found in almost every sector of the industry (Baysal & Kasapbası, 2017). Exposure to isocyanates is widely known as a common cause for developing occupational asthma (Ameille et al., 2003; Lefkowitz et al., 2015). Although cases of diisocyanate work-related asthma have been decreasing globally in recent years (Buyantseva et al., 2011; Paris et al., 2012; Reilly et al., 2020), exposure considerations are still important. Diisocyanates are irritants of the upper airways, skin, and eye, as well as dermal and respiratory sensitizers (European Chemicals Association (ECHA), 2021a). High peak exposures have been implicated in the induction of respiratory sensitization (Plehiers et al., 2020). In the case of the diisocyanate considered in the present work, methylene diphenyl diisocyanate (MDI), the No Observed Adverse Effect Level for histopathological pulmonary effects found in mammalian inhalation toxicology studies was 0.2 mg/m3 (Feron et al., 2001).
Occupational exposure to isocyanates is regulated by authorities at the national level and isocyanates are among the group of compounds having the lowest occupational exposure limits (OEL) (ECHA, 2019); a typical OEL for MDI is 0.05 mg/m3 as an 8h time weighted average (Deutsche Forschungsgemeinschaft, 2020).
Reaction of isocyanates with water results in the formation of polymeric ureas, and if the isocyanate concentration is low enough, can additionally lead to the formation of low levels of the corresponding diamine (methylene diamine (MDA) in the case of MDI). MDA is classified as a carcinogen presumed to have carcinogenic potential for humans (IARC, 1986), to which human exposure should be kept as low as possible. A typical OEL for MDA is 80 μg MDA/m3 [10 ppb] (United States Department of Labor (2019)).
In addition to industrial and professional applications, diisocyanates are also used in ‘do-it-yourself’ (DIY) products that are intended for use in the renovation of homes by both professional and non-professional users (consumers). For studying emission of hazardous compounds and evaluating different exposure scenarios from various consumer products, controlled-environment chamber studies have been used (Even et al., 2020; Schaeffer et al., 1996; Vojta et al., 2017). Emissions of diisocyanates have been tested in controlled-environment chambers during curing of different PU adhesives (Wirtz et al., 2003; Wirtz & Saltzhammer, 2002) and from other PU products such as various foams, varnishes and sealants (Kelly et al., 1999). It was observed that during curing, the isocyanate emission was temperature dependent and occurred in two phases: surface emission and internal diffusion. Typically, chamber volumes of less than 10 L have been used for product testing. Full scale testing of work operations using DIY consumer products was evaluated for emission of hazardous compounds (MEFD, 2018). That study included a one-component PU sealing foam based on MDI, noting an applicator breathing zone concentration of 6 μg MDI/m3 (compared to typical occupational exposure limit of 50 μg MDI/m3 in the EU). The risk assessment indicated a risk for developing airway and skin allergy when applying the foam based on the content of MDI in the product, and the use of protective gloves and respiratory protection during application was recommended. It was noted as well that there was no risk in remaining in the room during curing.
The aim of this study was to provide representative emission data for different applications when using MDI-based DIY consumer products such as different kinds of glues and a one-component foam. Concentration measurements in the work environment focused on the emission of diisocyanates and diamines, and the tests were conducted at high humidity conditions since these would be expected to promote any potential amine formation from the isocyanate.
Materials and methods
Chemicals
Dibutyl amine (DBA), acetonitrile, acetic acid, formic acid, methanol, toluene, 4,4’-methylene dianiline (MDA) and ethyl chloroformate (ET) were obtained from Merck (Darmstadt, Germany). Technical grade MDI (containing 2,4’-MDI and 4,4’-MDI at a ratio of 0.04/1) were obtained from Huntsman (Everberg, Belgium). Deuterium-labelled DBA [NH(C4H9)(C4D9)] and dideuterium-labelled 4,4’-MDA [CD2(C6H4NH2)2] were obtained from Ramidus (Lund, Sweden).
Reference solutions
Solutions of the urea and carbamate ester derivatives of MDI and MDA, respectively, were prepared and quantitatively characterised using LC-Chemiluminescence detection (CLND) and LC-Mass spectrometry (MS) according to the method developed, described and validated by Marand et al. (2004), Karlsson et al. (2002) and Gylestam et al. (2014) that forms the basis for ISO 17334-1 and 17334-2 (ISO, 2013a, 2013b). This method uses DBA to derivatize the isocyanates, which are then measured as the corresponding bis-ureas. The amines are reacted with ET and are measured as the corresponding acetyl derivatives.
The solutions of the urea and carbamate ester derivatives of MDI and MDA were diluted in acetonitrile to appropriate concentrations for the use of calibration standards and internal standards.
Instrumentation
A triple quadrupole mass spectrometer (MS), Quattro Micro (Waters, Altrincham, Cheshire, UK) was used for analysis of air samples. Isocyanate–DBA and amine-ET derivatives were monitored using positive electrospray ionization (ES+) and multiple reaction monitoring (MRM). Quantitative measurements were made by monitoring the reaction [MH]+→[DBAH]+ for the isocyanate derivatives and [MH]+→[M-46]+ for the diamine derivatives and for the corresponding deuterium-labelled internal standards ([MH]+→[d9-DBAH]+ and [d2-MH]+→[d2-M-46]+). For the MS instrument, the capillary voltage was 3 kV. The temperature of the ion source was 130°C and the desolvation gas temperature was 250°C. The desolvation gas flow was set to 700 L/h. Argon was used as collision gas. Entrance cone voltage and collision energies were optimized individually for the different derivatives. For chromatographic separation of the derivatives, gradient elution was performed, using Shimadzu LC10ADVP micro-LC pumps (Shimadzu Corporation, Kyoto, Japan). The gradient was linear from 40/60/0.05 (v/v/v) acetonitrile/H2O/formic acid to 95/5/0.05 (v/v/v) acetonitrile/H2O/formic acid in 10 min at a flow rate of 0.4 mL/min. Sample injections of 1 μL was made with an LC-Pal autosampler (CTC Analytics AG, Zwingen, Switzerland). The analytical column used was an Ascentis Express 5 cm × 2.2 mm, 2.7 µm C18 (Supelco, Bellefonte, PA, USA). A more detailed description of the instrumental parameters for the analysis of isocyanate–DBA and amine-ET derivatives using LC-MS is available elsewhere (Gylestam et al., 2014; Karlsson et al.,2002; ISO, 2013a, 2013b).
Products tested
Summary of the different products used and handling operations undertaken during the air sampling.
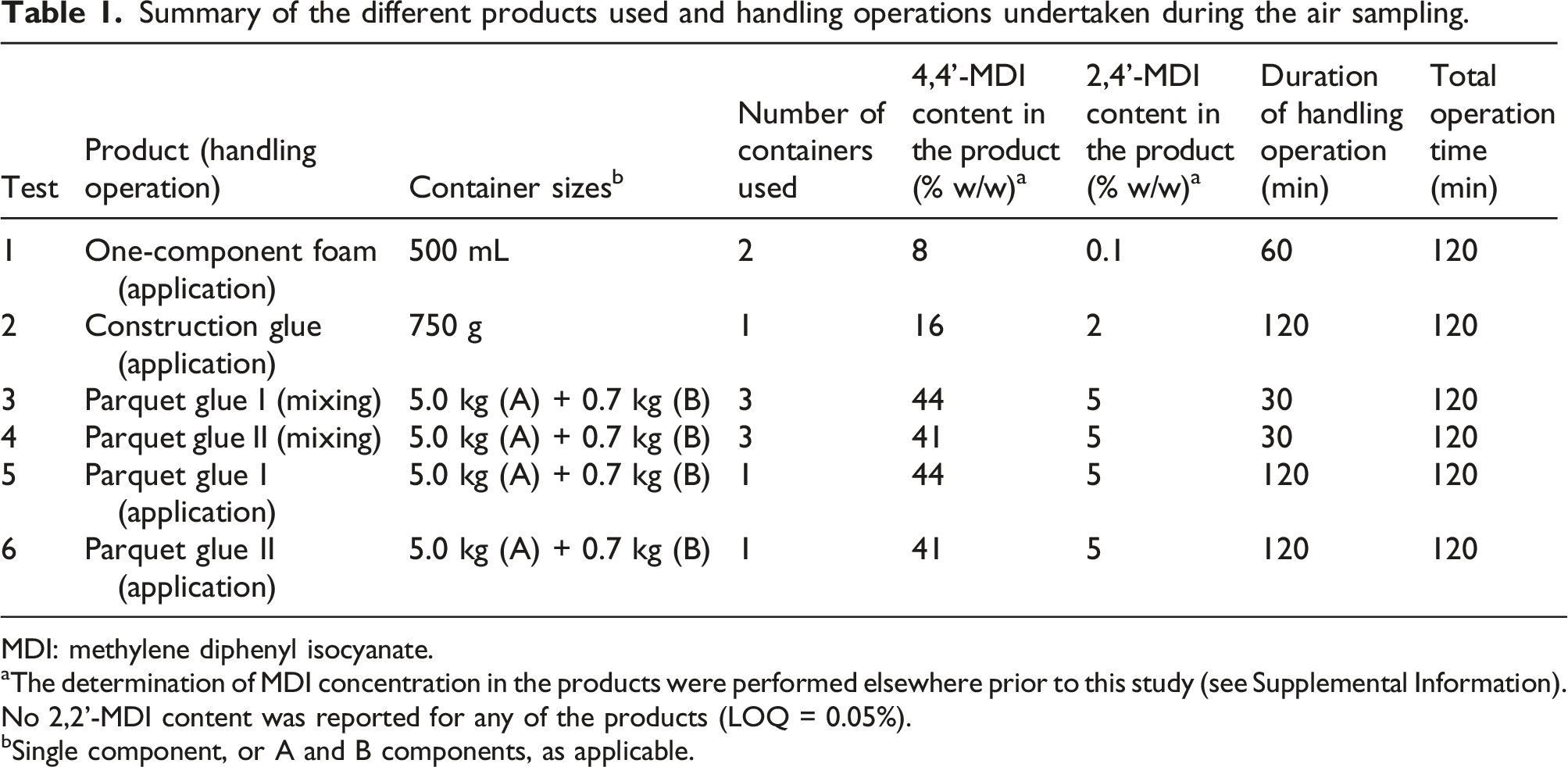
MDI: methylene diphenyl isocyanate.
aThe determination of MDI concentration in the products were performed elsewhere prior to this study (see Supplemental Information). No 2,2’-MDI content was reported for any of the products (LOQ = 0.05%).
bSingle component, or A and B components, as applicable.
Specifically, two kinds of two-component parquet glue (Parquet glue I and Parquet glue II; the main difference between those was the proportion of fillers to active ingredient used in the recipe), an OCF and a one-component construction glue were used. All products were tested during application and the two different parquet glues were also tested during mixing of the two components.
Controlled-environment test chamber
The controlled-environment chamber used for the emission testing was a full-scale chamber with an inside volume of 20.25 m3 (depth = 3.0 m, width = 2.8 m and height = 2.4 m). The inner walls, floor, ceiling and ventilation channels were all made of stainless steel. The air exchange rate was set to 10 m3/h. The air humidity was maintained in the range of 70–80% RH with temperatures in the range of 21.1–22.5°C. Due to the system set-up and to enable the low air exchange rate, there was a small variation in air humidity during and within the tests. However, the humidity remained always within the range of 70–80%.
Emission testing
Emission testing (air sampling) was performed during six different work operations using different PU products (Table 1). The different work tasks included 1: application of OCF; 2: Application of construction glue; 3: Mixing of two component parquet glues A; 4: Mixing of two component parquet glues B; 5: Application of parquet glue I and 6: Application of parquet glue II. Each of the six work operations were performed in duplicates (A & B) resulting in a total of 12 emission tests. All work operations were performed according to the manufacturer’s instructions, modelling real life situations.
Wood used during the testing was conditioned at 20°C and 70–80% RH for at least 24 h prior start of the testing.
Test 1: One-component foam application
The OCF was applied in a wooden frame with spaces simulating filling of cavities or fixing and insulating door/window frames. The frame consisted of wooden roof batten with an approximate width between the batten of 5 cm (Figure 1). The OCF was repeatedly applied for 15 s, paused for 45 s, then reapplied for 15 s; this cycle was repeated continuously until two OCF cans were emptied. The total application time, with the repeated pausing included, was approximately 1 h (each can could be used approximately 30 min before it was empty). The total operation time was 120 min. Application of one-component foam.
Test 2: Application of construction glue
The glue was applied using a fine-grained spatula to one side of two 50-by-5 cm wood pieces before pressing them together, followed by a 5-min wait time (Figure 2). This was repeated for 120 min. Application of construction glue.
Test 3: Parquet glue I mixing and test 4: Parquet glue II mixing
The mixing of the two-component parquet glues was performed by opening the containers and loading the 0.7 kg isocyanate component into the 5 kg container containing the polyol component (an operation that took 5 min), followed by 10 min of mixing (Figure 3). After the mixing, the lid was placed on the container and a 15-min waiting time elapsed. The procedure was then repeated for a new set of two-component containers. A total of three sets of two-component containers were mixed for each test. Between the second and last mixing, the container was closed but after the last mixing procedure, the lid was not placed on the container and a 45-min waiting time elapsed to simulate material in container that is not fully cured yet would be present in the vicinity of the product user. This resulted in a total mixing time of 30 min and a total sampling time of 120 min. Mixing of two-component parquet glue.
Tests 5 and 6: Parquet glue I and II applications
The mixing of the parquet glue for these tests was performed outside the controlled-environment chamber to avoid contamination. The freshly mixed glue was applied in accordance with the manufacturer’s instructions. The glue was applied on three wood panels at a time (Figure 4), resulting in a total application surface of 0.43 m2 (1.6 m × 0.27 m). Five minutes after completion of the first glue application, three new wood panels were placed on top of the glue surface and a new glue application was performed on the second set of wood panels. The procedure was then repeated for a total operation time of 120 min. Application of parquet glue.
Air sampling
For diisocyanate monitoring, samples were collected using a solvent-free sampler (ASSET™ EZ4-NCO Dry Sampler, Supelco, Bellefonte, PA, US) at a flow rate of approximately 0.2 L/min. For amine monitoring, samples were collected using impinger flasks containing 10 mL 0.01 Layout of controlled-environment chamber.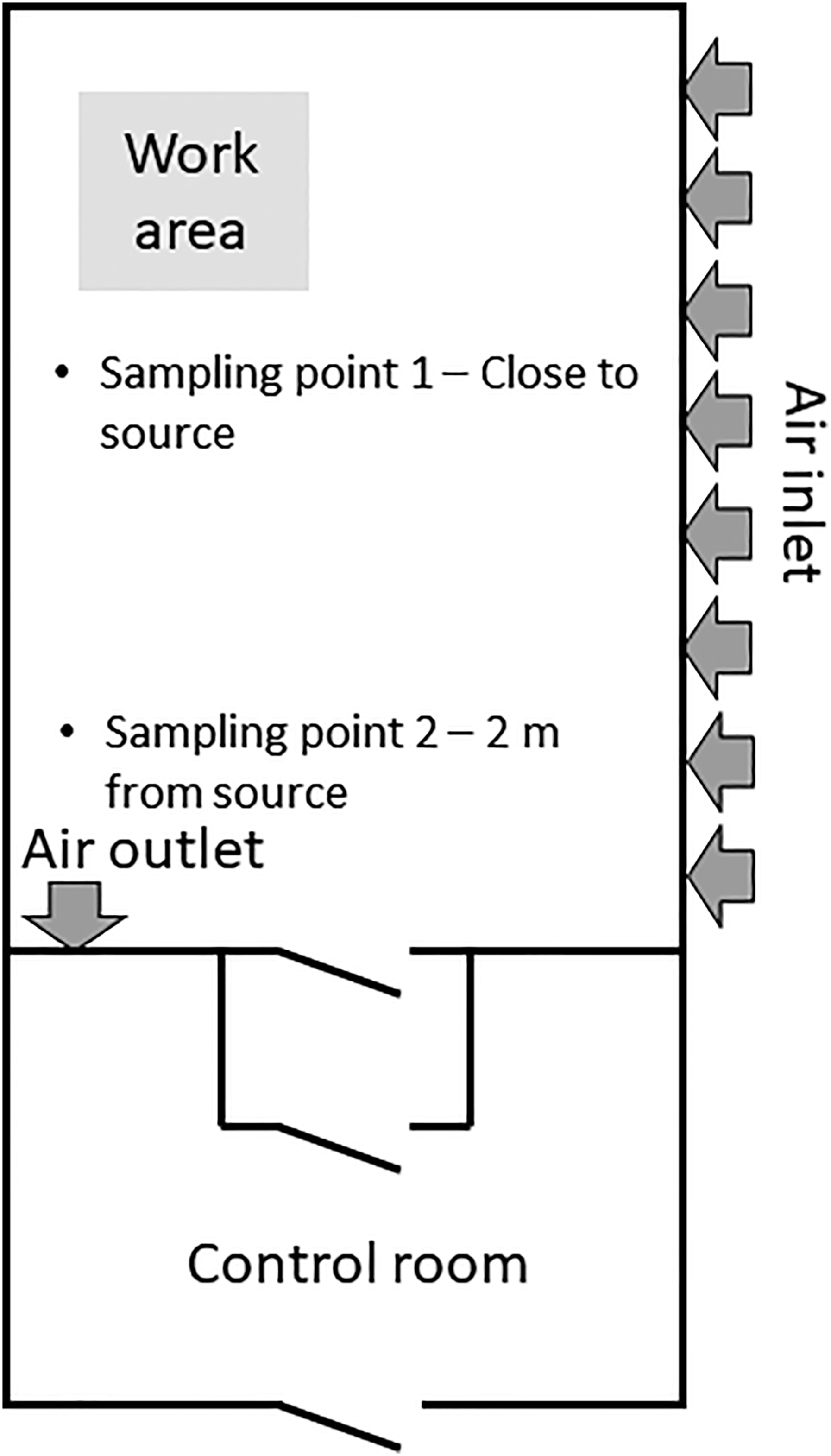
This sampling plan resulted in six ASSET samplers for isocyanate monitoring and six impinger-filter samples for diamine monitoring during the work operation for each of the 12 tests. In addition, immediately after the end of each work operation, new duplicate stationary sampling was performed (close to the work operation and 2 m away from the work operation) for an additional 120 min to assess the post-application curing phase. This resulted in an additional four ASSET samplers for diisocyanate monitoring and four impinger-filter samples for diamine monitoring and was done for all the emission tests except mixing of parquet glue (Test 3 and Test 4).
To ensure that there was no contamination inside the controlled-environment chamber, prior to the start of each test two ASSET samplers and two impinger samplers were collected for 120 min inside the chamber (these were all found negative).
During all the sampling with the impinger-filter samplers, the reagent solution (0.01
Work-up and analysis
After sampling of diisocyanates using the ASSET samplers, the isocyanate-DBA derivatives were extracted from the filters. Deuterium-labelled isocyanate-DBA derivative (used as internal standard) was added to the extraction solutions. The excess reagent and solvent were evaporated and the samples were re-dissolved in acetonitrile followed by LC-MS/MS analysis. Quantification was made by MRM. Calibration was performed in the range of 0.00008–0.028 μg for 2,4’-MDI and 0.002–0.7 μg for 4,4’-MDI. The limit of quantification (LOQ) was 0.002 μg for both 2,4’-MDI and 4,4’-MDI. Sampling at 200 mL/min for 120 min resulted in a LOQ of = 0.08 μg/m3 (0.03 μg NCO/m3). 2,2’-MDI was not individually quantified.
After sampling of MDA using the impinger-filter samplers, deuterium-labelled amine-ethyl chloroformate (ET) adducts (used as internal standards) were added to the sample solutions. Isocyanate groups were derivatised by DBA immediately upon being captured by the sampler during sampling. The amine groups were derivatised in subsequent two-phase derivatisation procedure with ET to form carbamate esters. The excess reagent and solvent were evaporated, and the samples were re-dissolved in acetonitrile. The samples were analysed using reversed-phase liquid chromatography (LC) and electrospray (ESP) mass spectrometric (MS) detection, monitoring positive ions. Calibration was performed in the range of 0.008–0.56 μg for MDA. Quantification was made by MRM. The LOQ was 0.01 μg for 4,4’-MDA. Sampling at 1000 mL/min for 120 min resulted in a LOQ of 0.09 μg/m3 (The MDA isomers were quantified as a sum and reported as 4,4’-MDA).
A more detailed description of the work-up and analysis procedures for the ASSET sampler and impinger-filter sampler is available elsewhere (Gylestam et al., 2014; Karlsson et al., 2002; ISO, 2013a, 2013b).
Results
Methylene diphenyl isocyanate (MDI) concentration in the collected samples from the different emission tests. Test 1: Application of one-component foam; Test 2: Application of construction glue; Test 3: Mixing of two-component parquet glue I; Test 4: Mixing of two-component parquet glue II; Test 5: Application of parquet glue I and test 6: Application of parquet glue II. The concentrations are given in μg/m3. Values that were less than the limit of quantification of 0.03 for MDI and 0.09 for methylene diphenyl diamine (120 min of sampling at 200 mL/min) are marked with ‘-'. ‘A’ and ‘B’ in the test numbers refer to duplicate experiments. The 120–240 min sampling was not performed for Tests 3 and 4; therefore, these are marked as N/A (not applicable) in the table.

MDI: methylene diphenyl isocyanate.
Discussion
It is important to obtain information regarding the risk of exposure from hazardous compounds during applications that involve the use of consumer products in general; this is even more important with DIY consumer products that involve reactive components (i.e. those that ‘cure’). Safety precautions such as ventilation, work routines and personal respiratory protection cannot be guaranteed in the same way for DIY applications as for handling of diisocyanate products in industrial or professional settings. Under the experimental conditions in this study, which were considered to be representative of typical DIY consumer use of these products, no MDA emission was measured above the LOQ, MDI was only measured above the LOQ in a few of the collected samples during the different tests performed and no MDI or MDA emissions were measured during the post-application/curing phase. The LOQ of 0.09 μg/m3 for MDA is three orders of magnitude below the existing occupational exposure limits (OELs) (typical occupational exposure limit of 80 μg MDA/m3 [10 ppb] (United States Department of Labor (2019)). In addition, a theoretical estimation based on the maximum level of MDA found in the curing products (Supplementary Material 2) indicates that the maximum possible MDA air concentration during curing of the products tested would be 0.002 μg/m3 (2 ng/m3).
The air concentrations of MDI that were greater than the LOQ were found in the breathing zone samples or the stationary samples close to the emission source. The levels found were almost two orders of magnitude below existing OELs (typical occupational exposure limit of 50 μg MDI/m3 in the EU) and the no observable effect level (NOEL) in animal tests was 0.2 mg/m3 for MDI (Feron et al. (2001). The DIY consumer products tested were selected as having the highest MDI concentration among products on the market in their category. The results of this study demonstrated that airborne exposure to MDI and MDA during preparation, application and post-application curing of MDI-based DIY consumer products is minimal. It is important to emphasize that, when handling DIY consumer products, instructions for safe handling of the products must be followed and adequate protective gloves and clothing should be used to minimize the risk of dermal exposure.
The diisocyanate and diamine emission found from the different work operations studied are most likely to occur as a vapour. Impinger-filter sampling can be used for simultaneous selective sampling of isocyanates and amines (ISO, 2013a, 2013b). However, there is a risk of underestimating MDI vapour concentrations using impinger flasks due to potential condensation of MDI in the impinger inlet glass tubing. To avoid this, ASSET samplers were used for isocyanate monitoring and impinger-filter samplers for selective amine monitoring after DBA derivatisation of any isocyanate groups captured during sampling. Sampling of isocyanates using ASSET samplers and the determination of their DBA derivatives using LC-MS/MS provides a convenient sampling approach with low detection limits. The LOQs presented in this study for diisocyanate monomers can be lowered even further by increasing the enrichment and optimisation of the MS data acquisition. This would be beneficial if there was a need to compare sampling results to a lower exposure limit or guideline.
Summary and conclusions
The air emission potential of the DIY consumer products studied was determined under extreme application conditions of high humidity (70–80% RH) in a specialized controlled-environment chamber. No concentrations of airborne MDA were found greater than the LOQ in any of the samples collected during the extreme-case conditions (i.e. high humidity) for any of the applications studied. In addition, airborne MDI concentrations from DIY consumer products were less than the LOQ for the mixing and foaming applications; airborne MDI concentrations were found to be just above the LOQ, in the range of 0.1–0.7 μg/m3 (0.03–0.2 μg NCO/m3), in the breathing zone and close to the work operation for the different gluing operations.
Since the MDI results were almost two orders of magnitude less than the relevant OEL of 50 μg MDI/m³, these results indicated that only incidental exposure to MDI at insignificant levels would be expected from application of MDI-based DIY consumer products. For MDA, the results indicated that no inhalation exposure at any significant levels would be expected during applications of such DIY consumer products. These results provide user exposure estimates under realistic exposure scenarios which could be used in risk assessment/characterisation.
Supplemental Material
sj-pdf-1-tih-10.1177_07482337221079433 – Supplemental Material for Emission of methylene diphenyl isocyanate and methylene diamine during use of cure-in-place methylene diphenyl isocyanate-based consumer products
Supplemental Material, sj-pdf-1-tih-10.1177_07482337221079433 for Emission of methylene diphenyl isocyanate and methylene diamine during use of cure-in-place methylene diphenyl isocyanate-based consumer products by Daniel Karlsson, Thomas Merz and Volker Nuthmann in Toxicology and Industrial Health
Supplemental Material
sj-pdf-2-tih-10.1177_07482337221079433 – Supplemental Material for Emission of methylene diphenyl isocyanate and methylene diamine during use of cure-in-place methylene diphenyl isocyanate-based consumer products
Supplemental Material, sj-pdf-2-tih-10.1177_07482337221079433 for Emission of methylene diphenyl isocyanate and methylene diamine during use of cure-in-place methylene diphenyl isocyanate-based consumer products by Daniel Karlsson, Thomas Merz and Volker Nuthmann in Toxicology and Industrial Health
Footnotes
Acknowledgements
The research reported in this work was sponsored by the International Isocyanate Institute. The views expressed are those of the author(s) and not necessarily of the sponsor or its members. The authors wish to express appreciation to Georg Pirkl (Covestro AG), Erik Vangronsveld (Huntsman Company) and Ronald van den Bosch (Dow, Inc.) for their assistance in conceptualizing and designing the study, and to Mark Spence (International Isocyanate Institute, Inc.) for assistance in manuscript preparation.
Declaration of conflicting interests
Daniel Karlsson declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Thomas Merz and Volker Nuthmann are employed by BASF Polyurethanes GmbH, a producer of MDI and TDI.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by International Isocyanate Institute.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
