Abstract
After being recovered from the Central Indian Ocean, core ABP-06, Station II, underwent a thorough micropaleontological analysis. Forty well-preserved radiolarian species were discovered and recorded from a total of 40 sediment samples. Each species’ taxonomy and vertical distribution of the core are thoroughly described in the paper. Two radiolarian biozones—the
INTRODUCTION
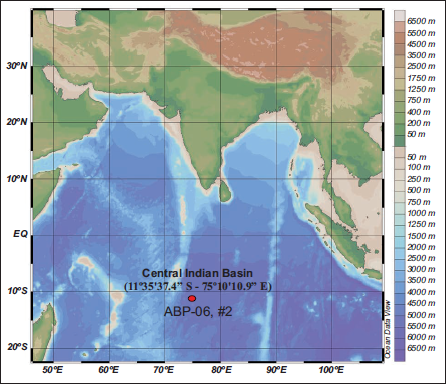
The study on late Cenozoic Radiolaria in the Central Indian Ocean is very scanty. The core ABP-06, Station II, is located between Latitude 11°35.374’ S and Longitude 75°10.109’ E, and samples were collected at a depth of 5179 m (Figure 1). Lithologically, the core consists of fine-grained clayey material, mainly siliceous ooze, with off white lensoid patches (Figure 2). Riedel originally laid the fundamental foundation for the natural classification of radiolarians in 1957. A significant turning point in micropaleontological study was reached 10 years later when Nigrini (1967) provided the first evidence of radiolarian dispersion in pelagic sediments from the Indian Ocean. Our knowledge of Neogene radiolarian biostratigraphy in the low-latitude Indian Ocean zones has been dramatically enhanced by later research. Riedel and Sanfilippo (1974), Sanfilippo and Riedel (1974), Gupta (1988, 1991), Johnson et al. (1989), Haslett et al. (1995, 2004), Nigrini (1971), Caulet (1979, 1986), Johnson and Nigrini (1980, 1982), and, more recently, Pandey et al. (2016), are important contributors in this field. Morley (1989) contributed important insights into the palaeoceanographic history of the southern Indian Ocean. Extensive research on radiolarian taxonomy and the development of biostratigraphic zones within the Indian Ocean was conducted by Sharma and Sharma (1988, 1989), Gupta and Srinivasan (1992), Nigrini (1974, 1991), and Gupta et al. (2002). To strengthen the link between radiolarian data and meteorological interpretations, Gupta (2003) examined how changes in radiolarian assemblages at the orbital scale affected the severity of the Indian summer monsoon. Furthermore, Rogers and De Deckker (2011) used radiolarians as paleoenvironmental proxies to reconstruct oceanographic and climatic conditions in the Indian Ocean throughout the past 40,000 years.
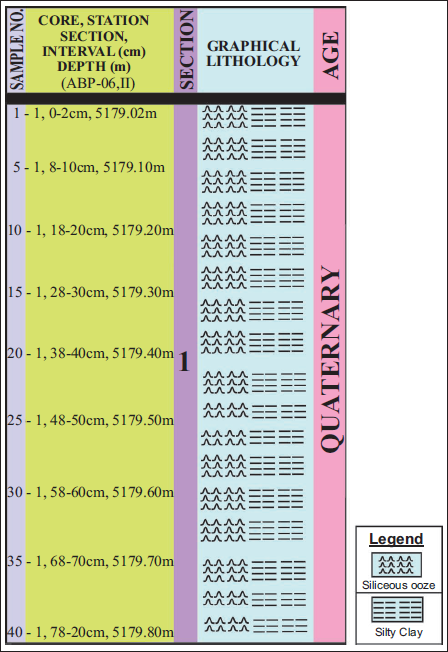
. Litho-column showing age, position of the sample, and depth of the core ABP-06.
MATERIALS AND METHODS
The core ABP-06, Station II, Section I, is 80 cm thick, and 40 samples (from ABP-06, II, 00-02, 5179 m to ABP-06, II, 78-80) of depth 5179.80 m were used to carry out this study. Each sample is taken at an interval of 2 cm. About 3–4 gm of the sample is soaked in water and an added disaggregating agent, that is, hydrogen peroxide (H2O2), to oxidise organic debris and facilitate physical disintegration. The samples are then boiled for an additional 4–5 hours and subsequently washed with water through a 63-micron mesh stainless-steel sieve, followed by drying. Dry residual sediment is sprinkled over the slide and covered with a cover slip using Canada balsam as a mounting medium. Four strewn slides are prepared for each sample for taxonomy and identification. The picked slide was also prepared for the precise marking of zones. For an accurate study, approximately 1000 radiolarian taxa were taken from the total radiolarian population of each sample.
TAXONOMY
The present study follows the classification proposed by Riedel (1967) and Sanfilippo and Riedel (1980) for the subclass Radiolaria. Distribution, abundance, and remarks are mentioned in each of the identified taxa. Semi-quantitative estimates of radiolarian abundance are designated as: Phylum: Sarcodina Hertwig and Lesser, 1874 Class: Actinopoda Calkins, 1909 Sub-class: Radiolaria Müller, 1858 Order: Polycystina Ehrenberg, 1838 emend Riedel, 1967 Sub-order: Spumellaria Ehrenberg, 1875 Family: Collosphaeridae Müller, 1858 Genus: (Plate 1, Figure 1)
1887
1979
Distribution: The species
Remarks: No remarks.
Abundance: Very rare to rare.
Age: Quaternary (mikrotax.org)
(Plate 1, Figure 2)
1887
1980
Biozonations of the core ABP-06, Station II, Section 1, showing comparison of zones (Berggren et al., 1985).

Distribution: Nigrini (1967) observed scanty distribution of
Remarks: No remarks.
Abundance: Very rare to few.
Age: Quaternary (mikrotax.org)
(Plate 1, Figure 3)
1887
1979
Distribution: Haeckel (1887) reported
Remarks: Based on morphological similarities, Riedel and Sanfilippo (1971) proposed
Abundance: Very rare to few.
Range: Miocene to Pliocene (mikrotax.org).
(Plate 1, Figure 4)
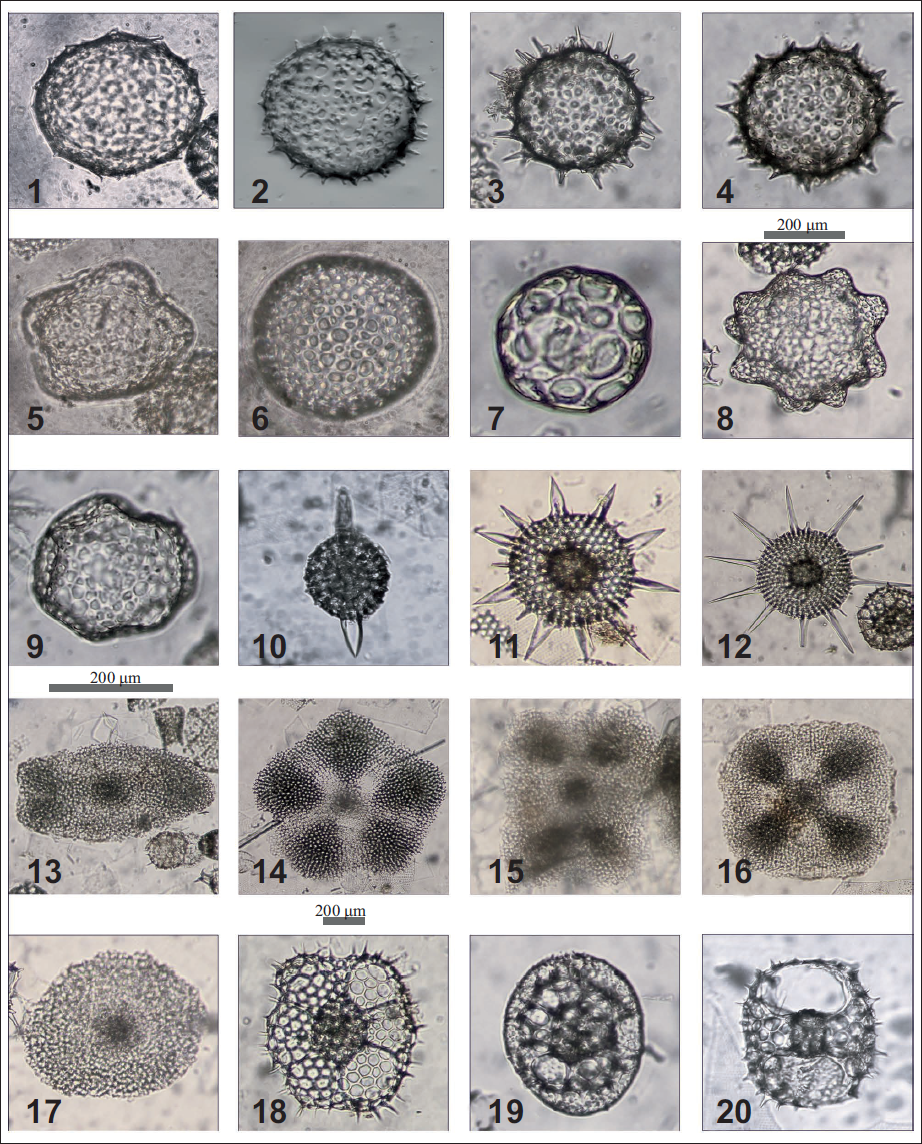
Plate 1. 1. Acrosphaera collina -Haeckel, focused on surface; 2. Acrosphaera lappacea Haeckel, focused on the periphery; 3. Acrosphaera spinosa echinoides (Haeckel), focused on spines; 4. Acrosphaera spinosa spinosa (Haeckel), focused on spines; 5. Buccinosphaera invaginata Haeckel, focused on surface; 6. Collosphaera sp., focused on surface; 7. Collosphaera macropora Popofsky, focused on surface; 8. Collosphaera orthoconus (Haeckel), focused on the periphery; 9. Collosphaera tuberosa Haeckel, focused on surface; 10. Stylatractus sp . Hays, focused on surface; 11. Heliodiscus asteriscus Haeckel, focused on surface; 12. Heliodiscus echiniscus Haeckel, focused on surface; 13. Spongaster berminghamii Campbell and Clark, focused on the surface; 14. Spongaster pentas Riedel and Sanfilippo, focused on surface; 15. Spongaster tetras Ehrenberg irregularis Nigrini, focused on surface; 16. Spongaster tetras tetras Ehrenberg focused on the surface; 17. Spongodiscus ambus Sanfilippo and Riedal, focused on surface; 18. Larcospira quadrangula Haeckel group, focused on surface; 19. Larcopyle buetschlii Dreyer, focused on surface; 20. Tetrapyle octacantha Müller, focused on the surface.
1862
1979
Distribution: Nigrini (1967) showed a sparse presence of
Remarks: Thin spines are present over a conical base, which are shorter than those of the
Abundance: Rare to few.
Age: Quaternary (mikrotax.org)
Genus: (Plate 1, Figure 5)
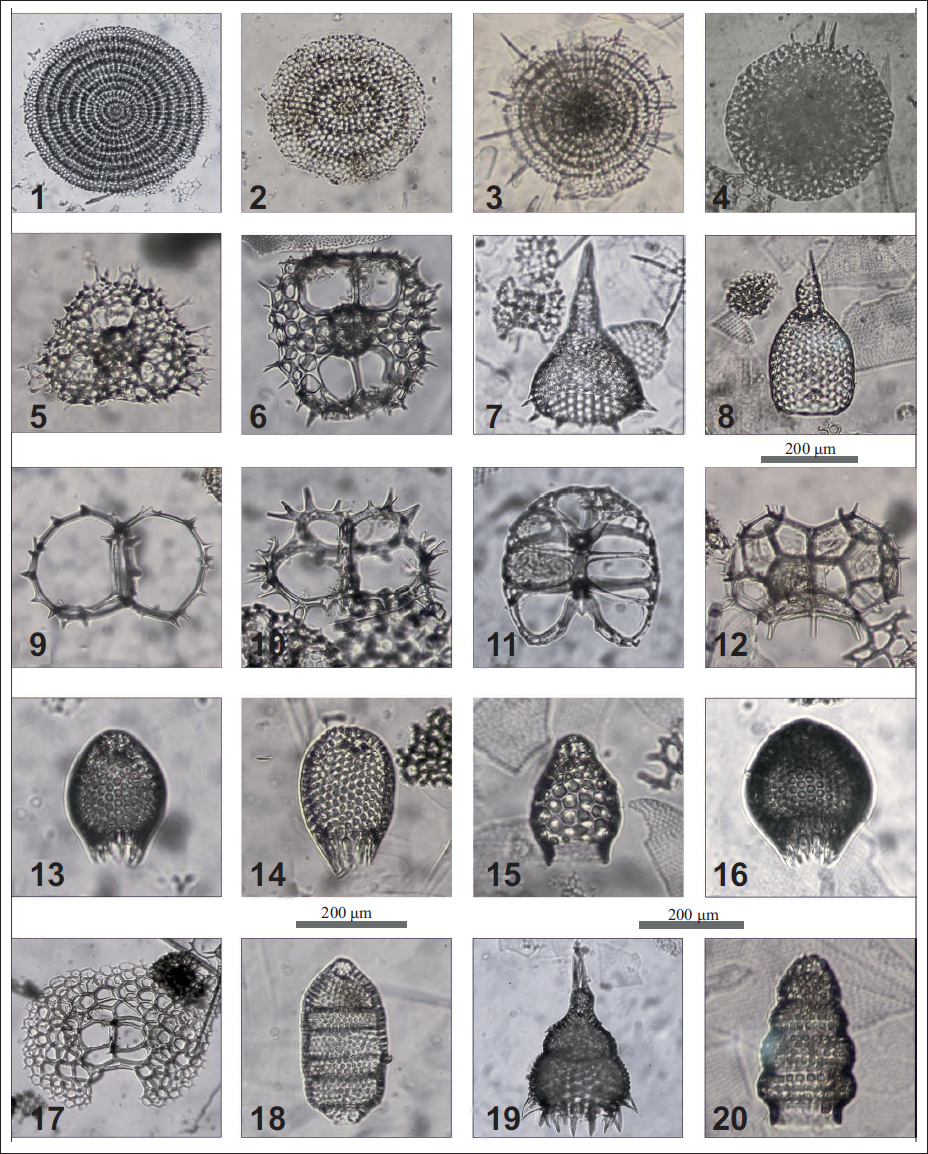
PLATE 2. 1. Stylodictya validispina Jӧrgensen, focused on surface; 2. Circodiscus ellipticus (Stoehr), focused on surface; 3. Stylochlamydium asteriscus Haeckel, focused on surface; 4. Spongopyle osculosa Dreyer, focused on the periphery; 5. Hexaplye dodecantha Haeckel, focused on surface; 6. Octopyle stenozona Haeckel, focused on surface; 7. Anthocyrtidium ophirense (Ehrenberg), focused on shell outline; 8. Anthocyrtidium zanguebaricum (Ehrenberg), focused on shell outline; 9. Giraffospyris angulata (Haeckel), focused on surface; 10. Giraffospyris circumflexa Goll, focused on spines; 11. Liriospyris reticulata (Ehrenberg), focused on surface; 12. Lophospyris pentagona pentagona (Ehrenberg) emend. Goll, focused on the surface; 13 Carpocanistrum spp ., focused on pores; 14. Carpocanistrum sp A. , focused on surface; 15. Carpocanarium papillosum (Ehrenberg), focused on pores; 16. Carpocanopsis cristata , focused on pores; 17. Nephrospyris renilla renilla , focused on surface; 18. Eucyrtidium acuminatum (Ehrenberg), focused on the periphery; 19. Lamprocyclas maritalis maritalis Haeckel, focused on shell outline; 20. Botryostrobus auritus/australis (Ehrenberg), focused on shell outline.
1887
2001
Distribution: Its wide yet selective range is demonstrated by the fact that
Remarks: Its first morphotypic appearance makes the base of the youngest Quaternary zone of
Abundance: Very rare to rare.
Age: Uppermost Quaternary (Sanfilippo et al., 1985).
Genus: (Plate 1, Figure 6)
Description: Large, single, thick shell with smooth surface, rounded to sub-rounded pores.
Distribution: Collosphaera sp. reported in the Central Indian Ocean.
Remarks: Showed its presence for the first time in the region.
Abundance: Very rare to rare.
Age: Quaternary (Blueford,1984).
(Plate 1, Figure 7)
1917
1980
Distribution: Johnson and Nigrini (1980, 1982) and Munir et. al (2020) observed the rare occurrence of C. macropora in the Indian Ocean.
Remarks: No remarks.
Abundance: Very rare to rare.
Age: Quaternary (mikrotax.org)
(Plate 1, Figure 8)
1887 C. orthoconus n. sp.-Haeckel, p. 221, Plate 12, Figure 2.
1979 C. orthoconus-(Haeckel) Bjørklund and Goll, p. 1317.
Distribution: Alexandrovich (1989) documented the presence of C. orthoconus in the eastern equatorial Pacific.
Remark: C. orthoconus is restricted to the base of C. tuberosa (Knoll & Johnson, 1975).
Abundance: Very rare to rare.
Range: Miocene-Pliocene (mikrotax.org)
(Plate 1, Figure 9)
1887 Collosphaera tuberosa-Haeckel, p. 97.
2001 Collosphaera tuberosa-Haeckel, Nigrini and Sanfilippo, p. 209.
Distribution: Bjørklund and Goll (1979) and Johnson and Nigrini (1980, 1982) reported C. tuberosa from the Pacific and the Indian Ocean, respectively.
Remarks: Nigrini and Sanfilippo (2001) showed its morphotypic first appearance as synchronous with the lower limit of the Collosphaera tuberosa zone.
Abundance: Very rare to rare.
Range: Late Quaternary-Recent (Sanfilippo et al., 1985).
Family: Actinommidae (Haeckel) Sanfilippo and Riedel, 1980 Genus: Stylatractus Haeckel, 1887
(Plate 1, Figure 10)
1965
Description: The authors included those specimens that have one longer, cylindroconical, non-bladed, and the other smaller, bladed polar spine.
Distribution:
Remarks: Similar to what is described by Hays (1965).
Abundance: Very rare.
Age:
Family: Phacodiscidae Haeckel, 1881 Genus:
(Plate 1, Figure 11)
1887
2025
Distribution: Johnson and Nigrini (1980, 1982) and Nigrini and Lombari (1984) reported
Remarks: Morley (1977) considered
Abundance: Very rare to few.
Age: Early Quaternary (mikrotax.org)
(Plate 1, Figures 12)
1887
1983
Distribution: Nigrini (1967) showed a sparse distribution of
Remarks: Morley (1977) considered
Abundance: Very rare to few.
Age: Quaternary (mikrotax.org)
Family: Spongodiscidae (Haeckel) Riedel, 1967 Genus: (Plate 1, Figure 13)
1944
1978
Distribution:
Remarks:
Abundance: Very rare.
Range: Late miocene to early pliocene (Sanfilippo et al., 1985).
(Plate 1, Figure 14)
1970
1984
Distribution: Sanfilippo et al. (1985) reported the occurrence of
Remarks:
Abundance: Very rare.
Age: Middle Pliocene (Sanfilippo et al., 1985).
(Plate 1, Figure 15)
1967
2020
Distribution: Haeckel (1887) found
Remarks:
Abundance: Very rare to rare.
Range: Early Miocene to Late Pliocene (mikrotax.org)
(Plate 1, Figure 16)
1861
1967
Distribution: Johnson and Nigrini (1980, 1982) reported it in the Indian Ocean.
Remarks: No remarks
Abundance: Rare to few.
Range: Pliocene-Recent (Sanfilippo et al., 1985).
Genus: (Plate 1, Figure 17)
1974
1984
Distribution: Sanfilippo and Riedel (1974) reported this species in the western Indian Ocean.
Remarks: No remarks.
Abundance: Very rare to rare.
Age: Lower Pliocene (Sanfilippo & Riedel, 1974).
Genus: (Plate 2, Figure 1)
1905
1991
Distribution: Nigrini and Lombari (1984) reported it from Miocene sections of tropical and temperate regions.
Remarks: Sachs (1973) and Robertson (1975) considered it a cold-water species.
Abundance: Very rare to few.
Age: Early Miocene (Nigrini & Lombari, 1984).
Genus (Plate 2, Figure 2)
1880
1975
Distribution: Chen and Tan (1996) showed its presence in the South China Sea (15°N).
Remarks: No remarks.
Abundance: Very rare to rare.
Age: Late Miocene (Abelmann, 1990).
Genus:
(Plate 2, Figure 3)
1887
1979
Distribution: Benson (1983) reported it in the Southern Ocean.
Remarks: Similar to what is described by Nigrini and Moore (1979).
Abundance: Very rare to few.
Age: Quaternary (mikrotax.org).
Genus:
(Plate 2, Figure 4)
1889
1990
Distribution: Riedel (1958) and Benson (1966) reported it as cosmopolitan.
Remarks: No remarks.
Abundance: Very rare to rare.
Age: Quaternary (mikrotax.org).
Family: Pyloniidae Haeckel, 1881 Genus:
(Plate 2, Figure 5)
1887
1981
Distribution: Haeckel (1887) and Benson (1966) reported
Remarks: Similar to what is described by Takahashi and Honjo (1981).
Abundance: Very rare to rare.
Age: Quaternary (mikrotax.org).
Genus:
(Plate 2, Figure 6)
1887
2024
Distribution: Benson (1966) and Gupta (1999) reported
Remarks: Very similar to what is described by Haeckel (1887).
Abundance: Rare to Few.
Age: Quaternary (mikrotax.org).
Genus:
(Plate 1, Figure 20)
1858
1987 Tetrapyle octacantha-Müller, Dworetzky & Morley, Plate 2, Figure 1
Distribution: Sachs (1973) and Robertson (1975) considered
Remark: Very similar to what is described by Müller (1858).
Abundance: Few.
Age: Quaternary (mikrotax.org).
Family: Litheliidae Haeckel, 1862 Genus: (Plate 1, Figure 19)
1889
1984
Distribution: Benson (1966) considered cosmopolitan and upwelled taxa.
Remarks: No remarks.
Abundance: Very rare to rare.
Age: Quaternary (mikrotax.org).
Genus
(Plate 1, Figure 18)
1887
1984
Distribution: Haeckel (1887) reported this species from the central Pacific.
Remarks: No remarks.
Abundance: Very rare to few.
Age: Quaternary (mikrotax.org).
Family
Genus: (Plate 2, Figure 9)
1887
1969
Distribution
Remarks: No remarks.
Abundance: Very rare to few.
Range: Late Miocene to Recent (Goll, 1972).
(Plate 2, Figure 10)
1969
1984
Distribution: Nigrini and Lombari (1984) showed its abundance in tropical latitudes.
Remarks: No remarks.
Abundance: Very rare to few.
Range: Miocene to recent (Mikrotax.org).
Genus: (Plate 2, Figure 11)
1872
1968
Distribution: Nigrini (1967) observed the presence of
Remarks: No remarks.
Abundance: Very rare to few.
Range: Middle Miocene-Quaternary (Goll, 1968).
Genus: (Plate 2, Figure 12)
1872
1976
Distribution: Nigrini (1967) observed a sparse presence of
Remarks: Nigrini (1967) and Goll (1976) considered it a cosmopolitan species.
Abundance: Very rare to rare.
Range: Miocene to Quaternary (mikrotax.org).
Genus
(Plate 2, Figure 17)
1887
1980
Distribution: Boltovskoy (1998) considered
Remarks:
Abundance: Very rare.
Age: Quaternary (Mikrotax.org).
Family Genus (Plate 2, Figure 13)
1887
1971
Distribution: Nigrini (1970) reported
Remarks: No remarks.
Abundance: Very rare to few.
Age: Quaternary (Microtax.org).
(Plate 2, Figure 14)
1966
1983
Distribution:
Remarks: No remarks.
Abundance: Very rare to few.
Age: Quaternary (Microtax.org).
Genus (Plate 2, Figure 15)
1872
1958
Distribution: Riedel (1958) observed
Remarks: No remarks
Abundance: Rare to few.
Age: Quaternary (mikrotax.org).
(Plate 2, Figure 16)
1908
1971
Distribution: Kamikuri (2022) reported
Remarks: No remarks.
Abundance: Very rare to rare.
Range: Lower Miocene to Upper Pliocene (mikrotax.org).
Family: Theoperidae (Haeckel) Riedel, 1967 Genus: (Plate 2, Figure 18)
1844
1970
Distribution: Nigrini (1967) observed the presence of
Remarks:
Abundance: Very rare to few.
Range: Early Palaeocene to recent (mikrotax.org).
Family: Pterocoryidae (Haeckel, 1881) Riedel, 1967 Genus: (Plate 2, Figure 7)
1872
1967
Distribution: Nigrini (1967,1970) observed
Remarks: No remarks.
Abundance: Very rare to few.
Age: Quaternary (mikrotax.org).
(Plate 2, Figure 8)
1872
1967
Distribution: Nigrini (1967) reported in both low and middle latitudes.
Remarks: No remarks.
Abundance: Very rare to few.
Age: Quaternary (mikrotax.org).
Genus: (Plate 2, Figure 19)
1887
1967
Distribution:
Remarks: Nigrini (1967) and Johnson and Nigrini (1980, 1982) observed
Abundance: Very rare to rare.
Range: Early Miocene to Late Pliocene (mikrotax.org).
Family: Artostrobiidae (Riedel) Foreman, 1973 Genus: (Plate 2, Figure 20)
1844
1977
Distribution: Popofsky (1913) reported
Remarks: No remarks.
Abundance: Very rare to few.
Age: Quaternary (mikrotax.org).
BIOSTRATIGRAPHY
The radiolarian assemblage of ABP-06, Section II, has typical tropical taxa. Two radiolarian biozones (Figure 3) are established based on morphotypic and evolutionary limits, consistent appearance, and common occurrence of stratigraphically important taxa. The two biozones, namely
Buccinosphaera invaginata Range-Zone (Nigrini, 1971) (=NR1)
Nigrini (1971) described the range zone of
Collosphaera tuberosa Interval-Zone (=NR2/NR3)
Nigrini (1971) defined the top of this zone by the first appearance of
The top of
Further, the
One important upwelled and one cold-water taxa are present:
REWORKING
Within the core samples, four reworked radiolarian species have been found, each with varied stratigraphic importance and abundance levels. Although they are extant,
A COMPARISON: PLANKTONIC FORAMINIFERAL BIOSTRATIGRAPHY
In the studied section, an integrated proposed radiolarian biozonal scheme is compared with the foraminiferal zonal scheme of McGowran (1974). It is observed that
CONCLUSIONS
A total of 26 species of Spumellarians and 14 species of Nassellarians have been identified and systematically described from core ABP-06, Station II, Section I, located in the Central Indian Ocean. Based on the radiolarian assemblages, two distinct biozones have been established, reflecting a tropical marine environment.
In addition to the primary assemblage, four reworked species—dating from the Miocene to Pliocene—are present throughout the core, indicating sedimentary mixing and possible reworking from older geological intervals. The presence of one species typically associated with upwelling systems and cold-water conditions suggests that the region currently experiences, or has historically experienced, upwelling activity and the influence of colder bottom waters.
Biostratigraphic analysis indicates that this section corresponds to the Upper Quaternary, with an estimated age range of approximately 0.18–0.5 million years. This time interval aligns with radiolarian zones NR1 and NR2. Furthermore, these radiolarian zones are biostratigraphically equivalent to the
Footnotes
Acknowledgements
GKS is grateful to Director NCAOR for attending the scientific cruise of RV Akademik Boris Petrov and collecting the samples in the Central Indian Ocean. We are also thankful to Dr D.V. Lazarus for the identification of specific taxa. Deeksha Bohra is grateful to the Government of Uttarakhand for partly providing the fellowship to carry out this work. Thankfully acknowledged to the Head, Department of Geology, Kumaun University, and Prof. Sharma is also thankful to the Head, Department of Geology, University of Delhi, Delhi, for providing the research facilities.
Author’s Contribution
All three authors have contributed to this research work in some way or another.
Data Availability Statement
All the data was generated in the Marine Micropaleontology Lab of the Department of Geology, Kumaun University, Nainital.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent Statements
All three authors have consented to participate in publishing this research paper, and this article does not contain any studies with human or animal participants.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: One of the authors (Mrs D. Bohra) got partial support in the form of a fellowship to carry out this work.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
