Abstract
Objective:
The imbalance in neurotransmitter and neuronal metabolite concentration within cortico-striato-thalamo-cortical (CSTC) circuit contributes to obsessive–compulsive disorder’s (OCD) onset. Previous studies showed that glutamate mediated upregulation of resting-state activity in healthy people. However, there have been few studies investigating the correlational features between functional and neurochemical alterations in OCD.
Methods:
We utilize a combined resting-state functional magnetic resonance imaging (rs-fMRI) and proton magnetic resonance spectroscopy (1H-MRS) approach to investigate the altered functional connectivity (FC) in association with glutamatergic dysfunction in OCD pathophysiology. Three regions of interest are investigated, i.e., medial prefrontal cortex and bilateral thalamus, for seed-based whole-brain FC analysis as well as MRS data acquisition. There are 23 unmedicated adult OCD patients and 23 healthy controls recruited for brain FC analysis. Among them, 12 OCD and 8 controls are performed MRS data acquisition.
Results:
Besides abnormal FC within CSTC circuit, we also find altered FCs in large-scale networks outside CSTC circuit, including occipital area and limbic and motor systems. The decreased FC between right thalamus and right middle occipital gyrus (MOG) is correlated with glutamatergic signal within right thalamus in OCD patients. Moreover, the FC between right thalamus and right dorsal anterior cingulate cortex (dACC) is associated with glutamate level in right thalamus, specifically in patient’s group. Finally, the FC between right thalamus and right MOG is correlated with patient’s Yale–Brown Obsessive Compulsive Scale (YBOCS) compulsion and total scores, while the right thalamic glutamatergic signal is associated with YBOCS-compulsion score.
Conclusion:
Our findings showed that the coupled intrinsic functional–biochemical alterations existed both within CSTC circuit and from CSTC to occipital lobe in OCD pathophysiology.
Keywords
Introduction
Obsessive–compulsive disorder (OCD), as a common psychiatric disorder, is characterized by unwanted obsessions and intrusive compulsions (Fontenelle and Hasler, 2008). The lifetime prevalence of OCD is approximately 2–3%, which badly influences patients’ social function.
Structural, functional and metabolic dysfunction within the cortico-striato-thalamo-cortical (CSTC) circuit of OCD has been reported by previous neuroimaging studies (Beucke et al., 2013; Milad and Rauch, 2012; Yin et al., 2018). The CSTC circuit includes thalamus (Brennan et al., 2013), caudate nucleus (Milad and Rauch, 2012), orbitofrontal cortex (OFC) and medial prefrontal cortex (mPFC)/anterior cingulate cortex (ACC). The mPFC/ACC is involved in affective/cognitive function primarily for motivation-related behaviors, while the OFC is responsible for the integration of emotional and limbic information into behavioral responses. The thalamus is important in integrating perception with thoughts. To improve the understanding of the neurobiology of OCD, complementary approaches have been proposed to further investigate the neurochemical variations within these areas.
Resting-state functional magnetic resonance imaging (rs-fMRI) has been widely applied in OCD studies. Hou et al. found that OCD patients had increased amplitude of low-frequency fluctuation (ALFF) values in OFC and ACC areas using rs-fMRI technology. Matilde et al. recently found that reduced FC of caudate-ventrolateral prefrontal cortex (VlPFC) was associated with reduced cognitive flexibility in OCD patients. Besides abnormal FC within CSTC circuit, increasing evidence showed that FC from CSTC circuit to occipital lobe and limbic system may be linked with mechanism of OCD. In an rs-fMRI study comparing OCD patients with their healthy first-degree relatives, the patients showed increased FC within the CSTC circuit, while decreased FC in the occipital cortex, temporal cortex and cerebellum. Therefore, the investigation of functional alteration from CSTC circuit to occipital/limbic system might add more insights into understanding the mechanism of OCD.
The imbalance in neurotransmitter and neuronal metabolite concentration within CSTC circuit has been shown contributing to OCD’s onset (Russell et al., 2003). Proton magnetic resonance spectroscopy (1H-MRS) has been applied to study OCD’s neurobiology alteration. Both animal and human studies have shown that the glutamatergic abnormality in CSTC circuit may lead to OCD symptomology (Kariuki-Nyuthe et al., 2014; Wu et al., 2012), which serves as a potential pharmacotherapy target (Milad and Rauch, 2012). Elevated Glx (Glutamate and Glutamine) concentration in cerebrospinal fluid (CSF) and caudate or reduced Glx level in ACC, mPFC and right thalamus of OCD patients have been reported (Yücel et al., 2008). Discrepant results were shown, such as elevated or unaltered Glx level in thalamus/mPFC/ACC regions, due to the difference in patient’s age or medication (Gnanavel et al., 2014; Simpson et al., 2012). Glutamate is a key excitatory neurotransmitter and plays a vital role within CSTC circuit (Milad and Rauch, 2012). A classical model of OCD pathophysiology is due to imbalance between the indirect and direct pathways in CSTC circuit, in which the indirect pathway contributes to thalamic inhibition of cortex and the direct pathway leads to thalamic stimulation of cortex. In OCD, it is postulated that disinhibition of CSTC circuit resulting from overactivity of the direct pathway causes consequent obsession behaviors and compulsion (Wu et al., 2012). The excessive activity in orbitofrontal–subcortical circuit and hyperactivity of glutamate that is related to an increase in glutamate level lead to the imbalance between direct and indirect pathways (Yücel et al., 2008).
Previous studies showed that glutamate mediated upregulation of resting-state activity in healthy people (Enzi et al., 2012). It is also supported that the glutamatergic neurons had a direct impact on blood oxygen-level dependent (BOLD) signal, while glutamate could mediate metabolic response to neuronal activity underlying functional imaging signal changes (Duncan et al., 2011). However, there have been few studies investigating the functional and neurochemical alterations simultaneously in OCD. Yücel et al. found greater deactivation of rostral anterior cingulate and activation of the supplementary motor area (SMA) during the MultiSource Interference Task in OCD patients. They also found decreased neuronal N-acetylaspartate (NAA) level in dorsal anterior cingulate cortex (dACC) negatively correlated with the dACC functional activation in OCD (Yücel et al., 2007). Brennan et al. showed reduced rostral anterior cingulate cortex (rACC) deactivation during an emotional counting Stroop task but did not find significant association between glutamatergic signal and rACC functional deactivation in OCD (Brennan et al., 2015).
Rs-fMRI signal reflects the brain intrinsic functional activity and the glutamatergic neurons have a direct impact on fMRI signal (Duncan et al., 2011). The combined MRS and task-related fMRI approach has been utilized to investigate the relationship between brain functional activation and neurochemistry of OCD. To the best of our knowledge, there is no study yet investigating the correlational characteristics of brain resting-state activity with metabolic biomarkers, specifically the glutamatergic abnormality, in OCD patients. In this paper, we utilized the combined rs-fMRI and 1H-MRS approach to investigate the coupling features between resting-state FC and glutamatergic dysfunction in unmedicated OCD patients. The MRS data are acquired in bilateral thalamus and mPFC areas, since they are core regions in the CSTC circuit (Brennan et al., 2013). We hypothesize that (1) OCD patients display altered FC features within CSTC circuit and to occipital/limbic system and (2) the FC changes may be associated with regional glutamatergic signaling in OCD patients.
Materials and methods
Subjects
We recruited 23 OCD patients and 23 control subjects from the Shanghai Mental Health Center, aged 18–54 years and matched in age, educational status, handedness and gender (Table 1). The healthy subjects were recruited by local advertising. Each participant signed the written informed consent by Shanghai Mental Health Center Ethics Committee prior to study participation. Each subject is right-handed and has senior middle school or higher education level. The OCD patients were psychotropic drugs free for at least 2 months. We used the Mini-International Neuropsychiatric Interview (MINI; Hergueta et al., 1998) to give Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) diagnosis of Axis I psychiatric disorders for each OCD patient. The exclusion criteria include diagnosis of traumatic brain injury, severe physical illness, comorbid Axis I psychiatric disorders or other nervous system disorders, suicide attempts, pregnancy, psychoactive substance abuse or lactation, dentures, pacemakers, stents, metal implants and other implanted magnetic resonance imaging (MRI)-incompatible devices.
Demographic and clinical information on participants.
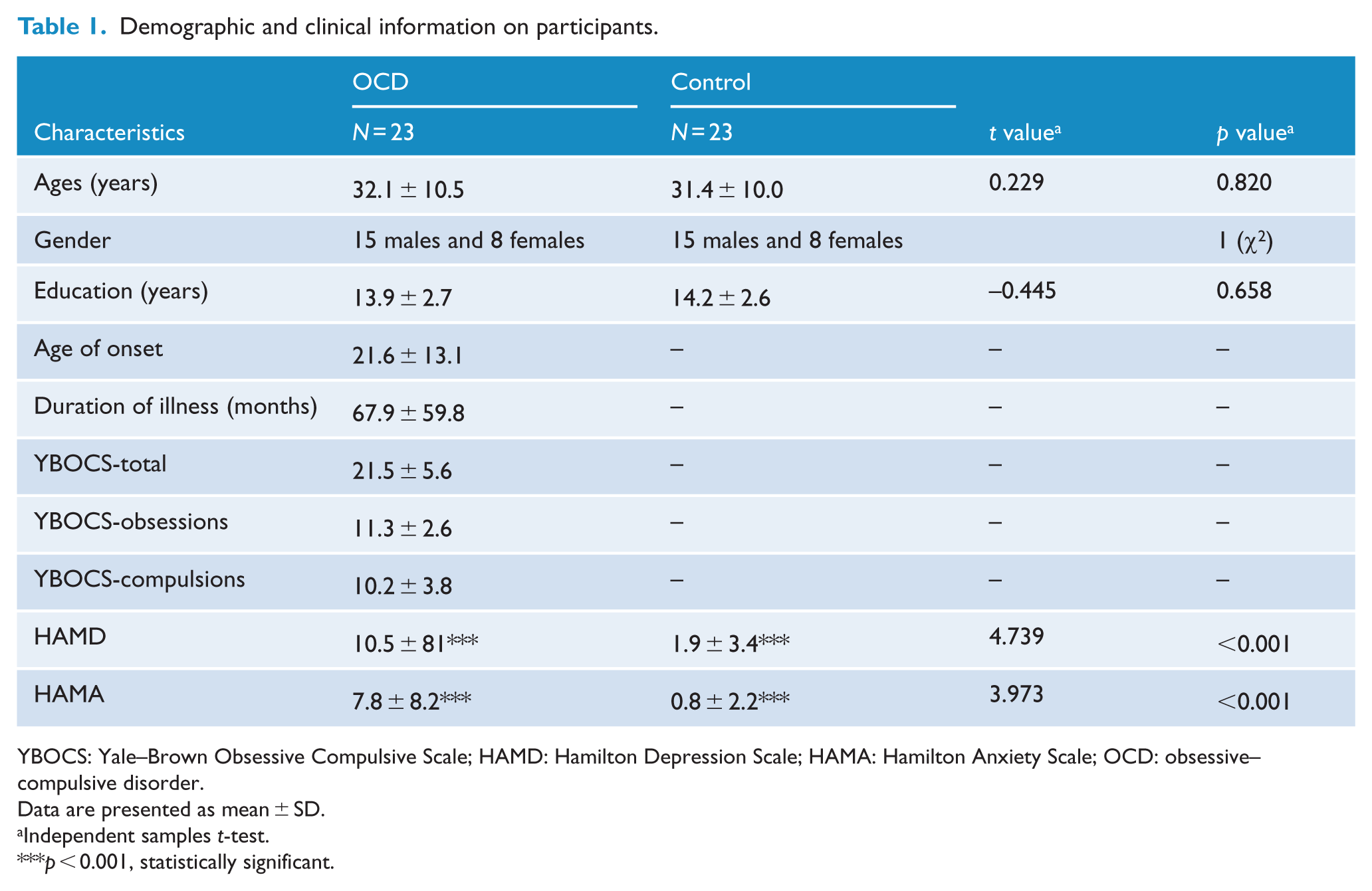
YBOCS: Yale–Brown Obsessive Compulsive Scale; HAMD: Hamilton Depression Scale; HAMA: Hamilton Anxiety Scale; OCD: obsessive–compulsive disorder.
Data are presented as mean ± SD.
Independent samples t-test.
p < 0.001, statistically significant.
Moreover, we used Yale–Brown Obsessive Compulsive Scale (YBOCS) to report the OCD symptom severity, with both compulsion and obsession features recorded. The Hamilton Depression Scale (HAMD; Williams, 1988) and Hamilton Anxiety Scale (HAMA) measurements were applied to score subjects’ anxiety and depression symptoms.
fMRI data acquisition and preprocessing
We collected the resting-state fMRI data on a 3.0-T Signa MRI scanner (Signa Horizon LX; General Electric Healthcare, Milwaukee, WI, USA) at Shanghai Ruijin Hospital. During MRI scanning, all participants laid supine with inflatable pillows placed between the head and coil to minimize the head motion. Functional MRI data acquisition was performed using gradient-echo echo-planar imaging (EPI) sequence with the following parameters: flip angle (FA) = 90°, field of view (FOV) = 24 cm, repetition time (TR) = 2100 ms, data matrix = 64 × 64, echo time (TE) = 30 ms, slice number = 33, thickness = 4.0 mm, voxel size = 3.75 × 3.75 × 4.6 mm3 and total number of volumes = 200. The subjects were required to lie still but keep awake with their eyes closed (Beucke et al., 2013; Harrison et al., 2009). We spent about 7 min for each fMRI scan.
For fMRI data processing, we used Data Processing Assistant for Resting-State fMRI (DPARSF) program for the resting-state fMRI image preprocessing, which is based on Resting-State fMRI Data Analysis Toolkit (REST v1.8, http://www.restfmri.net) and Statistical Parametric Mapping 8 (SPM8; http://www.fil.ion.ucl.ac.uk/spm). We discarded the first 10 volumes to allow the subjects to adapt to scanning noise and the signal equilibrium. The remaining 190 volumes were subsequently corrected for acquisition time to the middle slice. For each individual participant, we corrected head motion by adapting each time series to the first volume with a six-parameter (three for rotation and three for translation) and a least-square minimization linear spatial transformation. No patients were excluded for excessive head-motion of over 2° angular rotation or 2 mm translation in any direction. Next, we used the standard EPI template to spatially normalize the functional images and apply it to stereotactic space of Montreal Neurological Institute (MNI), with each voxel resampled to isotropic 3 × 3 × 3 mm3. Subsequentially, we used a Gaussian kernel of 4 mm full width at half-maximum (FWHM) to spatially smooth the data. Furthermore, we removed the linear trend and used a temporal bandpass filter (0.01–0.08 Hz) to the fMRI data to reduce the effect of very high-frequency physiological noise and low-frequency drift. We regressed out nine nuisance signals through linear regression, including the global mean signal, white matter signal, the estimated six head motion parameters and CSF signals.
1H-MRS data acquisition and processing
Among all subjects, 12 OCD patients and 8 healthy controls (HCs) underwent MRS scanning. Three regions of interest (ROIs) were investigated, i.e., bilateral thalamus and the mPFC, using a standard Point-Resolved Selective Spectroscopy (PRESS) pulse sequence (see Figure 1(B)). The mPFC voxel consisted of portion of bilateral medial orbital gyrus and the perigenual anterior cingulate gyrus (pACC). It was placed within the inferior border, superior to the olfactory sulcus and within the posterior border, as well as anterior to the corpus callosum sulcus. The following acquisition parameters were applied: TE = 35 ms, TR = 1500 ms, number of points = 4096 and number of scans = 64. Pre-scan calibrations involved an auto-shimming process to minimize the peak FWHM. The CHEmical Shift Selective (CHESS) radiofrequency (RF) pulse was applied for water suppression. The MRS data acquisition time was about 1.6 minutes.
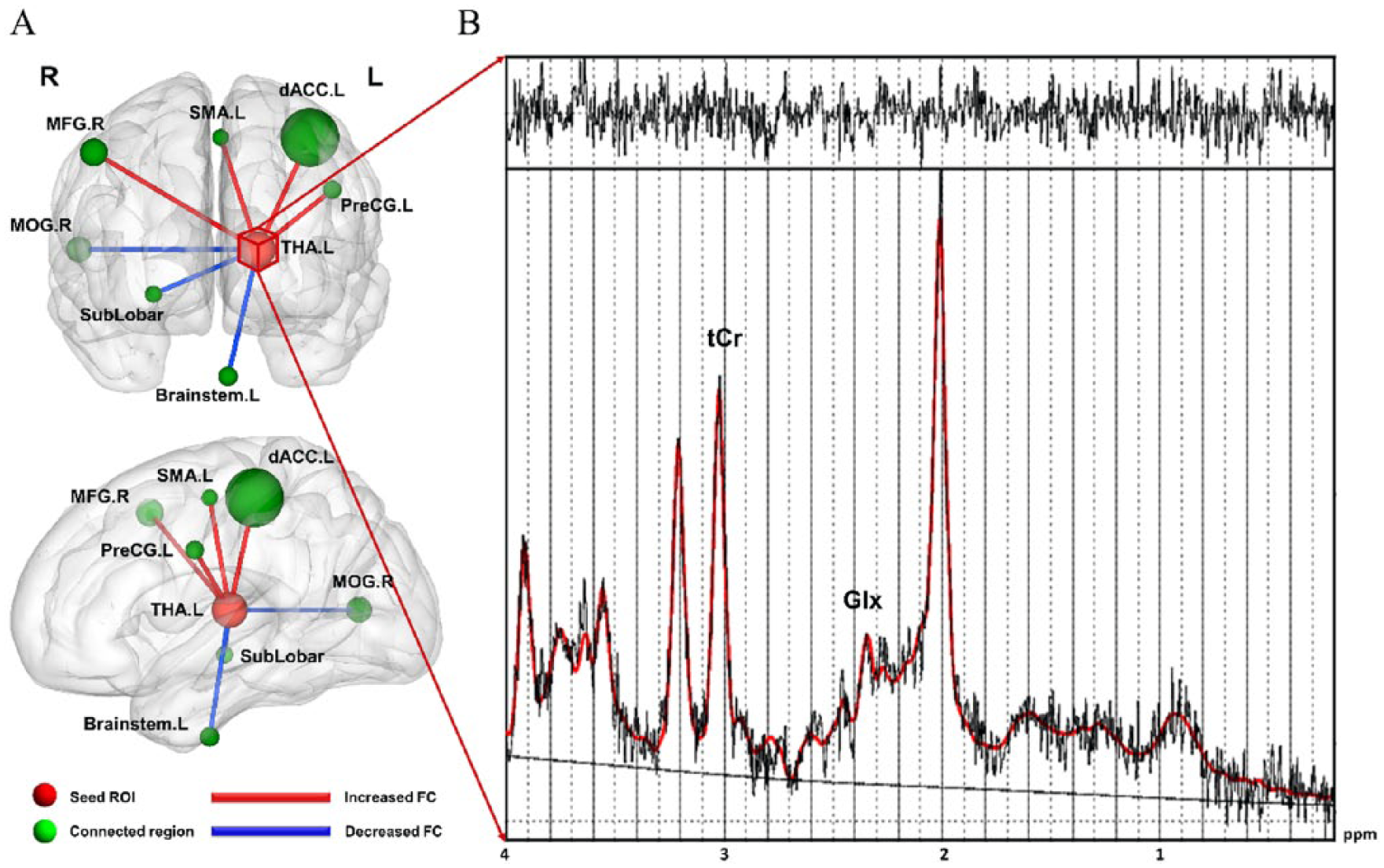
(A) Using the left thalamus as seed ROI, the functional connectivity (FC) difference between obsessive–compulsive disorder (OCD) patients and healthy controls. Red edges denote increased FC and blue edges denote decreased FC in OCD patients compared with healthy controls (p < 0.05, corrected with AlphaSim). The detailed statistics are listed in Table 2. The radius of each sphere is proportional to the corresponding cluster size. Each sphere center locates at the peak t-value position within the associated cluster. (B) A representative 1H-MRS spectrum acquired from the left thalamus fitted by LCModel. Metabolites: tCr—creatine and phosphocreatine; Glx—glutamine and glutamate.
Functional connectivity difference between OCD patients and healthy controls, left thalamus as seed region.
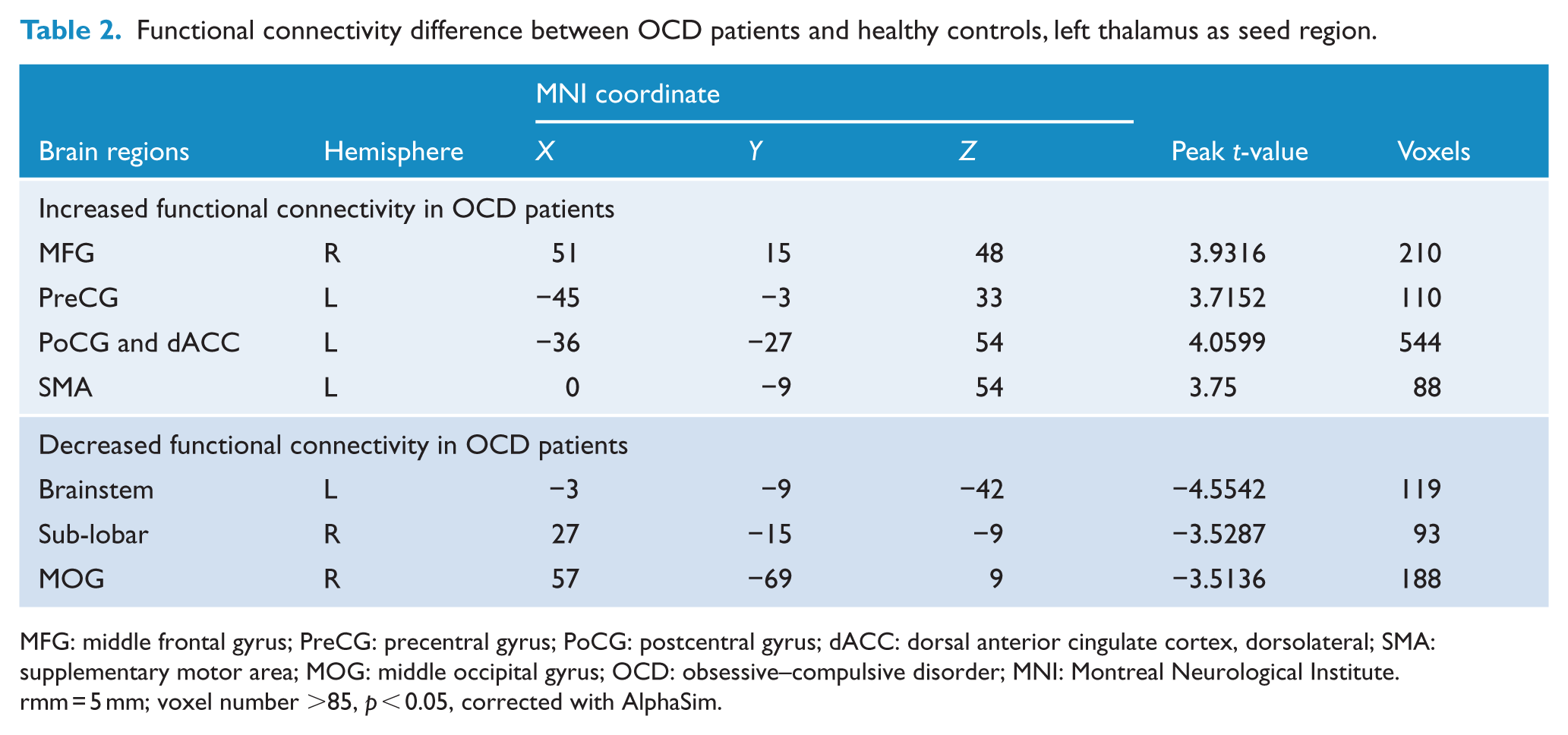
MFG: middle frontal gyrus; PreCG: precentral gyrus; PoCG: postcentral gyrus; dACC: dorsal anterior cingulate cortex, dorsolateral; SMA: supplementary motor area; MOG: middle occipital gyrus; OCD: obsessive–compulsive disorder; MNI: Montreal Neurological Institute.
rmm = 5 mm; voxel number >85, p < 0.05, corrected with AlphaSim.
The LCModel postprocessing software (version 6.3-0D; S.W. Provencher) was used to quantify the relative metabolite concentrations. We only included the quantification results with estimation errors (% standard deviation (SD)) below 20% for further correlation analysis. The metabolite ratios we considered Glx relative to tCr in this study.
Statistical analysis
Demographic measures of age, gender, educational status, age of onset and clinical scores such as YBOCS and duration of illness were compared between HCs and OCD patients. We used SPSS software (version 18.0; SPSS Inc., Chicago, IL, USA) to conduct two-tailed independent samples t-test. All the results were displayed with mean ± SD and the statistical threshold was set at p < 0.05 (see Table 1).
Seed-based method was applied to explore the FC between seed region and whole-brain voxels. Three spheres were defined centered within the corresponding ROIs in MRS, i.e., left thalamus: –15, –17, 9; right thalamus: 11, –17, 10 and the mPFC: 1, 49, 1 (as defined in MNI coordinates), r = 10 mm. The FC maps were constructed for all participants by calculating the correlation of mean time series in each ROI and time series of each brain voxel using Pearson’s correlation after Kolmogorov–Smirnov test. The FC map was subsequently transformed to Z-scores maps for between-group comparison. Two-sample t-tests were conducted to investigate the between-group difference using REST software. Statistically significant difference was considered at pcorrected < 0.05 and a minimum cluster size of 85 voxels, with multiple comparison correction performed using AlphaSim program (single-voxel p uncorrected = 0.05, FWHM = 5 mm, number of iterations = 1000, and within a default whole-brain mask). The results were displayed by BrainNet View software (Version 1.53; National Key Laboratory of Cognitive Neuroscience and Learning, Beijing Normal University). In addition, Pearson’s correlation analysis was performed to examine the relationship between metabolic level in different ROIs and FC in both HC and patient group. Spearman’s rank correlation analysis was conducted to explore the relationship between altered FC/metabolite concentration and OCD patients’ clinical symptom severity.
Results
Comparison of FC between OCD and HC groups
First, we compared the difference in FC between OCD and HC groups. When setting left thalamus as seed region, we found that OCD patients had increased FC values to right middle frontal gyrus (MFG), left precentral gyrus (PreCG), left dorsal anterior cingulate cortex (dACC) and left SMA, with decreased FC values to left brainstem, right sub-lobar and right middle occipital gyrus (MOG) compared with HC group (see Table 2 and Figure 1(A)). For right thalamus, we found that OCD patients showed increased FC values to right middle temporal gyrus (MTG), right SMA and left postcentral gyrus (PoCG), while decreased FC values to orbital part of superior frontal gyrus (ORBsup), right MOG and right dACC (see Table 3 and Figure 2). For mPFC, decreased FCs to left cerebellum and left SMA were shown and increased FCs were found to right median cingulate and paracingulate gyri (DCG; see Table 4 and Figure 3).
Functional connectivity difference between OCD patients and healthy controls, right thalamus as seed region.
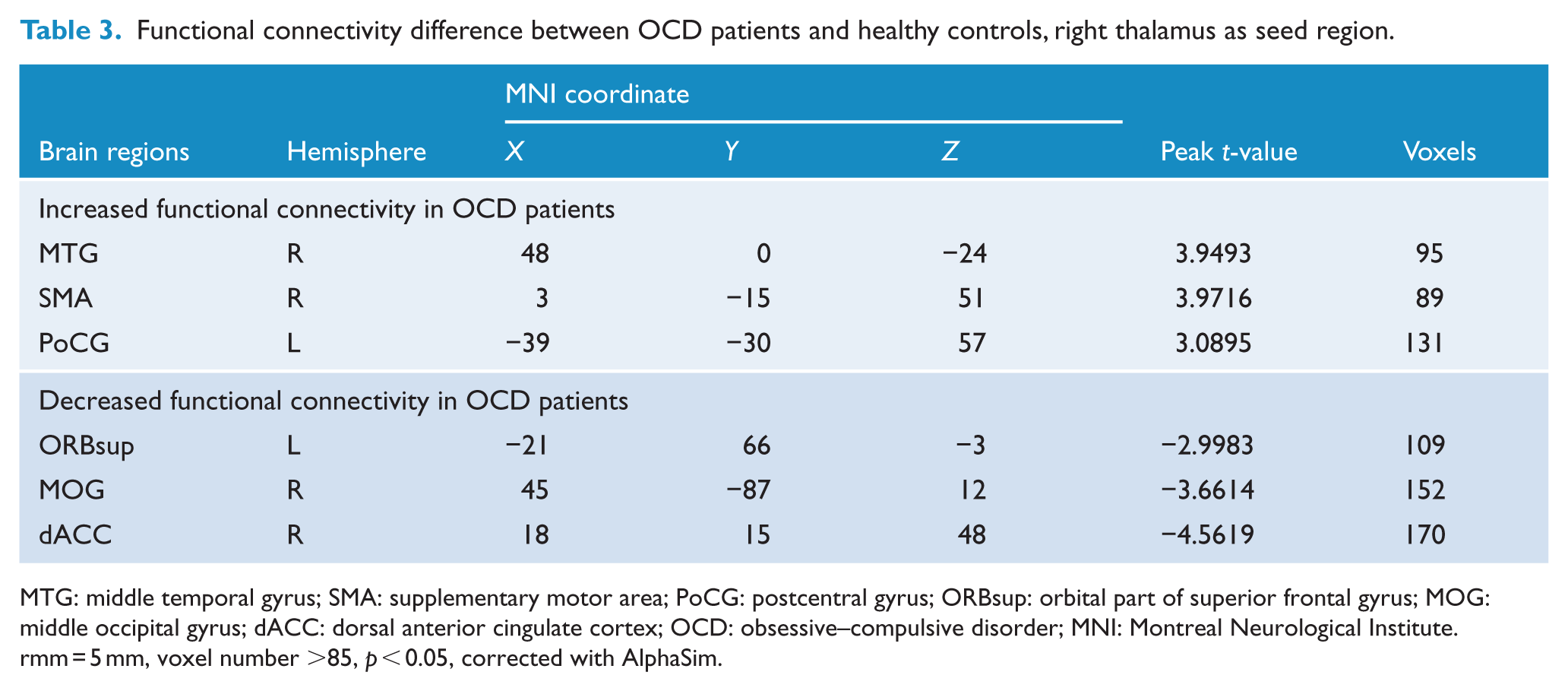
MTG: middle temporal gyrus; SMA: supplementary motor area; PoCG: postcentral gyrus; ORBsup: orbital part of superior frontal gyrus; MOG: middle occipital gyrus; dACC: dorsal anterior cingulate cortex; OCD: obsessive–compulsive disorder; MNI: Montreal Neurological Institute.
rmm = 5 mm, voxel number >85, p < 0.05, corrected with AlphaSim.
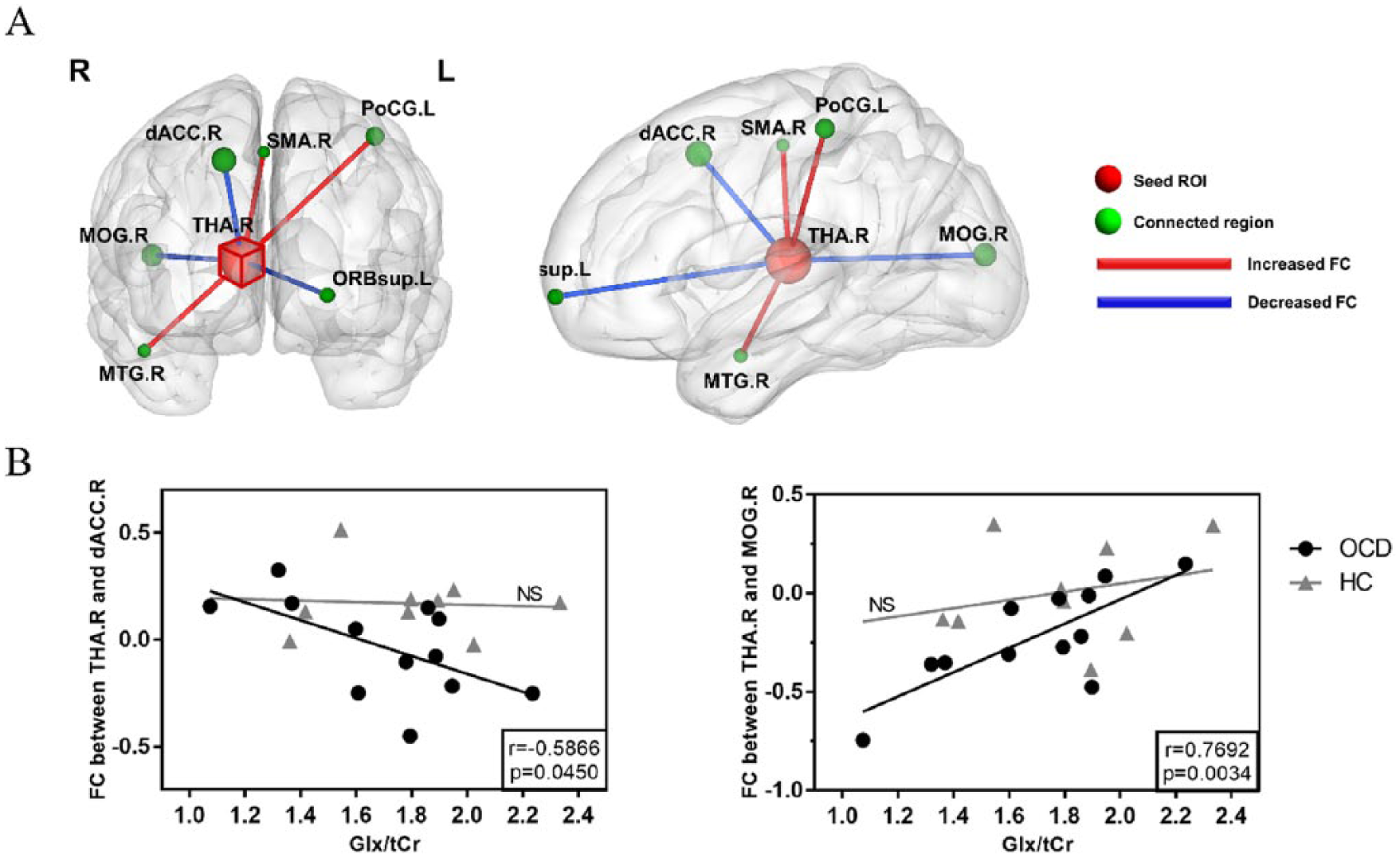
(A) Using the right thalamus as seed ROI, the functional connectivity (FC) difference between obsessive–compulsive disorder (OCD) patients and healthy controls (HCs). Red edges denote increased FC and blue edges denote decreased FC in OCD patients compared with healthy controls (p < 0.05, corrected with AlphaSim). The detailed statistics are listed in Table 3. The radius of each sphere is proportional to the corresponding cluster size. Each sphere center locates at the peak t-value position within the associated cluster. (B) Correlation between relative concentration of Glx in right thalamus and FC between right thalamus and right dorsal anterior cingulate cortex (left), and correlation between relative concentration of Glx in right thalamus and FC between right thalamus and right middle occipital gyrus (right), for both OCD and HC groups.
Functional connectivity difference between OCD patients and healthy controls, medial prefrontal cortex as seed region.
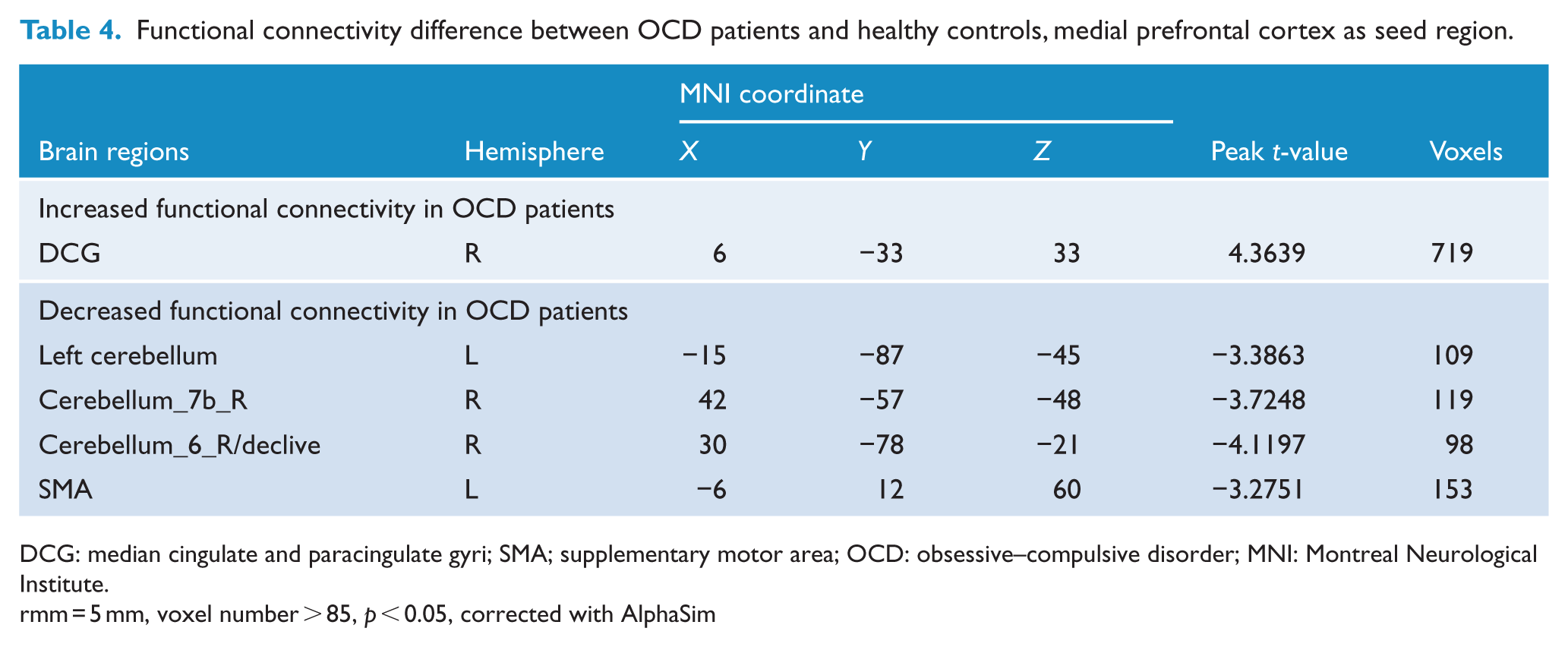
DCG: median cingulate and paracingulate gyri; SMA; supplementary motor area; OCD: obsessive–compulsive disorder; MNI: Montreal Neurological Institute.
rmm = 5 mm, voxel number > 85, p < 0.05, corrected with AlphaSim
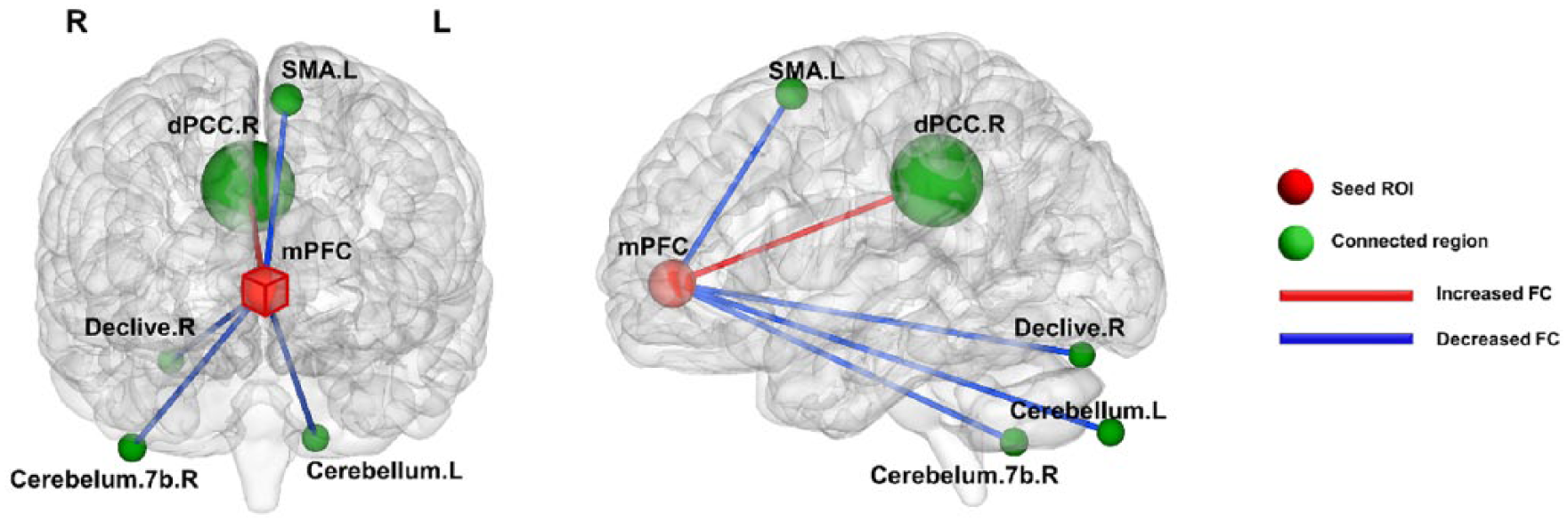
Using the medial prefrontal cortex as seed ROI, the functional connectivity (FC) difference between obsessive–compulsive disorder (OCD) patients and healthy controls (HCs). Red edges denote increased FC and blue edges denote decreased FC in OCD patients compared with healthy controls (p < 0.05, corrected with AlphaSim). The detailed statistics are listed in Table 4. The radius of each sphere is proportional to the corresponding cluster size. Each sphere center locates at the peak t-value position within the associated cluster.
Correlation analysis between regional metabolic level and FC
In OCD patients, the FC between right thalamus and right MOG was shown positively correlated with Glx (r = 0.77, p = 0.003) concentration in right thalamus (see Table 5 and Figure 2). Moreover, the FC value between right thalamus and right dACC was shown negatively correlated with Glx (r = −0.59, p = 0.045) in right thalamus. However, no such functional–biochemical correlation was found in HC group (see Table 5). Only the correlation between right thalamic Glx level and FC between right thalamus and right MOG survive after Bonferroni multiple comparison correction (p < 0.05/6).
The correlation between right thalamic Glx/tCr level and altered functional connectivity from right thalamus in OCD patients and healthy controls.
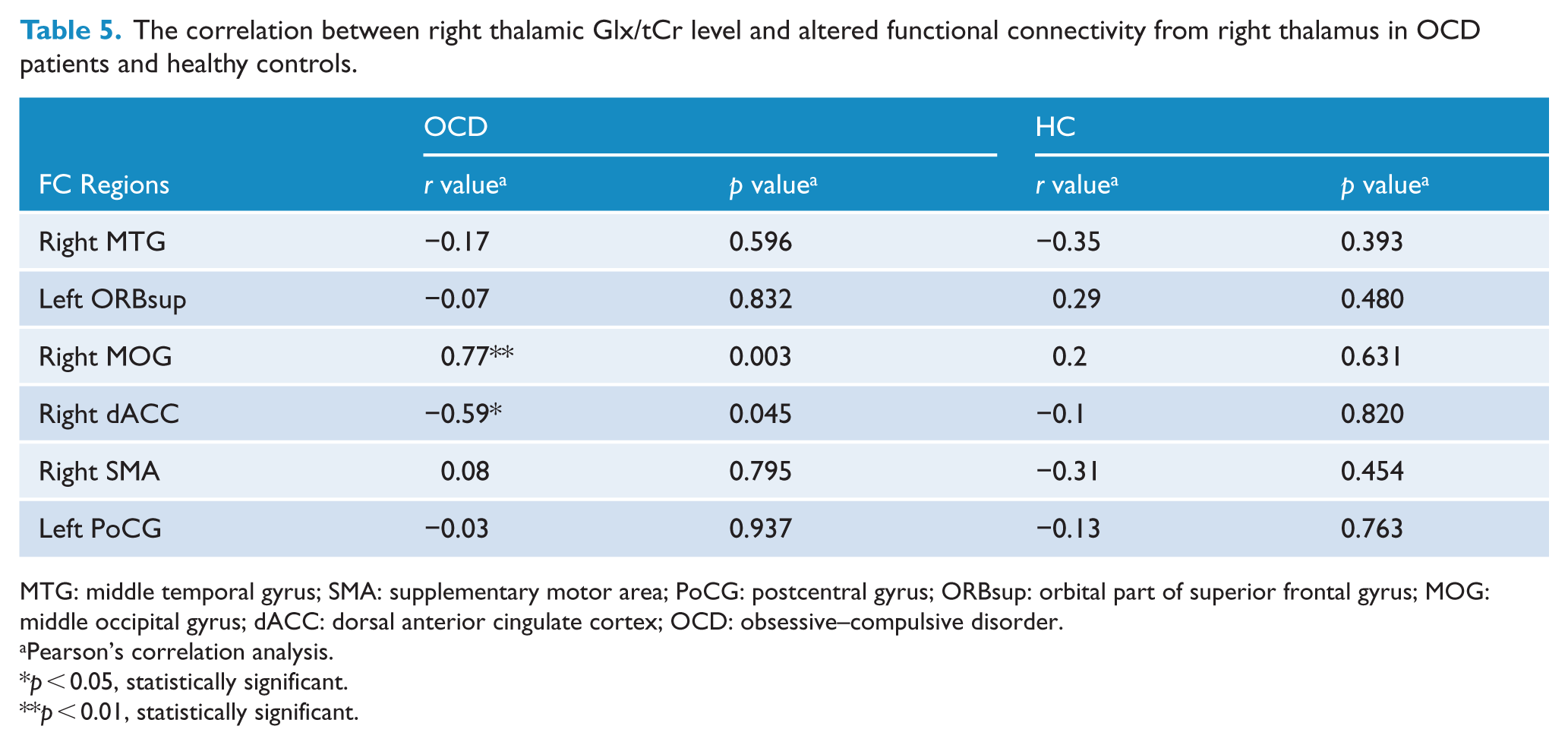
MTG: middle temporal gyrus; SMA: supplementary motor area; PoCG: postcentral gyrus; ORBsup: orbital part of superior frontal gyrus; MOG: middle occipital gyrus; dACC: dorsal anterior cingulate cortex; OCD: obsessive–compulsive disorder.
Pearson’s correlation analysis.
p < 0.05, statistically significant.
p < 0.01, statistically significant.
Correlation between clinical scores and metabolic level/FC
Finally, we investigated the clinical relevance of both regional metabolic level and altered FC in OCD patients. The FC value between right thalamus and right MOG was shown negatively correlated with YBOCS-compulsion score (r = −0.60, p = 0.002) and YBOCS-total score (r = −0.50, p = 0.01; see Figure 4(a) and (b)). The Glx level in right thalamus was negatively correlated with YBOCS-compulsion score (r = −0.65, p = 0.02) in OCD patients (see Figure 5).
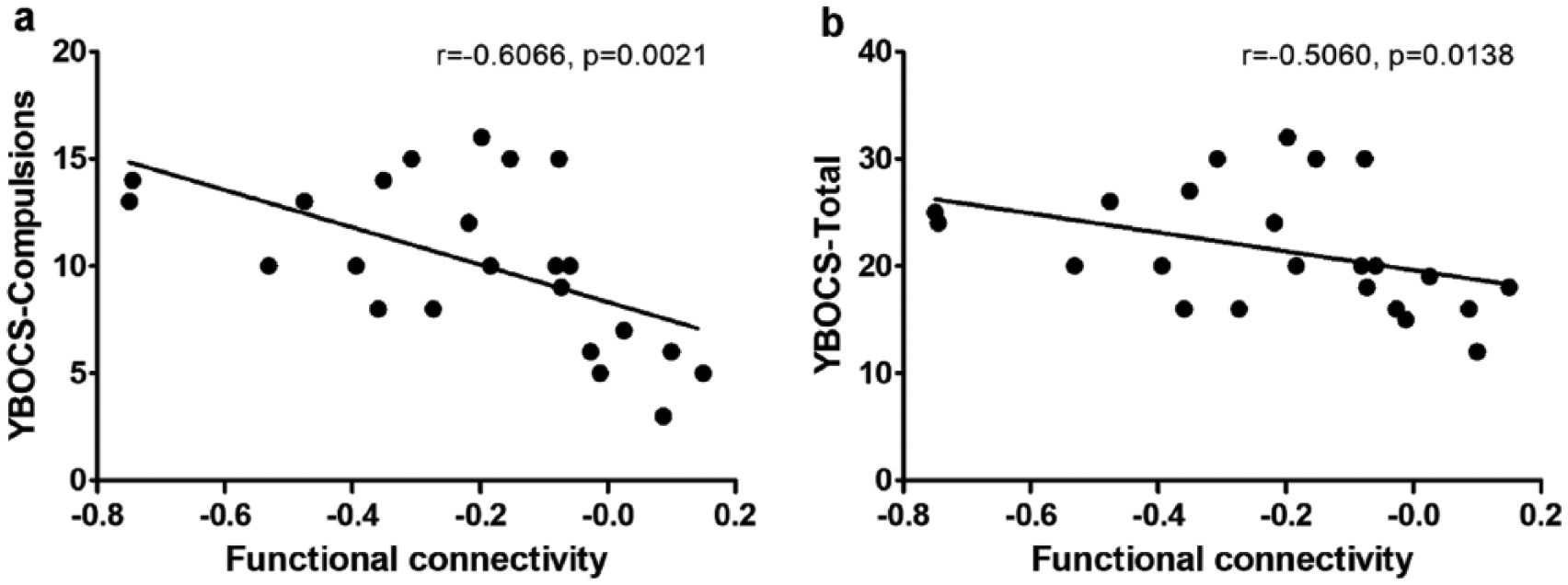
Scatter plots of Spearman’s correlation between the functional connectivity of right thalamus and middle occipital gyrus and (a) YBOCS-compulsions score and (b) YBOCS-total score for OCD patients.
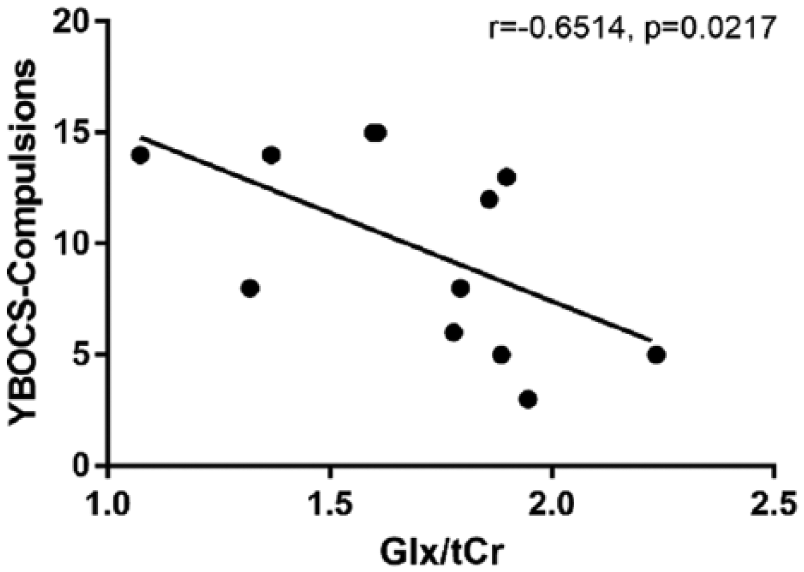
Scatter plot of Spearman’s correlation between YBOCS-compulsions score and the relative concentration of Glx in right thalamus for OCD patients.
Discussion
In this study, we used a combined fMRI-MRS methodology to investigate the alteration of FC within CSTC circuit in correlation with regional metabolite concentration in OCD. For the first time, we showed significant correlation between decreased FC from right thalamus to right MOG area with right thalamic glutamatergic signal, specifically in OCD patients. Besides, this FC was negatively correlated with YBOCS-compulsion and total scores in OCD. Interestingly, the glutamatergic signal in right thalamus was also negatively correlated with YBOCS-compulsion score. Moreover, altered FCs were shown between bilateral thalamus and dACC as typical connections within CSTC circuit.
We found that OCD patients had decreased/increased FC value between right/left thalamus and dACC compared to the HCs. A recent study also discovered reduced connectivity of right medial thalamus and dACC in children with OCD (Fitzgerald et al., 2011). The dACC is closely associated with OCD pathophysiology and also is within CSTC circuit (Diwadkar et al., 2015), which is anatomically connected with motor and frontal regions, playing a mediating function in control-related mechanism (Paus, 2001). The mPFC/ACC has affective and cognitive functions primarily contacted with motivation-related behaviors. The dACC is responsible for mediating cognitive process and rACC is thought to mediate affectively (Vogt et al., 1995). Diwadkar et al. (2015) found that OCD patients had significantly increased dACC modulation of thalamic, striatal and cortical targets during n-back working memory task, especially at the lower level of memory load. Thus, our finding reinforces the importance of dACC and thalamus within CSTC circuit in the pathophysiologic mechanism.
For the first time, we found that OCD patients had decreased FC values between bilateral thalamus and right MOG. The FC between right hemispheric thalamus and MOG was negatively correlated with YBOCS-compulsion and YBOCS-total scores in OCD patients. Decreased FC values within bilateral occipital lobe as well as to OFC area were shown in previous resting-state fMRI study using graph theory and seed-based analysis (Hou et al., 2014). Besides MOG, other altered FC values from bilateral thalamus to limbic system areas, e.g., MTG, brainstem and cerebellum, were found in our study. The limbic system is tightly linked to the prefrontal cortex, while the prefrontal limbic system is involved in affective processing, decision-making and regulation of memory (Roxo et al., 2011). Recently, increasing number of neuroimaging studies have shown that the pathogenesis of OCD may be related to functional abnormalities within other widely distributed large-scale systems, including the parietal (Yoo et al., 2008), temporal and occipital lobes (Togao et al., 2010). Moreover, a recent meta-analysis demonstrated that OCD had abnormality in large-scale networks outside CSTC circuit (Menzies et al., 2008). Our findings support the existence of system-level pathology in OCD, particularly in the limbic system.
As an excitatory neurotransmitter, the Glu and Glu-Gln recycling is closely related to brain energy metabolism (Mangia et al., 2009). It is also supported that the glutamatergic neurons had a direct impact on BOLD signal, while Glu could mediate metabolic response to neuronal activity underlying functional imaging signal changes (Duncan et al., 2011). The correlation between regional Glu concentration and BOLD-fMRI signal has been explored through a variety of neuroimaging studies. For example, Björn et al. demonstrated that the perigenual anterior cingulate cortex (pACC) Glu signal was positively correlated with the resting-state activity in pACC using a combined fMRI–MRS approach (Enzi et al., 2012). Falkenberg et al. investigated the correlation between Glu level in dACC and BOLD response evoked by cognitive control task. They found that low-glutamate subjects in the dACC showed an increased BOLD response when the task demands were high, whereas individuals with low-glutamate level showed the opposite pattern of an increased BOLD response when the task demands were low (Falkenberg et al., 2012). These results can be interpreted as an upregulation of neuronal activity mediated by glutamate (Enzi et al., 2012).
So far, there are two OCD studies utilizing both fMRI and MRS to analyze the CSTC metabolic and functional abnormality hypothesis. Yücel et al. found decreasing neuronal NAA concentration in dACC that was negatively correlated with the local BOLD signal activation in OCD patients (Yücel et al., 2007), which was proposed to link with cognitive impairment. A recent study showed rACC dysfunction during an emotional counting Stroop task, but did not find association between glutamatergic signal and rACC deactivation in the OCD group (Brennan et al., 2015).
In our previous study, we found a decreased glutamate level in right thalamus and mPFC of unmedicated adult OCD patients (Zhu et al., 2015). This finding supported the abnormal glutamatergic signaling hypothesis in OCD, specifically in the pathway from the thalamus to cortex. The excessive activity of direct pathway is dominant over the indirect pathway, in which the direct pathway projects an excitatory glutamatergic thalamic output to cortex and the indirect pathway plays an inhibitory role (Pauls et al., 2014). In this work, we further showed that the FC between right thalamus and right dACC was negatively correlated with right thalamic glutamatergic signal in OCD patients. This bimodal result reinforced the abnormal glutamatergic signaling hypothesis in OCD, specifically in the pathway from the thalamus to cortex (Pauls et al., 2014). The reduced glutamatergic signal in psychiatric disorder was reported to associate with impairment in cognitive control (Zhang et al., 2016). The dACC region played an important role in regulating the engagement of cognitive control process (Kouneiher et al., 2009) and handling cognitive conflict (Botvinick et al., 2001). It has strong connections to the frontoparietal and frontostriatal executive and cognitive control networks (Cools and D’Esposito, 2011), which constitute the CSTC circuit. Our results proposed that the impairment in cognitive control network in OCD (Javitt, 2004) might be mediated by glutamatergic signaling deficit (Enzi et al., 2012).
One more striking finding is that the FC between right thalamus and right MOG was positively correlated with Glx in right thalamus. More interestingly, the FC between right thalamus and right MOG was correlated with YBOCS-compulsion and YBOCS-total score, while the Glx level in right thalamus was negatively correlated with YBOCS-compulsion score. This finding is in line with the increasing evidence that showed the brain abnormalities of OCD within other widely distributed large-scale systems, such as the occipital lobe (Cohen et al., 1996). It is engaged in affective processing, decision-making and regulation of memories (Roxo et al., 2011). The occipital Glx was found negatively correlated with obsession in treatment-naive OCD patients (Ljungberg et al., 2017). The affective dysfunction in OCD was demonstrated in task-based fMRI study. Our results showed that the disrupted connection between thalamic and occipital area was closely associated with thalamic glutamate concentration, which might be linked to the affective dysfunction in OCD (Enzi et al., 2012). MOG was linked to the affective dysfunction[23] and dACC might be responsible for impairment in cognitive control network of OCD (Bush et al., 2000; Vogt et al., 1995). Falkenberg et al. showed that the glutamate level in dACC correlated with the fMRI activation evoked by a cognitive control task, which was interpreted as an upregulation of resting-state activity mediated by glutamate (Falkenberg et al., 2012). The glutamatergic signaling deficit has also been shown to mediate the affective dysfunction (Enzi et al., 2012). Our results indicated that the affective dysfunction and cognitive control impairment in OCD might be mediated by glutamatergic level, interestingly in an opposite correlation pattern.
Finally, several other interesting FC alterations were shown in this study, i.e., decreased FC between mPFC and left cerebellum, increased FC between right thalamus and right premotor cortex and right supplementary motor cortex. We found increased FC between left thalamus and left dACC, but decreased FC of right thalamus to right dACC. There have been studies demonstrating a lateralized dysfunction to right hemisphere in brain activity of OCD patients (Zhu et al., 2015). We speculate that increased FC in left hemisphere might serve as a compensatory role for dysfunction in right hemisphere of OCD patients. The detailed mechanism merits further investigation. Also, we found decreased FC values between right thalamus and left OFC in this study. The OFC is closely linked to limbic system and responsible for the integration of emotional and limbic information into behavioral responses (Bush et al., 2000). The deceased FC between thalamus and OFC may be related with functional alteration between CSTC circuit and limbic system in OCD patients. Increasing attention has been paid to the role of cerebellum in OCD, which was related with higher cognitive function’s task (Allen et al., 1997). There is a study showing increased FC between the bilateral cerebellum and medial prefrontal cortex in OCD patients (Zhang et al., 2011). The activation of right cerebellum through OCD pre-treatment was predictive to the improvement in patients’ YBOCS score during symptom provocation (Sanematsu et al., 2010). The motor and premotor cortex are hyperexcitable in OCD patients. The supplementary motor cortex serves as a key area for inhibitory stimulation in the treatment of OCD, for it extensively connects with brain regions for cognitive process and motor control (Mantovani et al., 2006). As primary motor cortex (M1), thalamus and cerebellum were parts of the motor system (Carper et al., 2015), our findings showed the motor system dysfunction was involved in OCD pathophysiology, which merits further investigation.
There are several limitations of this study. First, the number of subject was relatively small, especially in MRS study, which made the detection of weak physiological biomarker changes difficult. Second, we did not record the corresponding information, that is, smokers/nonsmokers (Wheelock et al., 2014), menstrual phase/hormone use (Batra et al., 2008), number of psychopharmacologic treatments in lifetime and duration of lifetime treatment (Dell’Osso et al., 2015) during data acquisition in this study. Smoking was associated with elevated Glx level in ACC region (Wheelock et al., 2014), while gender differences in Glu between men and women and across the menstrual cycle were reported (Batra et al., 2008). Duration of lifetime treatment resulted significantly higher in the aggressive/checking subgroup, compared to other subgroups (Dell’Osso et al., 2015). These factors should be controlled for future studies. Third, eye condition (‘eyes closed’ vs ‘eyes open’ or ‘cross fixation’) may also influence resting-state fMRI reliability and consistency (Patriat et al., 2013; Yan et al., 2009), so condition of ‘eyes open’ or ‘cross fixation’ should be taken into account in our future studies. In addition, our methods could not disentangle Gln successfully because of technical difficulty. Finally, we utilized metabolite ratio to Cr level rather than the absolute concentration. Generally, Cr is regarded as a stable metabolite biomarker and serves as an internal reference in many MRS studies. But future studies should be performed using absolute measures to consolidate our findings.
Conclusion
Besides abnormal FC within CSTC circuit, we found altered FC from CSTC circuit to occipital lobe and limbic and motor systems. The importance of limbic and motor systems was shown in our study, which provides new insights in understanding the OCD mechanism. The right thalamus glutamate was correlated with FC to MOG but anti-correlated with FC to dACC, which might be linked to affective dysfunction and impairment in cognitive control network mediated by glutamatergic signaling in OCD. Our findings showed that the coupled intrinsic functional–biochemical alterations existed both within CSTC circuit and from CSTC to occipital lobe in OCD pathophysiology. The multimodal approach integrating MRS and fMRI provides a new perspective to encourage future study with larger sample size to better understand the etiology of OCD.
Footnotes
Acknowledgements
Prof. Tianzi Jiang and Dr. Bing Liu from Institute of Automation, Chinese Academy of Sciences provided the MRI and fMRI parameters.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China (Grant No. 81771460, 81871083), Clinical medicine project of Shanghai Science and Technology Committee Project-No.18411952000, 16411965100), Scientific Project from Shanghai Municipal Health Bureau-No.201740086-and Innovation Research Plan supported by Shanghai Municipal Education Commission (ZXGF082101).
