Abstract
The present study reports the occurrence of diatom taxa, their distribution and ecology in Thamirabarani River between the study areas Naranammalpuram and Punnakayal, covering the districts of Tirunelveli and Thoothukudiin Tamil Nadu, India. The study employs diatoms collected from 10 different locations in the Thamirabarani River to monitor its quality. A total of 40 diatom taxa belonging to 20 genera were recorded in the study area. The dominant presence of diatom taxa, such as Aulacoseira ambigua, Cocconeis placentula, Cyclotella meneghiniana, Cymbella tropica, Discostella stelligera, Gyrosigma acuminatum, Navicula cryptonella, Nitzschia amphibia, Planothidium lanceolatum, Staurosirella pinnata, and the water quality index values (29–48) recorded at sites 1 to 7 indicate oligotrophic due to good flow of water. The abundant of diatom taxa, such as Diadesmis confervacea, Gomphonema parvulum, Nitzschia palea and Pinnularia gibba, were recorded at sites 8–10, and the water quality indices (100–110) indicate eutrophication attributable to less flow of water, high dispersion of cremation ashes (the casting ceremony) and anthropogenic activities. Particularly, locations around Punnakayal are most polluted due to the dumping of fish wastes as well as stagnant water.
Introduction
Diatoms are unicellular microalgae that live within all aquatic habitats around the world (Potapova & Charles, 2002). They are a diverse group that plays a significant role in the upper trophic levels for providing energy and stream food webs. They are the most reliable indicators of environment and climate change, which includes acidic precipitation, declining water quality and habitat loss (Hernández et al., 2013; Potapova & Charles, 2005; Rimet, 2012; Smol & Stoermer, 2010; Soininen et al., 2004). The environment in which we live is constantly changing due to the high human population and anthropogenic activity. Several pollutants end up in bodies of standing water, affecting aquatic systems. Local environmental parameters of water, such as pH, nutrients and conductivity, are the most significant determinants of the composition of benthic diatom populations at any given location (Smucker & Vis, 2011; Soininen & Weckstrom, 2009), which also record factors, like historical, climate or distribution dynamics (Verleyen et al., 2009). Rivers and streams are highly dynamic systems, which spatially and temporally vary in water quality (Grubbs et al., 2007). A stream’s quality is usually determined by factors that influence it, which also reflects the biodiversity it supports. The type of biota that it supports serves as a basis for estimating water quality (Camargo & Jiménez, 2007). Diatoms are commonly used to monitor ecosystem structures, function of current environments, and to reconstruct climate and soil conditions. Due to their silica cell walls, many species have specific biological optimum conditions and limited tolerances; as a result, they are usually found in fossil deposits (Jones et al., 2000; Sterken et al., 2012). A few organisms that are ecologically sensitive, including those such as Periphyton, serve as measures of system health and hotspots for biodiversity (Bere & Mangadze, 2014; Larned, 2010; Taylor et al., 2007b). The aim of the present study is to record the distribution of the diatom taxa present in the study area and correlate them with the water quality index (WQI) in the river to assess its environmental condition.
Study Area
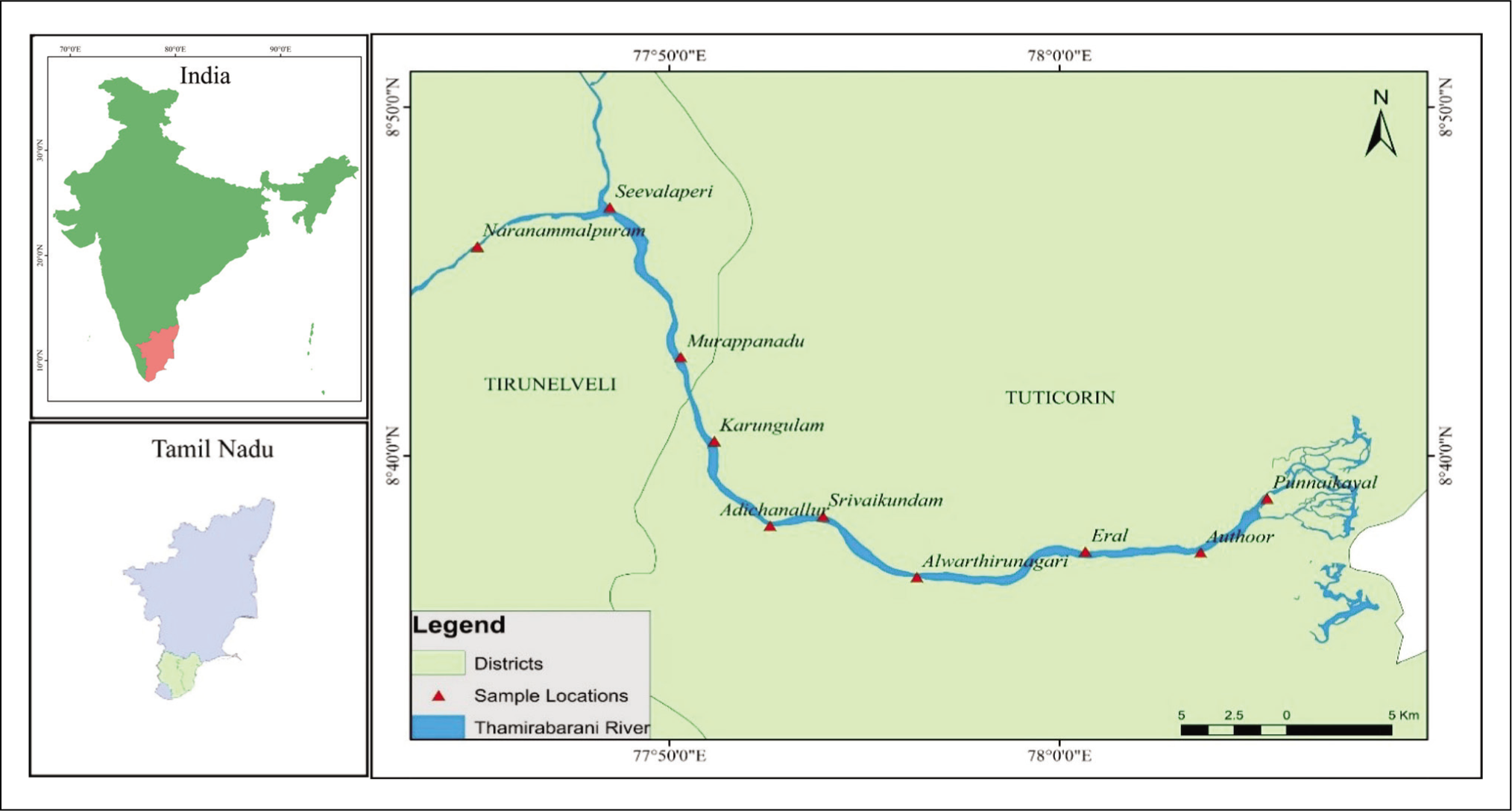
The Thamirabarani River is a perennial river at Tirunelveli, Tamil Nadu, India. It originates at the peak of the Pothigai Hills on the eastern slopes of the Western Ghats and flows eastward for about 128 km, passing through Tirunelveli and Thoothukudi districts before joining the Bay of Bengal in the Punnakayal region (Venkatachalapathy & Madhankumar, 2023). The Thamirabarani River is the main source of drinking water supply, irrigation, domestic usage and industrial processes in Tirunelveli and Thoothukudi Districts of Tamil Nadu (Figure 1).
Study area showing sampling locations.
Methodology
Sample Collection and Processing
Samples of macrophytes were collected for the current study at 10 different locations along a 62 km stretch of the Thamirabarani River between Naranammalpuram and Punnakayal, at a sampling interval of approximately 5–7 km. The diatom analysis protocols of the altered method proposed by Karthick et al. (2013) were used in the present study. Samples were collected in zip-lock plastic bags from all possible habitats, such as plants (epiphytic) and stones (epilithic) and preserved in formaldehyde (4%). Nitric acid (HNO3) and hydrogen peroxide (H2O2) were used to oxidise the diatom samples in the lab for three hours at 90oC to remove all the organic matter. The samples were then centrifuged at 3,000 rpm for 10 minutes, after which the supernatant was pipetted out. The pellet of diatom frustules remaining at the bottom was re-suspended in demineralised water. This process was repeated four times to ensure that all traces of nitric acid were removed (Karthick et al., 2013). The slides were heated on a hot plate until all the toluene had evaporated and only the mounting medium was left behind. The slides were taken off the hotplate once it was bubble-free and allowed to cool and harden at room temperature. To get the best representation of diatoms from each sample, three or five replicate slides were prepared from each sample. Each sample yielded at least 15–17 valves, which were counted and identified under a 40X light microscope.
Diatom Observations and Identification
For permanent slides, the washed material was air-dried on the coverslips and mounted with Naphrax mounting medium. Olympus BX 53 (Tokyo, Japan) microscope with differential interference contrast optics and a 100 × 1.4 oil immersion objective was used for microscopic observations, and photomicrographs were taken using an Olympus DP 73 digital camera and Cell Sense standard 1.16 imaging software. GIMP (version 2.8.14, GNU Image Manipulation Program) as well as Inscape (version 0.91) open-source software was used to create light microscopic plates (plates 1 to 5; scale bars = 10 µm). All recovered diatom taxa were identified using relevant monographs and papers listed under each taxon in the description and bibliography. All descriptions in this section are based on the previous literature (Karthick et al., 2013; Lange-Bertalot et al., 2017; Taylor et al., 2007a; Venkatachalapathy & Karthikeyan, 2014).
Water Quality Index (WQI)
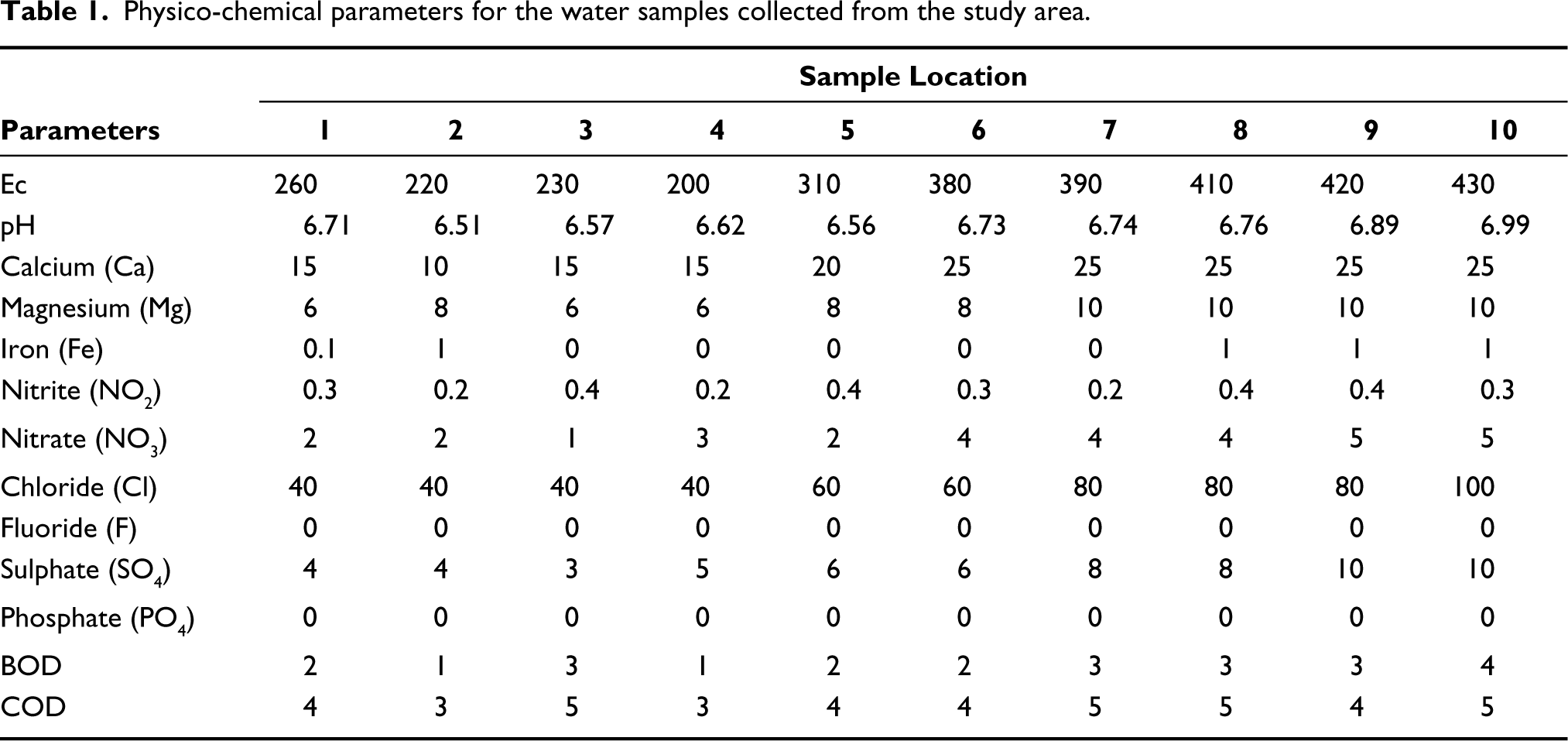
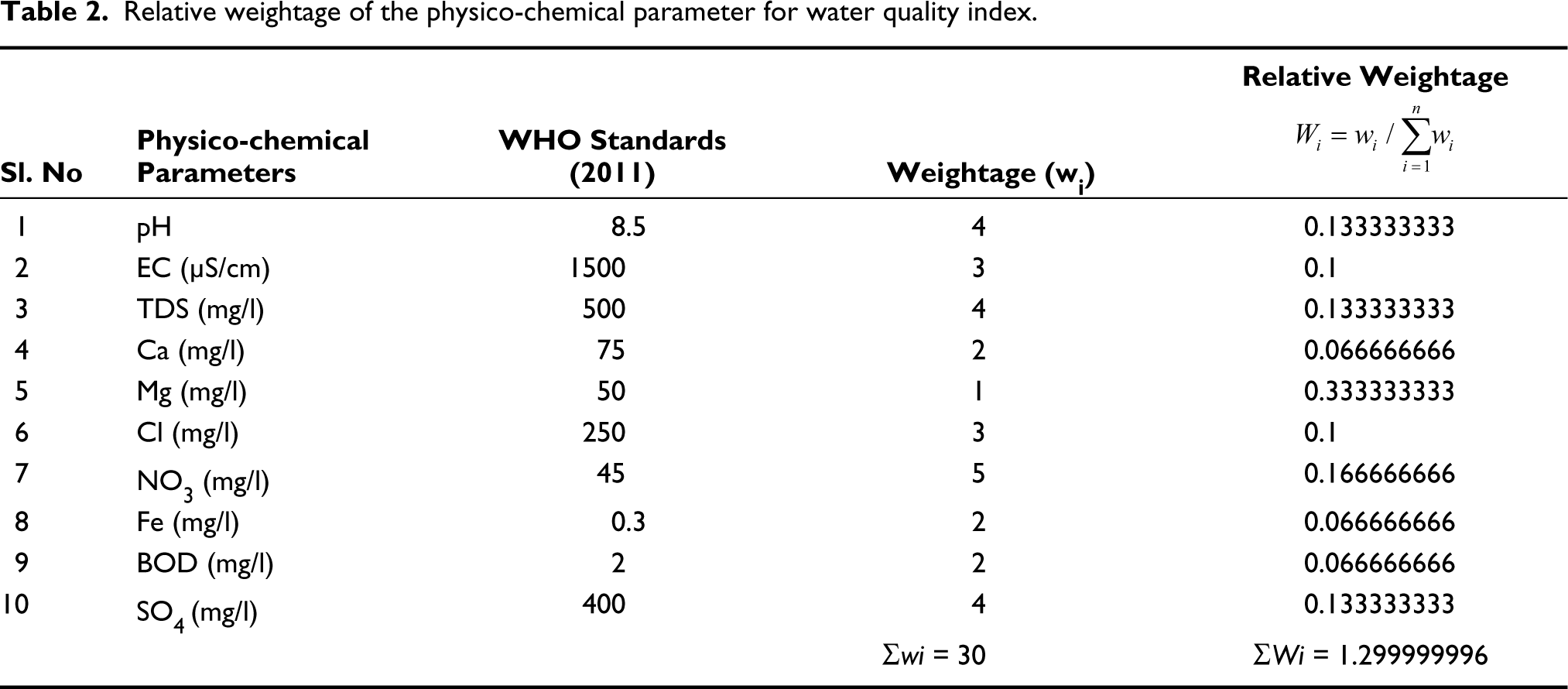
The best method for communicating the type of water resources available is by describing its WQI. In recent years, several global organisations have developed various water quality indices for evaluating water quality in specific cases. Much of the data can be reduced to a single value of WQI, which makes it simple and intelligent to communicate. The WQI was calculated for the samples under study based on the 10 parameters (physico-chemical information) listed in Table 1. This WQI was calculated using the following formula (Vasanthavigar et al., 2010) following the guidelines of WHO (2011), and the results have been denoted in Table 2:
Physico-chemical parameters for the water samples collected from the study area.
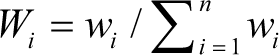
where Wi represents relative weight, wi represents the weight of each parameter and n represents the number of parameters.
In the second step, a quality rating scale qi for each parameter is consigned by dividing its concentration in each water sample and represented in (Table 2)
Relative weightage of the physico-chemical parameter for water quality index.
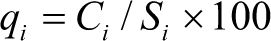
where qi is the quality rating and Ci is the concentration of each chemical parameter in mg/L.
For computing the WQI, sub-index of its parameter (SI) is the first determined for each physico- chemical parameters per the equation:
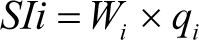
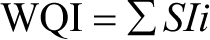
where SIi is the sub-index of its parameter, qi is the rating based on concentration Ith parameter and n is the number of the parameters.
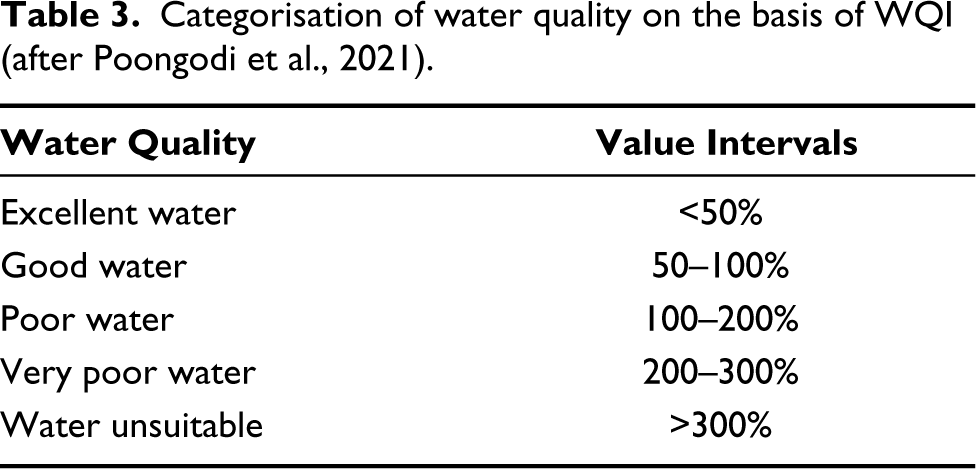
Water quality is categorised using the WQI shown in Table 3. The water quality results of relative weight are shown in Table 4.
Categorisation of water quality on the basis of WQI (after Poongodi et al., 2021).
Calculated water quality index of the sampled locations.

Description of Characteristic Features of the Identified Diatom Taxa
Light microscopy (LM): 1–3 Aulacoseira ambigua (Grunow); 4–5 Aulacoseira granulate (Ehrenberg); 6–8 Bacillaria paxillifera (Gmelin); 9–10 Colonies schumanniana (Grunow); 11–13 Cocconeis placentula (Ehrenberg; 14–16 Cyclotella meneginiana (Kutzing); 17–18, Cymbella tropica (Krammer); 19–21 Discostella stelligera (Houk and Klee); 22–23 Cymbella tumida (Berbisson); 24–26 Diadesmis confervacea (Kutzing); 27–29 Encyonema vulgare (Krammer); 30–32 Fragilaria crotonensis (Kitton); 33–35 Fragilaria rumpens (Kutzing) 36–37 Gomphonema tamilensis; 38–40 Gomphonema lacusrankala (Gandhi); 41–42Gomphonema affine (Kutzing); 43–45 Gomphonema lagenula (Kutzing); 46–48 Gomphonema parvulaum (Kutzing); 49–51 Gomphonema lacusrankaloides (Gandhi); Karthick & Kociolek; 52–54 Gomphonema pseudoaugur Lange-Bertalot, 1979; 55–57 Gyrosigma acuminatum (Kutzing) Rabenhorst, 1853; 58–59 Luticola geoppertiana (Bleish) Mann; 60 Luticola nivalis (Ehrenberg) Mann, 1990; 61–63 Navicula cryptonella (Lange-Bertalot, 1985); 64–66 Navicula erifuga (Lange-Bert, 1985); 67–69 Navicula rostellatta (Kutzing) 1844; 70–72 Nitzschia amphibia (Grown, 1862). Scale bars = 10 µm.

Ref: Faustino et al. (2016), figs. 2–3; Karthick et al. (2013), pl. 3.
Valves are cylindrical and form colonies. The mantle height to valve diameter is a ratio of more than 1. Valves are of length 13–27 and width 4–5. Valves spines are triangular and short. Valves are striae density 14–18/10 µm.
Habitat: Freshwater.
Ref: Karthick et al. (2013), pl. 3.
Frustules in girdle view, cylindrical, lengthy chains, join face-to-face and filamentous colonies. Valves are 5–7 µm in diameter, with a mantle height of 30–58 µm. The mantle has straight sides, and the valve face is flat. The mantle areolae are square. The valve face has small scattered areolae more densely distributed around the valve face margin. Spines are present around the circular margin, and valve faces are uniformly areolate. Species of the genus are mostly in mesotrophic or eutrophic but rarely in oligotrophic water bodies.
Habitat: Freshwater.
Ref: Karthick et al. (2013), pl. 109; Taylor et al. (2007a), pl. 136.
Valves have linear to linear-lanceolate with thin rostrate or capitates apices. Keel is present in the middle or adjacent to the middle, assisted by unequally spaced fibulae. Valves are of length 56–94 µm, width 6–7 µm and striae 20–22/10 µm. Striae are distinct, regular and parallel. Sometimes appeared as curved.
Habitat: Cosmopolitan species.
Ref: Stancheva et al. (2009), p. 165, pl. 18–38, fig. 24; Lange-Bertalot et al. (2017), pl. 70, figs. 5–6, p. 125.
Linear, linear-elliptical to lanceolate (Strongly), but centre almost always inflated; the margins are therefore frequently triangulated, more rarely convex. This species ends for obtusely rounded to wedge-shaped. Valves are of length 29–32 µm and width 8–11 µm. Striae 18–22/10 µm radiate somewhat in the centre, more powerfully radiate or parallel towards the poles, crossed by a longitudinal axis parallel to the valve edges. Central area moderately to very large, round and wider than half the valve width, or transapically—expanded as a fascia reaching more or less to the valve margins. The axial areas narrow to moderately broad, lanceolate, widening from the ends to the centre. This species is rare and usually sporadic; therefore, difficult to characterise ecologically. Most records are from carbonate freshwater low-land habitats with medium to high trophic states.
Habitat: Freshwater.
Ref: Tiffany and Britton (1971), p. 241, pl. 64, fig. 736; Prasad et al. (1992), p. 199, pl. 27, fig. 6.
Valves are relatively flat, elliptic to linear-elliptic and are of length 14–25 µm, width 8–24 µm, RV striae 20–23 µm and RL-valve striae 21–23 µm.
Habitat: Freshwater.
Ref: Prasad et al. (1992), p. 160, pl. 24, figs. 1–2; Karthick et al. (2013), pl. 5; Wojtal (2009), p. 135, pl. 1, figs. 14–15.
Valves have such a narrow mantle and a disc-shaped body. Round, flat and transversely undulating describe the valve face. The centre region is unique and separated from the striae by marginal chambers.
Habitat: Cosmopolitan distribution.
Ref. Krammer (2002), p. 61, pl. 44, figs. 1–10.
Valves are dorsiventral, broadly lanceolate and are of length 40–45 µm, width 10–12 µm. The axial area is linear and narrow. The middle striae end does have a single, elongated stigma. Striae density is 8/10 µm and radiate throughout the valve.
Habitat: Freshwater.
Ref. Tiffany and Britton (1971), p. 278, pl. 74, fig. 860; Karthick et al. (2013), pl. 91. Lange-Bertalot et al. (2017), p. 168.
Ventral margin convex, this species stria is coarse, radiate and unevenly bent. Valves are of length 50–54 µm, width 17–18 µm and striae density 9–10 µm.
Habitat: Freshwater.
Ref. Patrick and Reimer (1966), p. 476, pl. 45, fig. 9.
Valves are elliptical and rounded-protracted apices. The central area is rounded and broad. Valves are of length 17–22 µm, width 7–9 µm and striae 20–22 µm. During the process of valve formation, the raphe may be secondarily filled with silica. The central ending is distinctly period. Striae are variable in length. This species are cosmopolitan range.
Habitat: Polluted water.
Ref. Bishop et al. (2017), pl. 1, figs. 30–32; Solak et al. (2018), p. 109, figs. 75–78.
Valves have a distinct central area and a well-defined marginal ring. Valves are concave or convex.
Habitat: Freshwater.
Ref. Lange-Bertalot et al. (2017), pl. 89, figs. 30–32, p. 807.
Valves are lanceolate and semi-lanceolate. The dorsal and ventral are margins strong, weakly convex and more or less inflated in the centre. Striae uniseriate, lanceolate, slightly radiate all along the valve, central striae more distant. The central area is continuous with the linear axial area. One dorsal is sigmoid. Dimensions: Length 54–60 µm, width 8–14 µm and striae 7–9 µm.
Habitat: Freshwater.
Ref. Taylor et al. (2007a), pl. 16, Karthick et al. (2013), pl. 15.
Valves are linear-lanceolate, cells are centre frustules and attached in middle of the centre.
Ghost striae are present in the centre with parallel striae. The cell in range of length is 60–70 µm, width is 2–4 µm and striae density is 13–15 µm.
Habitat: Freshwater.
Ref. Lange-Bertalot et al. (2017), pl. 10, figs. 15–20, p. 274.
Valves are rounded ends and linear to linear-lanceolate; it has either not or only weakly drawn out. Cells form ribbon-like colonies or tufts sharing a common pad of mucilage. Dimensions: Length 34–65 µm, width 3–5 µm and striae 14–15/10 µm. Oligo-mesotrophic, electrolyte to poor streams and small rivers on the siliceous substrate.
Habitat: Freshwater.
Ref. Karthick et al. (2013), pl. 77; Taylor et al. (2007a), pl. 177.
The apices of the heteropolar, club-shaped valves are rounded bluntly and sharply. Valves are of length 62–72 µm and width 8–10 µm. Axial area is linear, slightly converging at the apices. Shortening of the central striae develops the thin central area. Striae density is 9–10/10 µm.
Habitat: Freshwater.
Ref. Karthick et al. (2013), pl. 83.
The apices of the broad to elliptical-lanceolate valves are enlarged and slightly rounded. Dimensions: Length 60–63 µm, width 14–18 µm and striae density 10–11/10 µm. Centre area that is unequal as the axial area narrows. Striae punctate the ends and radiate either weakly and strongly. At the end of a central stria, there is one stigma.
Habitat: Moderate water.
Ref. Karthick et al. (2013), pl. 84.
Valves have head poles, which are expanded to acute, linear and lanceolate-clavate. The axial region is narrow. Valves are of length 65–86 µm, width 11–15 µm and striae density 10–12/10 µm.
Habitat: Oligotrophic to mesotrophic.
Ref. Karthick et al. (2013), pl. 79; Taylor et al. (2007a), pl. 123.
Valves are uneven, broadly elliptical, thin heteropolar club-shaped, lanceolateoval. Apices, head pole and foot pole are distinctly capitates. The axial area is linear and narrow. Valves are of length 22–28.5 µm, width 7–8 µm, striae density 10–13/10 µm and the scale bar, LM images 10 µm. These species are poorly formed, with little information on ecology but are widely reported in moderate to high eutrophic conditions.
Habitat: Moderate to high polluted water.
Ref. Wojtal (2009), p. 153, pl. 14, figs. 8–12.
Valves are weakly heteropolar club-shaped; ends are rostrate to sub-capitate, slightly deflected and lanceolate to elliptical to oval. Valves are of length 18–22 µm, width 4–6 µm and striae density 14–15/10 µm. The axial area is narrow and linear. Apices protect weakly capitates. Striae are parallel to weakly radial and indistinctly punctate. The raphe is weakly curved, appearing filiform in small specimens. A widely spread species that thrives in a range of water reservoirs (small pools, lakes and rivers); it is typically thought to be tolerant of highly contaminated conditions.
Habitat: Highly polluted water.
Ref. Wojtal (2009), p. 153, pl. 14, figs. 15–16.
Valves are oval to lanceolate-shaped like a club. The largest width is usually above the centre. Ends foot pole more strongly narrowed, and head pole shortly protracted blunt. Valves are of length 55–60 µm, width 13–14 µm, striae density 10–12/10 µm, weekly throughout, punctate—indistinct. The unilateral shortening of one stria results in the undersized, poor construction of the axial region. Species are mainly meso-eutrophic lakes. Some similar taxa G. augur has typically larger valves with a more distinct head portion and the largest valve width just below the head pole.
Habitat: Polluted water.
Ref. Karthick et al. (2013), figs. 76–95.
Valves are clavate shapes, rounded head and foot poles. Dimensions: Length 26–27 µm, width 6.5–7 µm. Axial area tinny, central area irregular and raphe lateral undulate. Striae density 6.5–7.5/10 µm. Striae separately lineate and radiate throughout the valve.
Habitat: Moderate water.
Ref. Lange-Bertalot et al. (2017), pl. 63, p. 327, figs. 1–4; Karthick et al. (2013), pl. 35; Taylor et al. (2007a), pl. 35.
Valves are moderately sigmoid and deflected into broad, linear-lanceolate at the centre, bluntly rounded apices. Valves are of length 90–110 µm, width 13–14 µm and striae 18–20/10 µm. The central area is small, apically elliptic. Striae more or less parallel.
Habitat: Freshwater.
Ref. Karthick et al. (2013), pl. 48, Lange-Bertalot et al. (2017), pl. 46, p. 356; Taylor et al. (2007a), pl. 50.
Valves are elliptic-lanceolate, rhombic-lanceolate to linear-elliptic. Ends range from almost sharply or bluntly rounded, occasionally substrate. Striae radiate and are slightly denser than other taxa. A large stigma is found in one of the central areas. Valves are of length 29–32 µm, width 8–10 µm and striae 19 µm, radiate throughout. This species commonly occurs in electrolyte-poor sub-aerial environments.
Habitat: Polluted water.
Ref. Lange-Bertalot et al. (2017), pl. 46, p. 357.
Valve is linear, linear elliptical in the case of smaller specimens, with more or less tri-undulate margins. Valves are of length 11–20 µm, width 6–7 µm, one isolated, circular stigma present, striae 17–20 µm. Ends rostrate to capitates, with bluntly rounded apex.
Habitat: Polluted water.
Ref. Lange-Bertalot et al. (2017), pl. 33, p. 387, figs. 1–5.
Raphe filiform to weakly lateral, axial area narrow and linear, valves narrow to broadly lanceolate, apices acutely rounded. Valves are of length 27–32 µm, width 6–7 µm and striae density 15–16/10 µm. The central region was small and its border was unpredictable. Striae radiate outward, becoming convergent towards the pole and slightly bent at the valve centre.
Habitat: Freshwater.
Ref. Lange-Bertalot et al. (2017), p. 39, figs. 12–16; Krammer and Lange-Bertalot (1985), pl. 17, figs. 10–12.
Valves are lanceolate. Ends obtusely to acutely rounded, rarely weakly protracted. The raphe is filiform. Valves are of length 33–48 µm, width 8–10 µm and striae density 10–12/10 µm.
Habitat: Freshwater.
Ref. Lange-Bertalot et al. (2017), pl. 38, figs. 10–14; Foged (1980), p. 654, pl. 6, fig. 7; Rai (2006), p. 89, pl. 9, fig. 3.
Valves have sub-capitate ends that are often strongly protruded and lanceolate to virtually linear-lanceolate. Short axial area, moderately large, rounded and equal central area. Valves are of length 38–40 µm, width 8–10 µm and striae 13–14/10 µm. Raphe is filiform, straight, with proximal raphe, ends dilated slightly and bent towards one part.
Habitat: Freshwater.
Ref. Lange-Bertalot et al. (2017), pl. 119, figs. 9–15, Taylor et al. (2007a), pl. 163.
Valves feature rounded apices and are elliptical, lanceolate or linear-lanceolate to linear in structure. Dimensions: Length 15–23 µm, width 4–5 µm and striae density 15–18/10 µm. The central part sometimes has very slightly concave margins. Striae always appear coarsely punctate in LM. Cosmopolitan taxa are found in a wide range of waters from electrolytic poor or electrolytic rich waters.
Habitat: Indicator of very good ecological quality.
Light microscopy (LM): 1–3 Nitzschia obtusa var. (Kurzii Rabenhorst); 4–6 Nitzschia palea (Kützing) Smith, 1856; 7–9 Nitzschia sigma (Kutzing) Smith, 1853; 10–12 Nitzschia intermedia (Hantzsch in Cleve & Grunow 1880); 13–14 Pinnularia gibba (Ehrenberg, 1843); 15–17 Pinnularia nobilis (Ehrenberg, 1843); 18–20 Pinnularia acrosphaeira (Smith); 21–23 Planothidium lanceolatum (Brebisson ex Kutzing); 24–25 Staurosirella construens (Ehrenberg); 26–28 Seminavis strigosa (Hustedt); 29–31 Staurosirella pinnata (Ehrenberg); 32–33 Tryblionella calida (Grunow); 34 Tryblionella levidensis (Smith, 1856). Scale bars = 10 µm.

Ref. Karthick et al. (2013), pl. 112; Taylor et al. (2007a), pl. 146.
Valves with narrow rostrate or capitated apices that ranged from linear to linear-lanceolate. Valves are of length 78–83 µm, width 7.5–8, fibulae 5–8 and striae density 28–31. Keel is supported by irregularly spaced fibulae. Frustules occur in long chains, connected along their keel. It occurs as moderate in high electrolytic waters, mostly in sewage waters.
Habitat: Freshwater with low to moderate pollution.
Ref. Bishop et al. (2017), pl. 138, figs. 27–30; Lange-Bertalot et al. (2017), pl. 113, figs.1–20; Prasad et al. (1992), p. 288, pl. 35, fig. 5.
Valves are several varieties that exist, linear-lanceolate to linear, rarely lanceolate. Valves are lanceolate parallel and tapering rapidly at the poles, terming acting with sub-capitate apices. Valves are of length 12–40 µm, width 3–5 µm and striae density 23–40/10 µm. Striae is barely visible in LM.
Habitat: Polluted water.
Ref. Krammer (1988), p. 32, fig. 23.
Girdle view, the frustules sigmoid. Valve view, according to the position, is more or less sigmoid, centre part linear-lanceolate. The apices are small and capitates. The keel of the raphe is strongly eccentric. Valves are of length 35–40 µm, width 4–6 µm and striae 24–26/10 µm.
Habitat: Freshwater.
Ref. Lange-Bertalot et al. (2017), pl. 109, p. 448, figs. 1–6; Karthick et al. (2013), pl. 120; Taylor et al. (2007a), pl. 158.
Valves are having linear to linear-lanceolate shapes. Poles’ apexes are narrowly rounded and weakly capitated. Raphe and keel are supported by small narrow fibulae, with no central area. Valves are of length 53–65 µm, width 3–4 µm and striae density 23–32/10 µm.
Habitat: Medium to moderately water.
Ref. Taylor et al. (2007a), pl. 90; Lange-Bertalot et al. (2017), pl. 72, p. 482, figs. 1–5.
Valves are more or less rhombic—lanceolate to almost linear. The ends are broadly sub-capitate. Valves are of length 50–52 µm, width 11–13 µm and striae density 9–11/10 µm; from radiate to strongly convergent towards the ends.
Habitat: Poor fresh water.
Ref. Lange-Bertalot et al. (2017), pl. 76, p. 485, fig. 1.
Valves are moderately variable if varieties, linear, end inflated centre and ends. Valves are of length 36–54 µm, width 8–10µm and striae 12–13/10 µm. strongly radiate in the centre, convergent towards the ends. The valve axial area is a quarter to a third of the valve width. The central area is usually only weakly differentiated and raphe is complex.
Habitat: Moderate water.
Ref. Karthick et al. (2013), pl. 74.
Valves outline linear, margin straight, slightly convex, swollen in the middle. Ends broadly rounded, and sometimes looks capitates. Valves are of length 42–56 µm, width 9–11 µm and striae density 12/10 µm. Scale bar, LM images = 10 µm. The axial area is broad, linear and slightly widened near the central area.
Habitat: Cosmopolitan.
Ref. Lange-Bertalot et al. (2017), pl. 25, p. 509, figs. 27–33.
Valves are having elliptic to elliptic-lanceolate with slightly drawn-out apices. Valves end broadly rounded, occasionally broadly sub-capitate. Valves are of length 15–32 µm, width 6–8 µm and striae density 13–15/10 µm. The species’ most widely distributed diatoms are in freshwater habitats, particularly in running water.
Habitat: Freshwater.
Ref. Karthick et al. (2013), pl. 19.
Valves are cruciform with rostrate to sub-capitate ends. Valves are of length 10–15 µm, width 6–8 µm and striae density 12–14/10 µm. The valve’s ventral margin is inflated and narrowly or extensively curved. Axial are linear to lanceolate, and striae are alternate and composed of lineolae decreasing in size from the edge of the valve to the centre.
Habitat: Freshwater with moderate water.
Ref. Lange-Bertalot et al. (2017), pl. 94, p. 335, figs. 29–30, Karthick et al. (2013), pl. 97.
Valves are half-elliptical, dorsal margin strongly convex, ventral margin straight to weakly convex. Valves are of length 22–24 µm and width 4–4.5 µm. Raphe slightly arcuate, striae density 10–12/10 µm.
Habitat: Cosmopolitan.
Ref. Karthick et al. (2013), pl. 19; Lange-Bertalot et al. (2017), pl. 11, p. 651, figs. 30–35; Bishop et al. (2017), pl. 2, figs. 46–49.
Valves are broadly elliptical to ovoid in outline with rounded apices. The valve faces or slightly undulates due to raised costae. Frustules are rectangular, form colonies that resemble ribbon, and are linked by connecting spines in the valve girdle view. Valves are of length 7–10 µm, width 4–6 µm and striae density 8–10/10 µm.
Habitat: Freshwater.
Ref. Lange-Bertalot et al. (2017), pl. 105, p. 597, figs. 5–8.
Valves are linear with weakly concave margins in the centre, and tapering to the wedge-shaped, substrate ends. Valves are of length 50–78 µm and width 9–10µm. The apices are narrow, protracted and narrowly rounded. Keel is strongly eccentric. A narrow, indistinct fold runs along the longitudinal axis of the valve. Striae density is 28–32/10µm.
Habitat: Poor moderate water.
Ref. Lange-Bertalot et al. (2017), pl. 105, p. 599, figs. 1–4.
Valves are broadly linear-elliptical, valve ends wedge-shaped and obtusely rounded. Valves are of length 28–64 µm, width 14–23 µm and striae density 34/10 µm, difficult to resolve, masked by robust support ribs.
Habitat: Brackish water.
Results and Discussion
A total of 40 diatom taxa belonging to 20 genera were found in samples collected from 10 different locations sampled in the Thamirabarani River. Amongst the diatom taxa recorded, 36 are pennate diatoms and 4 are centric diatoms. The pennate diatom taxa are Bacillaria paxillifera, Caloneis schumanniana, Cymbella trophica, C. tumida, C. placentula, Diadesmis confervacea, Encyonema vulgare, Fragilaria crotonensis, F. rumpens, Gomphonema. affine, G. lagenula, G. lacusrankala, G. slacusrankalaoides, G. parvulum, G. pseudoaugur, Gomphonema tamilensis, G. acuminatum, Luticola geoppertiana, L. nivalis, N. cryptonella, N. erifuga, N. rostellatta, N. amphibia, N. incognita, N. obtusa, N. palea, N. sigma, Pinnularia gibba, P. nobilis, P. acrosphaeira, P. lanceolatum, Staurosirella construens, S. pinnata, Seminavis strigosa, Tryblionella calida and T. levidensis; whereas the centric diatoms recorded are A. ambigua, A. granulata, C. meneghiniana and D. stelligera.
As seen in Table 4, sites 1 to 7 possess a WQI between 29 and 48. These stations are characterised by abundant A. ambigua, D. stelligera, N. cryptonella, P. lanceolatum, S. pinnata. Based on these two observations, the stretch covered by sites 1–7 can be inferred to be Oligotrophic. This could be attributed to good flow of water and no industrial waste discharge into the stream. These results are consistent with those of Faustino et al. (2016) who describes the ecology of A. ambigua as oligotrophic. N. cryptonella is one of the most frequently found diatoms in rivers (Lange-Bertalot et al., 2017) and suggests oligotrophic to eutrophic conditions. P. lanceolatum has a wide ecological range and occurs in fresh water habitats abundantly. All freshwater habitats contain S. pinnata. The taxa at locations 1–7 thus are consistent with previous reports of their ecology.
The diatom taxa such as C. meneghiniana, Diadesmis confervacea, Gomphonema parvulum, N. amphibia and Nitzschia palea are abundant at sites 8 to 10 corresponding to a WQI of 100–110 categorised as poor water quality (Tables 3 and 4). Eutrophic waters are caused in the study area due to low water flow, high cremation-ash dispersion (the casting ritual) and anthropogenic activities. The high pollution in the can be attributed to high volumes of waste fish being discarded and the less water flows (Faustino et al., 2016; Kalyoncu & Şerbetci, 2013; Karthick et al., 2013; Lange-Bertalot et al., 2017). With its wide range and prevalence as benthos in lakes, rivers and streams, C. meneghiniana points to eutrophication. A globally distributed species called D. confervacea can be found in a variety of waterways; amongst them are eutrophic, electrolyte-rich conditions that are highly contaminated (Karthick et al., 2013). G. parvulum is a relatively common species at downstream eutrophic locations with heavy organic pollution (Kalyoncu & Şerbetci, 2013). N. amphibia is a cosmopolitan taxon found in a wide range of electrolyte rich poor waters (Karthick et al., 2013). N. palea is a species with a medium tolerance for pollution. This species is known to exhibit a high relative abundance in extremely polluted water, indicating that it can tolerate heavy pollution (Kalyoncu & Şerbetci, 2013).
Conclusion
The present study records 40 diatom taxa (36 pinnate and 4 centric) in the Thamirabarani River Naranammalpuram Tirunelveli District and Punnakayal in Thoothukudi District of Tamilnadu. The abundance of A. ambigua, D. stelligera, N. cryptonella, P. lanceolatum, S. pinnata at sites 1 to 7 characteriSed by WQI of 29 to 48 indicating oligotrophic waters in this stretch, due to good flow of water and no contamination sources. Diadesmis confervacea, Gomphonema parvulum, Nitzschia palea and Pinnularia gibba were recorded at sites 8 to 10 where the WQI varies between 100 and 110, indicating eutrophication due to anthropogenic activities.
Footnotes
Acknowledgements
This work was supported by the Periyar University, Salem, Tamil Nadu, India through providing the University Research Fellowship (URF). The authors are deeply grateful to the Department of Geology, Periyar University for providing sophisticated instrumentation facilities and constant support. The authors thank two anonymous reviewers for providing detailed comments that helped elevate the MS.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
