Abstract
Marine pollution due to heavy metals is an ever-growing concern globally, particularly in coastal environments affected by increased anthropogenic activities. The heavy metals are harmful to marine life because of their severe toxicity and easy bioaccumulation. We evaluate the toxicological impact of Arsenic on benthic foraminifera, a ubiquitous group of marine microorganisms in all types of marine environments. The juvenile specimens of shallow-water benthic foraminifera
INTRODUCTION
The rise in industrialisation, the discharge of untreated waste into the ocean, agricultural runoff, urbanisation, and other associated activities have led to heavy metal contamination in marginal marine regions. The heavy metals like Mercury, Cadmium, Arsenic, Lead, are very toxic, as they accumulate in the sediments, contaminating the areas on a large scale (Ganugapenta et al., 2018; Liu et al., 2020; Qian et al., 2015; Soliman et al., 2015; Tian et al., 2020). These heavy metals affect marine organisms, including fish populations, which, in turn, affect our primary economic resources. Therefore, in recent decades, studies on marine pollution have gained widespread attention worldwide. It stands as one of the most critical environmental issues of recent times. Marine pollution can be detected by various chemical, physical, and biological methods (Chakraborty et al., 2014; Suokhrie et al., 2017). Biologically, levels of pollutants can be detected in marine micro- and macro-organisms. In marine macro-organisms like fish and bivalves, toxic pollutants are not only bioaccumulated but also biomagnified as they move up the food chain (Chakraborty et al., 2016). Therefore, marine microorganisms that occupy the base of the food chain are well-suited to detect early signs of metal toxicity (Ramljak et al., 2024).
The unicellular marine microorganisms, such as benthic foraminifera, are sensitive to environmental conditions and have good preservation potential (Saraswat, 2023). Therefore, these organisms are extensively used to study heavy metal pollution (Alve, 1991; Amrutha et al., 2018; Dimiza et al., 2022; Ganugapenta et al., 2025; Nigam et al., 2006). Most of the above studies reported differences in species abundance and increased morphological deformities in foraminifera from polluted areas (Malek & Frontalini, 2024). The field-based studies have limitations, as at times the differences in species abundance and morphological deformities may not be solely due to heavy metal contamination. The deformities and differences in abundance can also be attributed to changes in ecological conditions (Prayudi et al., 2024). Therefore, culture studies under controlled laboratory conditions help assess the effects of individual or combined parameters to substantiate field-based studies (Filipsson, 2008; Nigam et al., 2009; Sagar et al., 2021; Saraswat et al., 2004; Schmidt et al., 2022). These studies reported distinct differences in morphological abnormalities, survival rates, reproduction, and distribution of benthic foraminifera subjected to different pollutants, thus confirming the extreme sensitivity of benthic foraminifera to pollutants. In the present study, the effect of Arsenic on shallow-water benthic foraminifera
Naturally, Arsenic originates from the weathering of rocks (mainly shale, sandstone, and volcanic rocks), volcanic eruptions, and certain biological activities (Nordstrom, 2002). Anthropogenically, Arsenic is obtained as a by-product of metal smelting and coal burning, and it enters the aquatic system through industrial waste and mining processes. Both Arsenic(III) and Arsenic(V) compounds are soluble in water. The primary oxidation state in which Arsenic enters the aquatic system is As(III), or Arsenite. China is the primary producer of Arsenic. Applications of Arsenic trioxide range from medicinal to industrial processes. In medicinal use, As2O3 is used in the treatment of acute promyelocytic leukaemia (Shen et al., 1997), under precautions. In industrial applications, it is used in glass manufacturing, electronic devices, and metal production. Previously, it was used in the preparation of pesticides, herbicides, and wood preservatives (Alamgir et al., 2004), which are now banned due to their toxicity. Arsenic enters the aquatic system through natural sources, such as the weathering of Arsenic-rich rocks and geothermal activities, and anthropogenically as a by-product of various industrial processes. Because it dissolves readily, Arsenic can find its way into soil, rivers, lakes, and other water sources. Groundwater in nearly 108 countries is contaminated with Arsenic, affecting more than 230 million people worldwide (Shaji et al., 2021). Arsenic concentration in the tropical coastal and estuarine regions of Goa ranged from <1 to >67 µg/L (Fondekar & Reddy, 1974). However, the concentration decreased in the open ocean and varied from <1 to >18.75 µg/L (Fondekar & Sen Gupta, 1976).
Arsenic is highly toxic and therefore should be handled very carefully. Its toxicity depends on the duration of exposure to and its concentration. Although Arsenic is highly toxic, very few studies have examined its toxicity to marine microorganisms, which form the base of aquatic food webs. Given its acute toxicity, we studied the effects of Arsenic on marine microorganisms benthic foraminifera in a controlled laboratory culture experiment.
MATERIALS AND METHODS
The living benthic foraminifera were collected from the shallow waters of the central west coast of India, specifically off the coast of Goa. The surface sediments and the marine algal samples were collected in pre-labelled zip-lock bags. The material was then brought to the shore and emptied into a tub containing 63 µm filtered seawater. The material was then shaken vigorously to detach foraminifera attached to the substrate and sieved through 1 mm and 63 µm sieves. The larger-pore-size sieve (1 mm) was used to remove the unwanted material. The material >63 µm but <1 mm was transferred to a beaker filled with filtered seawater and brought to the laboratory.
In the laboratory, these samples were observed under an Olympus SZX16 stereomicroscope to isolate living specimens of
Preparation of stock solutions
Solutions with different concentrations of Arsenic, ranging from 20 to 220 µg/L, were prepared at 20 µg/L intervals. The stock solution was prepared by dissolving Arsenic trioxide in a 1 N NaOH solution. The final media with different Arsenic concentrations were prepared from this stock solution.
Experimental set-up
The experiment was conducted using the six-welled culture plates. The setup included two complete independent sets. Each set contained one six-welled plate for the main experiment and a second six-welled plate as a replicate, with different concentrations of Arsenic solution (20–220 µg/L). One well each in two trays had normal seawater without any additional Arsenic. A total of 5 juvenile specimens of
RESULTS
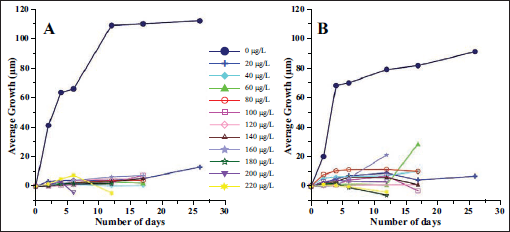
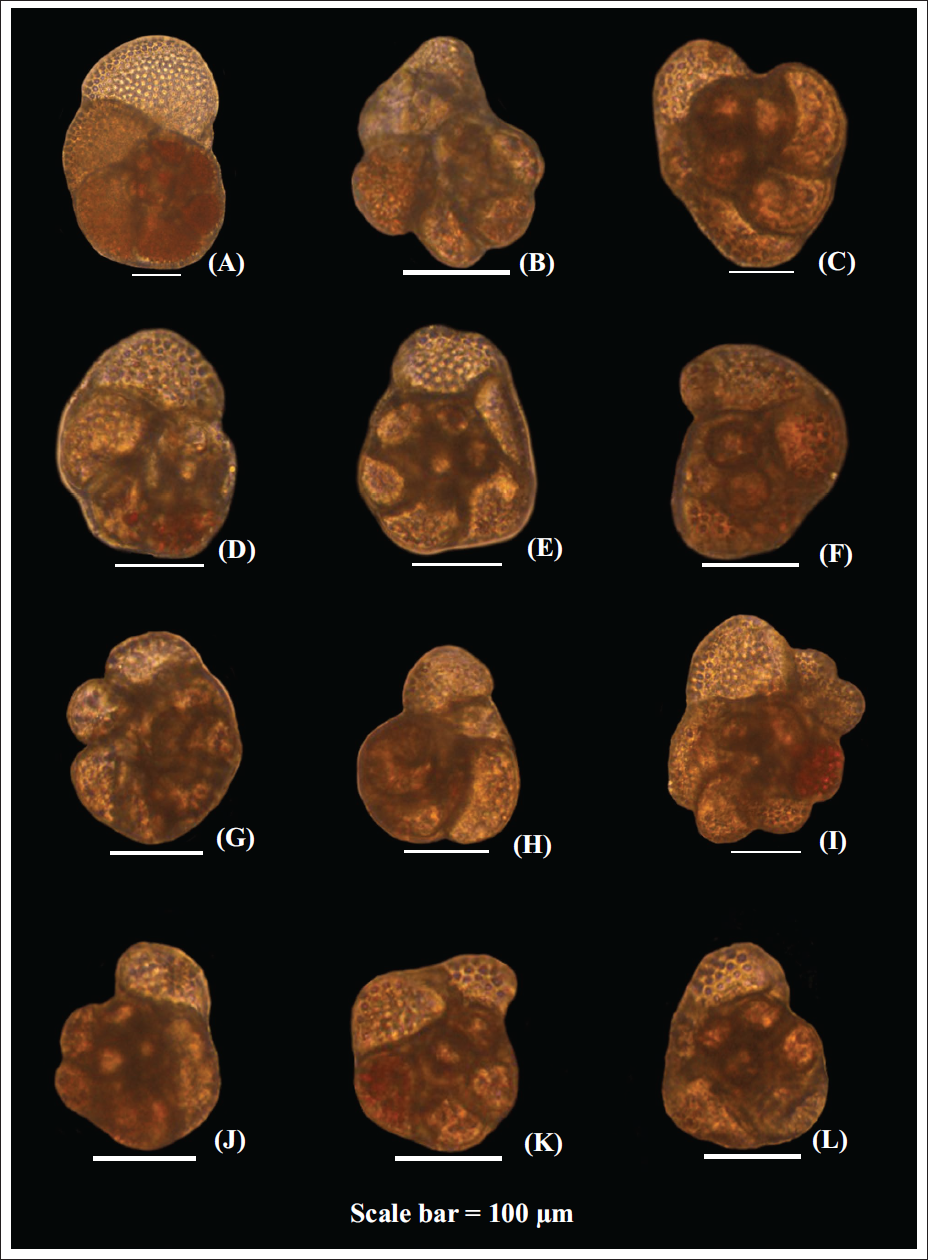
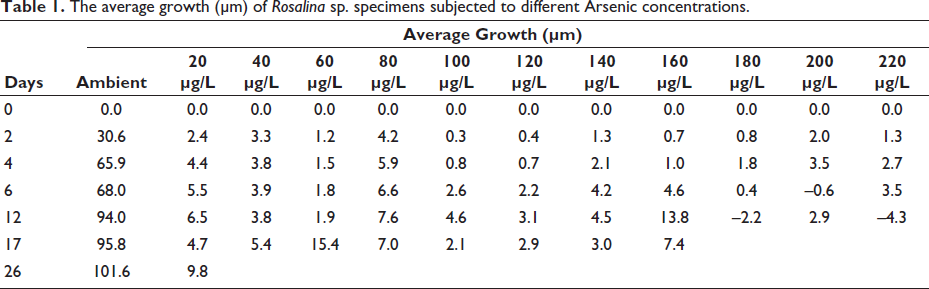
At the onset of the experiment, all the specimens showed extensive pseudopodial activity. Shell movement and food collection were also observed in a majority of the specimens. As this was a sudden stress for the specimens subjected to different Arsenic concentrations, growth was very low in all specimens exposed to Arsenic compared to those without additional Arsenic in the media (Figure 1). The average growth of the specimens subjected to normal conditions was 112 µm in Set A and 91 µm in Set B (mean 101.5 µm; Figure 1). The growth of specimens exposed to Arsenic was an order of magnitude lower than that of the control specimens (Table 1). Many of the specimens exposed to Arsenic were abnormal, and morphological abnormalities increased with increasing Arsenic concentration (Plate 1). The abnormalities included chambers that were smaller than the standard size. The orientation of the chambers was also abnormal compared with those in specimens subjected to seawater without additional Arsenic (Plate 1). After a few days, growth almost ceased in most specimens treated with Arsenic, accompanied by a decline in pseudopodial activity.
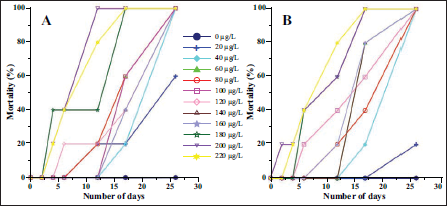
A few specimens with a higher concentration of Arsenic (180–220 µg/L) started to die after 3 days (Figure 2). Initially, one or two specimens died, but by the 12th day, all the specimens subjected to 200 and 220 µg/L died, and by the 16th day, all the specimens at 180 µg/L also died. The specimens subjected to other concentrations were alive for more than 12 days. All specimens exposed to 160 µg/L died after 17 days; a few specimens exposed to 60-140 µg/L also died. The remaining specimens exposed to 40–140 µg/L As2O3 died within 26 days of the experiment. A few specimens subjected to 20 µg/L died after 26 days. A few specimens exposed to 20 µg/L As2O3 remained alive for 26 days. Incidentally, none of the specimens reproduced during the course of the experiment.
The average maximum growth attained by benthic foraminifera Rosalina sp. at different concentrations of Arsenic in the laboratory culture experiment. The replicate sets are marked as A and B.
The abnormalities observed in specimens subjected to different concentrations of Arsenic. The specimen (A) is the normal specimen subjected to normal conditions (35‰ salinity and 27°C temperature, without any additional pollutant). The specimens (B–L) show the morphological abnormalities observed during the experiment.
The mortality rate (%) of Rosalina sp. at different concentrations of Arsenic in the laboratory culture experiment. The replicate sets are marked as A and B.
The average growth (µm) of Rosalina sp. specimens subjected to different Arsenic concentrations.
DISCUSSION
The high prevalence of abnormalities and mortality in specimens exposed to different concentrations of Arsenic suggests its severe impact on benthic foraminifera. The increased abnormalities in foraminiferal tests from polluted sites were reported by many workers (Bhalla & Nigam, 1986; Carpenter, 1856; Debenay et al., 2001; Frontalini & Coccioni, 2008; Jayaraju et al., 2008; McCrone & Schafer, 1966; Naidu et al., 1985, 2000; Orabi et al., 2017; Romano et al., 2008; Samir, 2000; Setty & Nigam, 1984; Stouff et al., 1999; Yanko et al., 1999). However, morphological abnormalities in foraminifers have also been reported in the specimens subjected to different naturally ecologically stressed environments (Boltovskoy et al., 1991; Kurtarkar et al., 2011; Saraswat et al., 2015). To delineate the abnormalities caused by naturally ecologically stressed conditions and those caused by anthropogenic pollutants, several laboratory culture studies have been conducted (Nigam et al., 2009; Qiao et al., 2025; Saraswat et al., 2004; Stouff et al., 1999; Yanko et al., 1999). In this laboratory culture experiment, conducted to study the effect of the heavy metal Arsenic on benthic foraminifera
The abnormalities in the shells included stunted specimens with chambers oriented in planes other than the normal plane of chamber addition. The shells with chambers added in plains other than the normal plain of chamber addition are the dominant abnormality in regions polluted with heavy metals (Martínez et al., 2023). The culture study thus confirms that heavy metals are the primary factor driving the formation of abnormally oriented chambers in benthic foraminifera. The newly formed chambers in many of the specimens subjected to Arsenic were either too small or too large compared to the preceding chamber, leading to an odd shape in the shells. Similar abnormalities were reported in benthic foraminifera subjected to higher Mercury concentrations, under controlled laboratory conditions (Saraswat et al., 2004). Interestingly, similar abnormalities have also been reported in benthic foraminifera from usually ecologically stressed environments, without any anomalously high element-specific pollution (Boltovskoy et al., 1991; Kurtarkar et al., 2011; Saraswat et al., 2015). However, the relative abundance of abnormal specimens in such environments is much lower than that observed in this study.
The abnormalities in benthic foraminifera subjected to heavy metal pollution can be either because of physiological stress or odd-sized heavy metals altering the regular crystal lattice pattern of calcareous shells. In culture experiments with benthic foraminifera subjected to a combination of higher concentrations of several elements (Lead, Zinc, Mercury, Silver, Nickel, Manganese, Cadmium, Chromium and Tin), the newly formed chambers contained elevated concentrations of a few of these elements. However, no abnormality was reported in such specimens subjected to elevated concentrations of these elements (Schmidt et al., 2022; Titelboim et al., 2021). Therefore, it is likely that the heavy metal-induced physiological stress affecting the normal metabolic activities of benthic foraminifera resulted in abnormal shells.
Incidentally, none of the specimens in this experiment reproduced. Many factors control reproduction in benthic foraminifera. Previous studies have reported that higher or lower than optimal temperatures, as well as food availability (Bradshaw, 1957; Hemleben & Kitazato, 1995; Myers, 1935; Ross, 1977), affect reproduction in benthic foraminifera. A few studies have reported that benthic foraminiferal species may reproduce under stressed conditions (Ernst et al., 2006; Le Cadre & Debenay, 2006; Nigam et al., 2009), whereas other species may not reproduce (Moodley et al., 1998; Saraswat et al., 2004). However, the absence of reproduction even in the control set specimens in this experiment suggests that the experiment may have lasted less than the lifespan of this species, or that the juveniles may not have been fully matured to reproduce. Future experiments with a longer duration may help resolve this issue.
CONCLUSIONS
Based on a laboratory culture experiment, we report that Arsenic is severely toxic to shallow-water benthic foraminifera. The increasing Arsenic concentration resulted in morphological abnormalities and mortality in benthic foraminifera. The specimens subjected to higher concentrations (180–220 µg/L) showed chamber deformities and early death within 12–16 days. Based on this study, we report that the benthic foraminifera
Footnotes
Acknowledgements
The authors thank the Director of the CSIR-National Institute of Oceanography for the infrastructural support and permission to publish the findings. We also thank the Guest Editor, Prof. Ashutosh Singh, for inviting us to submit this manuscript. SRK thanks the Department of Science and Technology for the financial support in the form of the Women Scientist Scheme [DST/WOS-A/EA-51/ 2021 (G)]. The authors declare that they have no conflict of interest. We thank the two anonymous reviewers for their valuable comments and suggestions, which helped improve this manuscript.
Author Contributions Statement
RS designed the research, reviewed and edited the original draft; SRK and ED performed the research; SRK wrote the original draft, and all authors contributed to the manuscript.
Data availability statement
The data are included as a table in the manuscript.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors received financial support for this research and/or publication of this article by the Department of Science and Technology, Government of India, Women Scientist Scheme (DST/WOS-A/EA-51/ 2021 (G)).
Statements and Declarations
Ethical considerations: Not applicable; Consent to participate: Not applicable; Consent for publication: Not applicable.
