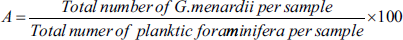Abstract
A significant modulation and adaptive response are observed in Globorotalia menardii from the Bay of Bengal (BoB), reflecting its sensitivity to critical and environmentally stressful conditions. The extreme variations in relative abundance indicate a strong influence of temperature and stratification-driven productivity at the regional scale. The size parameters (maximum diameter (D1), intermediate diameter (D2), and keel thickness) of the species reflect its response to climate and ecological changes over the ~3,4000 years BP. Abundance peaks during warm intervals, such as the Bølling–Allerød (B/A), Holocene, and Medieval Warm Period (MWP), and declines during colder phases, including the Last Glacial Maximum (LGM) and Heinrich events, consistent with the species’ thermophilic nature. The size parameters also increase during warm periods, though their variability suggests stronger control by nutrient availability than by temperature alone. Depth-wise morphological patterns reveal robust, symmetrical, and well-developed tests in the younger intervals, contrasting with irregular and underdeveloped forms in the deeper core samples, reflecting ecological stress. Correlation analysis indicates strong positive interrelationships among the test D1, D2 and keel thickness, signifying proportional morphological development, while abundance shows a moderate negative correlation with core depth. These findings demonstrate that both climatic phases and depositional depth regulate the ecological and morphological expressions of G. menardii, underscoring its reliability as a palaeoceanographic proxy for reconstructing past oceanographic variability in the BoB.
Keywords
INTRODUCTION
Background on Planktic Foraminifera and its importance
Planktic foraminifers are single-celled marine microorganisms that mostly live in the open ocean and usually in the upper water column. They are free-floating and pelagic life forms whose distribution and assemblage variability in surface water and sediments in recent times are strongly influenced by specific temperatures, salinity ranges and surface water masses (Anbuselvan & Senthil Nathan, 2021; Boltovskoy et al., 1996, 2000; Fairbanks et al., 1980, 1982; Thunell, 1978). The interplay of biological, physical, and chemical processes governs the depth, seasonal, and geographical distributions of planktic foraminifera in the ocean waters and sediments (Bé & Hutson, 1977). They are used to infer water mass properties, paleoclimate, and palaeoceanography, and their long palaeontological record, good preservation in surface sediments, and easy collection in turn make them a good indicator in understanding various seawater properties (Anbuselvan & Senthil Nathan, 2021). Variation in the morphological parameters of the species reflects the evolutionary and ocean environment over the period (Besiou et al., 2023). However, its ecological significance is determined through experimental manipulation of its occurrences and population size (DesRoches et al., 2018). So, the specific changes in the morphological characteristics of particular species can provide critical insights into the environmental conditions prevailing at the time and location where the changes occur, aiding in the interpretation of the temporal and spatial ecological variations. The morphological changes and other revolutionary aspects of the species Globorotalia menardii have been extensively studied by researchers (Antonarakou et al., 2004; Besiou et al., 2023; Bhonsale & Saraswat, 2012; Broecker & Pena, 2014; Brown & Kingdom, 2007; Friesenhagen, 2022; Knappertsbusch, 2007; Regenberg et al., 2010; Reynolds et al., 2025; Stainbank et al., 2021) across several oceanic regions at different time frames.
About Globorotalia menardii
G. menardii is a tropical-subtropical, thermocline species that lives preferentially in subsurface waters below 50 m and most likely descends to bathypelagic depths in late ontogeny(Bhonsale & Saraswat, 2012; Tolderlund, 1971; Toledo et al., 2007). It is a non-spinose perforate planktic foraminiferal species, with a pore size of about 1–2 µm. The species is distinguished by its low trochospiral, compressed, and relatively large test with a lobulate equatorial outline. The axial periphery is sharply acute and features a prominent keel. The final whorl consists of five to six wedge-shaped chambers that increase progressively in size. On the spiral side, sutures are strongly curved and raised, while on the umbilical side, they are radial to slightly curved and depressed. The test surface is smooth and densely perforated with circular pores. The umbilicus is broad and shallow, and the aperture is intramarginal (Parker et al., 1865; Reynolds et al., 2025). G. menardii has been used as an important tool for studying palaeoceanographic and paleoclimatic reconstructions. As its abundance is more affected by temperature and salinity compared to primary productivity (Besiou et al., 2023), these species serve as effective markers for understanding changes in seawater properties, such as sea surface temperatures and salinity, thereby providing insight into how the climate may have changed in the past.
STUDY AREA
Bay of Bengal: Oceanographic and climatic importance
The Northern Indian Ocean provides a distinctive setting to investigate the processes governing the distribution of planktic foraminifera in deep-sea sediments. Surface water directly influenced by the monsoonal climate and oceanographic conditions of the area is a habitat for the evolution of planktic foraminifera. The calcareous tests of foraminifera species accumulate at various depositional sites and seafloor depths; hence, the diverse physiography of the North Indian Ocean can be reflected in their tests (Cullen & Prell, 1984). The northeastern Indian Ocean, specifically the Bay of Bengal (BoB), is predominantly composed of fine-grained sediments such as silt and clay, reflecting a strong influence of terrigenous (Cullen, 1981; Cullen & Prell, 1984; Li et al., 2020; Sijinkumar et al., 2021) input from surrounding landmasses (Nagasundaram et al., 2014). BoB is the world’s largest bay (Achyuthan et al., 2014), opening southward into the Indian Ocean. Seasonal insolation changes over the Indian subcontinent, Tibetan Plateau, and the Northern Indian Ocean drive the reversal of the Indian monsoon system (Haridas et al., 2022; Rashid et al., 2011; Webster et al., 1998). This reversal alters wind patterns and precipitation, leading to strong continental runoff and enhanced freshwater discharge from the Indian subcontinent into the BoB, the Andaman Sea and adjoining basins (Dixit & Tandon, 2016; Haridas et al., 2022; Ota et al., 2019). The opposing summer and winter monsoon wind regimes also regulate the seasonal reversal of the East Indian Coastal (EICC) current along the eastern Indian margin (Haridas et al., 2022), transporting freshwater and sediments between the BoB and the equatorial Indian Ocean. Seasonal changes in surface circulation patterns throughout the year affect surface hydrography and reflect variations in temperature and salinity (Figure 1b–1e). In modern-day BoB (Schlitzer, 2022), isosurface temperature ranges from ~28.6°C to 29°C during summer (July–September) and from ~27.8°C to 28.3°C during winter (October–December). Isosurface salinity during the summer season is an average of 31 psu and decreases during winter to between 31 and 32 psu. Monsoon controls the intensity of weathering and erosion in source regions, leading to sediment deposition in adjacent ocean basins. The marine sedimentary records spanning glacial-interglacial timescales are valuable archives for understanding past climatic variations. A previous study in the northeastern Indian Ocean examined the morphological characteristics (diameter) of G. menardii latitudinally across the glacial/interglacial transition. From the present sedimentary core of BoB, we observed distinct temporal changes in the size and abundance of G. menardii during census counts. To develop a deeper understanding of these changes, we are attempting to measure the key morphometry parameters, including test maximum diameter (D1), test intermediate diameter (D2) and keel thickness, of G. menardii across the ~34000 to present. The morphometric parameters of the species were measured during major geological events to understand how oceanographic conditions in the BoB have evolved and influenced the morphology of the thermocline in the G. menardii species over time. The BoB was selected as the study area due to its seasonally significant physico-chemical changes in the ocean. These variations influence the morphology and abundance of planktic foraminiferal species. The study site is situated on the western margin of the Oxygen Minimum Zone (OMZ), off the coast of Visakhapatnam in the BoB.
(continued)
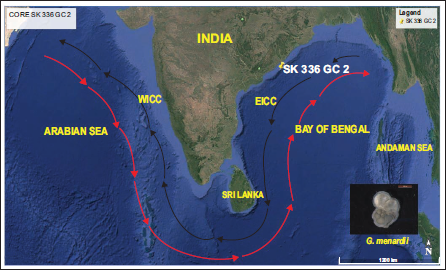
a) Map of the study area depicting the Gravity core SK 336 GC 2 and surface currents. Red arrows show the Southwest Monsoon Current (SMC) and black arrows show the Northeast Monsoon Current (NMC). Base map plotted in Google Earth Pro app (2025). b) and c) Modern BoB map of surface salinity variation during the summer and winter seasons, respectively. d) and e) Modern BoB map of surface temperature variation during the summer and winter seasons, respectively. (data source https://odv.awi.de2023 ).
MATERIALS AND METHODOLOGY
Sample processing
Sediment core SK 336/GC-2 collected from the western BoB (17°37.036ʹN, 83°52.175ʹE), at water depth ~814 m with a total 5.56 m core length. Each sample was processed with essential provisions to avoid contamination, followed by standard procedures (Gupta & Thomas, 1999). Samples were soaked in distilled water with a few drops of 3%–5% hydrogen peroxide (H2O2) for 8–12 hours and washed with a jet of clean tap water through a 63 µm sieve. The washed samples were oven-dried at 50°C and transferred to labelled glass vials.
Species analysis
Quantitative analysis of planktic foraminifera was examined under a stereomicroscope. Aliquots of the samples were taken for the census count, 300 species (>150 µm) of planktic foraminifera from each depth were identified (Nirmal et al., 2021). Data on the relative abundance of G. menardii were derived from a census count of 343 samples.
Morphometry analysis
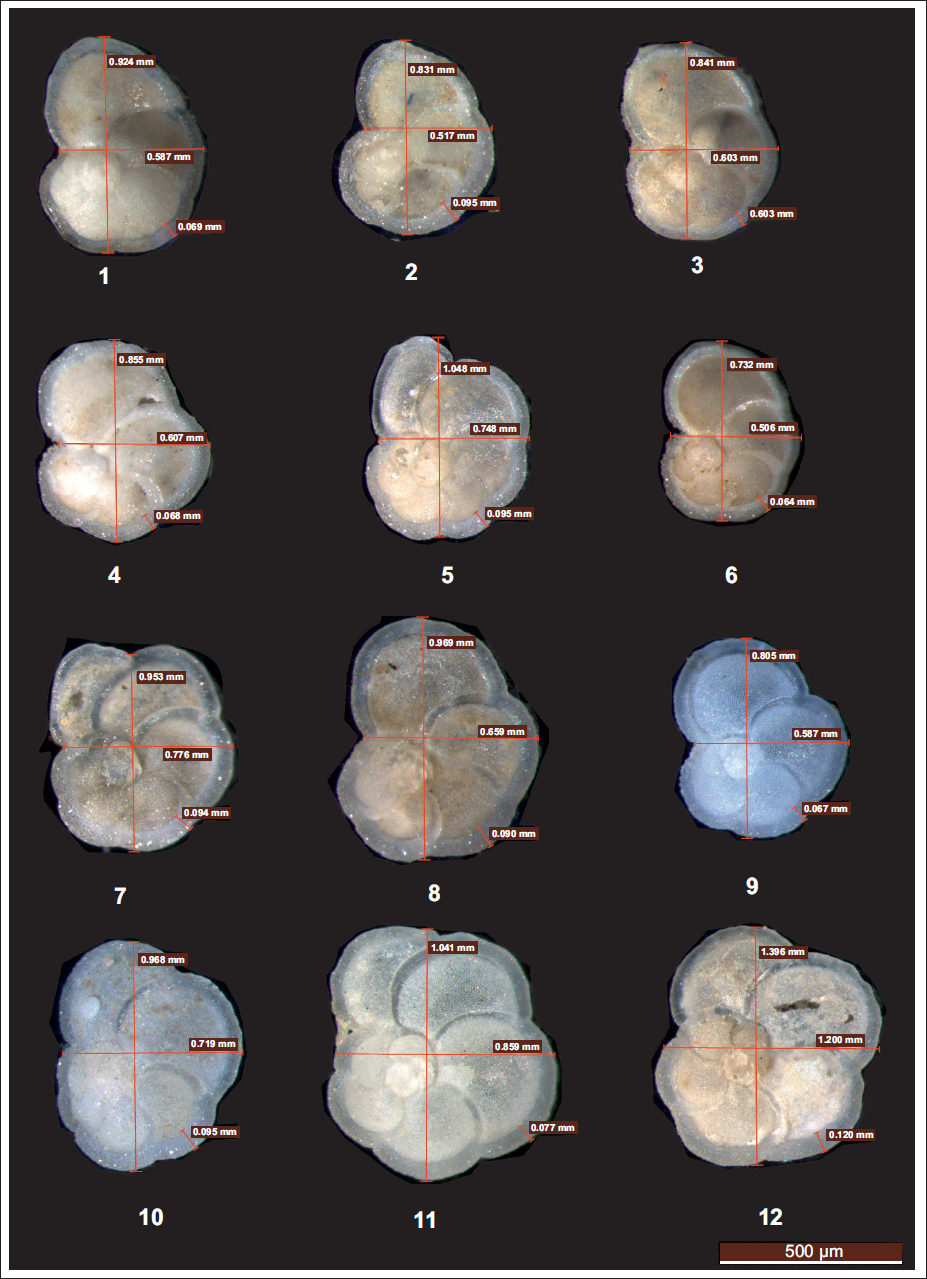
A subset of 30 samples from a total of 343 was selected to assess variations in G. menardii morphometry across intervals corresponding to significant geological events. well-preserved specimens displaying an intact, well-developed keel and undamaged chambers were chosen for measurements. For morphometric analysis, >150 µm fraction representing fully developed adult G. menardii specimens was considered to minimise bias from juvenile forms. For each stratigraphic level, three to five adult G. menardii specimens (mean = 4) were measured; in total, 100 specimens were analysed to ensure reproducibility and consistency of the morphometry dataset. All specimens were digitally imaged in spiral (dorsal) view, where chambers are visible coiling outward from the proloculus. This orientation was selected to measure the maximum diameter (D1) and intermediate diameter (D2) within the spiral plane following (Regenberg et al., 2010). To evaluate keel development, the upper and lower edges of peripheral distance measures (i.e., keel thickness). High-resolution images were captured using the Leica application suite (LAS v4.12) software at a 500 µm scale. All the measurements are converted into millimetres.
Age Model
The age for the core was established from the mixed planktic foraminifera, and the analysis of AMS dating was carried out in IUAC, Delhi, following the procedures from (Kumar et al., 2022). The age from top to bottom of the core was obtained and calibrated with the online OXCAL age model with (∇R)-195 ± 51 (Agnihotri et al., 2002) Marine reservoir values. The core expanding from ~34000 years BP to the Present.
Statistical analysis
To understand the overall distribution of three variables maximum diameter (D1), intermediate diameter (D2), and keel thickness and to evaluate their dimensional significance, the box whisker plot was generated in PAST 4.03 software. Second, to further understand the correlation between species size parameters and age, the Pearson correlation coefficient is calculated in Microsoft Excel. The abundance of G. menardii (A) is calculated by the following equation.
RESULTS
Relative abundance of G. menardii and morphological distribution
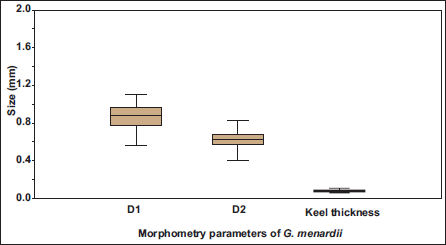
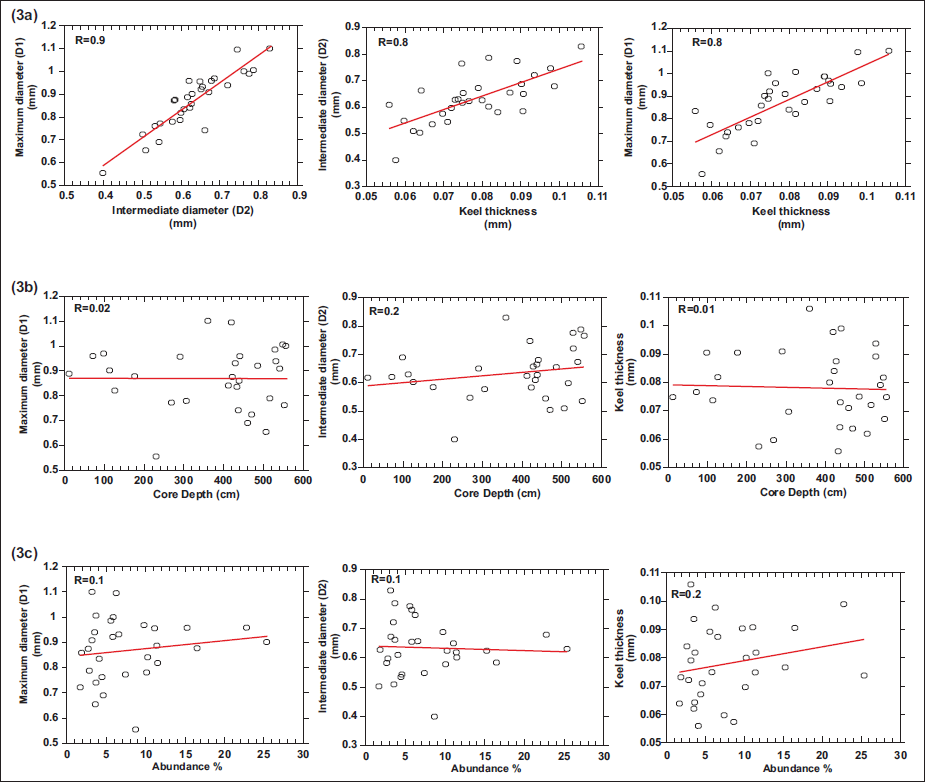
The relative abundance of G. menardii is lower during the last glacial period than in the Holocene. It ranges from 25% to 2%, with an average of 10%. The relative abundance of G. menardii (%) from all samples is also presented for comparative analysis. During the glacial phase, the size parameters exhibit a fluctuating pattern, with a noticeable decline around 25,000 years BP. Throughout this period, species abundance remains consistently low, with only minor variations. A significant increase in abundance occurs after ~15,000 years BP, corresponding to the mid-deglacial phase, followed by a sharp decline around ~12,000 years BP. From approximately 12,000 to 5,000 years BP, abundance remains low and relatively stable, then increases steadily and reaches another peak at approximately 1,000 years BP. The size parameters follow a similar trend, with a distinct rise around 15,000 years BP, followed by a fluctuating decline between ~13,000 and ~4,000 years BP. A sharp drop is observed around 2,000 years BP, marking the lowest point in size, after which they gradually increase again. The box whisker plot analyses three morphological variables: maximum diameter (D1), intermediate diameter (D2), and keel thickness, showing distinct differences in their central tendency and dispersion (Figure 2). The largest magnitude is shown in maximum diameter (D1), showing considerable variability with a median value of ~0.90, an interquartile range (IQR) of ~0.78–0.97, and an overall range of ~0.56–1.10. The intermediate diameter (D2) of the species has a lower median (~0.62) and a narrower IQR (~0.57–0.69), ranging from ~0.43 to ~0.83. Keel thickness shows the smallest absolute values, with a median of ~0.07, a relatively narrow IQR (~0.06–0.08), and an overall range of ~0.05–0.09, indicating the least variability. The lack of extreme outliers and the symmetry of the distributions imply that all three parameters exhibit stable and consistent measurements across the dataset.
Box plot of morphometric parameters of Species G. menardii
Correlation between the size parameters and the abundance of G. menardii with each other and with the core depth.
The Pearson correlation between the morphological parameters and abundance, as well as with core depth, was analysed to assess their relationships (Figure 3). A correlation coefficient (R) of 1 indicates a perfect positive linear relationship, 0 indicates no correlation, and −1 represents a perfect negative correlation. The result reveals a strong positive correlation was observed among the morphological parameters: maximum diameter (D1) and intermediate diameter (D2) (R = 0.9), and keel thickness (R = 0.8), and intermediate diameter (D2) and, keel thickness (R = 0.8), suggesting a consistent and proportional growth pattern of G. menardii. In contrast, the morphological parameters showed weak to negligible correlations with abundance and core depth, with values such as maximum diameter (D1) and abundance (R = 0.1), maximum diameter (D1) and core depth (R = 0.02), intermediate diameter (D2) and core depth (R = 0.2), keel thickness and abundance (R = 0.2), keel thickness and core depth (R = 0.01), and intermediate diameter (D2) and abundance (R= −0.1). A moderate negative correlation was observed between abundance and core depth (R = −0.6), indicating that abundance tends to decrease with increasing core depth. Hence, the abundance and core depth showed negligible influence on test size.
DISCUSSION
The Late Quaternary record of G. menardii from BoB reflects a complex interplay of temperature, nutrient dynamics, and regional hydrography. Rather than viewing its response purely in chronological phases, three overreaching ecological controls emerge: (a) thermal control of abundance, (b) Nutrient Modulation of morphology, and (c) Regional and global Climate Imprints of G. menardii.
Thermal control on abundances
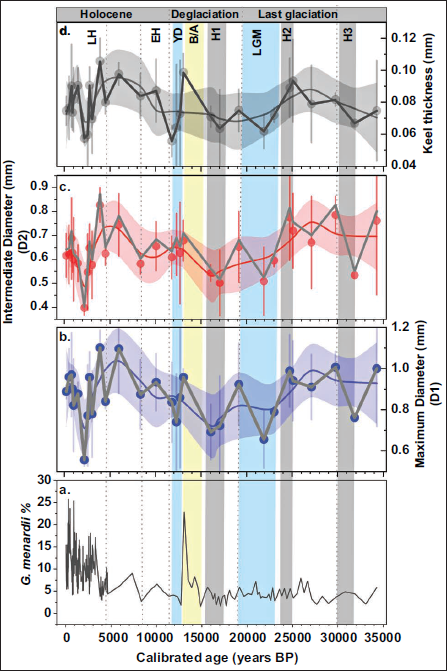
As a thermophilic species, G. menardii abundance is strongly controlled by sea surface temperature. The other studies have well explained the synergistic effects of abiotic and biotic environmental impacts on the shells of planktic foraminifera (Besiou et al., 2023, 2025; Bhonsale & Saraswat, 2012). In contrast, the geographic variations in G. menardii suggest that the physico-chemical environment controls the species. It is recognised that typical thermocline-dwelling species (Bé & Hutson, 1977; Bhadra & Saraswat, 2021; Cullen & Prell, 1984). They are susceptible to vertical changes in regional thermal, salinity, and productivity-driven stratification within the upper water column, which affect their reproduction, abundance, and morphological characteristics. The relative abundance of this species has often correlated with warm water in past (Bhadra & Saraswat, 2021; Govil et al., 2022; Sijinkumar et al., 2021; Verma et al., 2022). This relationship is evident in the current study (Figure 4), where the relative abundance of G. menardii remains consistently low throughout the last glacial period, reflecting the cold climatic conditions of that time. Its population minimums coincide with cold intervals, such as the Last Glacial Maximum (LGM) (~23,000–19,000 years BP) and Heinrich events (H1, H2, and H3), when cooler sea-surface conditions rendered the BoB unsuitable for sustained populations. In contrast, abundance peaked during warm intervals, including the Bølling–Allerød (B/A), the Holocene warm phase. Another prominent peak in abundance occurs during the Medieval Warm Period (MWP) (~1,100–700) years BP till the recent ~200 years BP. This trend is interrupted by the onset of the Little Ice Age (LIA: ~700–500 years BP), reflecting the influence of colder conditions. This consistent relationship underscores G. menardii as a robust proxy for surface hydrographic warmth in the tropical Indian Ocean. Its near absence during the glacial episodes serves as a clear marker of thermal stress, while resurgences during interglacial and warm anomalies highlight its ecological preferences for elevated temperature.
a) Correlation among the morphometric parameters, b) Correlation between morphometric parameters and core depth, c) Correlation between morphometric parameters and relative abundance of G. menardii.
Temporal variation in the morphometric parameters and relative abundance of G. menardii over the past ~34000 years BP. (a-d) show changes in (a) G. menardii relative abundance (%), (b) Maximum diameter (D1), (c) intermediate diameter (D2), and (d) keel thickness plotted against calibrated age (year BP). Major climatic intervals and events, including (Vertical shaded bars indicate geological events. YD: Younger Dryas, B/A: Bølling-Allerød period, LGM: Last Glacial Maximum, H: Heinrich events, EH: Early Holocene, LH: Late Holocene). The dark grey straight line represents the average of each morphometric parameter.
Nutrient Modulation of morphology
Although larger individuals tend to appear when species are abundant (Hecht, 1976; Schmidt et al., 2004), several studies demonstrate that size and abundance are not necessarily correlated (Adebayo et al., 2023; Rillo et al., 2020; Weinkauf et al., 2016). These works explain that the multiple environmental drivers, such as stratification intensity, nutrient supply, temperature, and salinity, regulate the test size in planktic foraminifera. However, the relative importance of these factors varies regionally. For instance, in the Mediterranean, elevated salinity and temperature restrict average test sizes to ~385 µm (Besiou et al., 2024). In contrast, in the nutrient-rich, freshwater of the Gulf of Mexico (Schmidt et al., 2004), enhanced productivity linked to Mississippi River discharge supports much larger individuals (~500–600 µm). In the northeastern Indian Ocean, north-south transect studies indicate that G. menardii abundance and size are more strongly influenced by temperature and salinity in the northern BoB. At the same time, nutrient productivity exerts a comparatively weaker control. In the present core, specimens of G. menardii exhibit clear morphological variability with core depth reflecting both paleoenvironmental changes and the influence of preservation in the western BoB (Plate Figures, 1–12). Greater morphological variability is evident at deeper intervals. Some specimens show thick, well-developed keels and prominent perforation (~5,980 years BP to 21,900 years BP), others exhibit irregular growth, distorted chamber arrangements, or reduced keel development. From ~268 cm (~2,560 years BP), the test morphology shifts noticeably; the specimen appears underdeveloped, with a smaller test size and a barely visible keel, possibly indicating juvenile forms or environmental stress during calcification. Shallower intervals (11–126 cm) tend to have moderately sized tests with smoother surfaces, evenly arranged chambers, and moderately developed keels, features consistent with relatively stable conditions favourable to normal growth. Positive correlation among the size parameters further indicates balanced ontogenetic development. However, the negative correlation between size and abundance contrasts with earlier BoB studies.
Occasional large test sizes (maximum diameter D1, intermediate diameter D2, and keel thickness) during the last glacial, when G. menardii abundance was low, suggest that nutrient supply rather than thermal conditions played a significant role in shaping morphology. As episodic nutrient delivery through intensified winter monsoon winds and weakly stratified surface waters promoted the upward advections of nutrient-rich subsurface waters during the last glacial period (Verma et al., 2022). These short-lived productivity events may have sustained sufficient nutrient levels for surviving individuals to grow larger despite a reduced overall population. Conversely, during the Late Holocene, although the climatic conditions were generally warm (Mayewski et al., 2004; Walker et al., 2019), the test size gradually declined. This likely reflects reduced nutrient flux into the BoB due to a strong monsoon, altered river discharge, and reduced nutrient availability in a stratified upper ocean. Maximum diameter (D1) in our records shows a rapid increase at several intervals (during the last glacial ~29,770, ~24,740, and 19,500; the deglacial ~13,070 years BP; the Holocene periods ~5,980, ~3,900, ~2,760, ~1,000 years BP, and during the MWP). This indicates that D1 does not track temperature directly but instead responds to nutrient availability, which can increase during both cool- and warm-climate intervals. Interestingly, peak test sizes in our datasets (~13,070 years BP) indicate that the transition from the B/A to YD corresponds to enhanced nutritional availability during the deglaciation transition (Thirumalai et al., 2025). The keel appears to balance hydrodynamic and ecological pressures, mirroring trends in D1 but with lower amplitude, indicating adaptive responses of G. menardii as it sought preferred habitats under shifting palaeoceanographic conditions across different water depths (Besiou et al., 2023; Ezard et al., 2011). The intermediate diameter (D2) of species reflects longer-term adaptive adjustments and a decline under nutrient-limited, highly stratified Holocene conditions. At a deepest interval (~25,000–29,770 years BP), specimens commonly show shiny surfaces and visible perforation pores, pointing to better preservation in some intervals. Thus, the overall abundances primarily capture large-scale thermal regimes, whereas morphology provides an additional signal of nutrient dynamics in the upper ocean. Therefore, it implies that the morphology of G. menardii serves as a sensitive proxy for nutrient modulation, complementing abundance trends in reconstructing paleo-productivity variability in the BoB.
Regional Climate Imprints
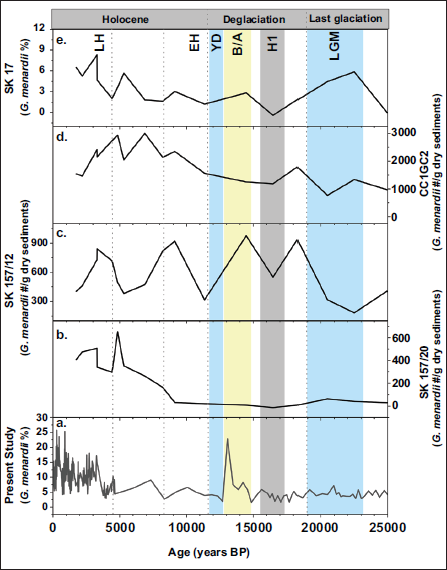
The distribution and morphological evolution of G. menardii show a strong sensitivity to regional and global climate oscillations. This species is widely used as an indicator of past thermocline structure, as changes strongly influence both abundance and test size in upper-ocean stratification. In the Northern Indian Ocean, records from the Arabian Sea (SK 17) (Singh et al., 2018), BoB (SK218/1, CC1GC2, SK157/12), and Andaman Sea (SK168) (Sijinkumar et al., 2011) studies consistently report reduced G. menardii abundance during major cooling episodes such as the LGM and Heinrich events (Figure 5). According to a study on test size evolution in the eastern tropical Atlantic Ocean (Friesenhagen, 2022), relatively cool mixed-layer and warm subsurface layers develop a weak thermocline, leading to reduced chlorophyll and organic matter accumulation and ultimately reducing the growth of G. menardii. Comparable patterns are evident in the BoB core SK 218/1 (Verma et al., 2022), which recorded a weak thermocline between ~44,000 and 19,000 years BP, with extremely weak conditions at the LGM (22,000–19,000 years BP), the YD, and Heinrich events, associated with high salinity and diminished freshwater input. However, periods of moderate to strong thermocline development were observed at 16,000–15,000 years BP, B/A and during the Holocene. Similarly, SK 308/2 (Suokhrie & Saraswat, 2024) revealed variations in the mixed-layer/thermocline (M/T) index, showing a deep thermocline from 24000 to 18000 years BP, maximum depths at ~18,000–14,500 years BP and shoaling between ~14,500 and 10,500 years BP and during the Late Holocene. These regional observations closely match the trends in our study, in which G. menardii abundance increases sharply during warm intervals, such as the B/A and Holocene, and declines during cold phases. The shoaling thermocline coincides with strong Northeast monsoon winds and deepened mixed-layer depths, as well as reduced temperature. During the YD, this resulted in a pronounced decline in G. menardii in the Northern Indian Ocean, including our sites, reflecting hydrographic changes, even though BoB SST records indicate surface warming due to an AMOC slowdown (Panmei et al., 2017). Although the ecology model predicts that abundance and size factors are correlated, this has not been observed in our studies, suggesting that species can selectively adapt in high-stress situations through stabilisation selection. Such anomalies emphasise the importance of considering regional oceanographic settings. Monsoon-driven runoff, freshwater influx and circulation dynamics can significantly modulate habitat conditions, sometimes overriding global climate forcing. G. menardii thus integrates both global scale and basin-specific signals, making it a nuanced recorder of climate variability.
Comparison of the temporal variation in G. menardii relative abundance across adjacent cores. a) Present study (SK 336/GC2), b) 157/20 (Bhonsale and Saraswat, 2012), c) 157/12 (Bhonsale and Saraswat, 2012), d) CC1GC2 (Bhonsale and Saraswat, 2012) and e) SK 17 (Singh et al., 2018).
Globorotalia menardii recorded from the present studied core SK 336 GC 2, Bay of Bengal. Each specimen within a plate is numbered sequentially from 1 to 12, corresponding to its stratigraphic order from top to bottom. The series documents the morphological variations of G. menardii preserved in core SK 336 GC2 from BoB spanning up to 34,000 years BP. The Scale bar represents 500 µm for all the figures. 1. SK 336/2; 10–11 cm; Spiral (dorsal) view 2. SK 336/2; 70–71 cm; Spiral (dorsal) view 3.SK 336/2; 112–113 cm; Spiral (dorsal) view 4. SK 336/2; 266–268 cm; Spiral (dorsal) view 5. SK 336/2; 358–360 cm; Spiral (dorsal) view 6. SK 336/2; 426–428 cm; Spiral (dorsal) view 7. SK 336/2; 438–440 cm; Spiral (dorsal) view 8. SK 336/2; 468–470 cm; Spiral (dorsal) view 9. SK 336/2; 514–516 cm; Spiral (dorsal) view 10. SK 336/2; 528–530 cm; Spiral (dorsal) view 11. SK 336/2; 546–548 cm; Spiral (dorsal) view 12. SK 336/2; 554–556 cm; Spiral (dorsal) view
CONCLUSIONS
The present study on the temporal and depth-wise variation in the size and abundance of G. menardii in the BoB reveals a clear relationship between these parameters and past climatic fluctuations over the last ~34,000 years BP. Abundance peaks during warm intervals, such as the B/A and MWP, while remaining low during colder phases (LGM and Heinrich events), reaffirming the thermophilic nature of the species. Size parameters similarly increase during warm periods but appear to be more strongly influenced by nutrient availability than by temperature alone at the present site. In addition to temporal trends, morphological variation reveals further ecological insights. younger specimens tend to be more robust, symmetrical, and well-developed, suggesting favourable conditions for growth. In contrast, older specimens often exhibit irregular forms, distorted chamber arrangements, and underdeveloped keels, likely reflecting environmental stress, nutrient availability, or variable preservation. Correlation analysis shows that size parameters (D1), (D2), and keel thickness) are positively and strongly interrelated, indicating proportional morphological development. Thus, both climate and local conditions play critical roles in shaping the ecological and morphological trends of G. menardii, confirming its utility as a reliable paleoenvironmental proxy for reconstructing past oceanographic conditions in the BoB.
Footnotes
Acknowledgement
We thank the National Centre for Polar and Ocean Research (NCPOR) for providing the sub-sampled core SK 336/GC-2 of the Bay of Bengal. We thank the 29th ICMS for allowing us to contribute to this special volume. KS is supported by Vellore Institute of Technology, Chennai, for a fellowship.
Authors’ Contribution
KS conceptualised, generated data and wrote the article; MK conceptualised, wrote, reviewed, and PN generated the data.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical/Patient Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement Regarding Plagiarism or Assisted Content from AI (If Taken)
No.