Abstract
Dissolved oxygen, a vital parameter for a majority of marine organisms, may decline in the near future due to anthropogenic eutrophication in coastal waters. The effect of depleting dissolved oxygen on the marine benthic community is difficult to assess from field studies, as dissolved oxygen often co-varies with several other parameters, especially organic matter. The controlled laboratory culture experiments can help to assess the effect of a specific parameter on marine organisms. Foraminifera constitute a substantial fraction of the marine benthic organisms, and a few species are vulnerable to depleted oxygen. A majority of the previous culture experiments have used isolated specimens of individual benthic foraminiferal species. Although such studies helped to understand the response of individual species to a particular parameter, they have to be scaled up to understand the community-level response of benthic foraminifera. Here, we assess the community-level response of the shallow subtidal benthic foraminiferal community to five different oxygen concentrations (1.67 mL/L to 5.01 mL/L) in a laboratory culture experiment. The living benthic foraminiferal abundance was considerably high at the intermediate oxygen concentration, whereas it decreased at both the lower and higher concentrations. The decreasing dissolved oxygen caused poor preservation of dead benthic foraminifera due to a drop in pH. The relative abundance trend of the species belonging to the same genus was different, suggesting a species-specific response to the dissolved oxygen. The maximum abundance at 2.91 mL/L and the varying response of individual species clearly suggest a non-linear response of the benthic foraminiferal community to the dissolved oxygen.
Introduction
Dissolved oxygen concentration in seawater is one of the major environmental factors that control marine community structure (e.g., Gooday et al., 2010; Nigam et al., 2007). Anthropogenic eutrophication has depleted the dissolved oxygen in the ocean, especially on the shallow continental shelves (Keeling & Garcia, 2002; Meier et al., 2011; Rabalais et al., 2014; Sarmiento et al., 1998). Oxygen is vital for marine organisms, especially benthic organisms. The unicellular benthic foraminifera constitute a substantial fraction of the benthic community on the shelf and slope (Alves Martins et al., 2019; Lei et al., 2014; Suokhrie et al., 2021; Yamashita et al., 2020). Foraminifera play a largely unquantified but potentially significant role in carbon cycling (Gooday et al., 1992; Kurbjeweit et al., 2000). Their abundance and diversity are strongly influenced by the ambient conditions, including the temperature, salinity, sediment texture, food availability and oxygen (e.g., Jorissen, 1995; Jorissen et al., 2007; Kaithwar et al., 2020; Kurbjeweit et al., 2000; Kurtarkar et al., 2019; Langlet et al., 2020; Lei et al., 2017; 2019; Stewart et al., 2021; Venturelli et al., 2018). The vertical distribution of benthic foraminifera is also affected by oxygen, except for some deep infaunal species (Ernst & van der Zwaan, 2004). A few species of benthic foraminifera are vulnerable to low oxygen (Gomaa et al., 2021). However, the abundance of infaunal benthic foraminifera increases in low-oxygen environments (Bernhard & Sen Gupta, 1999; Kaiho, 1994; Singh et al., 2006, 2015). As the bottom water oxygen decreases below 2 mL/L, the infaunal foraminifera start to migrate upwards in the sediment, and this response is species-specific (Alve & Bernhard, 1995).
The dissolved oxygen also modulates benthic foraminiferal morphology. The infaunal angular asymmetrical benthic foraminifera (e.g., Bolivina dilatata, Bulimina marginata, Fursenkoina bradyi, Uvigerina asperula) dominate the oxygen-deficient zones along the western continental margin of India (Nigam et al., 2007). A few distinct morphological characteristics were observed among calcareous benthic foraminiferal species living in dysaerobic conditions, namely the smaller, thin-walled tests (e.g., Spiroplectammina, Textularia) and tests being more porous as compared to specimens from the oxic waters (e.g., Bolivina seminuda) (Bradshaw, 1961; Gooday et al., 2000; Perez-Cruz & Machain-Castillo, 1990; Phleger & Soutar, 1973; Sen Gupta & Machain-Castillo, 1993; Tetard et al., 2017). Further, foraminifera in an oxygen-deficient environment are characterised by high pore density (Glock et al., 2011; Kuhnt et al., 2013; Tetard et al., 2017). Given the strong influence of dissolved oxygen, benthic foraminifera are often used to trace the changes in the extent and intensity of both the coastal and deep-water oxygen-deficient zones (Bernhard & Sengupta, 1999; den Dulk et al., 2000; Kranner et al., 2022; Palmer et al., 2020; Platon et al., 2005; Richirt et al., 2022; Sen Gupta et al., 1996). The benthic foraminifera are also suggested to have an added advantage of delineating oxic (>1.5 mL/L O2) and suboxic (0.3–1.5 mL/L O2) facies, which is problematic from the organic carbon/sulphur ratio, sulphur isotopes and trace elements (Kaiho, 1994).
The application of benthic foraminifera to reconstruct dissolved oxygen concentration in the past is complicated because the dissolved oxygen often co-varies with organic matter in the majority of the marine settings. The higher productivity increases the organic matter flux. The increased availability of organic matter supports a large microbial population. Excessive microbial respiration is one of the factors responsible for the development of oxygen-deficient zones. Several authors suggest that both dissolved oxygen and organic matter flux are the critical factors controlling the abundance and distribution of benthic foraminifera (Bernhard, 1986; Caralp, 1984; Caulle et al., 2014; Corliss & Chen, 1988; Douglas, 1981; Gooday, 1993; Jorissen, 1999; Jorissen et al., 1992; Kranner et al., 2022; Lutze & Coulbourn, 1984; Nienstedt & Arnold, 1988; Sen Gupta et al., 1981; Singh et al., 2018, 2021; Sjoerdsma & Van der Zwaan, 1992; Suokhrie et al., 2020; Wahyudi & Minagawa, 1997). Therefore, it is not easy to delineate the effect of dissolved oxygen from that of the organic matter on the benthic foraminifera community from the field studies. The widely accepted Trophic Conditions and Oxygen Concentrations model also incorporated both the dissolved oxygen and organic matter content to explain the changes in the benthic foraminifera community (Jorissen et al., 1995). A more efficient application of foraminifera to reconstruct the past dissolved oxygen concentration requires an assessment of the effect of only dissolved oxygen on the benthic foraminiferal community. Additionally, increasingly hypoxic to anoxic conditions have been reported from the marginal marine regions of several oceans (Diaz, 2001; Naqvi et al., 2010). The implications of such hypoxic to anoxic regions on benthic communities have to be assessed (Vaquer-Sunyer & Duarte, 2008; Bernhard et al., 2021). The objective of this work was to understand the effect of different concentrations of dissolved oxygen on shallow inter-tidal benthic foraminiferal communities in a controlled laboratory culture experiment. We also studied the specific response of the dominant species to different dissolved oxygen concentrations.
Methodology
The living benthic foraminiferal community was collected from the intertidal and subtidal waters of Dias Beach, off Goa, on the west coast of India (Figure 1). The beach is surrounded by the Mandovi Estuary to the north and the Zuari Estuary to the south. It is ~110 m long and has rocky cliffs on two sides. The seawater temperature varies from 25°C to 35°C, with the lowest during the winter season and the highest during the pre-summer monsoon season (Kurtarkar et al., 2011). A large variation is also observed in the salinity (11–36 psu). The lowest salinity is during the summer monsoon season, and the highest is during the pre-summer monsoon season (Kurtarkar et al., 2011). The sample was collected from a water depth of 2–5 m. Both the surface sediments and the seaweeds attached to the rocky patches were collected to retrieve the living benthic foraminifera. The seaweeds attached to the rocks were gently removed and stirred vigorously in a plastic bucket to detach living benthic foraminifera. The material was then sieved by using 63 µm and 1,000 µm sieves to concentrate living benthic foraminifera and to get rid of the unwanted material. The >63 µm and <1000 µm fraction as transferred to glass beakers partially filled with seawater. The fine-grained sediments (<63 µm) were also collected separately and brought to the laboratory to be used in the experiment. Additionally, the top half centimetre of the sediments were carefully scrapped by using a sterile spatula to avoid contamination. The sediments with seawater were transferred to glass beakers and covered with parafilm to avoid excessive evaporation while maintaining the gas exchange. Along with the living foraminiferal community and sediments, seawater was also collected in 20 L jerry cans. The seawater temperature at the sampling site was 27.15°C, and the dissolved oxygen concentration was 5.43 mL/L. The sediments containing the entire benthic community, including benthic foraminifera, brought from the field were incubated at 27°C and ambient air oxygen concentration (20%, dissolved oxygen ~5.0 mL/L) for two weeks before starting the experiment to acclimatise the specimens.
The location of Dias Beach, off Goa, on the western coast of India. The seaweeds attached to the rocky patches and sediments were collected by diving in the inter-tidal and subtidal region, to retrieve the living benthic foraminifera for the experiment. Source: Google Earth.

The seawater brought from the field was filtered at 0.4 µm by using a Millipore vacuum pump. The filtered seawater was transferred to five pre-leached, washed and dried wide-mouth 2,000-mL glass reagent bottles. The bottles were rinsed three to four times with filtered seawater. After rinsing, 2,000 mL of the filtered seawater was filled in each glass stock bottle. The bottles with seawater were placed in four different multi-gas incubators (Sanyo MCO 19M) with different concentrations of O2 (1%, 5%, 10% and 15%). The reported oxygen controllability was ±0.2%. The desired air oxygen concentrations were obtained by purging the air inside the incubators with nitrogen. The circulating system in the experiment was maintained with the help of double-headed Cole-Parmer peristaltic pumps. One bottle was kept in a cooling incubator (MIR 154) as the control set (ambient oxygen, ~20%). The dissolved oxygen concentration in the seawater in different incubators was measured by the Winkler titration method. The water for dissolved oxygen measurement was taken with the help of a syringe attached to a tube inserted in the stock bottles. The dissolved oxygen was titrated with Metrohm 865 Dosimat. The average dissolved oxygen concentration in the seawater media at these air oxygen concentrations was 1.67, 2.04, 2.91, 4.35 and 5.01 mL/L, respectively.
The sediments brought from the field were homogenised and split into 12 aliquots by using a Folsom splitter. One aliquot was stained with rose-Bengal ethanol solution (2 g rose-Bengal in 1 L ethanol) to determine the living foraminifera at the time of the start of the experiment. Another aliquot was used for grain size and organic carbon analysis. The remaining ten aliquots were transferred to a 500-mL Büchner flask with a sidearm. The conical flasks were sealed with corks wrapped in cling film. The water in the conical flask was continuously pumped from the 2,000 mL glass stock bottle, with the help of a Masterflex L/S peristaltic pump, by inserting a tube through the cork. The water overflowing through the side arm drained back to the stock bottle, thus creating a re-circulating system. The water in the 2000-mL stock bottle was continuously aerated to equilibrate the dissolved oxygen with the modified ambient air (with the desired oxygen concentration) in the incubator (Figure 2).
The setup of the experiment to understand the community-level response of the living tropical shallow-water benthic foraminiferal community to different levels of dissolved oxygen. The replicate sets of the benthic foraminiferal community were kept in a 500-mL Büchner flask with a sidearm on the upper rack. The flasks were sealed, except for the inlet for the inflowing seawater. The modulated seawater media with different dissolved oxygen concentrations was pumped into the flasks from the stock bottle with the help of a peristaltic pump kept at the lower rack. The overflowing media was collected in the stock bottle, thus maintaining a circulating system.

All the incubators were maintained at 27°C temperature, 35 psu salinity and a 12-hour light and 12-hour dark cycle throughout the experiment. The seawater salinity, pH and dissolved oxygen were measured after transferring the sediment samples to the flasks. These parameters were also measured every fourth week. The measurement frequency was optimised to avoid frequent changes in the incubation conditions. The incubator oxygen concentration rapidly increased upon opening the door for sampling, and thus it also affected the dissolved oxygen concentration in the culture media. The pH was measured in the conical flasks that contained sediments. The freeze-dried in-house cultured diatom, Navicula, was fed once a week to the foraminifera. The experiment was conducted in a replicate for about four months. At the end of the experiment, the sediments were split into two parts. For splitting, excess overlying water was carefully removed, and the sediments were gently stirred and split into two aliquots by a Folsom splitter. One part of the sediments was stained with rose-Bengal ethanol solution, and the other part was stored for grain size and organic carbon analysis. For the grain size analysis, the sediments were freeze-dried. The completely freeze-dried sediments were carefully weighed. The dried and weighed sediments were then soaked and subsequently wet sieved by using a 63 µm sieve. The fraction retained on the sieve (>63 µm, Coarse Fraction, CF) was dried, weighed and stored in vials. A small aliquot of the original freeze-dried sediment was finely powdered for carbon analysis. The total carbon was measured using a Thermo Fisher Scientific CN analyser, and the inorganic carbon was measured by a UIC CM 5015 coulometer. The organic carbon was estimated by subtracting the inorganic carbon from the total carbon.
Processing and Picking of Samples
The wet sediments from each experimental set were poured over a 63 μm mesh and washed with gentle water pressure. The coarse fraction (>63 μm) retained on the mesh was transferred into pre-weighed beakers and dried. The dried, coarse fraction was weighed and stored in neatly labelled plastic vials. After conning and quartering, a representative coarse fraction was weighed and used for picking. A minimum of 300 specimens (including both the dead and rose-Bengal stained living benthic foraminifera) were picked from each sample with the help of an Olympus SZX16 stereo zoom microscope. Thus, benthic foraminifera were picked from a total of 22 samples. The total number of foraminifera (TFN) was standardised to 1 g of dry sediment to compare different samples. A t-test for independent variables was performed to test the significant difference between the control and the experimental sets (p values <0.05 were considered significant). The t-test helps in evaluating whether the difference between the means of replicates of different experimental setups is statistically significant. The test morphology (including the number and orientation of the chambers, aperture, suture and periphery) was used to identify the specimens. The generic-level identification was done with the help of Loeblich and Tappan (1988). The species were confirmed by using the previous literature and the Ellis and Messina Foraminifera Catalogue (EMFORCAT).
Results
Physico-chemical Parameters
The dissolved oxygen concentration in the seawater media was measured at the respective oxygen concentration in the incubator air (Table 1). The dissolved oxygen concentration in the media co-varied with ambient air oxygen (Figure 3(A)). The linear relationship between ambient air and dissolved oxygen concentration was confirmed with the t-test. The dissolved oxygen concentration varied from 1.67 mL/L at 1% to 5.01 mL/L at a 20% air oxygen concentration. The purging with nitrogen can also affect the concentration of gases other than oxygen in the ambient air and, thus, the gases dissolved in seawater, as well. An important gas for the carbonate system is carbon dioxide. However, we observed a decrease in pH with decreasing oxygen concentration (increased purging), which suggests that the CO2 system was not significantly affected by the purging with nitrogen. The pH decreased from 7.98 to 7.78, with a drop in oxygen percentage from ambient air concentration to 1% (Figure 3(B)). The coarse fraction in different experiment sets varied within a narrow range (81–83%), without any trend (Figure 4(A)). The organic carbon, however, significantly increased (R = 0.73) with decreasing oxygen concentration (Figure 4(B)). The salinity was maintained at 35 ± 0.5 psu.
The details of the ambient dissolved oxygen concentration and salinity in different experimental setups at various intervals of the experiment.
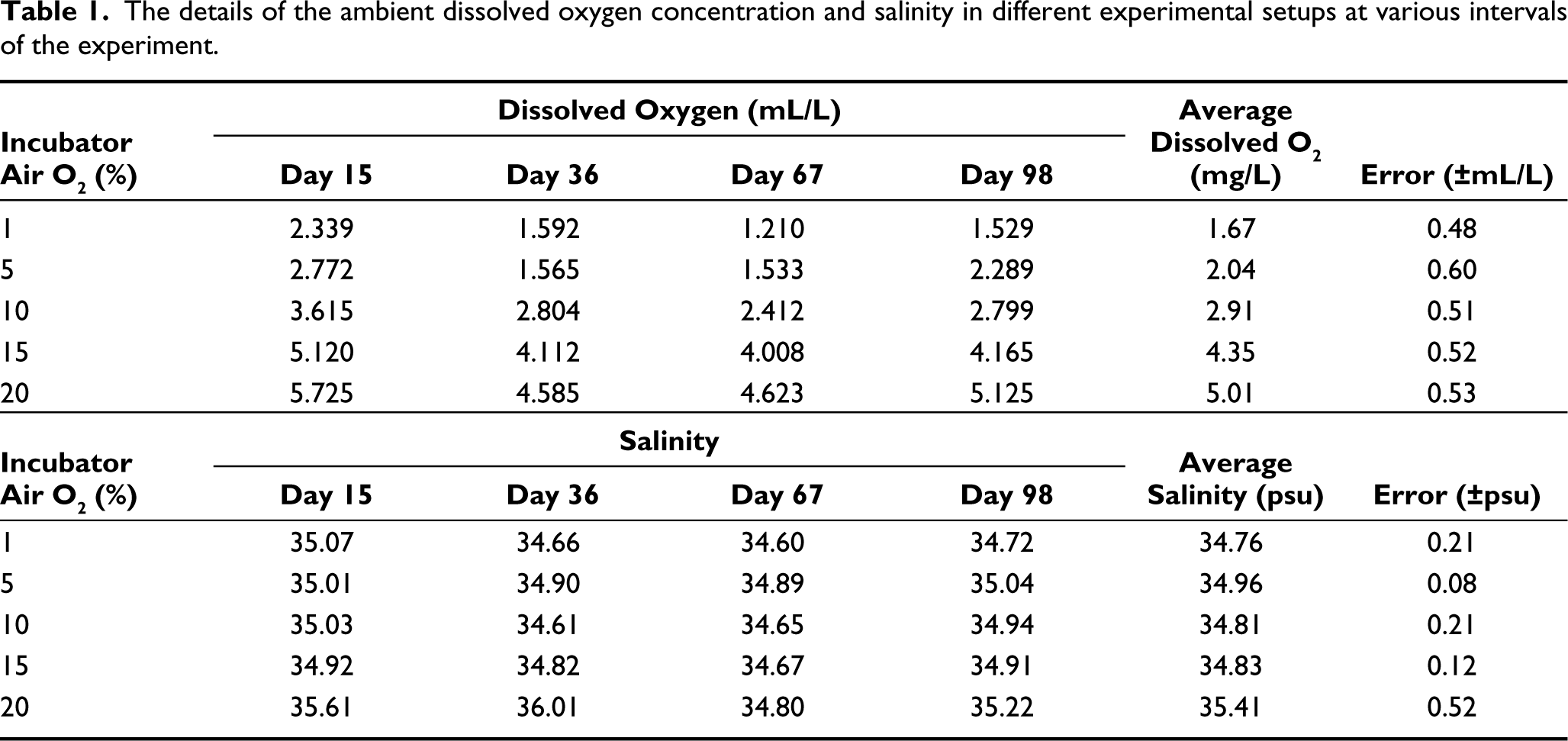
Seawater properties at different oxygen concentrations; (A) Dissolved oxygen concentration in the seawater media, and (B) pH of the seawater media at different percentages of oxygen in the air inside the incubators.
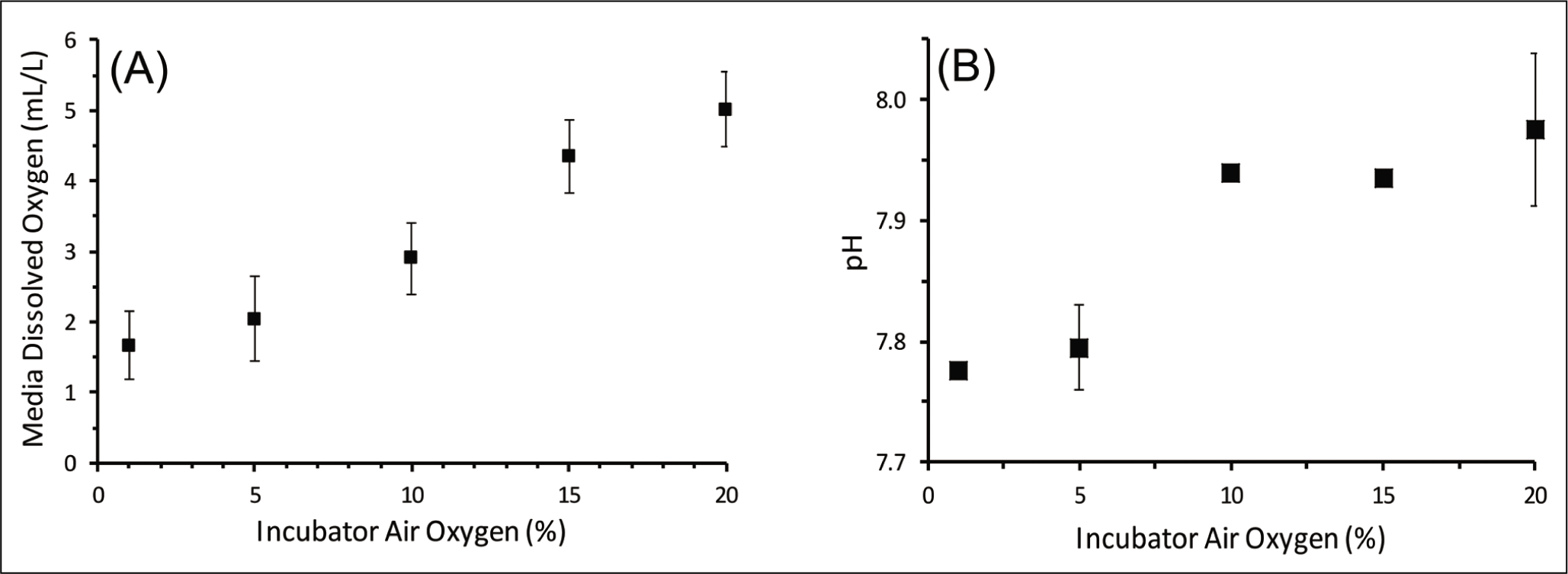
Sediment properties at different oxygen concentrations; (A) Coarse fraction (%) and (B) organic carbon (Corg) in sediments at different percentages of oxygen in the air inside the incubator.
Dead and Living (Rose-Bengal Stained) Benthic Foraminifera
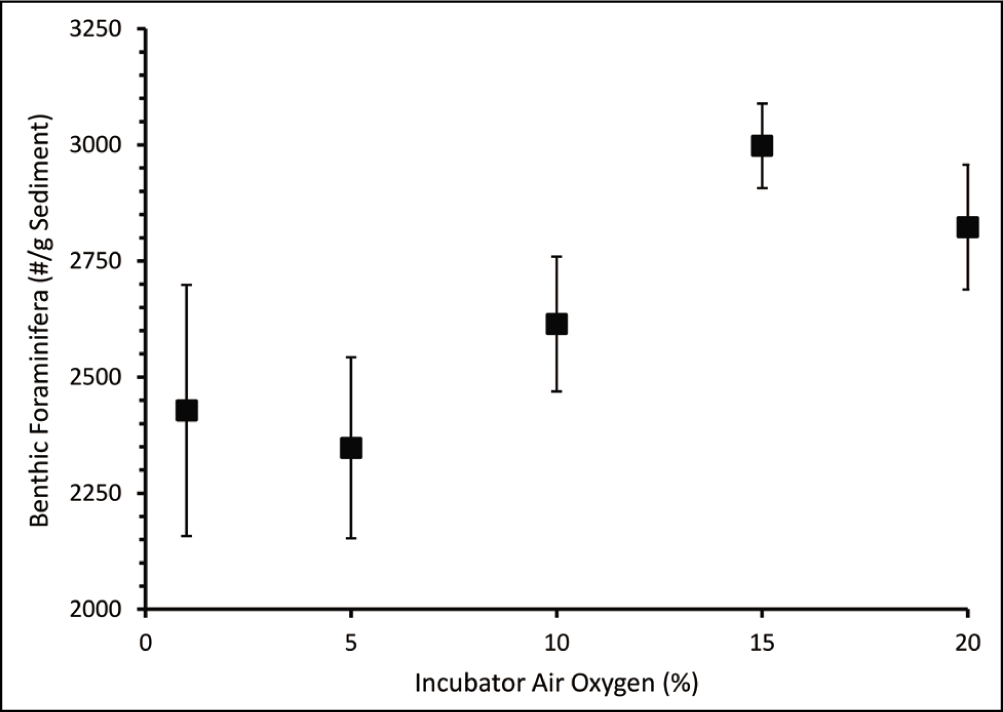
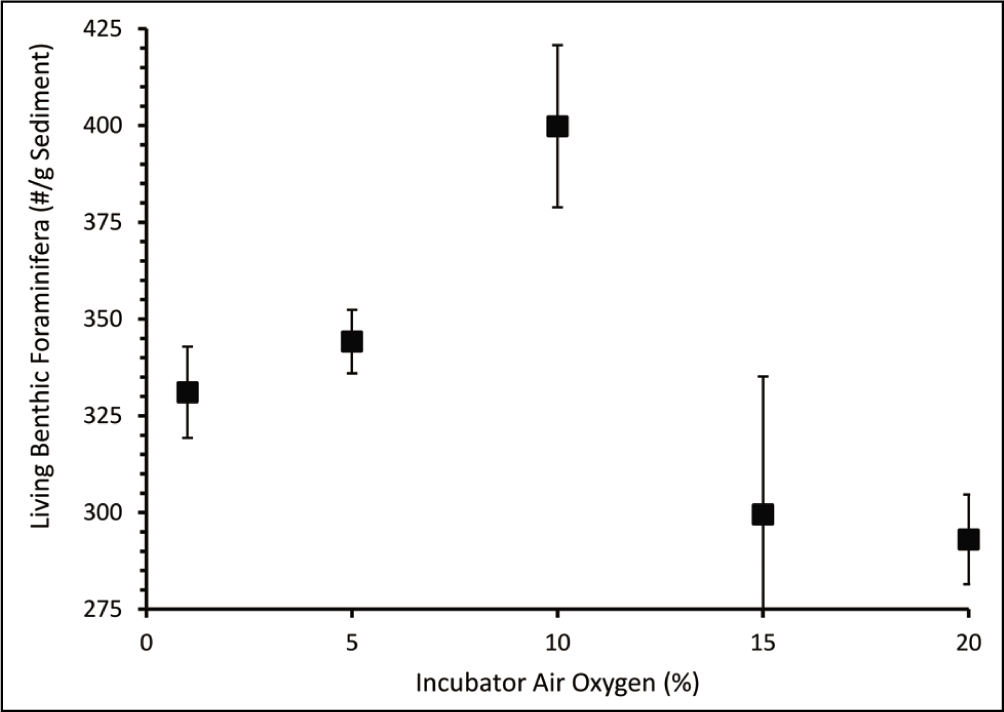
The absolute abundance of dead benthic foraminifera varied from a minimum of 2,348 individual/g sediment at 2.04 mL/L dissolved oxygen concentration to a maximum of 2,998 individual/g sediment at 4.35 mL/L dissolved oxygen concentration (Table 2). The dead benthic foraminiferal abundance decreased with decreasing dissolved oxygen concentration (Figure 5). The living benthic foraminifera were an order of magnitude lower than the dead benthic foraminifera (Figure 6). The living benthic foraminifera were abundant at 2.91 mL/L oxygen (400 individual/g sediments) and the lowest in the control set (293 individual/g sediments). The living benthic foraminifera population increased from 1.67 mL/L to 2.91 mL/L in oxygen concentration. However, a decreasing trend was observed in sets subjected to 4.35 mL/L and 5.01 mL/L oxygen concentration (Figure 6). A significant variation from the t-test for independent variables was seen at 1%, 5% and 10% air oxygen, compared to the control set. The living benthic foraminiferal abundance in the field samples was 696 individual/g sediment.
The coarse fraction (>63 µm) abundance and absolute abundance of total and living benthic foraminifera picked from each experimental setup.
Absolute abundance of benthic foraminifera (individual/g sediment) at different oxygen concentrations.
Absolute abundance of living benthic foraminifera (individual/g sediment) at different oxygen concentrations.
(A) Living benthic foraminifera found in the sediments used for the experiment. 1. Murrayinella murrayi: a—dorsal view; b—ventral view; 2. Murrayinella nicaraguaensis: a—dorsal view; b—ventral view; 3. Cornuspira involvens; 4. Pararotalia nipponica: a—dorsal view; b—ventral; 5. Pararotalia sp.; 6. Cornuspira sp.; 7. Elphidium crispum; 8. Ammonia tepida: a—dorsal view; b—ventral view; 9. Ammonia sobrina: a—dorsal view; b—ventral view; 10. Elphidium advena; 11. Rosalina leei: a—dorsal view; b—ventral view; 12. Rosalina globularis: a—dorsal view; b—ventral view; 13. Fissurina multicostata; 14. Rosalina sp: a—dorsal view; b—ventral view; 15. Cymbaloporetta plana: a—dorsal view; b—ventral view. The scale bar is 100 µm, unless stated otherwise.
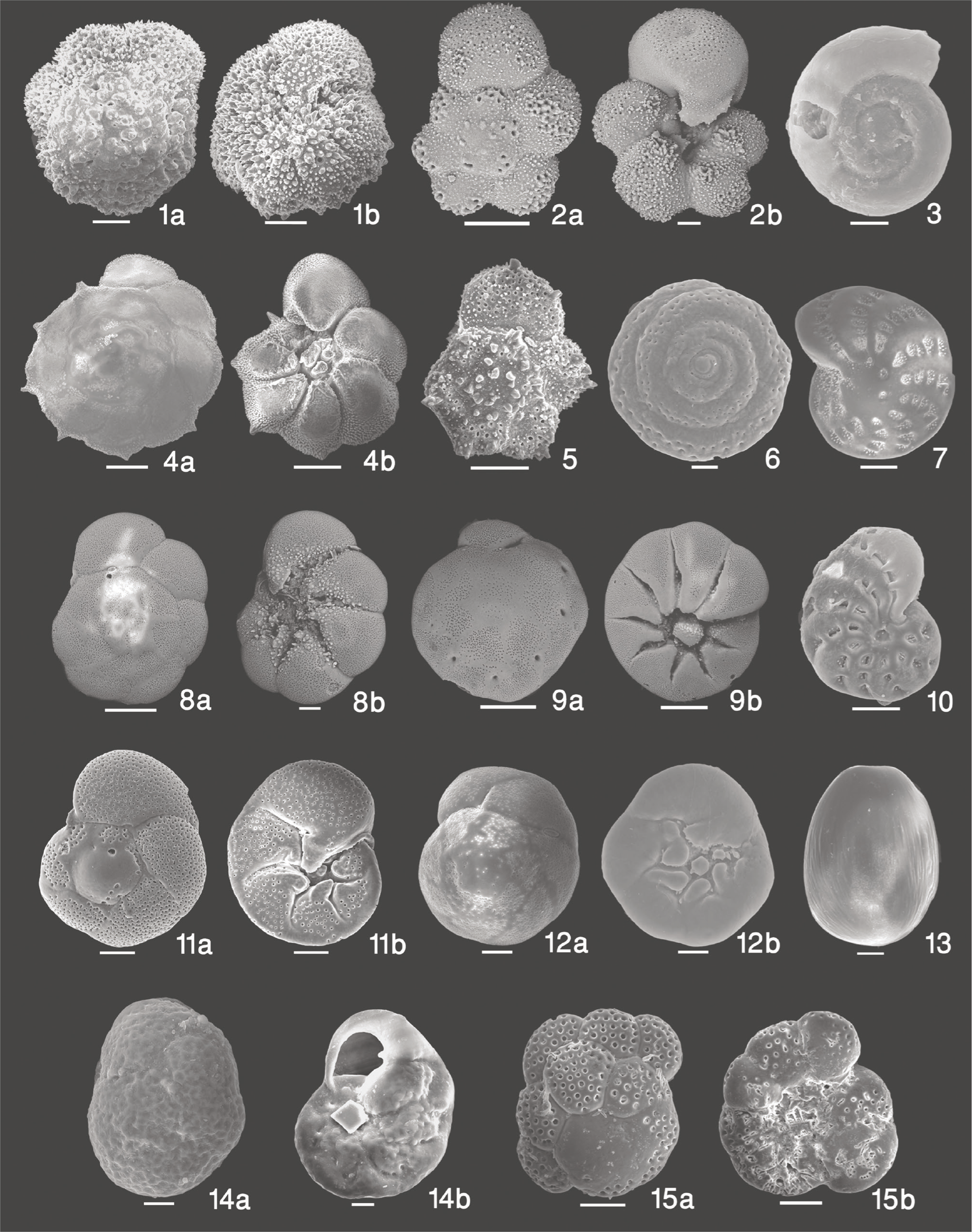
(B) 1. Cibicides refulgens: a—dorsal view; b—ventral view; 2. Eponides repandus: a—dorsal view; b—ventral view; 3. Quinqueloculina parkeri; 4. Quinqueloculina seminulum; 5. Quinqueloculina oblonga; 6. Quinqueloculina sp.; 7. Bolivina striatula; 8. Bolivina dilatata; 9. Bolivina subspinescens; 10. Bolivina sp.

Relative Abundance of Benthic Foraminifera
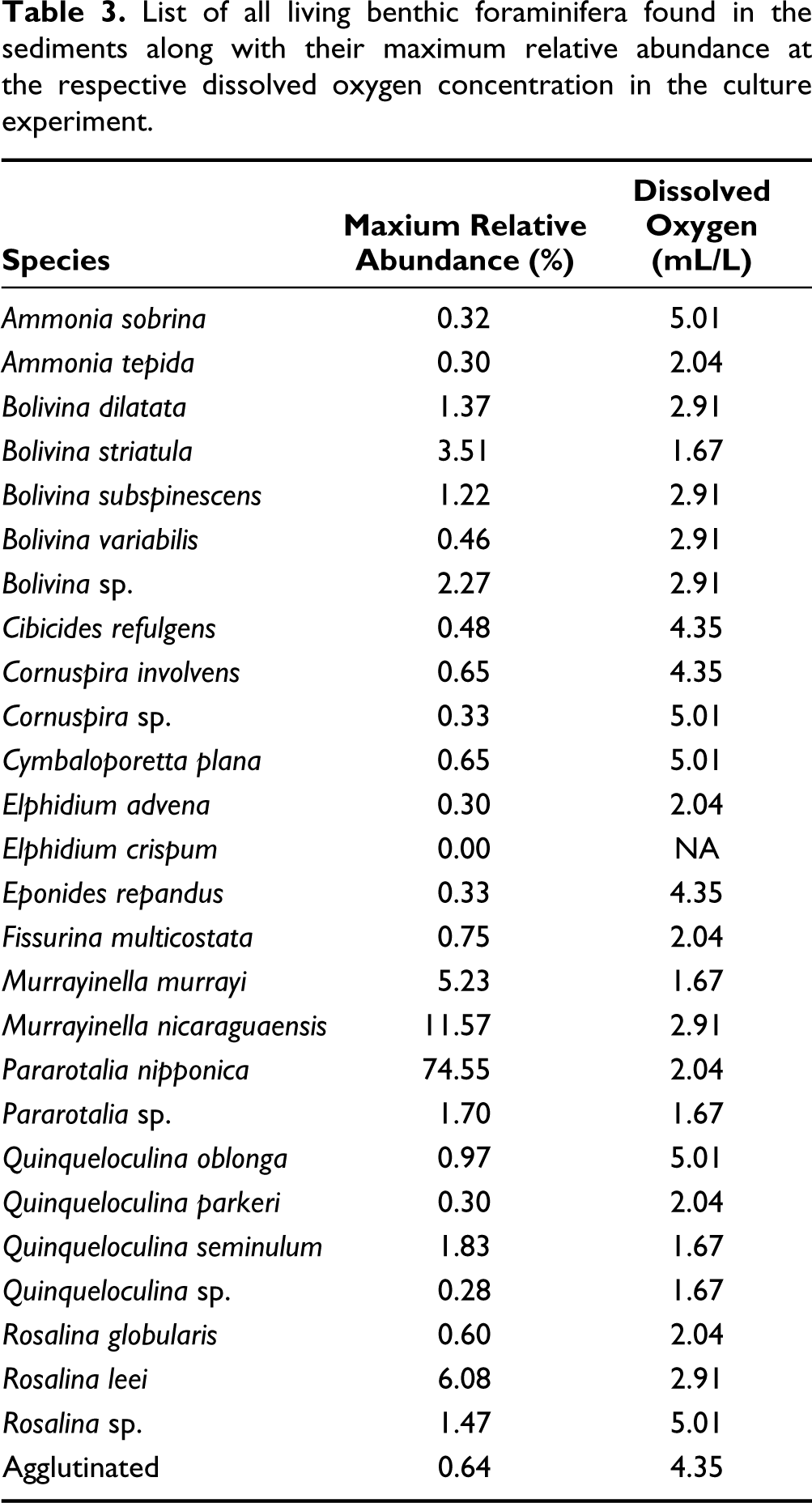
The living benthic foraminifera found in the sediments belong to 12 genera, namely Ammonia, Bolivina, Cibicides, Cornuspira, Cymbaloporetta, Elphidium, Eponides, Fissurina, Murrayinella, Pararotalia, Rosalina and Quinqueloculina. The agglutinated specimens could not be identified. A majority of these genera are widely reported from the shallow-water inner shelf regions. A total of 26 species (Ammonia tepida, Ammonia sobrina, Bolivina variabilis, Bolivina striatula, Bolivina subspinescens, Bolivina dilatata, Bolivina sp., Cibicides refulgens, Cornuspira involvens, Cornuspira sp., Cymbaloporetta plana, Elphidium advena, Elphidium crispum, Eponides repandus, Fissurina multicostata, Murrayinella murrayi, Murrayinella nicaraguaensis, Pararotalia sp., Pararotalia nipponica, Quinqueloculina seminulum, Quinqueloculina parkeri, Quinqueloculina oblonga, Quinqueloculina sp., Rosalina leei, Rosalina globularis, Rosalina sp.) of calcareous living benthic foraminifera, other than agglutinated ones, were found (Table 3; Plate 1A and 1B). As the agglutinated specimens were extremely rare, they were all clubbed together. Thus, out of these 27 species (26 calcareous and other agglutinated), Pararotalia nipponica constituted more than half of the total living benthic foraminifera at all dissolved oxygen concentrations, as it was naturally abundant in the sediments at the sampling site. The relative abundance of P. nipponica varied from 65.5% at 2.91 mL/L dissolved oxygen to 74.5% at 2.0 mL/L dissolved oxygen concentration. M. nicaraguaensis was the second most dominant living benthic foraminifera, with the relative abundance varying from 6.7% at the highest dissolved oxygen concentration to 11.6% at 2.91 mL/L dissolved oxygen.
List of all living benthic foraminifera found in the sediments along with their maximum relative abundance at the respective dissolved oxygen concentration in the culture experiment.
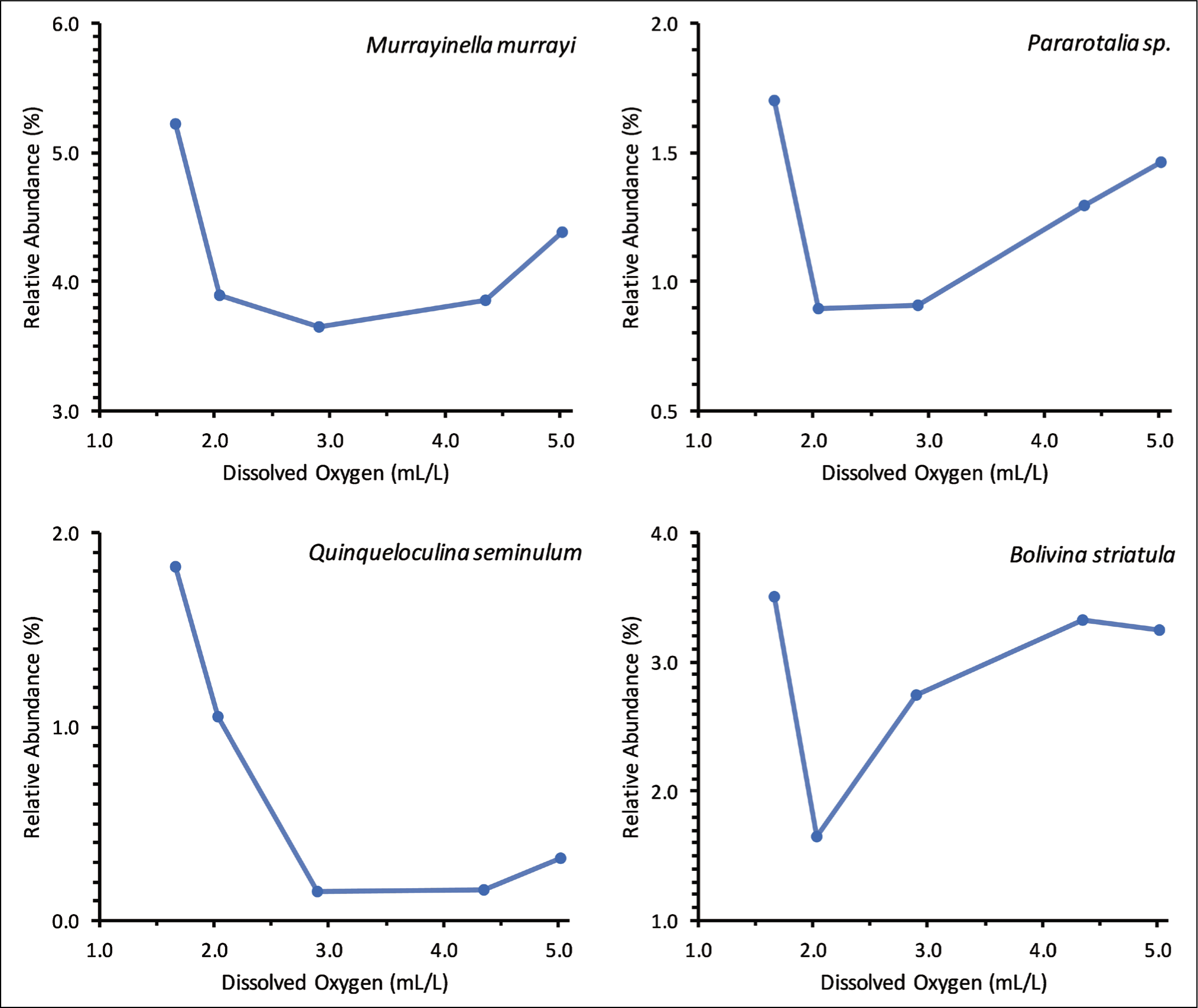
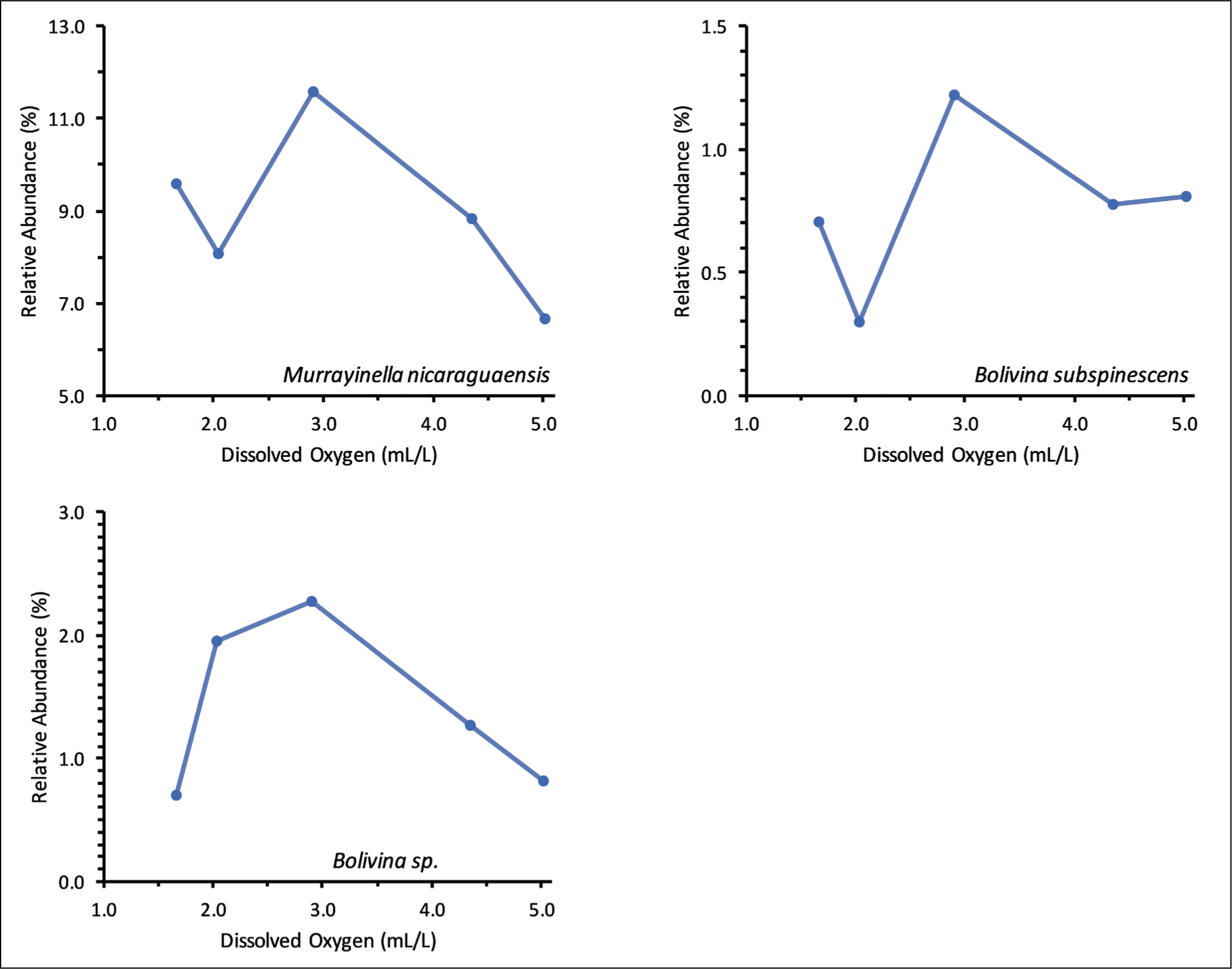
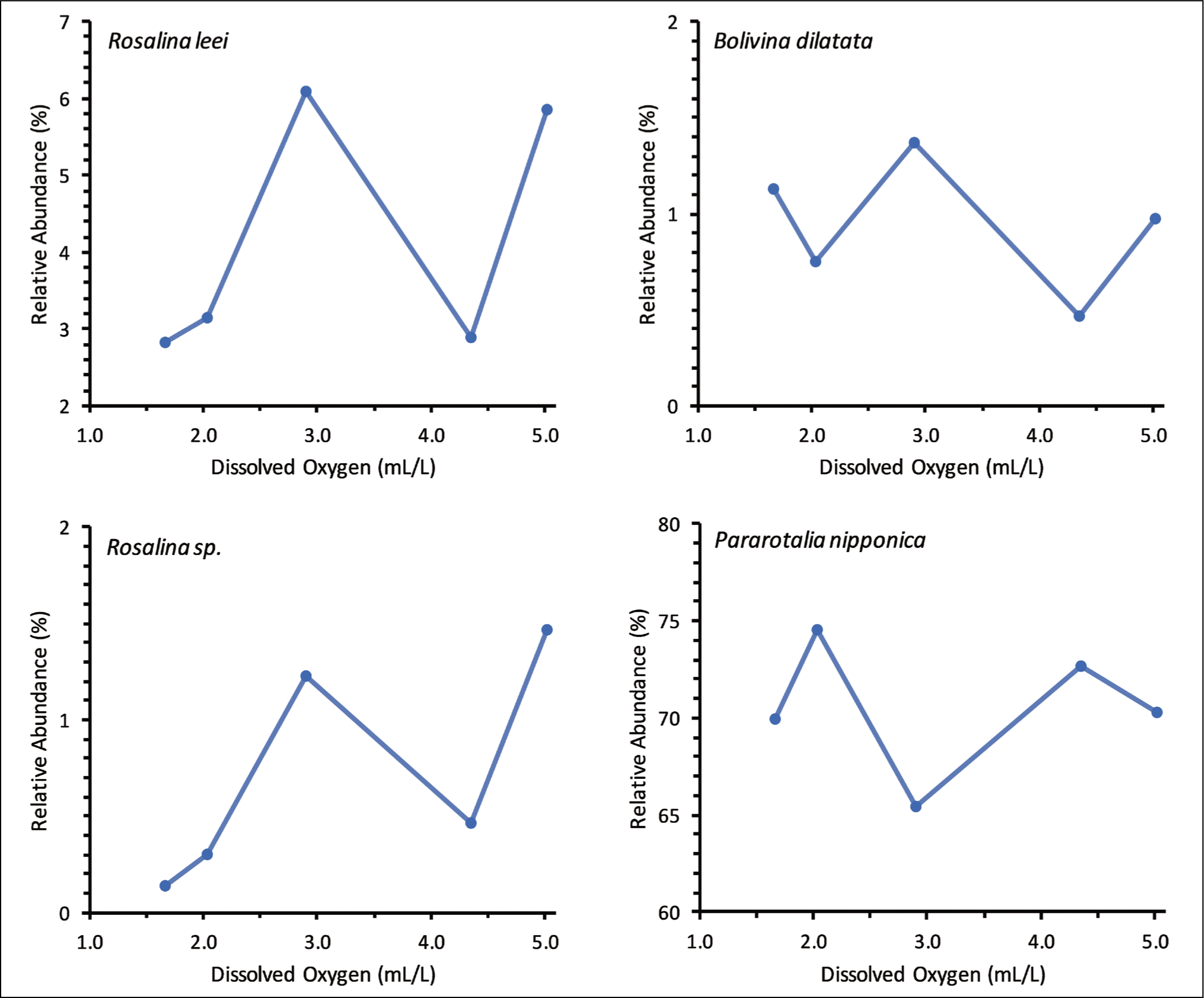
Three different patterns of benthic foraminiferal responses to different concentrations of dissolved oxygen were observed. The relative abundance of B. striatula, M. murrayi, Q. seminulum and Pararotalia sp. decreased with the initial decrease in dissolved oxygen concentration but peaked at the lowest dissolved oxygen concentration (Figure 7). The lowest relative abundance of all these species was at 2.04 mL/L or 2.91 mL/L dissolved oxygen concentration. In contrast to these species, the relative abundance of three living benthic foraminifera, namely M. nicaraguaensis, Bolivina subspinescens and Bolivina sp., was the maximum at the intermediate dissolved oxygen concentration (2.91 mL/L) (Figure 8). A few other dominant species, P. nipponica, Rosalina leei, Rosalina sp. and Bolivina dilatata, did not have any definite trend with different dissolved oxygen concentrations (Figure 9).
Relative abundance of Bolivina striatula, Murrayinella murrayi, Quinqueloculina seminulum and Prarotalia sp. decreased with the initial decrease in dissolved oxygen concentration but peaked at the lowest dissolved oxygen concentration.
Relative abundance of three living benthic foraminifera, namely Murrayinella nicaraguaensis, Bolivina subspinescens and Bolivina sp. was the maximum at the intermediate dissolved oxygen concentration (2.91 mL/L).
Relative abundance of Rosalina leei, Rosalina sp., Bolivina dilatata and Pararotalia nipponica does not show any definite trend with different dissolved oxygen concentrations.
Variation Within the Replicate Sets
The variation in dead benthic foraminiferal abundance was large within the replicates subjected to the same oxygen concentration. However, the response of living benthic foraminifera was more consistent with limited variability in the replicates at the majority of oxygen concentrations. The variation was also very low in the organic carbon content of the replicate samples subjected to the same oxygen concentration. It is worth noting here that the organic carbon content was very low in the sediments. The difference in the coarse fraction abundance within the replicates of 5%–15% air oxygen concentration (<5%) was also well within the acceptable limits.
Discussion
Benthic Foraminiferal Abundance at Different Oxygen Concentrations
The difference in both the living and dead foraminiferal abundance at different oxygen concentrations suggests a strong influence of experimental conditions on foraminiferal ecology. Kaiho (1994) proposed five categories depending on the oxygen concentration, namely anoxic (0–0.1 mL/L), dysoxic (0.1–0.3 mL/L), suboxic (0.3–1.5 mL/L), low oxic (1.5–3.0 mL/L) and high oxic (3.0–6.0 mL/L). Therefore, the experimental sets ranged from low-oxic to high- oxic conditions (1.67–5.01 mL/L). The distribution and abundance of benthic foraminifera in shallow water regions are influenced by the grain size, temperature, salinity and organic matter (Bouchet et al., 2009; Dong et al., 2019; Lei et al., 2014; Mendes et al., 2004; Murray, 2006; Scott et al., 2001). The grain size, temperature and salinity were similar for all the sets, thus ruling out any differential influence of these parameters on living benthic foraminifera in the experiment. The estimation of benthic foraminiferal abundance in sediments is strongly influenced by the coarse fraction percentage (Suokhrie et al., 2021). The coarse fraction abundance in the field changes with the terrestrial supply. However, here we worked in a controlled laboratory setup, and therefore there were no factors that could significantly change the coarse fraction percentage. The coarse fraction percentage at all setups was comparable (varying between a narrow range of ~81–84%), without any systematic trend. Therefore, the difference in benthic foraminiferal abundance at different oxygen levels is not influenced by the coarse fraction. The seawater salinity in all the sets was in the range of 34.5–35.5 psu. Therefore, any change in the size, loss of ornamentation, or dissolution of the foraminifera test because of salinity changes (Boltovskoy et al., 1991; Kurtarkar et al., 2011) were ruled out.
The increased pH from the experimental set to the control set was expected to significantly influence benthic foraminiferal assemblage, as low pH is detrimental to foraminiferal tests (Saraswat et al., 2015). A clear decrease in the absolute abundance of benthic foraminifera (dead + living) is attributed to a drop in pH with decreasing dissolved oxygen concentration. The thin-walled, fragile benthic foraminifera tests are highly susceptible to dissolution in low-pH seawater. Thus, decreasing dissolved oxygen concentration will result in poor preservation of dead benthic foraminifera tests due to a drop in pH.
The abundance of both the dead and living benthic foraminifera was similar at the two lowest dissolved oxygen concentrations. The similar foraminiferal abundance at the two lowest oxygen concentrations is attributed to the small difference in dissolved oxygen (0.37 mL/L) at the lowest two setups. The foraminifera abundance at 1% and 5% air oxygen, although lower than that at 10% air oxygen, was higher than that at 15% and 20% air oxygen. Interestingly, the lowest abundance of living benthic foraminifera was at the highest oxygen concentration. This non-linear response of living benthic foraminiferal abundance is intriguing. The salinity and temperature were similar and within the error limits in all the sets and thus are not the factors influencing the observed patterns in living benthic foraminifera. The low foraminiferal abundance at the highest oxygen concentration is attributed to the difference in organic matter and predatory pressure (Figure 4(B)). Increased benthic foraminiferal abundance in a region is often attributed to the high organic carbon content of the sediments (Cappelli et al., 2019; Chaturvedi et al., 2000; Singh et al., 2021). The prevalence of macrobenthic communities also adversely affects benthic foraminifera by inducing high predatory pressure (Enge et al., 2016; Levin, 2003). We did observe bigger vagile ciliates and therefore it is likely that the predatory pressure by macrofauna also affected the benthic foraminifera. The dissolved oxygen at the two highest concentrations was sufficient (high oxic) to support the meiobenthic and microbenthic communities. The dissolved oxygen concentration at 10% air oxygen dropped to low oxic levels. The low oxic concentration of dissolved oxygen is detrimental to the macrobenthic community, thus leading to its decreased population and reduced predatory pressure. With this argument, the living benthic population should peak at the lowest dissolved oxygen levels. However, the living benthic foraminiferal abundance decreased at the two lowest oxygen concentrations. The decline in the living benthic foraminiferal population is attributed to the inability of the epifaunal benthic foraminifera to survive under low oxic and suboxic conditions. Unlike the previous observation that benthic foraminiferal abundance is independent of oxygen once it increases beyond 1.0 mL/L (Murray, 2001), here we report that the low and high oxic conditions also affect benthic foraminifera.
Effect of Oxygen on Benthic Foraminiferal Diversity
The living benthic foraminiferal population was dominated by epifaunal P. nipponica. The other abundant species were M. murayi, M. nicaraguaensis, Pararotalia sp. and B. striatula. Pararotalia nipponica is a robust species and was present at almost all oxygen concentrations with >60% abundance. The epifaunal foraminifera are adapted to well-oxygenated waters. Kumar and Manivannan (2001) reported the optimum oxygen concentration for P. nipponica to be 2.8–3.6 mL/L. The drop in oxygen to suboxic levels (0.3–1.5 mL/L) adversely affected P. nipponica, leading to higher mortality, and thus, an overall low-living benthic foraminiferal population. In marine oxygen-deficient regions, the population of infaunal forms increases significantly (Venturelli et al., 2018). However, such a significant increase in the infaunal benthic foraminifera was not observed in this experiment. The reason for this difference is that the infaunal foraminifera were rare in the original shallow inter-tidal marine benthic foraminiferal community. Therefore, the overall response of benthic foraminifera to different oxygen concentrations will depend on the community structure, especially the abundance of infaunal and epifaunal foraminifera. As robust epifaunal species dominate the shallow inter-tidal marine benthic foraminiferal community, the initial impact of low dissolved oxygen will be minimal unless the levels decrease to a drastically low concentration.
Only those species, with a relative abundance of ≥1%, are discussed in detail. Based on their relative abundance, species were categorised into a few distinct groups. One group is comprised of the species with the maximum relative abundance at the lowest dissolved oxygen concentration. Although the relative abundance of B. striatula, M. murrayi, Q. seminulum, and Pararotalia sp. initially decreased with the depleting oxygen concentration, their highest relative abundance was at the lowest dissolved oxygen concentration (Figure 7). Bolivina is infaunal–epifaunal and abundant in muddy marine sediments on the inner (0–100 m) and outer shelves (100–200 m) (Murray, 1991). The Bolivina species prefer low light (Kitazato, 1981). The specimens of Murrayinella are common in shallow waters (Nomura & Takayaniagi, 2000). Incidentally, the relative abundance of these species peaked at the lowest dissolved oxygen concentration. The relative abundance pattern suggests either that the species belonging to this group prefer an oxygen-deficient environment or the inability of other species to thrive at the lowest dissolved oxygen concentration. The severely detrimental effect of low dissolved oxygen on other species resulted in the dominance of B. striatula, M. murrayi, Q. seminulum and Pararotalia sp., although the overall population of these species also declined.
Another group includes the species with the maximum relative abundance at the medium dissolved oxygen concentration. The relative abundance of three living benthic foraminifera, namely M. nicaraguaensis, Bolivina subspinescens and Bolivina sp., increased with the decrease in dissolved oxygen concentration from 5.01 mL/L to 2.91 mL/L (Figure 8). Subsequently, the relative abundance decreased with a further drop in dissolved oxygen to 1.67 mL/L. The increase in the relative abundance of M. nicaraguaensis, B. subspinescens and Bolivina sp. with the initial decrease in dissolved oxygen concentration is attributed to reduced bioturbation due to declined macrobenthic activity. The subsequent drop in relative abundance suggests that M. nicaraguaensis, B. subspinescens and Bolivina sp. do not prefer dissolved oxygen concentration ≤2.04 mL/L. No apparent trend was observed in the relative abundance of Rosalina leei, Rosalina sp., Bolivina dilatata and Pararotalia nipponica at different dissolved oxygen concentrations (Figure 9). Rosalina specimens are epifaunal, mostly clinging to or attached to hard substrates. They are both herbivores and omnivores, inhabiting temperate to warm marine waters on the inner shelf (Murray, 1991). Pararotalia nipponica is the most dominant species at all dissolved oxygen concentrations. Pararotalia is epifaunal and mostly found in very fine sand in the inner-shelf region (Murray, 1991). Pararotalia can survive in temperatures ranging from 22°C to 29°C. It is interesting to note that different species belonging to the same genus had different responses to various dissolved oxygen concentrations. Thus, the benthic foraminiferal response to dissolved oxygen is species-specific.
Conclusion
We assessed the response of the shallow inter-tidal benthic foraminiferal community to different oxygen concentrations under controlled laboratory conditions. We report a non-linear response of the living benthic foraminiferal assemblage to different levels of oxygen concentrations. The pattern is attributed to the combined effect of the presence of macrobenthic community as well as the dominance of epifaunal benthic foraminifera in the population. We report that the change in seawater pH at different oxygen levels also strongly affects the abundance of benthic foraminifera. The dominance of epifaunal Pararotalia nipponica at all oxygen concentrations is attributed to its very high abundance in the original community as well as its resilience to a wide range of oxygen levels. The increase in the living benthic foraminiferal population up to a certain level of decrease in dissolved oxygen suggests an overall increased carbon sequestration by benthic foraminifera under low dissolved oxygen concentration. However, the response will be region-specific, primarily driven by the existing benthic foraminiferal diversity and community structure. The findings are useful to assess the response of the marginal marine benthic foraminiferal community to global warming-induced changes in dissolved oxygen concentration.
Footnotes
Acknowledgements
Authors thankfully acknowledge the Department of Science and Technology, Government of India for funding the WOS—A Project. The authors declare no conflict of interest. We thank the anonymous reviewers for their valuable comments and suggestions that helped to improve a previous version of this manuscript. Authors thank Dr Thejasino Suokhrie and Science and Engineering Research Board for the financial support (Sanction No. EEQ/2023/000526). It is the National Institute of Oceanography, India contribution number 7236.
Authors Contributions
RS designed the research; SRK and AK performed the research; SRK wrote the first draft and all authors contributed to the manuscript.
Availability of Data and Materials
Data have been included as tables in the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed the following financial support for the research, authorship and/or publication of this article: Department of Science and Technology, Government of India.
