Abstract
Objective:
To explore the effect of long-term antipsychotics use on the strength of functional connectivity (FC) in the brains of patients with chronic schizophrenia.
Method:
We collected resting-state functional magnetic resonance imaging from 15 patients with continuously treated chronic schizophrenia (TCS), 19 patients with minimally TCS (MTCS), and 20 healthy controls (HCs). Then, we evaluated and compared the whole-brain FC strength (FCS; including full-range, short-range, and long-range FCS) among patients with TCS, MTCS, and HCs.
Results:
Patients with TCS and MTCS showed reduced full-/short-range FC compared with the HCs. No significant differences in the whole-brain FCS (including full-range, short-range, and long-range FCS) or clinical characteristics were identified between patients with TCS and MTCS. Additionally, the FCS in the right fusiform gyrus, right inferior temporal gyrus, and right inferior occipital gyrus negatively correlated with the duration of illness and positively correlated with onset age across all patients with chronic schizophrenia.
Conclusions:
Regardless of the long-term use of antipsychotics, patients with chronic schizophrenia show decreased FC compared with healthy individuals. For some patients with chronic schizophrenia, the influence of long-term and minimal/short-term antipsychotic exposure on resting-state FC was similar. The decreased full- and short-range FCS in the right fusiform gyrus, right inferior temporal gyrus, and right inferior occipital gyrus may be an ongoing pathological process that is not altered by antipsychotic interventions in patients with chronic schizophrenia. Large-sample, long-term follow-up studies are still needed for further exploration.
Introduction
A majority of patients with schizophrenia in chronic stage can hardly achieve full clinical and social functional recovery, 1 which imposes serious mental and economic burdens 2,3 on patients and their families, and deserves our increasing attention. 4 Antipsychotic medication is one of the primary interventions for schizophrenia. 4 –6
The effects of antipsychotics on controlling psychotic symptoms have been consistently recognized as useful in both patients with first-episode psychosis and the relapsed stages. 5,7 The mechanisms of antipsychotics vary by type. Most antipsychotics block the dopamine D2 receptor to relieve positive symptoms. 8 –10 Others act on glutamate receptors to alleviate psychotic symptoms. 4,9,10 Regarding the improvement in emotional, negative, and cognitive symptoms, potential pharmacological mechanisms are described below. 4,9,10 Atypical antipsychotics also exert effects by blocking α2 receptor (risperidone and paliperidone) and serotonin 5-HT2A receptor, activating serotonin 5-HT1A receptor (quetiapine and ziprasidone), or functioning as serotonin 5-HT2C receptor antagonists (olanzapine, clozapine, and quetiapine) to relieve emotional, negative, and cognitive symptoms. 9,10 However, during chronic periods, the effects of antipsychotics on improving psychotic symptoms diminishe over time. 11 Furthermore, long-term use of first-generation antipsychotics or high doses of second-generation antipsychotics potentially produces additional psychotic symptoms 12,13 due to drug-induced dopamine receptor hypersensitivity, which is one of the underlying causes of treatment resistance. 14,15
An understanding of the effects of antipsychotics on brain function may help us identify the brain regions targeted by antipsychotics and psychotic symptoms. 16 –18 For first-episode schizophrenia (FES), abnormal functional indexes in some brain regions were altered after the short-term use of antipsychotics and were associated with the severity of psychotic or cognitive symptoms. 19 –21 Furthermore, the cerebral functional indexes of certain regions are potentially important in predicting the therapeutic effects of antipsychotics. Functional connectivity (FC) in specific brain regions, that is, the ventral tegmental area/midbrain, striatum, and the sensory-motor network, may predict treatment responses to antipsychotics. 20,22 –24
Resting-state functional magnetic resonance imaging (rs-fMRI) reflects the organization of spontaneous cerebral fluctuations, which are maintained during the task and play important roles in diverse cerebral responses. 25 Because a task is not required and the data are convenient to acquire, rs-fMRI has been applied to study the effect of antipsychotics on brain function in patients with schizophrenia. 25,26
Most recent studies have focused on changes in cerebral function after short-term (several weeks or months) use of antipsychotics, particularly, in FES patients. 27 The indexes assessed with rs-fMRI in some regions or networks, including the prefrontal cortex, striatum, hippocampus, superior temporal gyrus, thalamus, default mode network, and dorsal attention network, were changed after short-term exposure to antipsychotics and were associated with clinical characteristics. 18,19,22,28 –30 Long-term (>2 years) exposure to antipsychotics was reported to be associated with gray matter (GM) loss controversially. 31 –33 However, rs-fMRI studies exploring the effect of long-term antipsychotic use on brain function are mostly limited to animal models and have rarely involved human patients. 27
The influence of more than 5 years of continuous exposure to antipsychotics on cerebral function in patients with chronic schizophrenia remains unclear. We compared the differences in whole-brain resting-state FC between patients with continuously treated chronic schizophrenia (TCS) and minimally TCS (MTCS) to explore the potential effects of long-term exposure of antipsychotics on brain function. According to the Treatment Guideline for Schizophrenia of the American Psychiatric Association (APA), over 70% of FES patients reach full remission after treatment for 3 to 4 months and require approximately 1-year treatment. 34,35 We defined the minimally treated group as patients receiving a continuous therapeutic dose of antipsychotics for less than 3 months, accumulating a total therapeutic dose of antipsychotics for less than 1 year, and having not received continuous antipsychotics for the last 3 months. The guideline recommends the indefinite use of antipsychotics for patients with 2 episodes within 5 years; 34,35 thus, we regarded patients who used antipsychotics for more than 5 years as the TCS group. We investigated the differences in the long-range and short-range FC, based on anatomical distance, in these patients. Furthermore, as a supplementary evaluation, we collected and compared the clinical characteristics between the TCS and MTCS patients. Finally, we measured the associations between rs-fMRI FC measures and clinical characteristics. Based on the diminished effects of antipsychotics with time 14,36 –38 and the theory of inefficient neural system stabilization that posits that the brain may undergo spontaneous resolution after onset of psychosis to finally restore the sparse connectivity with low efficiency over time, 39,40 we hypothesize that the long-term use of antipsychotics may exert a limited influence on resting-state functional connection patterns in certain patients with schizophrenia.
Materials and Methods
Participants
We enrolled the TCS patients, MTCS patients, and healthy controls (HCs). Both the TCS and MTCS patients had been diagnosed with schizophrenia according to the Structured Clinical Interview for DSM-IV, had the duration of 5 years or more, and had never been treated with electroconvulsive therapy. Additionally, patients with TCS had received continuous antipsychotic treatment after the onset of the disease, with the requirement that withdrawal from antipsychotic drugs had not exceeded 3 months and cumulative drug withdrawal did not exceed 1 year. Patients with MTCS had received a continuous therapeutic dose of antipsychotics for less than 3 months, had accumulated a total therapeutic dose of antipsychotics for less than 1 year, and had not received continuous medication for the last 3 months. All participants, including patients with TCS, MTCS, and HCs, met the following criteria: (1) 18 to 45 years old, (2) right-handed, (3) Chinese Han ethnicity, (4) no serious physical illness, (5) no history of consciousness disturbance for more than 5 min and no medical history of drug abuse, (6) completed at least one cognitive test, (7) no metal in the body, (8) no pregnancy, and (9) no organic causes after neuroradiological assessments.
Fifty-four subjects were included, including 19 MTCS patients, 15 TCS patients, and 20 HCs. The process of recruiting patients and treatments of TCS and MTCS patients is described in Supplementary data (Sections 1 to 3).
The Positive and Negative Syndrome Scale (PANSS) was used to evaluate clinical psychiatric symptoms. The MATRICS Consensus Cognitive Battery (MCCB) plus the grooved pegboard test were applied to evaluate the cognitive function of schizophrenia. The original scores for the cognitive domains were acquired from scores for several subtests and converted into T scores based on the Chinese norm formula. The social function of all patients was assessed using the Personal and Social Performance (PSP) Scale.
The study was approved by the Medical Research Ethics Committee of Peking University Institute of Mental Health. All participants or their legal guardians provided written informed consent.
Image Acquisition
All participants were scanned by a General Electric (GE) 3.0-T scanner at Peking University People’s Hospital using an 8-channel sensitivity encoding head coil. All participants were required to close their eyes and stay awake during the scan. Earplugs and foam pads were used to reduce the impact of scanner noise and minimize head motion. All functional images were acquired across 240 time points with the following parameter settings: field of view = 240 × 240 mm2, thickness/gap = 5 mm/1.2 mm, repetition time = 2,000 ms, echo time = 40 ms, flip angle = 90°, matrix = 64 × 64, and 22 to 24 slices (2 participants were scanned with 23 slices and 1 participant was scanned with 24 slices, respectively).
Functional Data Preprocessing
Data preprocessing was performed using Statistical Parametric Mapping (SPM12, https://www.fil.ion.ucl.ac.uk/spm/) and the Data Processing Assistant for the R-fMRI (DPARSF) tool kit (http://rfmri.org/DPARSF). 41 First, the first 10 functional volumes were discarded to allow for stabilization of scanner equilibrium and the participants’ adaptation to the scanner circumstances. Then, the remaining fMRI data were performed by temporal correction for slice acquisition time delay and intervolume geometrical displacement due to the head motion were further performed successively. All fMRIs were then spatially normalized into the Montreal Neurological Institute space using the echo planar imaging (EPI) template and resampled to 3-mm isotropic voxels. Next, the following nuisance variables from each voxel’s time series were regressed out, including 24 head motion parameters, 42 cerebrospinal fluid, and white matter time series and the linear trend, followed by filtering all residual time series within a frequency range of 0.01 to 0.1 Hz to reduce the effect of low-frequency drifts and high-frequency noise. 43,44 Finally, the residual signals were used for the following analysis. Notably, we did not regress the effect of the global signal since this signal may contain significant information in patients with schizophrenia. 45,46 Additionally, participants with movement over 2.5 mm of translation or 2.5° of rotation were removed from the following analysis.
Resting-State FC Strength (FCS)
A whole-brain resting-state FCS analysis was performed on the above-preprocessed data. We calculated Pearson’s correlation coefficients between the time series of all pairs of gray matter voxels to obtain a whole-brain FC matrix for each subject. Then, we transformed individual correlation matrices into z-score matrices by applying Fisher’s r-to-z transformation. Because of the vague explanation for negative and weak correlations, 47,48 we conservatively restricted the FC analyses at a threshold of 0.2. Thus, the full-range FCS of a certain voxel was computed as the mean of the connections (z values) between this voxel and all other voxels. Moreover, we divided full-range FCS into long-range FCS and short-range FCS that reflect the effects of anatomical distance on connectivity. The mean of the connectivity (z values) between this given voxel and other voxels with Euclidean distances less than 75 mm was regarded as short-range FCS. Meanwhile, the mean of the connections (z values) with Euclidean distances greater than 75 mm were defined as long-range FCS. 49,50 Finally, all the FCS maps were smoothed with a 4-mm full width at half maximum Gaussian kernel.
Statistical Analysis
Demographic and clinical components
One-way analysis of variance (ANOVA) was applied to compare age among the three groups. Gender distribution was compared with the Pearson χ2 test. Onset age, the sum scores for the total, positive, negative, and general psychotic subscales of the PANSS, neuropsychological domain scores (except emotional intelligence), and PSP were compared using two-sample t tests between TCS and MTCS patients. Years of education, duration of disease, emotional intelligence and mean framewise displacement (FD) did not conform to the normal distribution. The Kruskal–Wallis H test was used to compare years of education and mean FD between the 3 groups. The Mann–Whitney U test was applied to compare emotional intelligence and duration of disease between the TCS and MTCS patients.
Resting-state fMRI
To examine differences between the TCS, MTCS, and HC groups in full-range, long-range, and short-range FCS, voxel-wise one-way analyses of covarianc based on a general linear model were performed, with age, gender, and years of education included as covariates. Considering the potential influence of head motion on FC, we calculated the FD values for each participant 51 and included the mean FD as an additional covariate. 52 Subsequently, a voxel-wise post hoc analysis was used to perform pair-wise tests of all significantly different voxels among the three groups. The statistical threshold was set at P < 0.05 for multiple comparisons corrected with Gaussian random field (GRF) theory (voxel significance: P < 0.001, cluster significance: P < 0.05, two-tailed) using the Data Processing & Analysis for (Resting-State) Brain Imaging (DPABI_V3.0_171210) software. 53 Moreover, we saved the abnormal clusters identified in patients with MTCS and TCS after voxel-wise post hoc analysis, respectively. Then, we calculated the common/overlapping regions between the abnormal regions identified in MTCS and TCS and saved the overlapping regions. We integrated TCS and MTCS patients as a group of patients with chronic schizophrenia. Partial correlation analysis was applied between the patients’ (including the TCS and MTCS patients) mean z values of the overlapping clusters and duration of illness, onset age, PSP, PANSS scores (including total and subscale scores), and scores for cognitive domains. We controlled age, gender, years of education, and mean FD. The statistical threshold was P < 0.05 using a two-tailed test.
Results
Demographic and Clinical Characteristics
As shown in Table 1, no significant differences were observed in gender distribution, age, or mean FD. Years of education in the TCS were significantly higher than in the MTCS and HCs. No significant differences existed between the MTCS and HC groups, consistent with the recruitment process.
Demographic and Clinical Characteristics.
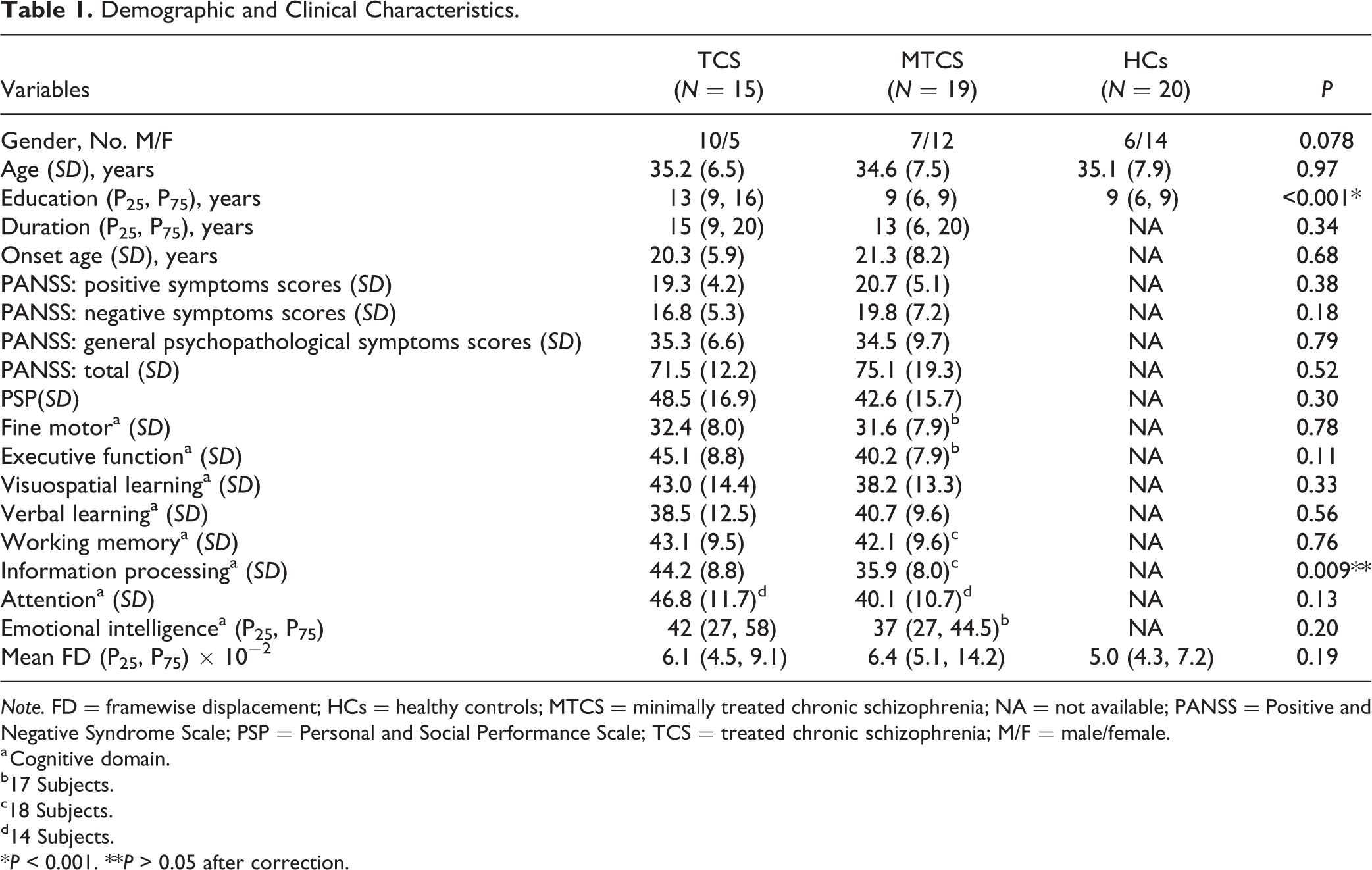
Note. FD = framewise displacement; HCs = healthy controls; MTCS = minimally treated chronic schizophrenia; NA = not available; PANSS = Positive and Negative Syndrome Scale; PSP = Personal and Social Performance Scale; TCS = treated chronic schizophrenia; M/F = male/female.
a Cognitive domain.
b17 Subjects.
c18 Subjects.
d14 Subjects.
*P < 0.001. **P > 0.05 after correction.
No significant differences were identified in duration, onset age, PSP, any cognitive domains scores (P > 0.05 after correction) and the sum of the scores for the total, positive, negative, and general psychopathology sections of PANSS between the TCS and MTCS groups.
Abnormal full-range FCS
Three regions including the right fusiform gyrus, right inferior temporal gyrus, and right inferior occipital gyrus showed significant differences among the three groups (Section 4. Table 1S in Supplementary data; GRF corrected, voxel significance: P < 0.001, cluster significance: P < 0.05, two-tailed).
Compared to the HCs, both the TCS and MTCS patients displayed lower full-range FCS in the right fusiform gyrus, right inferior temporal gyrus, and right inferior occipital gyrus. However, no significant differences existed between TCS and MTCS patients (Figure 1; Section 4. Table 2S in Supplementary data; GRF corrected, voxel significance: P < 0.001, cluster significance: P < 0.05, two-tailed).
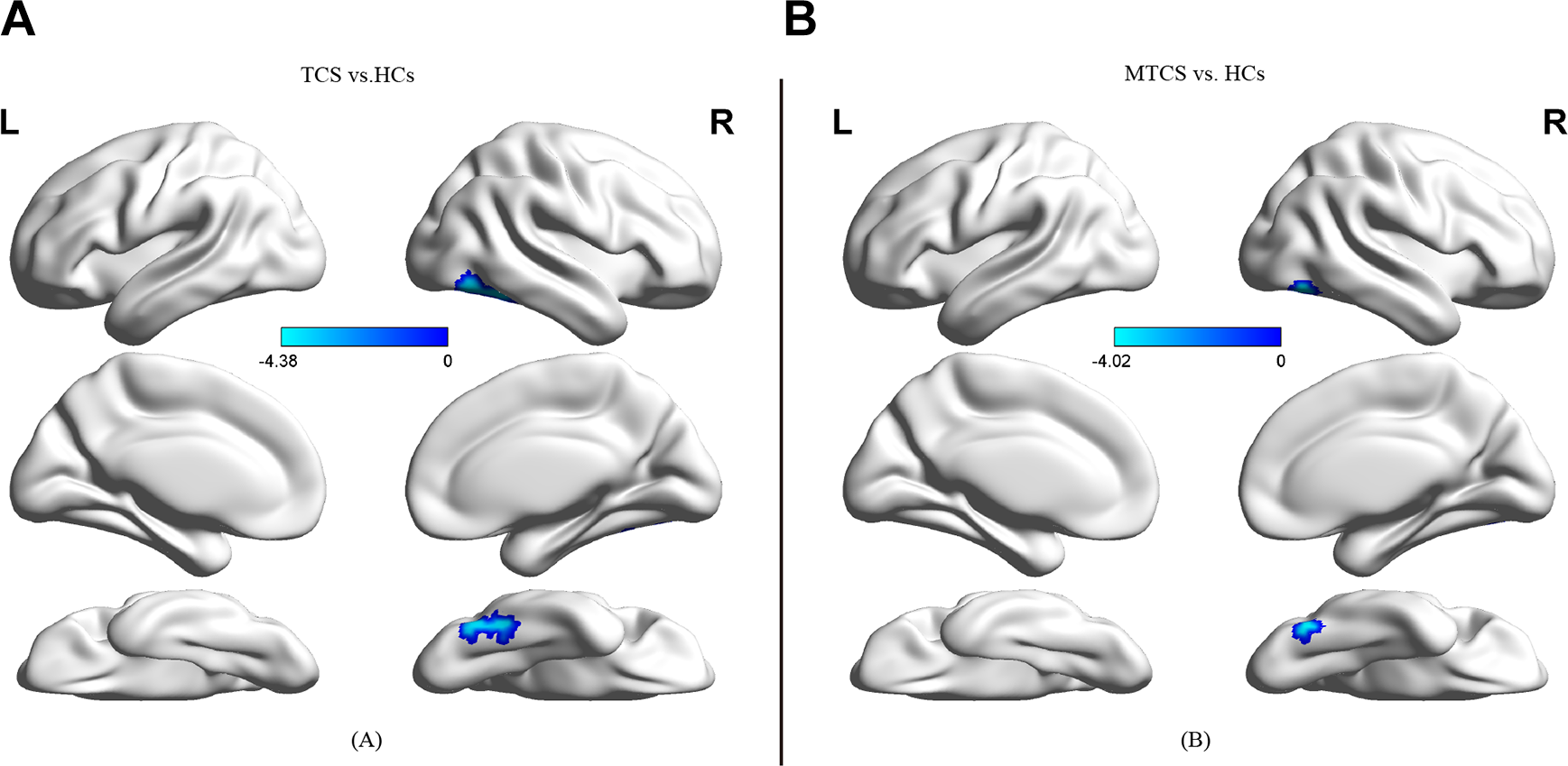
Full-range FCS maps. The left column (A) displays the brain regions with aberrant full-range functional connectivity in patients with TCS (n = 15) compared to HCs (n = 20). The right column (B) displays the brain regions with aberrant full-range FC in patients with MTCS (n = 19) compared to HCs (n = 20). Clusters represent brain regions with a decreased FCS in patients with TCS/MTCS compared to HCs. Colored bars indicate T scores. The surface map was acquired using BrainNet Viewer (http://www.nitrc.org/projects/bnv/. FC = functional connectivity; FCS = functional connectivity strength; HCs = healthy controls; MTCS = minimally treated chronic schizophrenia; TCS = treated chronic schizophrenia; vs = versus.
Abnormal short-range FCS
Several regions showed significant differences among the three groups. These regions included the right inferior temporal gyrus, bilateral fusiform gyri, bilateral inferior occipital gyri, bilateral insula, bilateral rolandic operculum, bilateral Heschl gyri, left lingual gyrus, left postcentral gyrus, left superior temporal gyrus, and left supramarginal gyrus (Section 4. Table 1S in Supplementary data; GRF corrected, voxel significance: P < 0.001, cluster significance: P < 0.05, two-tailed).
Compared to the HCs, both the TCS and MTCS patients exhibited lower short-range FCS in the bilateral fusiform gyri, bilateral inferior occipital gyri, right inferior temporal gyrus, and left lingual gyrus. No significant differences were observed between the TCS and MTCS groups (Figure 2; Section 4. Table 2S in Supplementary data; GRF corrected, voxel significance: P < 0.001, cluster significance: P < 0.05, two-tailed).
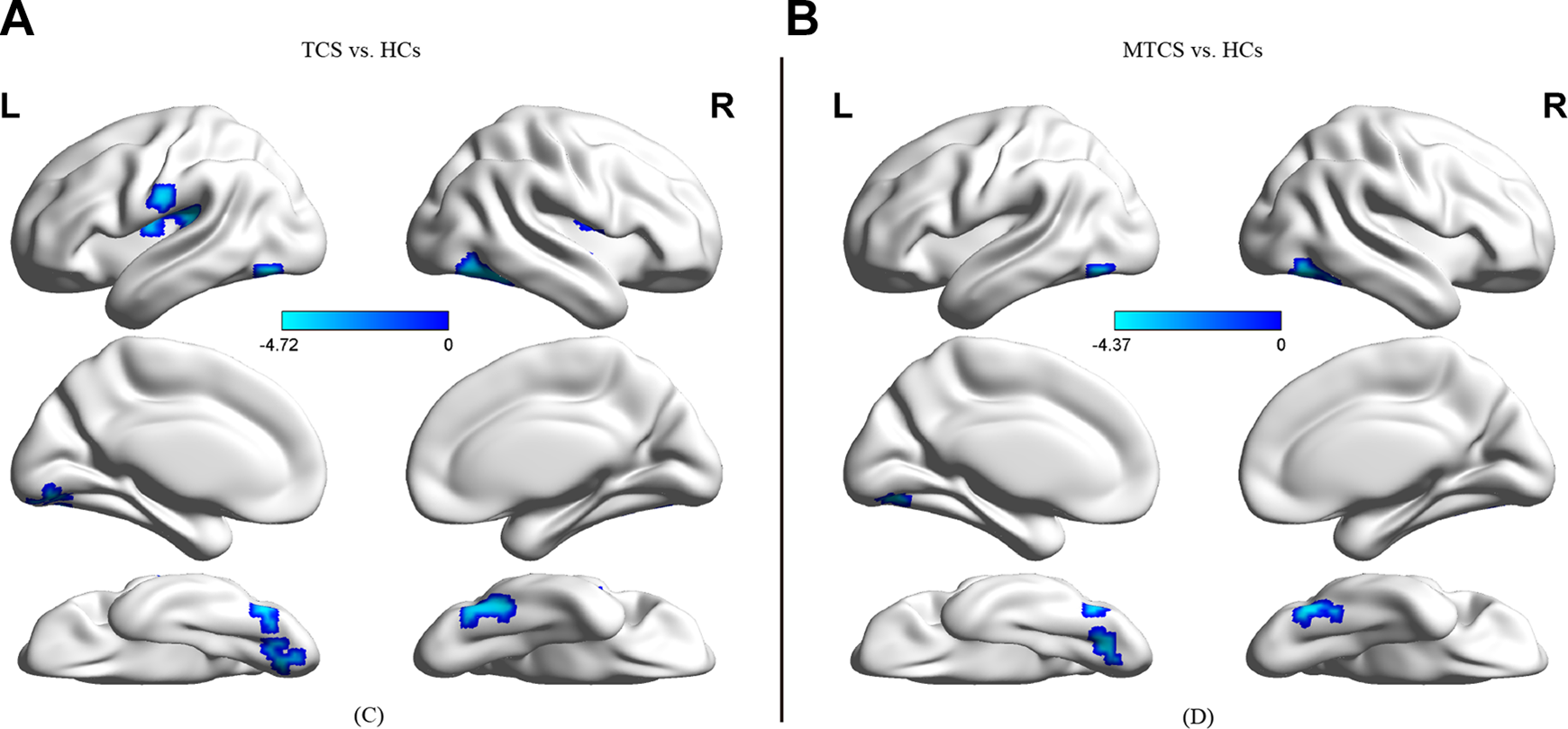
displays the brain regions with aberrant short-range functional connectivity in patients with TCS (n = 15) compared to HCs (n = 20). The right column (B) displays the brain regions with aberrant short-range functional connectivity in patients with MTCS (n = 19) compared to HCs (n = 20). Clusters represent brain regions with a decreased FCS in patients with TCS/MTCS compared to HCs. Colored bars indicate T scores. The surface map was acquired via BrainNet Viewer (http://www.nitrc.org/projects/bnv/). FC = functional connectivity; FCS = functional connectivity strength; HCs = healthy controls; MTCS = minimally treated chronic schizophrenia; TCS = treated chronic schizophrenia; vs = versus.
Abnormal long-range FCS
None.
Aberrant regions overlapping between the TCS and MTCS patients
As shown in Table 2, the aberrant regions overlapping between the TCS and MTCS groups consisted of the right fusiform gyrus, the right inferior temporal gyrus, and the right inferior occipital gyrus.
Overlapping Clusters between Patients with TCS and MTCS.
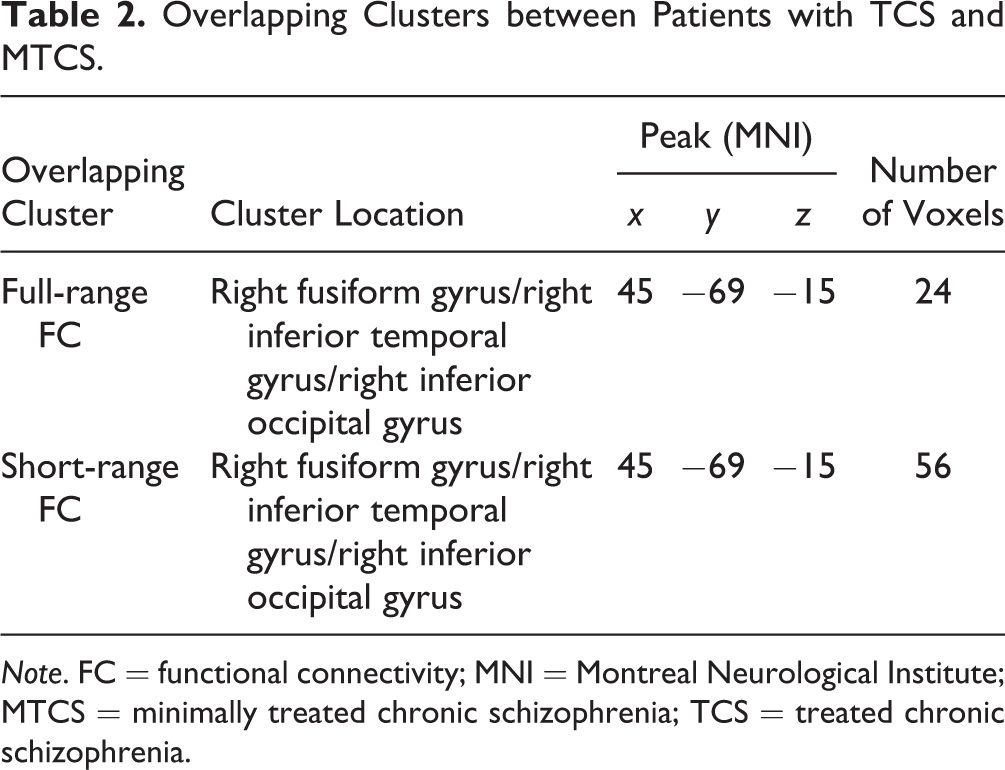
Note. FC = functional connectivity; MNI = Montreal Neurological Institute; MTCS = minimally treated chronic schizophrenia; TCS = treated chronic schizophrenia.
Correlations between the FCS of overlapping aberrant regions and duration of illness
As shown in Figure 3, the mean z values of overlapping regions in terms of full-range FC were negatively correlated with the duration of illness in all patients with chronic schizophrenia (r = −0.439, P = 0.015). The mean z values of overlapping regions in terms of short-range FC were negatively correlated with the duration of illness in all patients with chronic schizophrenia (r = −0.376, P = 0.041).
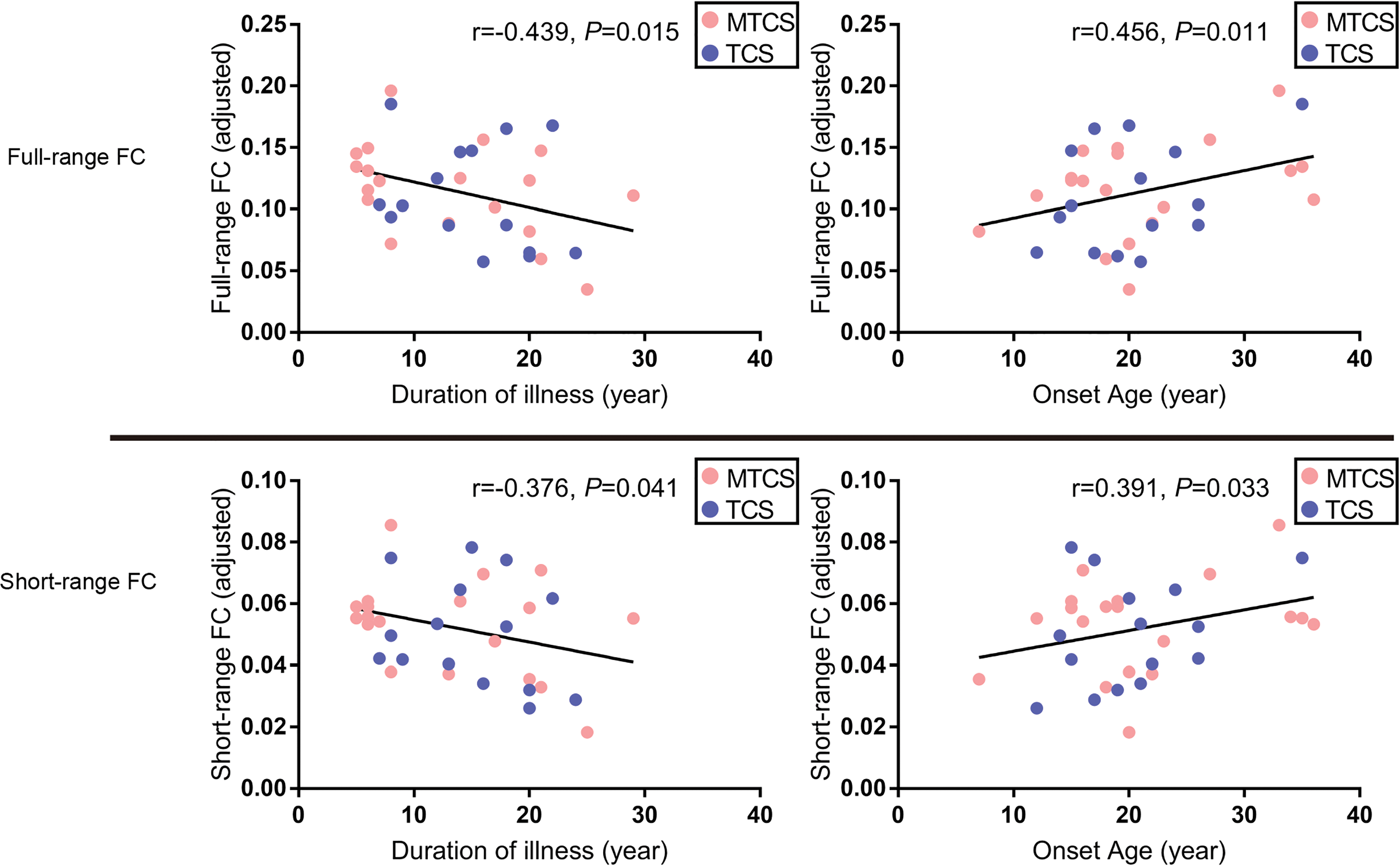
Correlations between mean z values for overlapping clusters and the duration of illness/onset age. The overlapping clusters included the right fusiform gyrus, right inferior temporal gyrus, and right inferior occipital gyrus. The red spots represent the patients with MTCS. The blue spots represent the patients with TCS. FC = functional connectivity; MTCS = minimally treated chronic schizophrenia; TCS = treated chronic schizophrenia.
Correlations between the FCS of overlapping aberrant regions and onset age
The mean z values of overlapping regions in terms of full-range FC, as shown in Figure 3, were positively correlated with onset age in all patients with chronic schizophrenia (r = 0.456, P = 0.011). The mean z values of overlapping regions in terms of short-range FC were positively correlated with onset age in all patients with chronic schizophrenia (r = 0.391, P = 0.033).
Correlations between the FCS and other clinical variables
No significant correlations were observed between the FCS and other clinical variables.
Discussion
In this study, we did not observe significant differences in resting-state FC and clinical characteristics between the TCS and MTCS patients. Consistent with previous studies, 54 –56 TCS and MTCS patients showed decreased FC patterns compared with HCs in our study, which also supports the disconnection hypothesis of schizophrenia. 54,57 Furthermore, in all patients with chronic schizophrenia, signals of full-range and short-range FC in the right fusiform gyrus, right inferior temporal gyrus, and right inferior occipital gyrus were associated with the onset age and the duration of illness.
Comprehension of the Lack of Significant Differences in Resting-State FC between the TCS and MTCS Patients
The effects of antipsychotics on some patients with schizophrenia may become limited over time. The psychotic symptoms of many continuously treated patients with schizophrenia appeared to stabilize through time and rarely achieved an overall improvement in a study. 38 More treatment options have become available in recent decades. However, it was reported that the proportion of patients achieving recovery rarely rose across time, and instead, it remained relatively stable. 1 Meanwhile, long-term exposure to antipsychotics may not restrain pathophysiologic processes and even reduce brain volume of schizophrenia. 31 A large proportion of patients with chronic schizophrenia who underwent antipsychotic therapy experienced a recurrence of positive symptoms. 11 In a 20-year follow-up study, researchers found that long-term antipsychotic medications could not reduce the frequency or severity of psychosis, although antipsychotics could relieve psychiatric symptoms in acute period. 37 For neuroimaging studies, limited improvements in resting-state network FC were observed after 6 weeks of risperidone treatments. 18 Besides, psychopathologists noted that long-term exposure to antipsychotics may induce the hypersensitivity of dopamine receptors and lead to a weakening effect. 14
In our study, we did not observe significant differences between the resting-state FC of TCS and MTCS, indicating that the influences of long-term treatment on resting-state FC were not different from short-term/minimal treatment in patients with chronic schizophrenia analyzed in the this study. Importantly, patients with schizophrenia who were enrolled in our study did not include individuals with severe psychotic symptoms, due to the need for safe transport MRI scanning (Section 2 in Supplementary data). Our results were not object to the important role of antipsychotics in the acute stage and the beneficial role of long-term use of antipsychotics in reducing of mortality and preventing of relapse. 7,15,58
Decreased Full-Range and Short-Range FC in Overlapping Regions between the TCS and MTCS Patients
Regardless of long-term use of antipsychotics, we found the overlapping decreases in full/short-range FC in the right fusiform gyrus, right inferior temporal gyrus, and right inferior occipital gyrus in patients with chronic schizophrenia. Thus, these gyri may be regions where long-term antipsychotics might not exert significant effect. In addition, long-range FC and short-range FC were distinguished by a threshold. 49,50 Based on our previous study 54 and other researchers’ results, 59 short-range FC may reflect the tendency or the basic disconnection in schizophrenia. Consistently, decreased short- and full-range, but not long-range, FC was observed in this study, indicating that alterations in full-range connectivity were mainly attributed to the changes in the short-range connectivity.
The overlapping regions play important roles in neuropsychiatric diseases. The inferior temporal gyrus is involved in processing visual stimuli, which is more sensitive to complex shapes and is the anatomical basis of prosopagnosia. 60 The fusiform gyrus plays a major role in processing facial signals, and an aberrant state of the fusiform gyrus is related to abnormal facial processing in schizophrenia. 55,61,62 The alterations in the inferior occipital gyrus may be involved in facial processing in schizophrenia. 63 The altered connectivity in these regions correlated with the onset age and the disease course in schizophrenia, suggesting that decreased full- and short-range FC of the overlapping regions may reflect an ongoing pathological process of chronic schizophrenia.
Limitations
First, although the sample size is small, after the rigid analysis of neuroimage, we also obtained significant and important results that may provide some scientific evidence for further research into chronic schizophrenia. Additionally, we made several attempts to analyze the correlations between clinical characteristics and FC rather than combining all significant correlations to draw a conclusion. We preferred to explain the possible meaning of the overlapping regions from the perspective of the onset age or disease duration. Second, patients in our studies do not represent individuals with recovery or with serious psychotic symptoms. Third, almost all TCS patients had experienced a combination of one or more antipsychotics or switching to another antipsychotic drug. In this study, it is very difficult for patients and psychiatrists to avoid medication switching and treatment adherence may not be guaranteed in the real world. Future studies exploring the characteristics of patients with long-term exposure to certain antipsychotics may help us understand the mechanism of certain antipsychotics better. Meanwhile, this study is a cross-sectional comparison based in the real world. Longitudinal follow-up studies are needed to further explore the effects of long-term antipsychotic exposure on the chronic pathophysiological process of schizophrenia.
Conclusion
Regardless of the long-term use of antipsychotics, patients with chronic schizophrenia show decreased FC patterns compared with healthy people. For some chronic schizophrenia, the influences of long-term and minimal/short-term antipsychotic exposure on resting-state FC are not substantially different. Additionally, the decreased full- and short-range FC in the right fusiform gyrus, right inferior temporal gyrus, and right inferior occipital gyrus may present an ongoing pathological process in chronic schizophrenia. Longitudinal studies with larger sample size are needed for further exploration.
Supplemental Material
Supplementary_Material_a_clean_copy - Influence of More Than 5 Years of Continuous Exposure to Antipsychotics on Cerebral Functional Connectivity of Chronic Schizophrenia
Supplementary_Material_a_clean_copy for Influence of More Than 5 Years of Continuous Exposure to Antipsychotics on Cerebral Functional Connectivity of Chronic Schizophrenia by Qi Miao, Chengcheng Pu, Zhijiang Wang, Chao-Gan Yan, Chuan Shi, Qingjiu Cao, Xijin Wang, Zhang Cheng, Xue Han, Lei Yang, Yunyao Lai, Yanbo Yuan, Hong Ma, Keqing Li, Nan Hong and Xin Yu in The Canadian Journal of Psychiatry
Footnotes
Acknowledgments
The authors thank all individuals who served as the research participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Beijing Science and Technology Commission (No. D171100007017002).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
