Abstract
Neogloboquadrina pachyderma (Ehrenberg) is one of the few planktic foraminiferal species that thrive in polar waters and serves as an important palaeoceanographic indicator of polar water mass expansion. The species evolved from Neogloboquadrina continuosa during the late Miocene and has undergone several taxonomic revisions—initially described as Aristerospira and later reassigned to Neogloboquadrina. Traditionally, the ratio of its two coiling types (sinistral and dextral) has been used to distinguish glacial and interglacial periods. However, genetic studies have revealed that these two coiling forms actually represent distinct species. As a result, the dextral form was reclassified as Neogloboquadrina incompta. Despite this taxonomic change, earlier palaeoceanographic interpretations based on coiling ratios remain valid. The morphology of N. pachyderma is highly variable, influenced by water mass characteristics and climatic conditions. Variations are observed in aperture shape, chamber arrangement, chamber packing, and other phenotypic traits. In the present study, we identify and describe eight distinct morphotypes of N. pachyderma from the Southeast Atlantic Ocean, based on detailed morphometric measurements and surface ultrastructure analyses.
INTRODUCTION
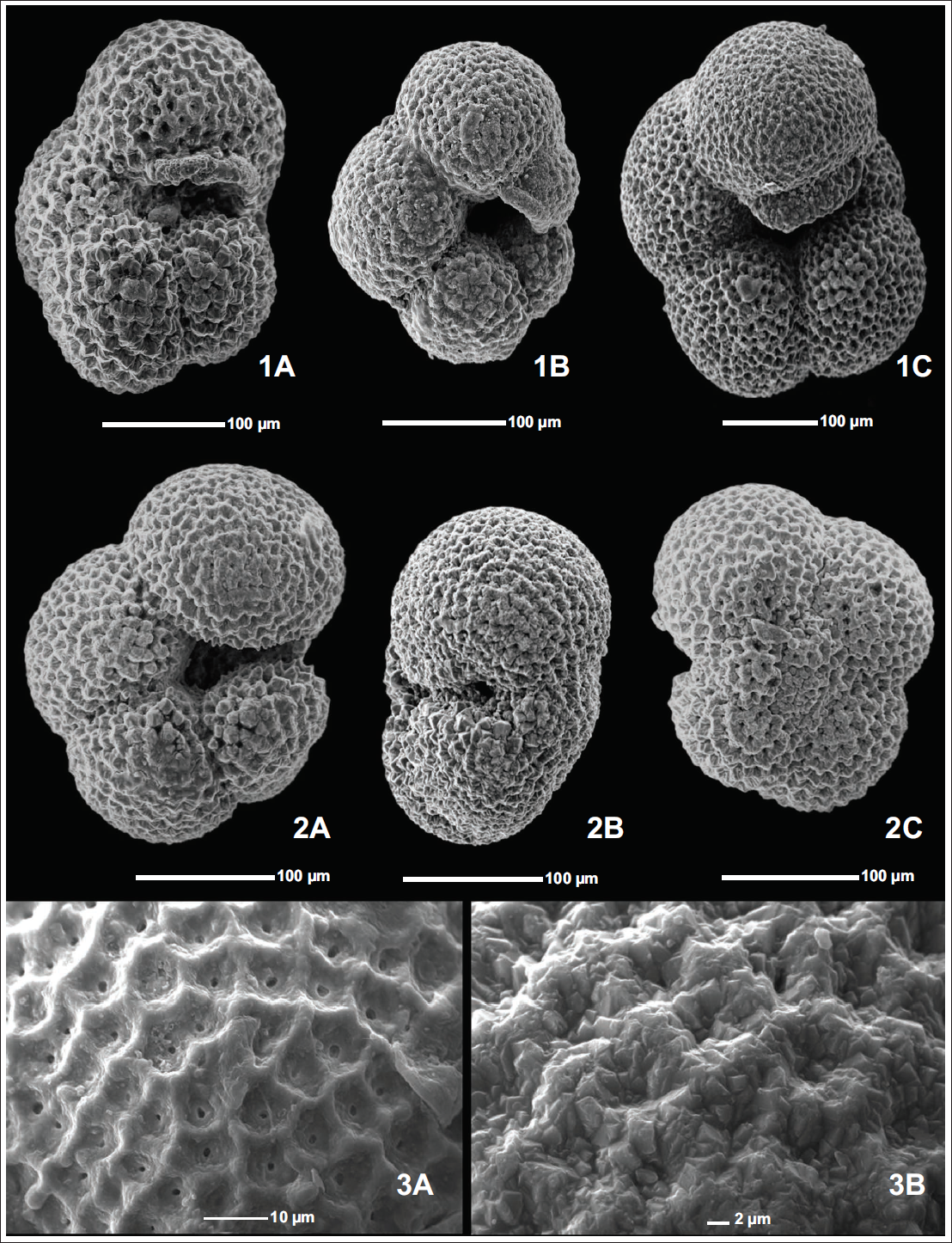
Neogloboquadrina pachyderma was initially established by Ehrenberg (1861, 1873) as Aristerospira pachyderma based on specimens collected from northern latitudes. N. pachyderma (Ehrenberg) is an extant planktic foraminifera residing primarily in the polar to subpolar oceans. N. pachyderma is also occasionally found in the temperate and sometimes even in the tropical latitudes due to the temporal shifts in the oceanic fronts and upwelling (Eynaud et al., 2009; Sahoo et al., 2022; Tolderlund, 1971). Jenkins (1967) observed that N. pachyderma shows morphological features similar to those of Neogloboquadrina continuosa (Blow), including aperture orientation and size, and has a similar apertural lip. Still, most importantly, they both show an unusual coiling pattern in which the number of chambers added per whorl reduces from 5 to 6 in initial whorls to four chambers in the final whorl. This led to the conclusion that N. pachyderma evolved from N. continuosa during the late Miocene. Further, evidence of this evolution was suggested by Stott and Webb (1989), who observed several specimens representing the intergradation of N. continuosa and N. pachyderma in the fossiliferous marine sediments along the continental margin of southern Chile. These authors believed that N. pachyderma evolved through cladogenesis of the parent population rather than gradual phyletic transformation from N. continuosa. Even though N. pachyderma shows four chambers in the final whorl, some morphotypes even show five chambers, and some show kummerform chambers in the final whorl (Plate 1, Figures 1A–1C) (Altuna et al., 2018; Bandy, 1972). Berger (1969) gave the concept of kummerform and normalform varieties, with the explanation that, if the ultimate/final chamber in a foraminifer is of the same size or smaller than the penultimate chamber, where chambers generally show an increase in size, they are termed as kummerforms (Plate 1, Figure 1B). While the final chamber of the last whorl follows size increase, these are termed as normalforms (Plate 1, Figure 1A). N. pachyderma population in the world oceans is dominated by the Kummer form varieties (Bandy, 1972). Olsson (1973) extended Berger’s work and stated that kummerforms phenotypes are an indication of complete growth in an individual, that is, the individual has reached the full adult size or growth limit of its species. He also stated that individuals that have reached the maximum size show a slowing of growth prior to termination, resulting in the diminutive final chamber. While N. pachyderma shows wide variation, its basic taxonomy can be described as four to four and a half subglobular chambers, increasing in size as they are added, with a low trochospiral coiling, forming an overall subquadrate shape with a rounded axial periphery and a lobulate equatorial periphery (Kennett & Srinivasan, 1983). It shows radial and depressed sutures on both the umbilical and spiral sides. Lastly, it has a laterally oriented interiomarginal aperture with a thick apertural rim (Plate 1, Figures 2A–2C). Its aperture shape, however, can vary from an elongated slit shape, a slightly arched shape, or even a rounded one due to the compactness of the test. During the cooler periods, the final chamber can even overgrow and partially cover the marginal portion of the aperture, resulting in a restricted aperture. Several authors have used N. pachyderma in palaeoclimatic and palaeoceanographic studies, as it exhibits variation in coiling pattern depending on the surrounding water mass. It can form chambers arranged in a clockwise-to-anticlockwise pattern (when viewed from the spiral side), representing a warmer water mass and a cooler water mass, respectively (Ericson, 1959). Setty (1977) was the first to conceptualise the existence of two separate subspecies of N. pachyderma representing dextral and sinistral coiling. Setty (1977) named them N. pachyderma dextralis typica and N. pachyderma sinistralis typica for dextral and sinistral varieties, respectively. The specimens for Setty’s (1977) study were collected from shelf-slope sediments of the Northern Indian Ocean. Years later, Darling et al. (2006), based on genetic studies of N. pachyderma’s dextral and sinistral populations, stated that the previously reported links between coiling and environmental factors in the two coiling varieties result from the different ecological preferences of the genetically distinct coiling types and preferred to use Neogloboquadrina incompta (Cifelli) for the dextrally coiling morphospecies.
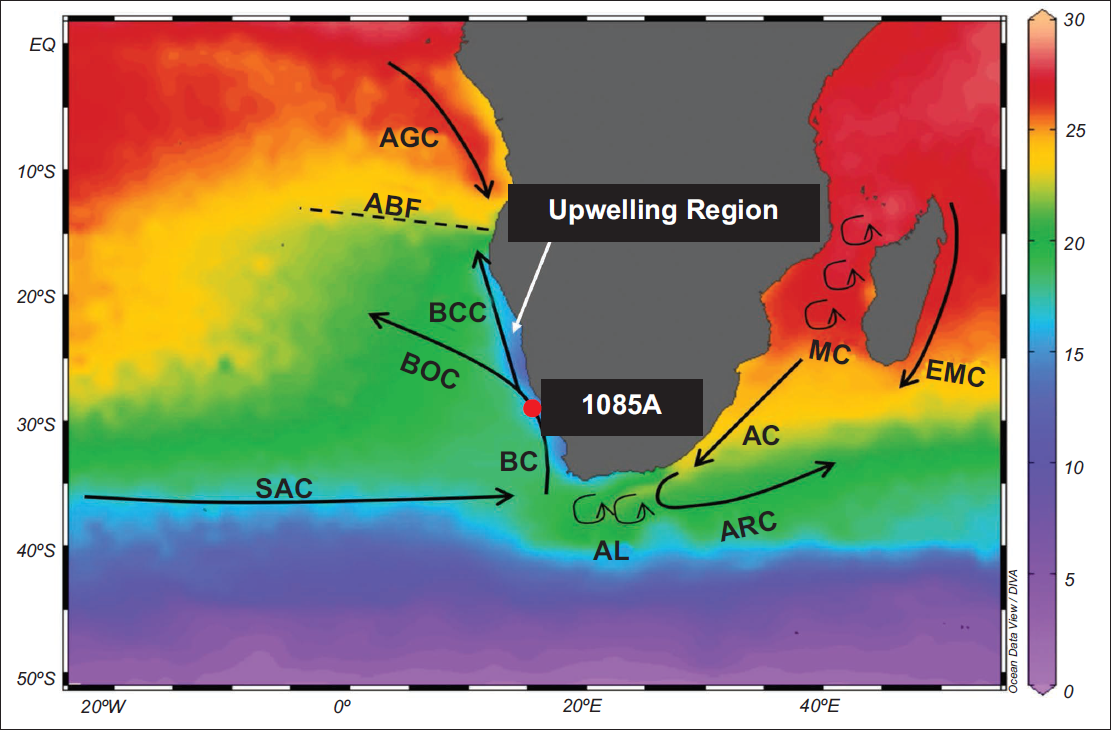
The map shows the annual SST of the Southeast Atlantic and Southwest Indian Ocean from the World Ocean Atlas 2023 (Reagan et al., 2024), visualised using Ocean Data View (Schlitzer, 2023). The ODP hole 1085A has been marked by a red circle and the modern oceanographic setup has been presented with major ocean currents (Agulhas Current (AC), Angola Current (AGC), Agulhas Leakage (AL), Agulhas Return Current (ARC), Benguela Current (BC), Benguela Coastal Current (BCC), Benguela Ocean Current (BOC), East Madagascar Current (EMC), Mozambique Channel (MC) and South Atlantic Current (SAC)) as well as the Angola-Benguela Front (ABF).
1A) 4 chambered Normalform N. pachyderma (175,1085A,31H-2,139-141cm). 1B) 4 chambered Kummerform N. pachyderma (175,1085A,29H-1,0-2cm). 1C) 5 chambered N. pachyderma (175,1085A,31H-2,139-141cm). 2A) N. pachyderma apertural view (175,1085A, 29H-6,144-146cm). 2B) N. pachyderma side view (175,1085A,28H-6, 115-117cm). 2C) N. pachyderma spiral view (175,1085A,31H-2,139-141cm). 3A) Reticulate Surface Ultrastructure in N. pachyderma. 3B) Crystalline Surface Ultrastructure in N. pachyderma.
In this study, eight morphotypes of N. pachyderma have been observed in the Southeast Atlantic Ocean, from ODP hole 1085A. These morphotypes were classified based on the number of chambers in the final whorl, kummerform or normalform variety, shell compactness, aperture shape and size, and the orientation of the ultimate chamber. Detailed morphometric analysis was carried out for each morphotype to quantify the test area and other key characteristics. Scanning Electron Microscopy (SEM) (JEOL JCM-6000 Plus) allowed for examination of the surface ultrastructure, including pore density and pore diameter. These results provide new insights into the morphological variability of N. pachyderma in the Southeast Atlantic and contribute to a broader understanding of its palaeoceanographic significance.
OCEANOGRAPHIC SETUP OF HOLE 1085A
ODP hole 1085A is located at 29°22.47′S, 13°59.41′E with a water depth of 1713.2 m in the Southern Benguela Region (SBR), which is a part of the Benguela Upwelling System (BUS) in the Southeast Atlantic Ocean (Figure 1). The atmospheric circulation in this region is influenced by an extensive, semi-permanent high-pressure system, the South Atlantic Atmospheric High (SAAH), and the Intertropical Convergence Zone (ITCZ). Both SAAH and ITCZ shift towards south (poleward) during the Austral Summer (December-February) and towards north (equatorward) during the Austral Winter (June-August) (Gilliland & Keim, 2018). These atmospheric conditions drive the southeast trade winds, which in turn fuel a large-scale coastal upwelling system (Figure 1) and the primary ocean current in the area, that is, the Benguela Current (BC) (Lutjeharms & Stockton, 1987; Rommerskirchen et al., 2011; Shannon, 1985). The BC forms the eastern boundary of the South Atlantic subtropical gyre and flows towards the north along the southwestern coast of Africa, extending from the Cape Agulhas (~35°S) in South to the Angola–Benguela Front (ABF) (~14–17°S) in the north (Figure 1) (Giraudeau et al., 2001; Gregor & Monteiro, 2013; Shannon et al., 1996).
MATERIALS AND METHODOLOGY
All the N. pachyderma (sinistral, Darling et al., 2006) studied were collected from the deep-sea core samples retrieved from ODP hole 1085A located in the SBR. All specimens were examined using a stereoscopic microscope (Nikon SMZ1000) equipped with an advanced image analyser. Specimens were then hand-picked and classified into morphotypes. The identification of morphotypes was based on several parameters, including the number of chambers in the final whorl, normalform or kummerform variety, test compactness, aperture shape and size, encrustation, and orientation of the final chamber. For quantitative morphometric analysis, 25 individuals of each morphotype were selected, and their morphological features were measured (Table 1). The average size of each morphotype was calculated by averaging the sizes of the 25 selected individuals. Similarly, their X/Y ratio was calculated to determine the kummerforms and normalforms morphotypes (Plate 2) (Altuna et al., 2018). The X/Y ratio > 1 shows the normal form variety, while the X/Y ratio < 1 represents the kummerform variety. Additionally, if the X/Y ratio ≈ 1 (0.95–1.05), it represents the quadrate forms. However, one of the morphotypes discussed here shows the X/Y ratio ≈ 1, but is described as pseudo-quadrate. This is because the ultimate chamber is actually larger in size than the penultimate chamber, meaning that it should belong to a normalform variety (X/Y ratio > 1). However, the ultimate chamber of this morphotype shows a dent-like deformation, resulting in an X/Y ratio ≈ 1. Representative specimens of each morphotype were also examined using SEM (JEOL JCM-6000 Plus). With the help of SEM imaging, the surface ultrastructure of all the specimens was studied, as well as the average pore size and pore density per unit area. This is measured to assess potential variations in the surface ultrastructure.
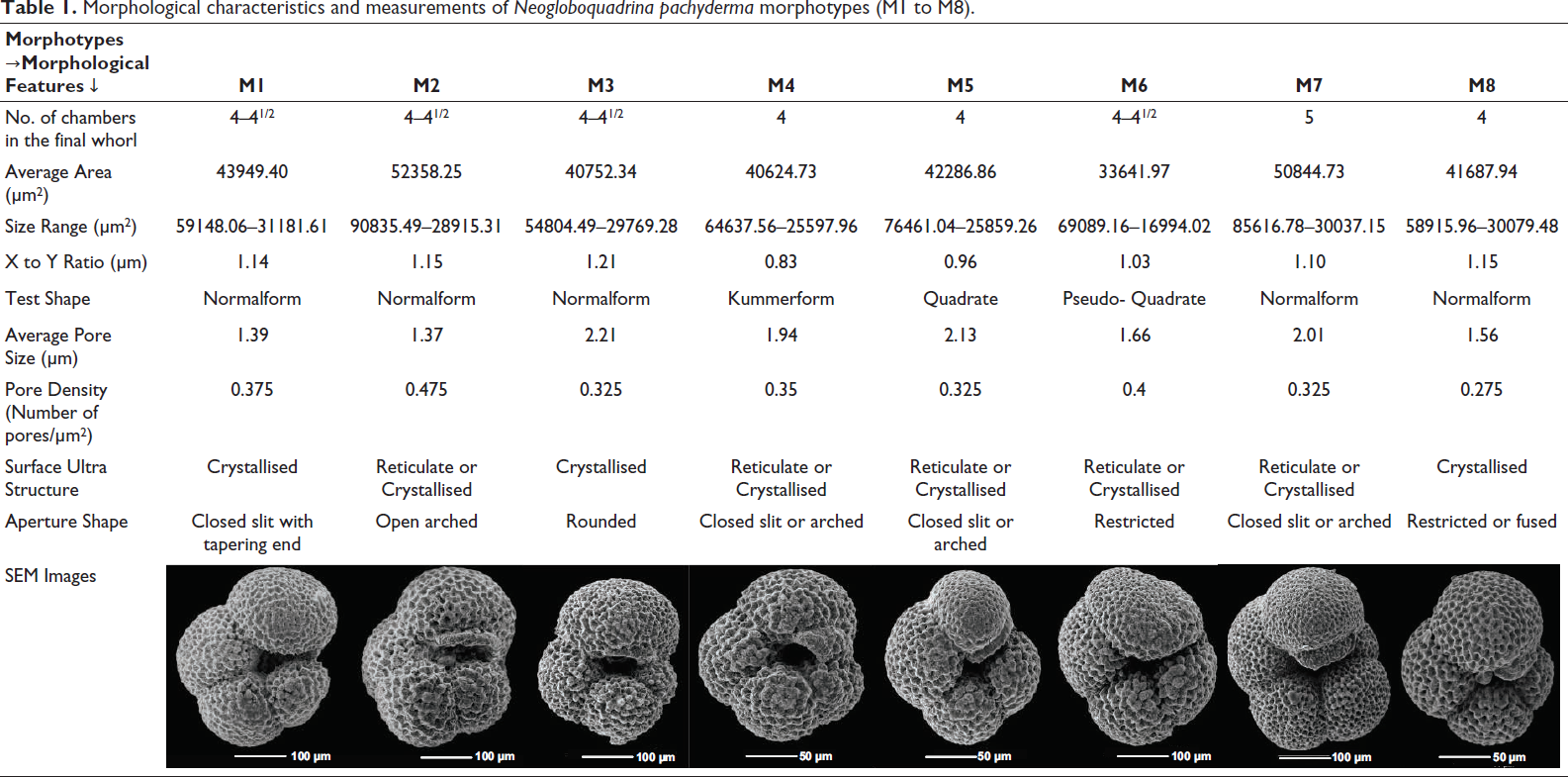
Morphological characteristics and measurements of Neogloboquadrina pachyderma morphotypes (M1 to M8).
X/Y ratio representation on N. pachyderma. X/Y < 1 represents a kummerform variety, X/Y ~ 1 represents a quadrate form, and X/Y > 1 represents a normalform N. pachyderma (175,1085A,29H-6,144-146 cm).

SURFACE ULTRASTRUCTURE OF N. pachyderma
As N. pachyderma inhabits the temperate-polar waters, its surface shows variation depending on whether the region is influenced by cold polar water masses or relatively warmer temperate-subtropical water masses, resulting in two types of surfaces–Reticulate and Crystalline (Srinivasan & Kennett, 1974).
Reticulate surface ultrastructure: Polygonal ridges of varying heights form a network of continuous and broken ridges, giving it a mesh-like appearance with distinct pore pits enclosed within this network of ridges (Plate 1, Figure 3A).
Crystalline surface ultrastructure: Individuals with a crystalline test are covered with large calcite crystals (Plate 1, Figure 3B). In the penultimate and final chambers, calcite clusters surround the pores. In contrast, in earlier chambers, the crystals begin to develop penetration twinning, leading to the formation of a stellate structure (Srinivasan & Kennett, 1974). Srinivasan and Kennett (1974) concluded that crystalline surface ultrastructure can be used to interpret cooler periods, whereas reticulate surface ultrastructure is associated with relatively warmer periods. Although Srinivasan and Kennett (1974) concluded the relation between surface ultrastructure and palaeoceanographic conditions, they did not incorporate the coiling direction in their results. Reynolds and Thunell (1986) combined the Srinivasan and Kennett (1974) surface ultrastructure studies with coiling direction and concluded that individuals with heavily calcified, thick tests are sinistrally coiled, while individuals with thin, reticulate tests are dextrally coiled. According to the results from these studies, the coiling direction can be used as a viable method for morphotype separation of N. pachyderma and ultimately for palaeoceanographic interpretation. This surface ultrastructural variation is not limited to N. pachyderma; all individuals of the genus Neogloboquadrina initially show a reticulate test. Later in their lives, depending on water depth and regional water masses, some individuals develop a secondary calcite coating, leading to a crystalline surface ultrastructure. In contrast, others form only microcrystals on interpore ridges (Srinivasan & Kennett, 1974).
Regarding the origin of this encrustation due to secondary calcification, Kohfeld et al. (1996) presented various hypotheses, some of which are:
N. pachyderma changes from uncrusted to encrusted forms as part of its life cycle. A similar case is also observed in the Globigerinoides sacculifer, which develops a sac-like chamber during gametogenesis. The crust of N. pachyderma may also be gametogenic. Encrustation could also be a gradual process beginning when the N. pachyderma starts to sink. The average increase in shell mass in N. pachyderma as it descends also supports this suggestion. Alternatively, encrustation could occur all at once but at different depths in the water column. Another alternate hypothesis is that encrusted and non-encrusted N. pachyderma are two different morphotypes occupying two distinct environments. The non-encrusted morphotype occupies the mixed layer well above the pycnocline, whereas the encrusted morphotype occupies the main pycnocline.
APERTURE
The interiomarginal aperture of N. pachyderma can exhibit different shapes and sizes depending on the type of shell and the water mass (Kipp, 1976). Several authors, including Bandy (1972), Reynolds and Thunell (1986), and Altuna et al. (2018), have conducted morphological studies of N. pachyderma, and the specimens they have described exhibit a variety of apertures. Here, we summarise all the variations in aperture that can be seen in N. pachyderma.
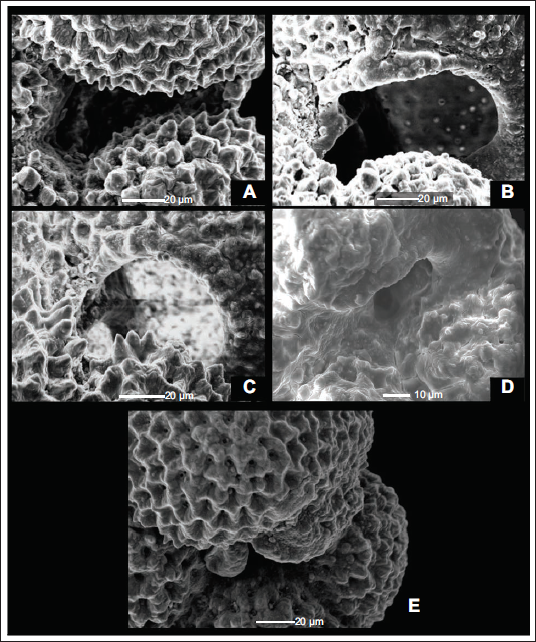
Elongated and lobate N. pachyderma generally shows an elongated slit like aperture extending from its umbilicus to the periphery of the individual (Plate 3, Figure A). However, in some individuals, this elongated slit begins to curve before extending to the margin. This curvature of the aperture forms a slightly arched shape resembling a bottomless pit (Plate 3, Figure B). In compact-shelled N. pachyderma, the aperture can take a roughly rounded form (Plate 3, Figure C). Some N. pachyderma also show a restricted aperture due to deformation of the final chamber (Plate 3, Figure D); this aperture restriction can also result from the overgrowth of the final chamber in the individual. When a final chamber grows, it can sometimes cover the marginal portion of the aperture (Plate 3, Figure E). This closed aperture results from the heavy crystallisation of the shell of N. pachyderma.
A) Long Slit Aperture (175,1085A,29H-6,144-146cm). B) Curved Aperture (175,1085A,28H-6,79-81cm). C) Rounded Aperture (175,1085A,31H-2,139-141cm). D) Restricted Aperture (175,1085A,29H-6,144-146cm). E) Aperture covered due to overgrowth of final chamber (175,1085A,34X-3,17-19cm).
RESULTS AND DISCUSSION
N. pachyderma shows various morphotypes due to the variations in the chamber arrangement, aperture size and shape, normalform-kummerform variety, surface ultrastructure, etc. Many authors have reported morphotype studies on N. pachyderma, including Keller (1978), Reynolds and Thunell (1986), Eynaud et al. (2009), and Altuna et al. (2018). However, a detailed description of these morphotypes has yet to be established for the South Atlantic. Furthermore, they combined many independent morphological variations into one morphotype, such as the N. pachyderma Form 1 of Keller (1978), which includes forms with a compact test, 4 to 5 chambers in the final whorl, normalform, and the Kummerform variety.
Here, we present eight morphotypes of N. pachyderma from the Southeast Atlantic Ocean that exhibit distinct morphological variation and are abundant in this region (Table 1). All eight morphotypes are present throughout the studied cores of the ODP hole 1085A, spanning from the Late Miocene to the Recent.
Morphotype I (M1)
This morphotype of N. pachyderma shows four to four and a half chambers in the final whorl, with the final chamber larger than the penultimate (X/Y ratio > 1), that is, it is a normalform variety (Table 1). M1 shows an average size of 43949.40 µm², ranging from 31181.61 µm² to 59148.06 µm² (Table 1). It has a crystallised surface ultrastructure, suggesting the intrusion of cold polar waters into the Southeast Atlantic. Furthermore, it shows an average pore size of 1.39 µm and a density of 0.375 pores per µm² (Table 1). The characteristic feature of M1 is its narrow, low slit like aperture that shows a tapering end at the periphery (Plate 4, Figures 1A–1C).
1A) Apertural view of the N. pachyderma (Ehrenberg) morphotype M1 showing a long-slit aperture (175,1085A,29H-6,144-146cm). 1B) Spiral view of the N. pachyderma (Ehrenberg) morphotype M1 showing sinistral coiling (175,1085A,31H-2,139-141cm). 1C) Surface Ultrastructure of the N. pachyderma (Ehrenberg) morphotype M1 showing pores and thick polygonal ridges (175,1085A,31H-2,139-141cm). 2A) Apertural view of the N. pachyderma (Ehrenberg) morphotype M2 showing a curved aperture (175,1085A,31H-2,139-141cm). 2B) Spiral view of the N. pachyderma (Ehrenberg) morphotype M2 showing sinistral coiling with a lobate test (175,1085A,28H-6,79-81cm). 2C) Surface Ultrastructure of the N. pachyderma (Ehrenberg) morphotype M2 showing reticulate surface with high pore density (175,1085A,28H-6,79-81cm). 3A) Apertural view of the N. pachyderma (Ehrenberg) morphotype M3 showing a rounded aperture with a highly compact test (175,1085A,31H-2,139-141cm). 3B) Spiral view of the N. pachyderma (Ehrenberg) morphotype M3 showing sinistral coiling (175,1085A,31H-1,30-32cm). 3C) Surface Ultrastructure of the N. pachyderma (Ehrenberg) morphotype M3 showing pores and a thick polygonal ridges network (175,1085A,31H-1,30-32cm). 4A) Apertural view of the N. pachyderma (Ehrenberg) morphotype M4 showing a small kummerform chamber covering the aperture (175,1085A,31H-2,139-141cm). 4B) Spiral view of the N. pachyderma (Ehrenberg) morphotype M4 showing the small kummerform chamber and a sinistral coiling (175,1085A,28H-6,115-117cm). 4C) Surface Ultrastructure of the N. pachyderma (Ehrenberg) morphotype M4 showing pores and polygonal ridges (175,1085A,28H-6,115-117cm).
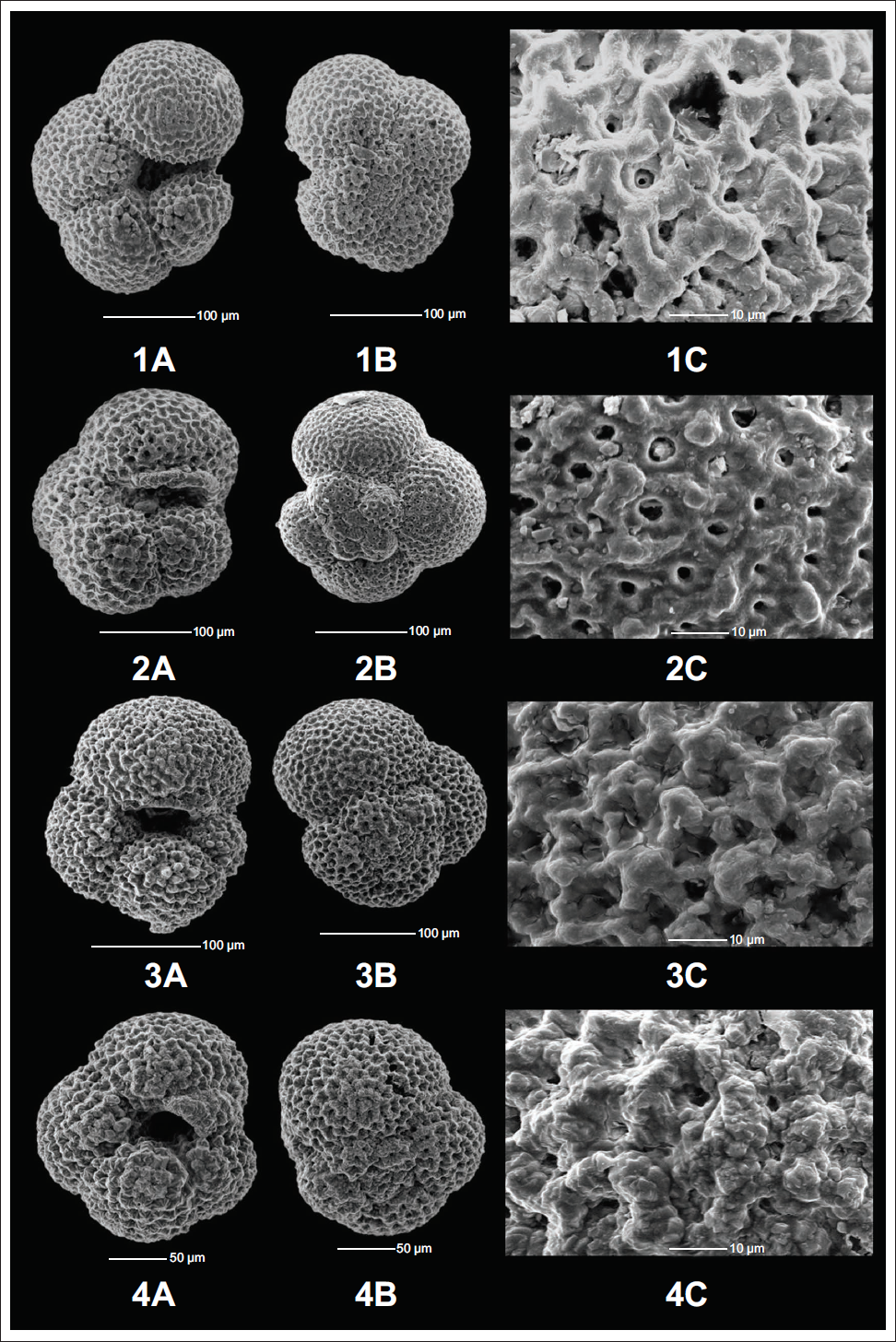
Morphotype II (M2)
M2 shows a test shape similar to that of M1. It has four to four and a half chambers with an X/Y ratio > 1 (normalform) (Table 1), but it shows an entirely different type of aperture. Instead of a narrow slit like aperture in M1, the M2 shows a relatively open, low-arched aperture with a curved end. M2 shows, on average, the largest specimens out of all eight morphotypes, together with an extensive range of variation from 28915.31 µm² to 90835.49 µm² with an average size of 52358.25 µm² (Table 1). It also shows both reticulated and crystallised surface ultrastructure. M2 shows the smallest average pore size of 1.37 µm with the highest average pore density of 0.475 pores per µm² (Table 1). These morphometric measurements support the findings of Moller et al. (2013) and Altuna et al. (2018), which suggest that larger morphotypes are associated with higher pore concentrations. This morphotype also shows the 3rd highest population size in the Southeast Atlantic Ocean (Plate 4, Figure 2A–2C).
Morphotype III (M3)
M3 varies from M1 and M2 in its aperture and has a relatively compact test. Similar to M2, it shows a curved aperture, but the test is too compact, resulting in the aperture being roughly rounded and restricted to the interiomarginal region; that is, it does not arch at the periphery but near the umbilicus itself. M3 shows four to four and a half chambers in the final whorl with the largest X/Y ratio of 1.21 (normalform) (Table 1). The M3 size ranges from 29,769.28 µm² to 54,804.49 µm², with an average of 40,752.34 µm² (Table 1). It shows a crystallised surface ultrastructure with the largest average pore size of 2.21 µm and an average pore density of 0.325 pores per µm² (Table 1). M3 is the 2nd most abundant morphotype of the studied area (Plate 4, Figure 3A–3C).
Morphotype IV (M4)
M4 morphotype shows 4 chambers in the final whorl. However, it contains an additional smaller chamber at the end of the final whorl, which can be partially or fully grown, that is, it shows a kummerform chamber with the lowest X/Y ratio of 0.83 (Table 1). This kummerform chamber covers the aperture of the individual, resembling an umbilical plate. The size of this morphotype ranges from 25,597.96 µm² to 64,637.56 µm² with an average size of 40,624.73 µm² (Table 1). M4 shows both reticulated and crystallised surface ultrastructure depending on the water mass characteristics. The surface ultrastructure study shows an average pore size and average pore density of 1.94 µm and 0.35 pores per µm² (Table 1). M4 is the most abundant morphotype of N. pachyderma in this region (Plate 4, Figures 4A–4C).
Morphotype V (M5)
This morphotype shows the perfect quadrate shape of the N. pachyderma with four chambers in the final whorl, and the final chamber is roughly similar in size to the penultimate chamber, with an X/Y ratio of 0.96, resulting in the quadrate periphery (Table 1). M5 shows a size range of 25,859.26 µm² to 76,461.04 µm², with an average of 42,286.86 µm² (Table 1). Furthermore, the surface ultrastructure study of M5 shows both reticulated and crystallised surfaces with an average pore size and density of 2.13 µm and 0.325 pores per µm², respectively (Table 1) (Plate 5, Figures 5A–5C).
5A) Apertural view of the N. pachyderma (Ehrenberg) morphotype M5 showing a perfect quadrate-shaped test (175,1085A,28H-6,79-81cm). 5B) Spiral view of the N. pachyderma (Ehrenberg) morphotype M5 showing sinistral coiling and a quadrate test (175,1085A,31H-2,139-141cm). 5C) Surface Ultrastructure of the N. pachyderma (Ehrenberg) morphotype M5 showing pores and polygonal ridges (175,1085A,31H-2,139-141cm). 6A) Apertural view of the N. pachyderma (Ehrenberg) morphotype M6 showing a dent-like deformation of the final chamber (175,1085A,27H-2,42-44cm). 6B) Spiral view of the N. pachyderma (Ehrenberg) morphotype M6 showing sinistral coiling with the deformed final chamber (175,1085A,31H-1,30-32cm). 6C) Surface Ultrastructure of the N. pachyderma (Ehrenberg) morphotype M6 showing reticulate surface with high pore density (175,1085A,31H-1,30-32cm). 7A) Apertural view of the N. pachyderma (Ehrenberg) morphotype M7 showing a 5-chambered test (175,1085A,31H-2,139-141cm). 7B) Spiral view of the N. pachyderma (Ehrenberg) morphotype M7 showing sinistral coiling with 5 chambers (175,1085A,34X-2,106-108cm). 7C) Surface Ultrastructure of the N. pachyderma (Ehrenberg) morphotype M7 showing pores and a well-formed network of polygonal ridges (175,1085A,34X-2,106-108cm). 8A) Apertural view of the N. pachyderma (Ehrenberg) morphotype M8 showing an overgrown final chamber covering the aperture (175,1085A,34X-3,17-19cm). 8B) Spiral view of the N. pachyderma (Ehrenberg) morphotype M8 showing sinistral coiling (175,1085A,31H-2,139-141cm). 8C) Surface Ultrastructure of the N. pachyderma (Ehrenberg) morphotype M8 showing pores and thick polygonal ridges (175,1085A,31H-2,139-141cm).

Morphotype VI (M6)
M6 has four to four and a half chambers in the final whorl, with the final chamber being aberrant. The presence of such an aberrant chamber is observed in a large population, ruling out the possibility that this is ‘random’ and suggesting that these are a separate morphotype altogether. The abnormal final chamber resembles a dent-like shape, sometimes resulting in a restricted aperture. The degree of this abnormality can vary, but the type was found to be similar in all individuals. This aberrant final chamber results in an X/Y ratio close to 1, suggesting a quadrate shape (Table 1). However, the final chamber is actually larger than the penultimate chamber, and the quadrate shape is due to the abnormal chamber. We suggest the term ‘pseudo-quadrate’ for this variety of specimens. M6 shows the smallest average size of 33,641.91 µm², ranging from 16,994.02 µm² to 69,089.16 µm² (Table 1). Additionally, the surface ultrastructure study shows both reticulated and crystallised surfaces with an average pore size of 1.66 µm and an average pore density of 0.4 pores per µm² (Table 1) (Plate 5, Figures 6A–6C).
Morphotype VII (M7)
This is the only morphotype with five chambers in its final whorl, with the ultimate chamber larger than the penultimate, and an X/Y ratio of 1.1; that is, it is a normalform variety with a low slit or arched-shaped aperture (Table 1). The size of M7 varies from 30,037.15 µm² to 85,616.78 µm² with an average of 50,844.73 µm² (Table 1). The surface ultrastructure study shows that M7 varies its surface from reticulated to crystallised with changes in water mass characteristics, with an average pore size and pore density of 2.01 µm and 0.325 pores per µm², respectively (Table 1). M7 can be distinguished from the other members of Neogloboquadrina as it shows a closed umbilicus in contrast to the relatively open umbilicus of N. dutertrei, N. acostaensis and N. humerosa (Plate 5, Figures 7A–7C).
Morphotype VIII (M8)
M8 morphotype shows 4 chambers in the final whorl, with the final chamber being larger than the penultimate, with the X/Y ratio of 1.15 (Table 1), but it also shows an overgrowth of calcite. Unlike M4, which shows a kummerform chamber covering the aperture, M8 shows signs of heavy calcification and an overgrown final chamber, which can be fused with the first chamber of the final whorl in some individuals. In contrast, in others, it can be partially fused, resulting in a restricted aperture. M8 shows an average size of 41,687.94 µm² with a size range of 30,079.48 µm² to 58,915.96 µm² (Table 1). The surface ultrastructure shows prominent polygonal ridges with heavy calcification, indicating a heavily crystallised surface. It shows an average pore size of 1.56 µm (Table 1). However, due to the much larger and heavily calcified polygonal ridge, it results in the lowest pore density of 0.275 pores per µm² (Table 1) out of all the morphotypes of N. pachyderma (Plate 5, Figures 8A–8C).
CONCLUSIONS
In this study, the morphological variability of N. pachyderma (Ehrenberg) was examined along with the surface ultrastructure of the specimens from ODP Hole 1085A in the SBR of BUS in the South Atlantic Ocean. By performing morphometric analysis and a surface ultrastructure study, eight distinct morphotypes were identified. These morphotypes were categorised by variations in chamber arrangement, kummerform-normalform variety, compactness, test encrustation, and aperture type. All eight morphotypes were consistently observed throughout ODP hole 1085A, from the Late Miocene to the Recent. M6 shows a deformed ultimate chamber, but all the individuals assigned to the M6 show a similar type of deformation and are present in a significant abundance. This deformed final chamber also resulted in its X/Y ratio being ~1, and the name pseudo-quadrate has been assigned to it. Five of the morphotypes (M2, M4, M5, M6, and M7) exhibit both reticulate and crystalline surface ultrastructure, reflecting the effects of changing water mass characteristics.
These findings contribute to a more refined classification of the N. pachyderma morphotypes in the Southeast Atlantic Ocean and highlight the potential of morphotype-based studies for reconstructing the palaeoceanographic conditions. By integrating morphometric analysis with surface ultrastructure, this study provides a framework for further investigations into the ecological and palaeoceanographic significance of N. pachyderma across different oceanic settings.
Footnotes
Acknowledgements
DC thanks the Joint CSIR-UGC JRF (NTA ref no. 231610004628) in the form of a Junior Research Fellowship. DC, AKS and DKS thank the Department of Geology and the Delhi School of Climate Change and Sustainability, IoE, University of Delhi, for infrastructural support. AS thanks the Department of Geology, Mohanlal Sukhadia University. Thanks to IODP for providing deep-sea core samples. DC and AKS conceptualised the research problem and prepared the manuscript. DKS and AS contributed to drafting the manuscript and provided valuable feedback.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data Availability
Data are available as a supplementary file in the online version of the MS.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was funded under the Palaeoclimate Programme of the Ministry of Earth Sciences, Government of India (Sanction No. MoES/CCR/Paleo-4/2019).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
