Abstract
Objective
To investigate the central mechanism of cognitive training in patients with stroke, using resting state (RS) functional magnetic resonance imaging (fMRI).
Methods
Patients with stroke and executive function and memory deficit were randomized to receive computer-assisted cognitive training (treatment group; total 60 h training over 10 weeks) or no training (control group). All participants received neuropsychological assessment and RS fMRI at baseline and 10 weeks.
Results
Patients in the treatment group (
Conclusions
Increased RS FC of the hippocampus with the frontal and parietal lobes may be an important mechanism of cognitive recovery after stroke.
Keywords
Introduction
Cognitive impairment (such as memory disorder, executive dysfunction, and attention or orientation disorder) is present in ∼61% of patients at 1 week after acute ischaemic stroke. 1 Of these patients, 30% continue to exhibit a degree of cognitive impairment for a further 3–15 months. 1 Executive dysfunction and memory disorder are the most common clinical manifestations of stroke,2,3 and are key factors in preventing patients from achieving a complete recovery. Cognitive training has been shown to be beneficial for rehabilitation of patients with cognitive impairment,4,5 but the underlying mechanisms remain unclear.
Functional magnetic resonance imaging (fMRI) is widely used in cognitive neuroscience research.6,7 Motor cortex fMRI signals have been found to be synchronously correlated with brain regions related to motor function, indicating the presence of functional connectivity (FC) in these regions. 8 Analysis of FC in different regions of the brain, before and after treatment, allows study of pathology-related changes in cortical networks;9,10 it also indicates correlations between brain function modifications and clinical changes. 11 It is possible that improvements in cognitive function brought about by cognitive training would result in clinical changes in cognition-related brain regions of patients. The aim of the present study was to assess resting state (RS) FC before and after cognitive training in patients with stroke, using RS fMRI and neuropsychological assessment, in order to explore the central mechanism of action of cognitive training.
Patients and methods
Study population
The study recruited patients with stroke attending the Department of Rehabilitation Medicine, Rehabilitation Hospital, Fujian University of Traditional Chinese Medicine, Fujian, China, between July 2011 and November 2012. Inclusion criteria were: (i) confirmed diagnosis of first stroke; (ii) deficits in both executive function and memory (defined as
The study was approved by the Medical Ethics Committee of Fujian University of Traditional Chinese Medicine, Fuzhou, China, and all participants provided written informed consent prior to inclusion in the study.
Experimental design
Patients were randomized between the treatment and control groups using a random number table. All patients underwent neuropsychological assessment and fMRI at baseline and at 10 weeks. Patients in the treatment group underwent computer-assisted executive function and memory training using the RehaCom software package (available at http://www.knowledge.scot.nhs.uk/media/CLT/ResourceUploads/4017079/RehaCom_en_Catalog_SCHUHFRIED.pdf) for six 1-h sessions/week for 10 weeks (total duration 60 h). Cognitive training was performed by two trained psychologists. Patients in the control group did not receive any cognitive training.
Neuropsychological assessment
Neuropsychological assessments were performed by two trained psychologists who were blinded to group assignment. Assessments included WMS 12 and TMT. 13 WMS is designed to assess memory and assesses seven distinct functions as well as providing a total MQ that takes into account age-related mnemonic variability. TMT comprises two parts: TMT-A and TMT-B, and is widely used to assess executive abilities in patients with stroke. 13
fMRI
Patients were placed in the MRI scanner, head movement was restricted with a band across the forehead and earplugs were provided to minimise the effect of noise. Patients were instructed to lie still and keep their eyes closed during scanning. Functional MRI was performed using a 3.0 T Signa MRI scanner (GE Healthcare, Little Chalfont, UK), acquiring 28 contiguous axial slices with a single-shot gradient-recalled echo planar imaging sequence (thickness 5.0 mm; gap 0 mm; repetition time 3000 ms; echo time 45 ms; flip angle 90°; matrix 96 × 96; and field-of-view 240 × 240 mm). A high-resolution T1-weighted structural scan was obtained using a three-dimensional (3D) magnetization-prepared rapid acquisition with gradient echo (3D MPRAGE) sequence.
Preprocessing of fMRI data was performed using Statistical Parametric Mapping, version 8 (SPM8; available at http://www.fil.ion.ucl.ac.uk/spm/). The initial five volumes were discarded to enable the signal to reach a steady state, and the remaining images were corrected for delays in slice acquisition and for any head movement. Patients with head motion >1.5 mm in any direction or with >1.5° of rotation were excluded from the study. The resulting images were spatially normalized into a standard Montreal Neurological Institute model, 14 resampled to 3 × 3 × 3 mm3 voxels and smoothed with a Gaussian kernel of 6 × 6 × 6 mm (full-width half-maximum). The resulting fMRI data were band-pass filtered (0.01 < ƒ < 0.08 Hz) to reduce low-frequency drift, and high-frequency physiological respiratory and cardiac noise.
Functional connectivity analysis
The hippocampus was selected as the region of interest using the Automated Anatomical Labeling template.
15
Patients with hippocampal injury were excluded, in order to ensure data quality. The correlation between reference time course and time series of each voxel in the brain was evaluated with Pearson’s correlation coefficient analysis, and the resulting correlation coefficients were transformed into
Statistical analyses
Neuropsychological assessment data were expressed as mean ± SD and analysed using SPSS® version 17.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Intergroup comparisons of continuous and categorical data were made using paired
Inter- and intragroup comparisons of FC data were made using SPM8, with a
Results
Demographic and clinical characteristics of patients with stroke included in a study to determine the effect of cognitive training on poststroke cognitive function.

Date presented as mean ± SD.
No statistically significant between-group differences (
Neuropsychological test scores of patients with stroke at study entry (baseline) and after 10 weeks of computer-assisted cognitive training (total 60 h; treatment group) or no training (control group).

Data presented as mean ± SD.
Paired
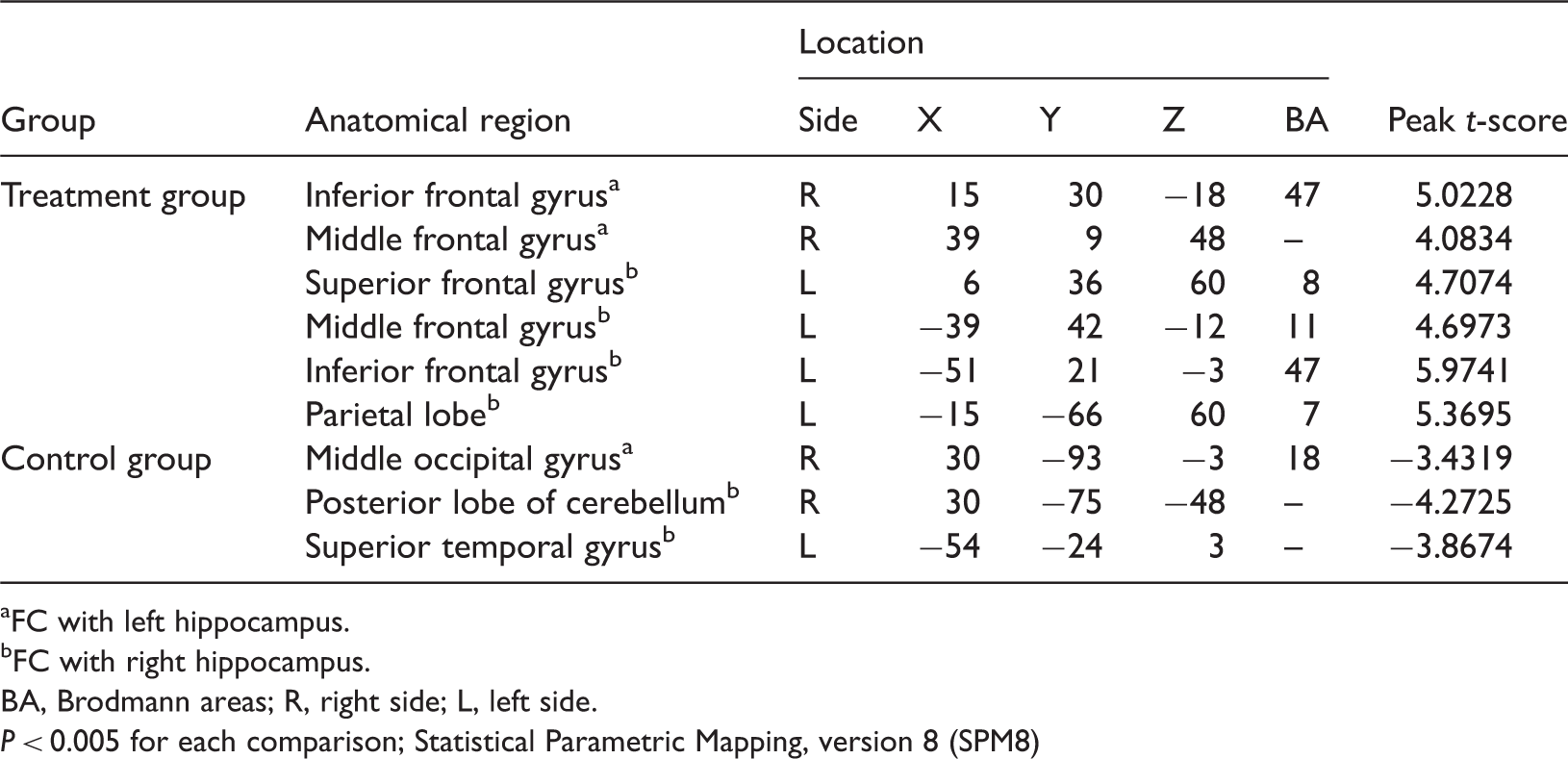
Data regarding changes in FC between baseline and 10 weeks are given in Table 3. There were no significant between-group differences at baseline (data not shown). Cognitive training resulted in significantly increased FC in the left hippocampus–right inferior frontal gyrus, left hippocampus–right middle frontal gyrus, right hippocampus–left middle frontal gyrus, right hippocampus–left inferior frontal gyrus, right hippocampus–left superior frontal gyrus and right hippocampus–left parietal lobe in patients in the treatment group ( Functional magnetic resonance imaging scans showing changes in resting state functional connectivity (FC) of the hippocampus, before and after treatment, in patients with stroke and executive function and memory deficit. Patients were randomized to receive 60 h of computer assisted cognitive training over a 10-week period. Significantly increased FC was observed post-treatment in (a) left hippocampus–right inferior frontal gyrus and left hippocampus–right middle frontal gyrus, and in (b) right hippocampus–left middle frontal gyrus, right hippocampus–left inferior frontal gyrus, right hippocampus–left superior frontal gyrus and right hippocampus–left parietal lobe; changes are denoted with arrows. The colour version of this figure is available at: http://imr.sagepub.com. Functional magnetic resonance imaging scans showing changes in resting state functional connectivity (FC) of the hippocampus in patients with stroke and executive function and memory deficit. Patients were randomized to receive no computer assisted cognitive training over a 10-week period (control group). Significantly decreased FC was observed post-treatment in (a) left hippocampus–right middle occipital gyrus and (b) right hippocampus–right posterior lobe of cerebellum and right hippocampus–left superior temporal gyrus; changes are denoted with arrows. The colour version of this figure is available at: http://imr.sagepub.com. Statistically significant changes from baseline in resting-state functional connectivity (FC) of the hippocampus in patients with stroke after 10 weeks’ computer-assisted cognitive training (total 60 h; treatment group) or no training (control group). FC with left hippocampus. FC with right hippocampus. BA, Brodmann areas; R, right side; L, left side.

In the treatment group, there were significant positive correlations between: (i) MQ and FC of the left hippocampus–right frontal lobe (
Discussion
An understanding of the mechanism underlying the effect of cognitive training on cognitive function in patients with stroke is essential. Cognitive training includes traditional and computer-assisted methods, and has been used in rehabilitation strategies for conditions including schizophrenia and multiple sclerosis.16,17 The use of computer-assisted methods can reduce training time compared with traditional methods, 18 and we therefore adopted this method of cognitive training in the present study.
Neuropsychological scales are regarded as a vital tool in studies on psychological behavioural changes caused by brain injury. 19 The present study investigated changes in WMS and TMT scores in patients with stroke following computer-assisted cognitive training, and found that cognitive training can improve memory (mainly short-term and instantaneous memory) and executive function in these patients.
The present study used RS fMRI to explore the central mechanism of cognitive training. After 10 weeks’ computer-assisted cognitive training, patients in the treatment group showed increased FC of the hippocampus with the frontal lobe (right inferior, right middle, left middle, left inferior and left superior frontal gyrus) and the left parietal lobe, compared with baseline. In addition, improvements in neuropsychological test scores were significantly correlated with FC of the hippocampus with the frontal and parietal lobes. In contrast, patients in the control group showed decreased FC of the left hippocampus–right middle occipital gyrus, right hippocampus–right posterior lobe of cerebellum and right hippocampus–left superior temporal gyrus versus baseline, and no correlations between neuropsychological test scores and FC.
The hippocampus plays an important role in memory, and short-term memory in particular. It is connected with extensive cortical and subcortical structures that form various neural networks associated with the frontal lobe. Memory encoding areas are mainly located in the anterior hippocampus, prefrontal lobe, temporal lobe, parietal lobe and thalamus; memory retrieval areas are mainly located in the prefrontal lobe, posterior hippocampus, thalamus, entorhinal cortex, perirhinal cortex and basal ganglia.20,21 Functional MRI studies have found that the RS hippocampus-associated brain network was insufficient in patients with memory disorder, 22 and that structural abnormalities of the hippocampus are correlated with executive dysfunction. 23 The present study therefore used the hippocampus as the seed brain area for study of cognitive impairment in patients with stroke.
The frontal lobe has a vital role in neurocognitive function, and is related to executive ability and memory. An fMRI study found that the middle and dorsolateral frontal cortex were activated during task switching, demonstrating the involvement of the frontal lobe in executive processes. 24 The frontal cortex is also important in episodic memory, especially in information recall with time. 25 Addressing complex cognitive problems involves not only the frontal lobe itself but also the frontal lobe function network, 26 with connectivity between the frontal lobe and hippocampus likely to sustain executive control functions. 27 The hippocampus–prefrontal lobe circuit is the key neural circuit in working memory:28,29 cognitive training can increase RS FC between the frontal lobe and other areas of the working memory and executive control networks in patients with multiple sclerosis. 11
The parietal cortex is functionally important in the execution of spatial judgement on visually presented material, 30 and the parietal–prefrontal lobe circuit plays a crucial role in manipulating spatial memory information. 21 The dorsolateral prefrontal cortex is thought to be involved in a domain-specific form of information retention, and the parietal lobe is involved in spatial computations.31,32 Decreased parietal–prefrontal lobe FC is an important physiological indicator of Alzheimer's disease. 33 The increased RS FC of the hippocampus with the frontal lobe and parietal lobe in the present study may have a compensatory role in patients who have completed cognitive training.
The present study has some limitations. The preliminary nature of the study and its small sample size meant that it was not possible to analyse changes in RS FC in subgroups of patients (e.g. stratified by sex, age or education level). Because of the small sample size and the difficulty in observing therapeutic effects, we chose a small threshold (10 voxels) in order to obtain images that demonstrated significance. However, this may not have been sufficient for obtaining significant results at a corrected threshold. Consequently, further research is required (using a larger sample size and a larger number of voxels, corrected for cluster size), to provide a more detailed analysis of central mechanisms of cognitive training, together with evaluations involving different patient subgroups.
In conclusion, the present preliminary study indicates that increased RS FC among brain regions may be associated with improved cognitive function. fMRI may help to elucidate the central mechanism of cognitive recovery and monitor the progress of treatment in patients with stroke and cognitive impairment.
Footnotes
Acknowledgements
We wish to thank Dr Li-fang Qiu and Dr Jia Huang, Department of Rehabilitation Medicine, Rehabilitation Hospital, Fujian University of Traditional Chinese Medicine, Fujian, China, for their assistance with cognitive training, and Dr Su-fang Cai and Dr Xie-hua Xue, Department of Rehabilitation Medicine, Rehabilitation Hospital, Fujian University of Traditional Chinese Medicine, Fujian, China, for their assistance with neuropsychological assessments.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was sponsored by the Key International S&T Co-operation Program of Fujian Science and Technology Department (No. 2010I0007) and the ‘Twelfth five-year’ National Technology Support Project (No. 2013BAI10B01).
