Abstract
Benthic foraminiferal assemblages are widely used as a proxy to monitor the coastal environment’s health. The current work involves the seasonal distribution of foraminifera in the outer channel sector of the Chilika Lagoon, Odisha during 2017–2018. The outer channel is marine as the sea mouth is nearby. Altogether 28 species of benthic foraminifera are identified, among which dominant are Ammonia spp. The other calcareous taxa are Elphidium spp, Quinqueloculina sp., Haynesina spp., Pararotalia sp., Hanzawaia sp., Nonionellina sp. and agglutinated forms such as Miliammina sp., Trochammina sp. and Textularina sp. are found in moderate abundance. Based on the pre-monsoon abundance, the cluster analyses show two distinct biofacies zones in the studied area. Total foraminiferal number (TFN) increases during pre-monsoon compared to post-monsoon months. A trend of decreasing abundance was observed in TFN, mainly calcareous taxa towards the inner part of the lagoon, that is, away from the sea mouth.
Introduction
Foraminifera, the shelled microorganism, dominate the modern ocean floor and are the most prolific benthic species well preserved in the sediment record (Gooday et al., 1992). They are eukaryotic unicellular microorganisms with a wide range of sizes, varying from <1 mm to 100 mm. As they have a short life span (from a few days up to one year, Murray, 1991; Nigam et al., 2003; Kurtarkar et al., 2019), they respond to small changes in the environment. Due to these characteristics, foraminifera is extensively used as a tool for monitoring the coastal environment along with different marginal environments like estuarine (Alve, 1995; Bhattacharjee et al., 2013; Luan & Debenay, 2005), marsh (Gehrels & Newman, 2004; Horton & Murray, 2007) and lagoonal environment (Martins et al., 2013; Samir, 2000).
The fluvial inputs and tidal variation with wave action highly impact the nearshore lagoons, which can affect the foraminiferal assemblages. Foraminiferal diversity can vary seasonally or periodically due to the monsoonal effects and tidal variation of the lagoon, respectively. These are the causes of low diversity accumulation of benthic foraminifera in the lagoonal environment. Only euryhaline and stress-tolerant species (Murray, 2007) dominate in these coastal waters.
Several researchers have studied the foraminiferal assemblages in different sectors of the Chilika Lagoon. Rao et al. (2000) reported 69 species from 15 sediment samples in the Chilika Lagoon. Kumar et al. (2014, 2015) identified 15 agglutinated forms in different sectors of the lagoon. Workers, such as Sen and Bhadury (2016), Sen et al. (2018) and Mishra et al. (2019), stated that the Chilika Lagoon has a low diverse foraminiferal assemblage and dominance of Ammonia sp. and agglutinated forms. The effect of low saline brackish water conditions due to the restricted inflow of seawater in the lagoon supports the dominance of Ammonia sp. (Barik et al., 2020). The total organic carbon, salinity and water depth are controlling factors for agglutinated forms (De & Gupta, 2010). Dasgupta and Ghosh (2021) identified four different Ammonia spp. and two morphotypes of Ammonia spp. Based on previous studies, a detailed investigation of foraminifera in the outer channel has yet to be carried out. The outer channel has the marine condition as sea water enters into this sector. This current study reports the seasonal foraminiferal distribution and its diversity from 45 sediment samples in the outer channel and presents the biofacies map of the area.
Study Area
Chilika is one of the large saltwater lagoons in Odisha, India. The coordinates of this lagoon are 19° 28′–19° 54′ N and 85° 05′–85° 38′ E (Figure 1). A 60 km long beach named Rajhansa isolates the lagoon from the adjacent Bay of Bengal. The lagoon is pear-shaped and has a width of 20.1 km and a length of 64.3 km. A ~32 km long zigzag passage connects the lagoon with the Bay of Bengal near a village named ‘Arakudha’ (Panigrahi et al., 2009) with a 100 m wide mouth, which is locally called the ‘Magarmukha’ (mouth of crocodile). It also connects with the sea by an artificial canal, ‘Palur Canal’ or ‘French Canal’, which links the southern sector of the lake and the Rushikulya estuary. The lagoon consists of numerous islands. Among them, six major islands constitute the Krishnaprasad revenue block of the Puri district. Also, the northern part of the lagoon is in the Khordha district, and the western part is in the Ganjam district, Chilika drainage basin and the lagoon covers 4,300 km2 of area (Das & Samal, 1988). Three hydrological sub-systems influence the lagoon, the northern areas are affected by the river Mahanadi, the western and southwestern parts are influenced by rivers and rivulets flowing from the Bhargavi and Rushikulya rivers, and the Bay of Bengal affects the eastern side (Ram et al., 1994). Apart from them, the lagoon also collects silt from the freshwater flow of 52 channels (Sahu et al., 2014).
(a) Chilika Lagoon and its four ecological sectors. (b, c) Locations of 45 sampling stations in outer channel (OC).
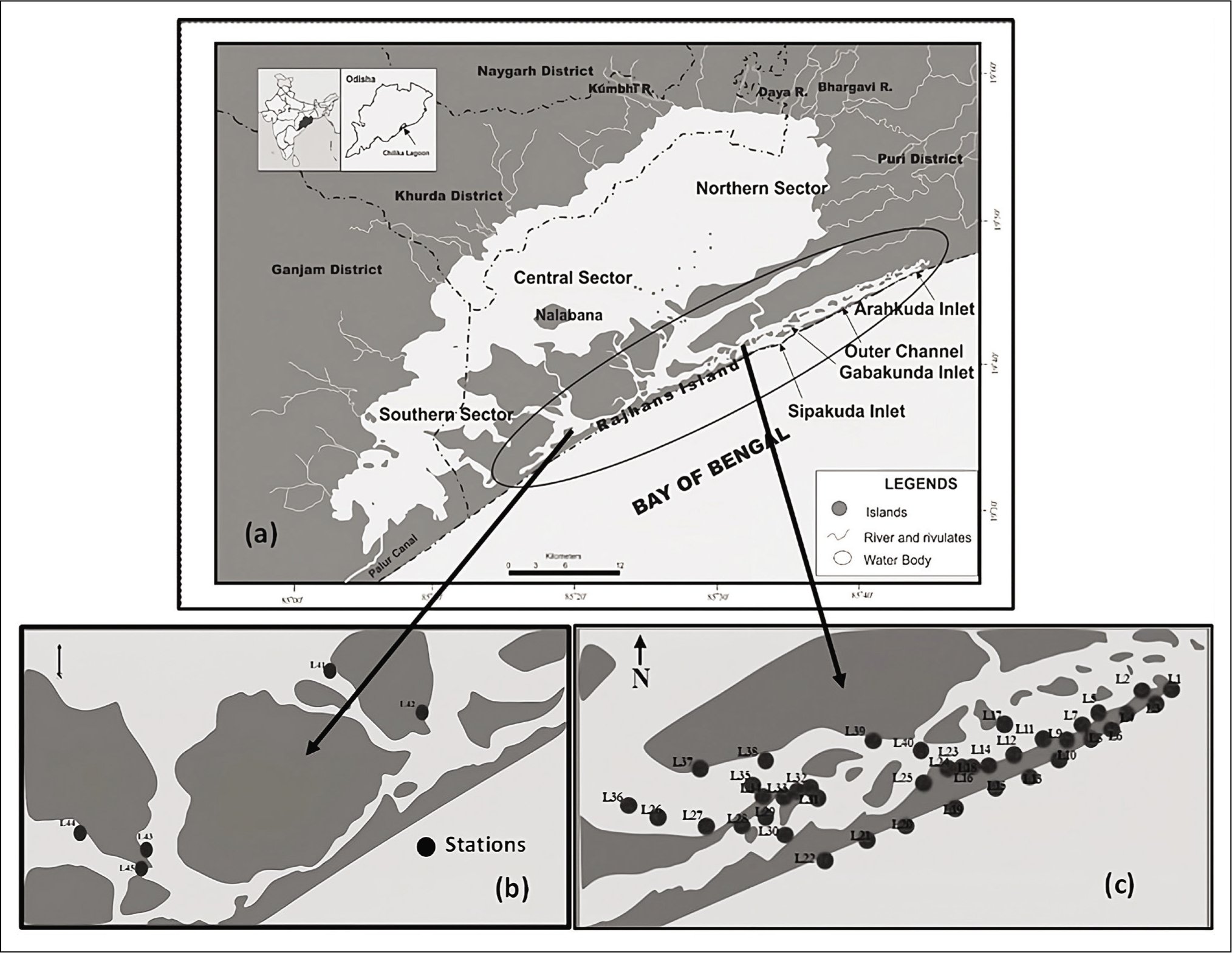
The overall water quality, depth and previously reported biological diversity divided the entire lake into four sectors, that is, the northern or freshwater zone, central or brackish zone, southern or marine and the outer channel, which is mainly marine in nature (Sahu et al., 2014). The depth of water at the outer channel (19° 39′–19° 41′ N; 85° 28′–85° 32′ E) is 1 m on average during summer but increases up to 2 m during monsoon (Rao et al., 2000). Three outlets also influence the sector. Two were naturally formed (2008 and 2012), and another was artificially created near Satapada (Sipakuda) in September 2000 (Sahu et al., 2014). The watershed area of the lagoon is 704 km2 during the summer (March to June) and 1020 km2 in the monsoon (Gupta et al., 2008), which was 905 km2 and 1165 km2 in the past (Annandale & Kemp, 1915). Southwestern monsoon (July to September) plays a critical role in changing the physical parameters of the lagoon. The freshwater flow from the rivers and rivulets and seawater intrusion through tides make an estuarine ecosystem in the lagoon. Due to high evaporation in the pre-monsoon months, the water body becomes shallow (Mohanty et al., 1996), while during monsoon and post-monsoon seasons, large freshwater comes from various rivers and canals because of precipitation, raising the water column of the lagoon. The process lowers the salinity of the water body (Jeong et al., 2008).
From July to September (Monsoon season), an average rainfall of 1240 mm was observed in the catchment area (Sahay et al., 2019). Panda and Mohanty (2008) reported a yearly influx of freshwater of about 5,090 million cubic meters (1999–2007) into the lagoon. The precipitation impacts the hydrography of the lagoon, and several rivers and rivulets contribute to the freshwater (Bhadury et al., 2020). During pre-monsoon, there is high evaporation and less freshwater influx from rivers and rivulets. The inflow of tide-propagated marine water through the mouth helps to increase the salinity in the outer channel (Barik et al., 2019). Maharana et al. (2019) reported that the salinity of this lagoon sector ranges between 23 PSU and 29 PSU, and the pH is between 8.5 and 8.7. The temperature recorded at the outer channel during the summer was 31.1oC, and in winter was 25oC, and the dissolved oxygen level varied between 6.5 and 8.5 mg/l (Maharana et al., 2019; Nayak et al., 2001). The sedimentation rate varies in different sectors of the Chilika due to the inflow from various rivers and rivulets. The outer channel shows low deposition due to its high energy flow of marine waters during the tides. Patnaik (1998) documented that the annual sedimentation rate was 1.8 million metric tons but rose to 2.94 million metric tons within three years (1998–2001), which was observed by Annon (2003).
Recently, the lagoon changed drastically due to the change in the width of the waterbody, lessening the depth due to deposition, reducing the salinity, macrophyte infestation, eutrophication and extinction of species (Ghosh et al., 2006; Mahapatro, 2013). The changing environment of the lagoon became a matter of concern to governments, policymakers and the local inhabitants (Mahapatro, 2013).
Materials and Methods
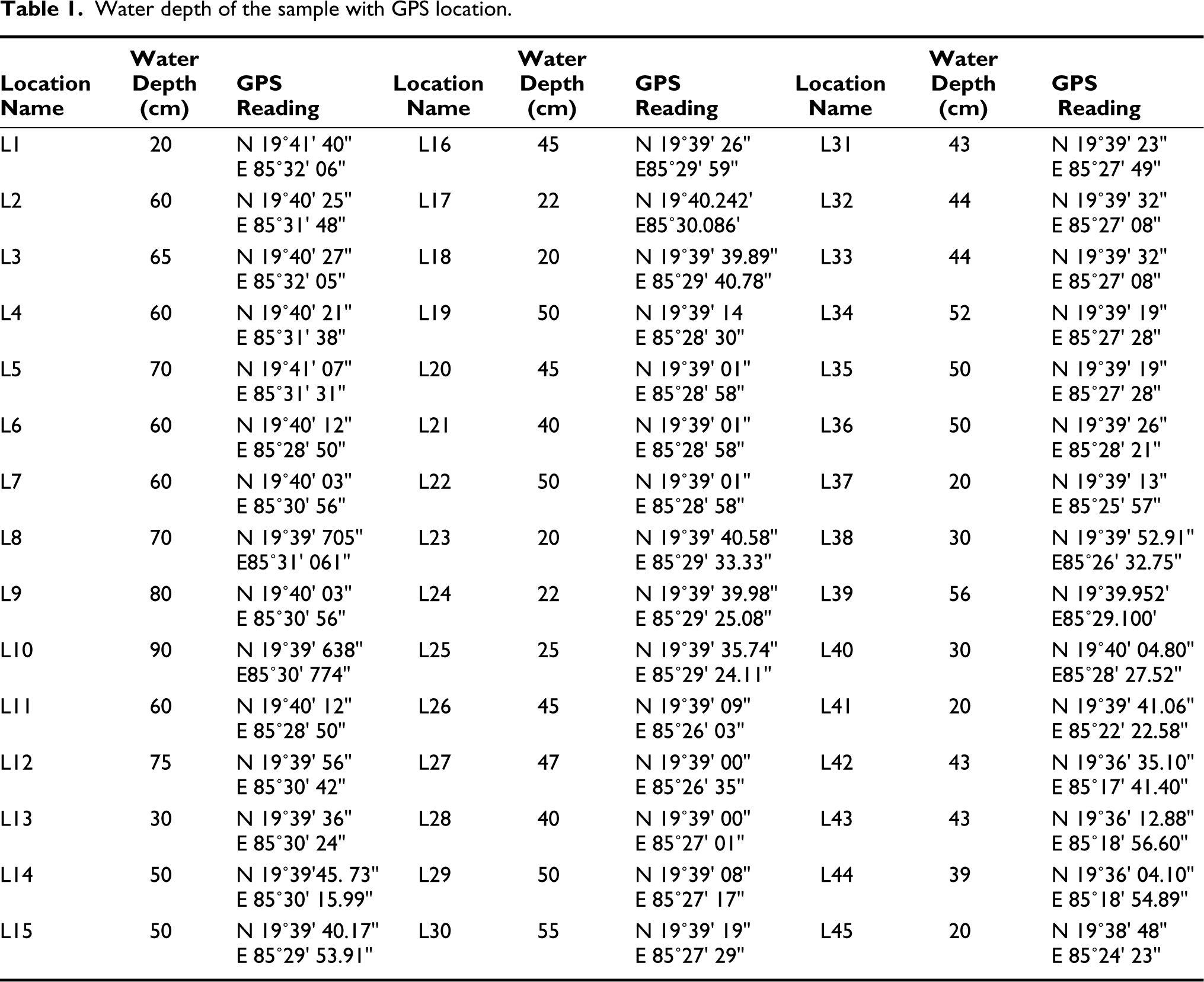
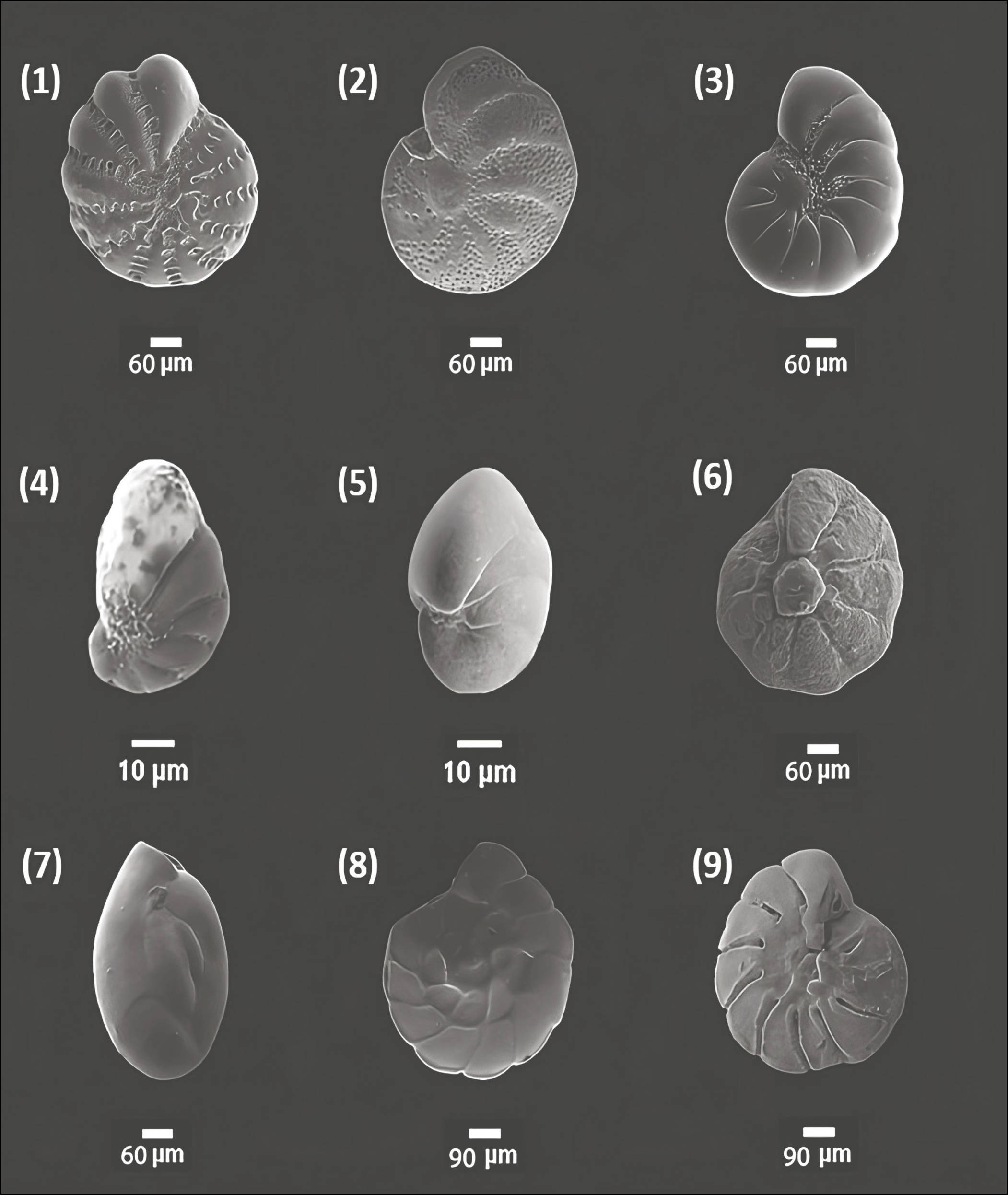
The present study comprises 45 sampling stations from the outer channel sector of the lagoon in the year 2017–2018 in both seasons (Figure 1). The samples were collected during March (pre-monsoon) and October (post-monsoon). A trowel was used to collect the sediments from the top sediment layer (0–1 cm) in a grid (10 cm × 10 cm × 1 cm). Samples were mainly collected from water depths varying from 20 cm to 90 cm (Table 1). The sample was then stored in a 250 mL plastic container where Rose Bengal solution (uniform mixture of 2 grams of Rose Bengal powder in 1-liter ethanol) was used as a stainer as well as a preservative. The samples were brought to the Micropaleontology Laboratory, Department of Geological Sciences, Jadavpur, and kept for 72 hours. After that, the collected sediments were cleaned using running water to remove the clay and silt particles using a 63-micron sieve. The >63 μm fraction residue was dried in an oven at 50oC. The dried residue was put in a micro-splitter, and 5 grams of each weighted sample was observed with the help of a stereo zoom microscope (model, Nikon SMZ-1000). This fraction is commonly used for benthic assemblages in ecological investigations (Murray, 2006; Gupta et al., 1987). The foraminifera were picked by using a fine brush. The images of the best-preserved species were captured with the help of Carl Zeiss EVO-18 Scanning Electron Microscope at Jadavpur University, India, for documentation. The observed foraminifera was primarily identified based on the works of Loeblich and Tappan (1974, 1988); Jorissen (1988); Holzmann and Pawlowski (1997); Rao and Srinath (2002); Kathal and Singh (2010); Kumar et al. (2014); Ghosh et al. (2014); Sen et al. (2018); Barbieri and Vaiani (2018) and Dasgupta and Ghosh (2021). All processed samples are kept in the Micropaleontology Laboratory, Department of Geological Sciences, Jadavpur University. The sand concentration of each sample was calculated from the resultant of the weight of dried unprocessed sediment and the weight of the dried residue left in >63 μm sieve after processing.
Water depth of the sample with GPS location.
Results
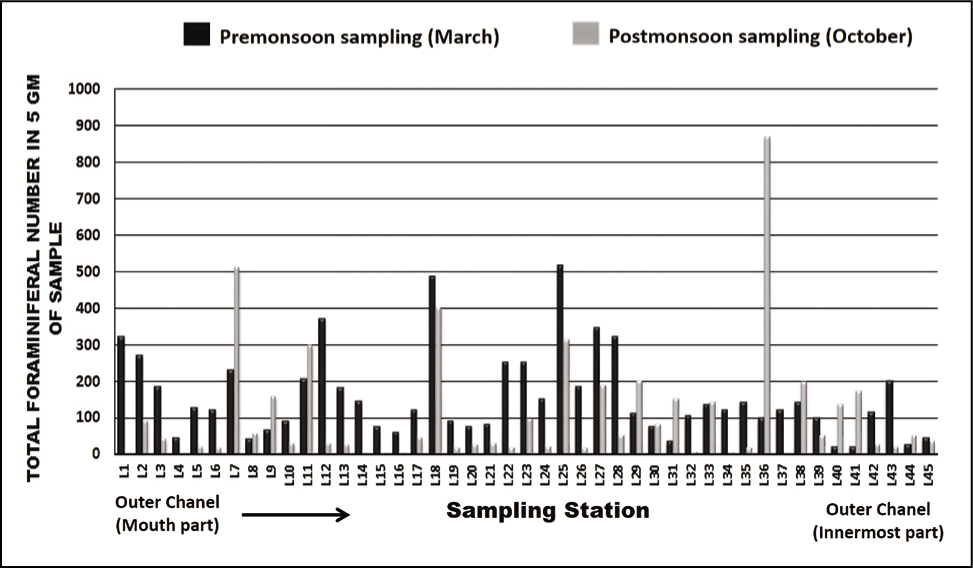
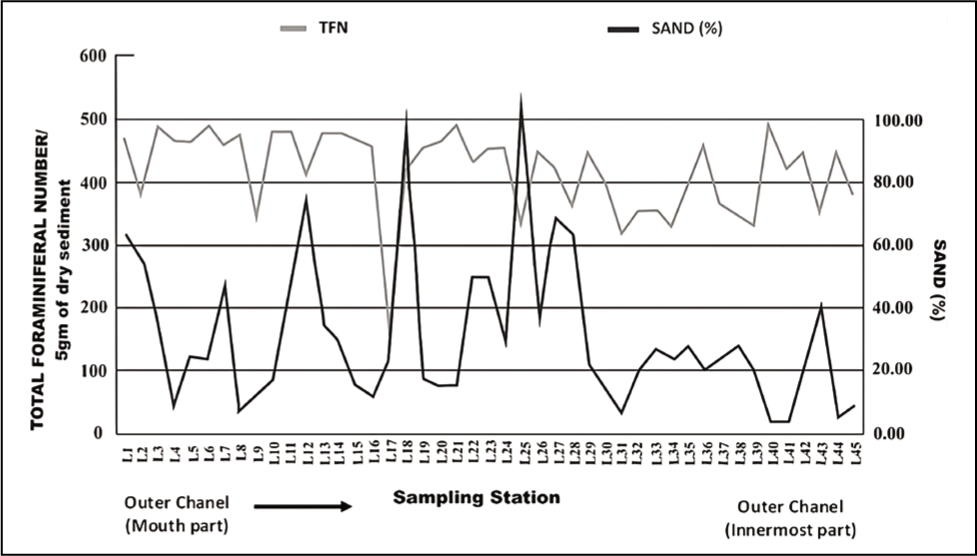
The seasonal distribution of total foraminiferal number (TFN) observed in all stations was plotted (Figure 2). In total, 28 species of benthic foraminifera are documented, of which 13 were hyaline, 14 were agglutinated and 1 had the porcelaneous test (Plate 1, 2 and 3). Ammonia spp. (Ammonia beccarii, Ammonia parkinsoniana and Ammonia tepida) is the most dominant taxa among all, followed by Pararotalia nipponica, Elphidium advenum, Cribroelphidium hispidulum, Elphidium williamsoni, Haynesina germanica, Rotalidium annctens, Hanzawaia sp., Elphidium sp., Nonion sp., Nonionellina labradorica, Miliammina fusca, Ammobaculites exiguus Textularia earlandi, Textularia agglutinans, Textularia sp., Ammobaculites agglutinans, Ammotium salsum, Miliammina obliqua, Trochammina inflata, Entzia sp., Haplophragmoides sp., Trochammina advena, Miliammina sp., Ammotium fragile and Quinqueloculina seminulum. The abundance of total foraminifera was high during pre-monsoon.
Seasonal distribution of the total number of foraminifera in the outer channel region.
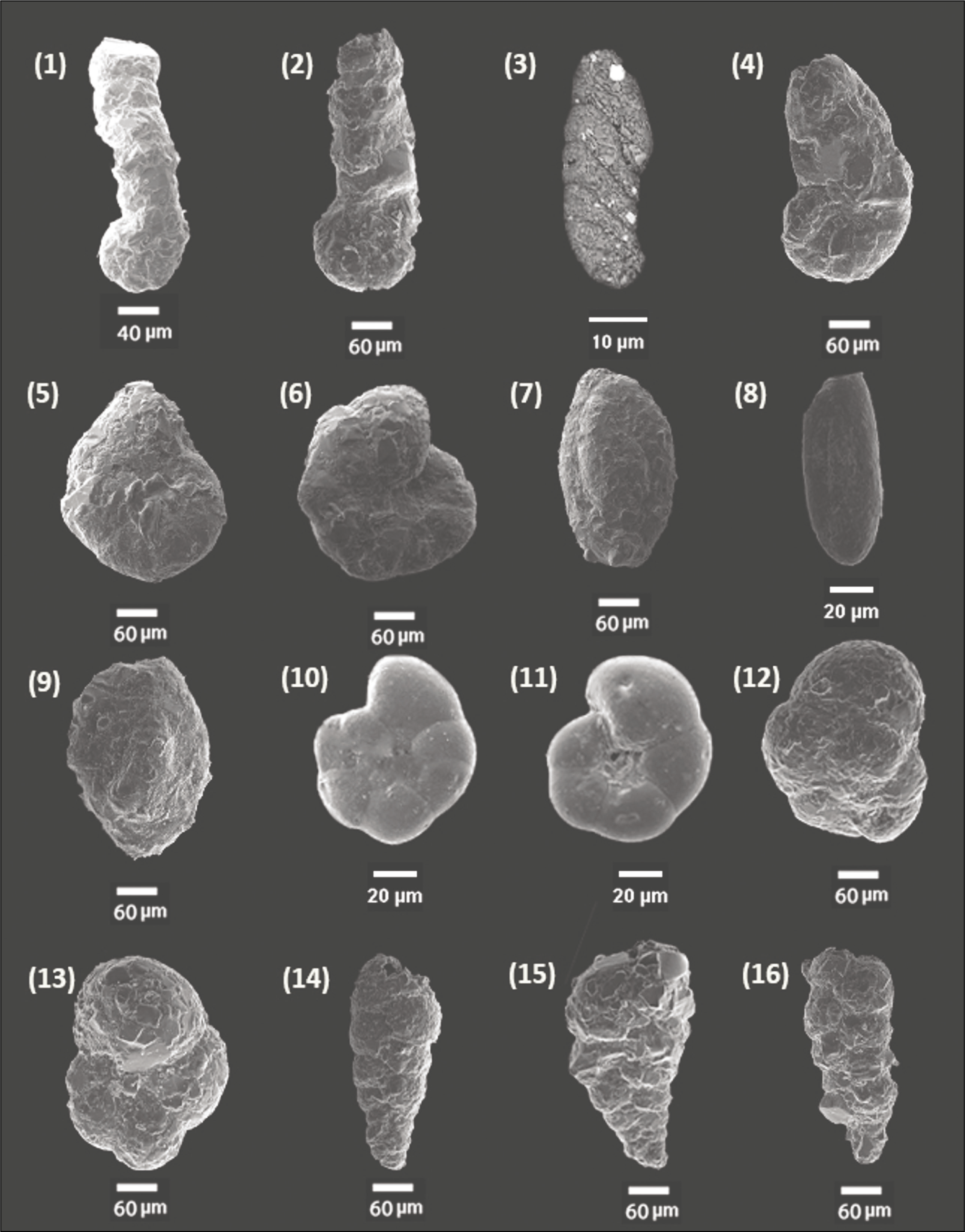
Scanning electron microscope images of foraminifera from Chilika Lagoon: 1. Ammobaculites agglutinans (Si); 2. Ammobaculites exiguus (Si); 3. Ammotium fragile (Si); 4. Ammotium salsum (Si); 5. Entzia sp. (Si); 6. Haplophragmoides sp. (Si); 7. Miliammina fusca (Si); 8. Miliammina obliqua (Si); 9. Miliammina sp. (Si); 10. Trochammina advena (U); 11. Trochammina advena (S); 12. Trochammina inflata (S); 13. Trochammina. inflata (U); 14. Textularia agglutinans (Si); 15. Textularia earlandi (Si); 16. Textularia sp. (Si). Legends, U: umbilical view; S: spiral view; Si: side view.
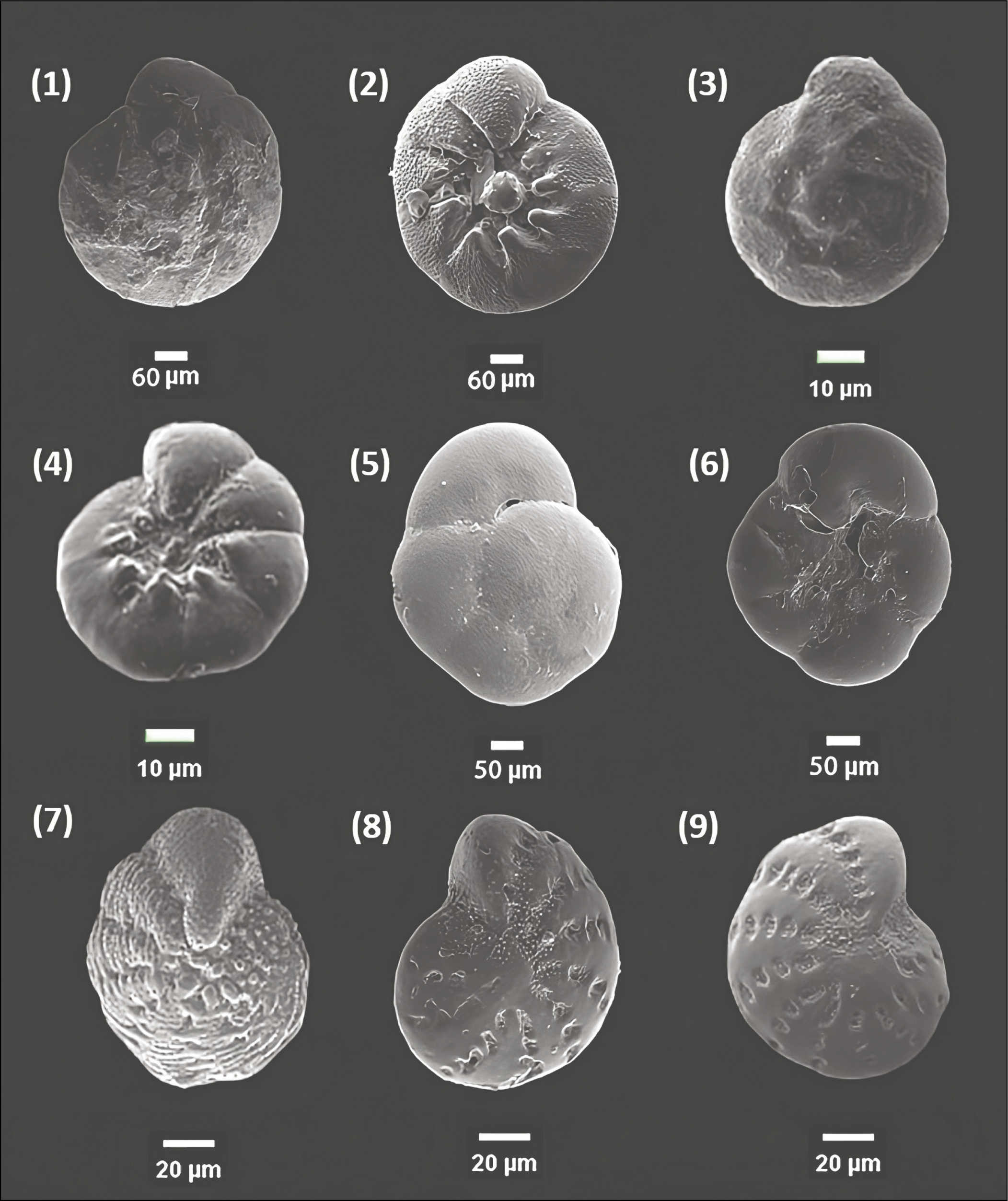
Scanning electron microscope images of foraminifera from Chilika Lagoon: 1. Ammonia beccarii (S); 2. Ammonia beccarii (U); 3. Ammonia parkinsoniana (U); 4. Ammonia parkinsoniana (S); 5. Ammonia tepida (U); 6. Ammonia tepida (S); 7. Cribroelphidium hispidulum (Si); 8. Elphidium advenum (Si); 9. Elphidium williamsoni (Si). Legends, U: umbilical view; S: spiral view; Si: side view.
Scanning electron microscope images of foraminifera from Chilika Lagoon: 1. Elphidium sp. (Si); 2. Hanzawaia sp. (U); 3. Haynesina germanica (Si); 4. Nonion sp.; 5. Nonionellina labradorica (Si); 6. Pararotalia nipponica (U); 7. Quinqueloculina seminulum (Si); 8. Rotalidium annctens (S); 9. Rotalidium annctens (U). Legends, U: umbilical view; S: spiral view; Si: side view.
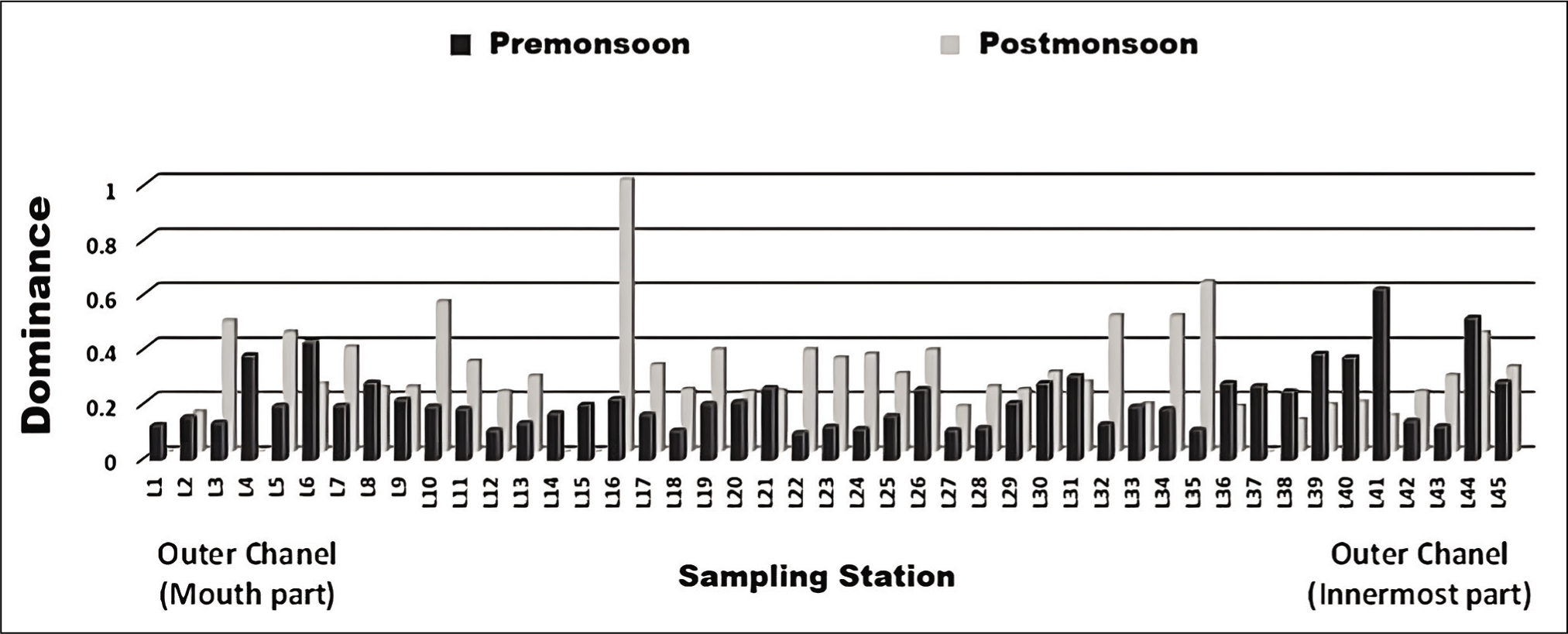
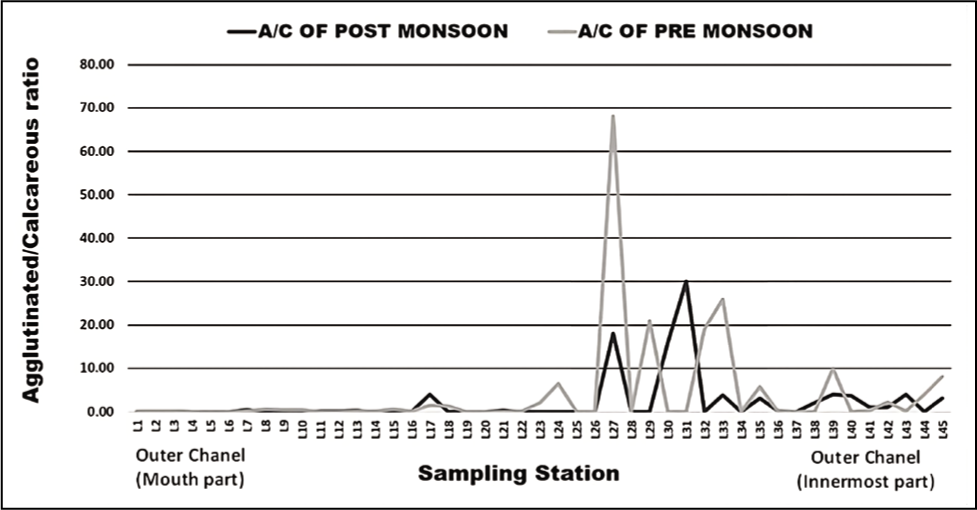
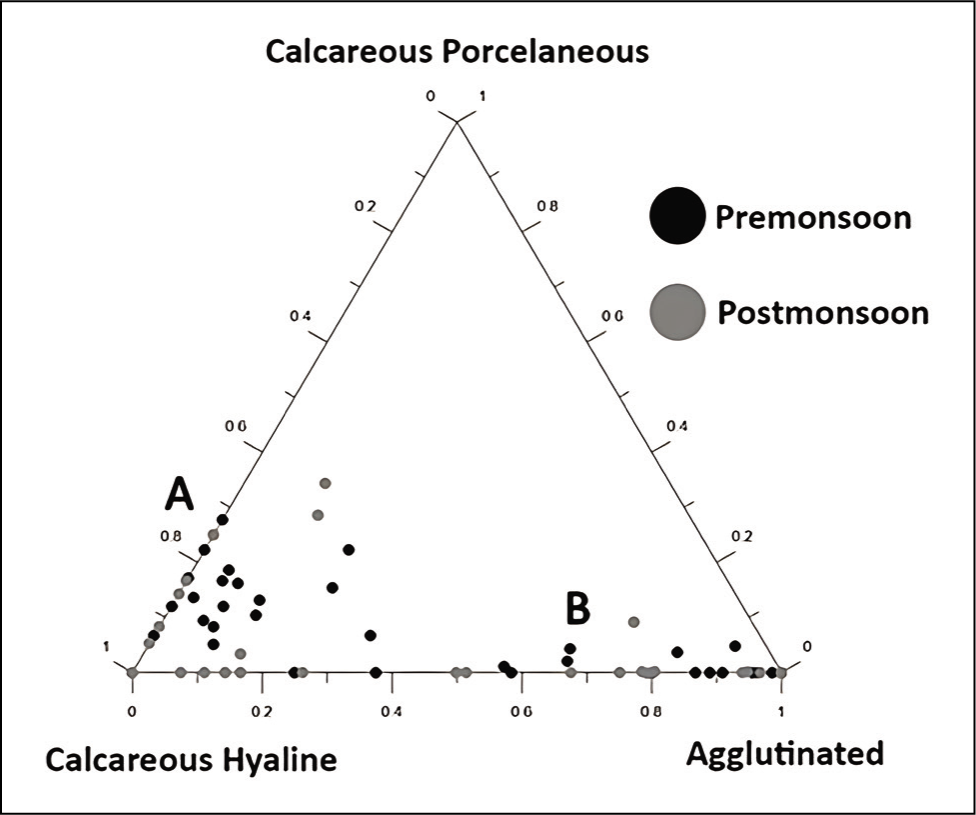
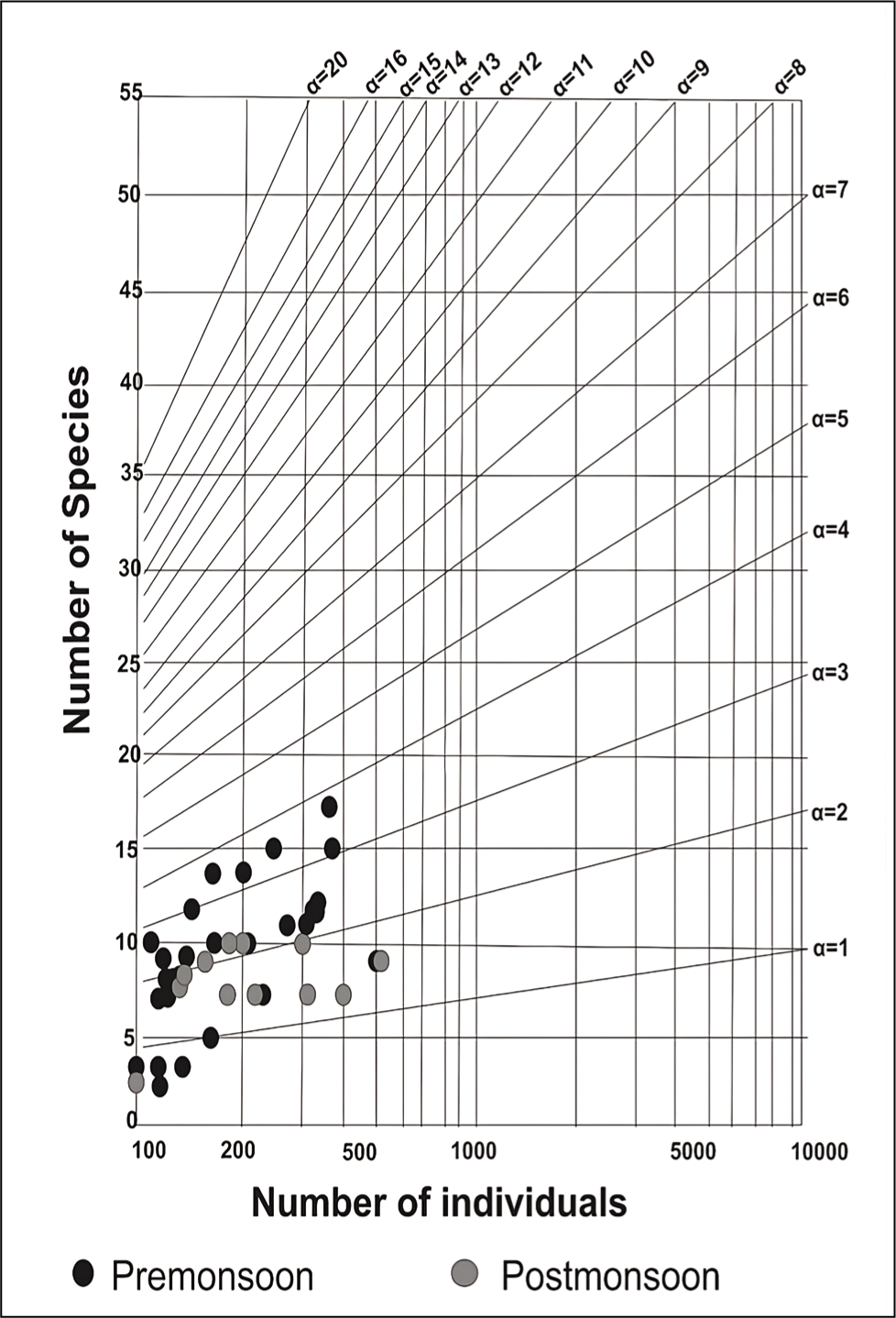
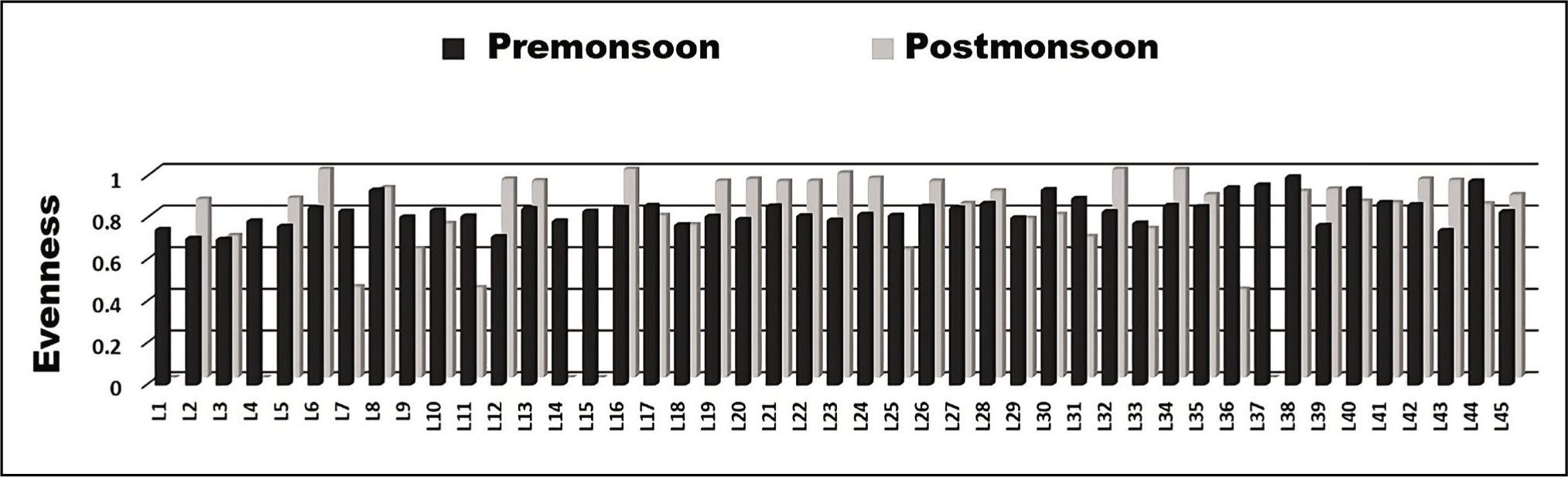
The outer channel shows an overall high sand percent-age varying between 70 and 100 (Figure 3). As we move away from its mouth, the assemblages of agglutinated foraminiferal forms are high (Figure 4). Murray’s Ternary diagram of pre- and post-monsoon shows two clusters (A and B) of foraminiferal populations (Figure 5). Cluster A represents the stations nearer to the mouth of the Chilika Lagoon dominated by calcareous tests. Cluster B includes the station away from the mouth, represented by the agglutinated tests. Fisher’s alpha species diversity value ranged between 1 and 4 for all the sampling stations (Figure 6). The Evenness index ranges between 0.70 and 1 with an average of 0.83 (Figure 7), and the Dominance index ranges from 0.09 to 0.62 with an average of 0.22 (Figure 8).
Trend of sand (%) and total foraminiferal number in the outer channel.
Agglutinated/calcareous foraminiferal ratio (A/C) in the outer channel.
Murray’s ternary diagram of pre- and post-monsoon along the study area of Chilika Lagoon.
Fisher-α diversity index.
Graph of evenness index.
Graph of dominance index.
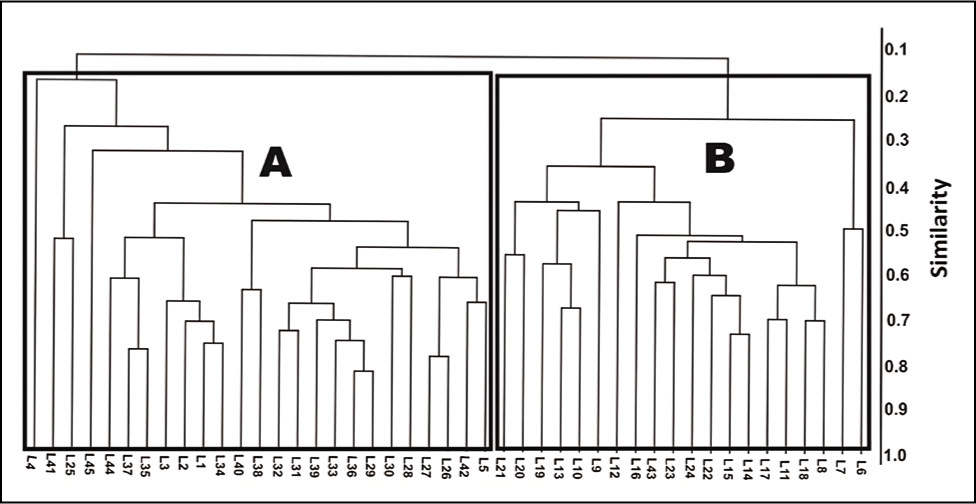
A multivariate clustering (Q-Mode) was performed with 28 species of foraminifera recorded from the sampling site to ascertain any grouping of similar taxa based on the stations (Figure 9). The data were entered into PAST software and analyzed (Hammer et al., 2001) for the cluster analysis. The Jaccard similarity index clustered them into two groups. The sampling station from the inner part of the outer channel constitutes Group A, while Group B represents the sampling stations near the lagoonal mouth
A multivariate cluster analysis (UPGMA paired group, Jaccard similarity index) based on species composition values.
Discussion
The TFN, including both living and dead forms, has been used for all the analysis. Previous studies (Kumar et al., 2015) reported an abundance of live foraminifera from the outer channel of Chilika, which is marine-influenced. However, the present study of the lagoon showed a poor number of living foraminifera. Here, it must be noted that the seasonally controlled hydrodynamics, intensified freshwater influx and vegetation growth impacted the composition of foraminifera. This has led to the increase of dead specimens or, at places, with rare living foraminifera (Dimiza et al., 2016b)
Among 14 species of agglutinated form, M. fusca, Ammotium salsa, Trochammina spp., Ammobaculites spp. and Haplophragmoides spp. indicate a stressed environment where water circulation is stagnant. Among these agglutinated forms of Chilika Lagoon, M. fusca (25%) was the most dominant one. They are strongly euryhaline and show high-temperature tolerance, and their mode of life is restricted in the marsh and shallow lagoonal environment (Murray & Alve, 2011). Trochammina sp. can sustain in low oxygenic conditions with high salinity (Kumar et al., 2014; Murray & Alve, 2011). Ammobaculites spp. are mud lovers. They like fine sediments. Hence, their presence indicates low oxygenic and muddy substrate (Murray & Alve, 2011). Haplophragmoides spp. are generally found in sandy to muddy substrates (Murray & Alve, 2011). So, these agglutinated species indicate temperate and high saline muddy substrate with low oxygenic conditions.
Benthic foraminiferal assemblages throughout the outer channel of Chilika Lagoon are dominated by A. beccarii, A. parkinsoniana and P. nipponica. Earlier studies reported that A. beccarii is opportunistic and pollution-tolerant. They can easily sustain the salinity variation in the shallow ocean, lagoons and delta (Debenay et al., 2005; Dimiza et al., 2016a, 2016b; Frontalini et al., 2009; Koukousioura et al., 2012; Martins et al., 2013). This species can sustain under abnormal oxygenic conditions and tolerate fluctuations in temperature (Walton & Sloan, 1990). The dominancy of this species increased with the low availability of primary production (Debenay et al., 1998). A. parkinsoniana is generally found in low-salinity zones (Schönfeld et al., 2021). Thus, Ammonia species across the stations indicate a stress-tolerant and cosmopolitan environment throughout the lagoon (Gupta et al., 2018). P. nipponica, the inner shelf benthic foraminifera, can sustain extensive saline conditions (Nigam et al., 2006). The presence of the genus Elphidium indicates a low oxygenic state in the water (Gupta et al., 1996). Also, Elphidium crispum is rich in a sandy substrate (Eichler et al., 2015; Gupta et al., 1996; Gupta & Platon, 2006). H. germanica is typically found in fine sediment and well documented in a stressed environment (Alve, 1995; Yanko et al., 1998; Debenay et al., 2001).
A low number (<5%) of Q. seminulum was recorded during our study along the sampling stations. This porcelaneous species is primarily found in low-vegetated zones and shallow marine waters (Dimiza et al., 2016a, 2016b; Jorissen, 1988; Murray, 2006; Sgarrella & Moncharmont Zei, 1993). Thus, the low population of miliolids tests indicates low saline and shallow brackish water habitat prevalence (Frontalini et al., 2011).
The present study observed that the abundance of total foraminifera was high during pre-monsoon. This might occur as the months before monsoon are considered the reproductive season of foraminifera (Ghosh et al., 2014). These assemblages also depend on different abiotic components like salinity, temperature and pH (Kumar et al., 2015; Sen & Bhadury, 2016). As the recorded salinity and temperature were high during pre-monsoon, it might be another cause for increased foraminiferal assemblages in the outer channel of the lagoon.
As the seawater intrudes into the outer channel by three inlets, the sand (%) remains high towards the mouth part of the outer channel. The abundance of foraminifera could be more noteworthy, with a few exceptions in stations away from the outer channel. As we move away from the mouth, the agglutinated foraminifera dominates with a high ratio of agglutinated/calcareous tests. The innermost part of the outer channel is characterised by relatively stagnant water and poor circulation (Rao et al., 2000; Sen & Bhadury, 2016; Sen et al., 2018) with higher salinity (Kumar et al., 2014). The substrate was dominated by mud towards the inner part of the outer channel. Thus, a low oxygen state with poor circulation and high salinity may be responsible for agglutinated forms in the channel. The mouth of the outer channel has no agglutinated form. Murray’s ternary plot (Figure 5) indicates that the study area is interpreted as a marginal marine environment. Fisher’s alpha index (Figure 6) interpreted that the lagoon has low to moderate foraminiferal diversity. The pre-monsoon periods show more abundant and diverse forms than the post-monsoon months. This is evident due to favourable salinity and temperature conditions during pre-monsoon.
Moreover, the evenness index indicates the high evenness of the population, while the dominance index is categorised as low to moderate. This result indicated that foraminifera is evenly distributed in this area with relatively equal abundance. It means no opportunistic taxa are present in the lagoon.
In the Q-Mode clustering, Group A shows low diversity with a high abundance of agglutinated taxa. In contrast, Group B shows high diversity and a high quantity of the calcareous hyaline group. This Q mode clustering showed that the innermost part of the outer channel was dominated by the agglutinated test, and calcareous forms characterised the lagoonal mouth region.
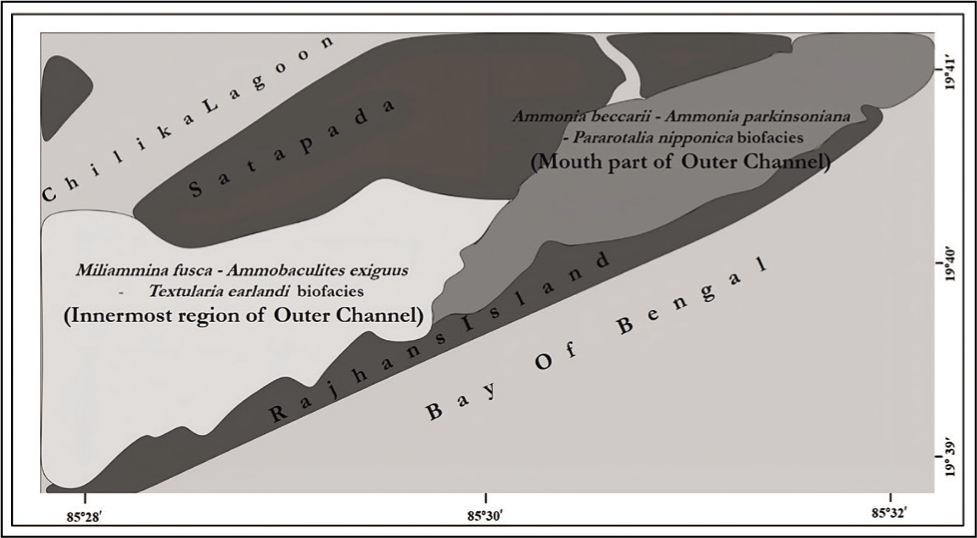
Depending on the significant abundance data of TFN along with the multivariate study by using similarity index for grouping of station and species, two distinct biofacies zones were identified (Figure 10). One was the innermost part of the outer channel, which was dominated by M. fusca, A. exiguus, T. earlandi and another was the mouth part of the outer channel, dominated by A. beccarii, A. parkinsoniana and P. nipponica. Low salinity, shallow water depth and low oxygen conditions are the major factors that affect this zonation of foraminifera. The inflow of freshwater from different streams and catchments, added with precipitation, was the leading cause of the lagoon’s salinity variation.
Biofacies map of the outer channel, Chilika Lagoon.
Conclusions
In total, 28 foraminiferal species are documented in the outer channel region. The dominant calcareous forms are A. beccarii, A. parkinsoniana and P. nipponica and agglutinated are M. fusca, Ammobaculites exiguus and T. earlandi.
Two biofacies zones are recorded in the outer channel: i. A. beccarii–A. parkinsoniana–P. nipponica biofacies—mouth part of outer channel; ii. M. fusca–Ammobaculites exiguous–T. earlandi biofacies—inner part of the outer channel.
The pre-monsoon assemblages are abundant and diverse as that of post-monsoon due to favourable abiotic parameters such as salinity and temperature and the onset of the reproductive season of the forms.
The dominance of calcareous forms in the outer part of the outer channel suggests favourable salinity, temperature and high carbonate input near the sea mouth.
The agglutinated tests found in the inner part of the outer channel indicate high total organic carbon, low water depth and low mixing of coastal waters.
Footnotes
Acknowledgements
The authors would like to acknowledge the DST FIST-sponsored SEM-EDS Laboratory of the Department of Geological Sciences, Jadavpur University for microphotographs.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors want to acknowledge Jadavpur University RUSA 2.0 Project Grant R-11/279/19 to support this study’s completion.
