Abstract
Coastal margins and their ecological setting are one of the magnificent landforms on globe. The complex interaction among the different biotic and abiotic components through ages has addressed that due to climatic changes the coastal regions, one of the pioneer regions, have been getting affected and this may continue in the future also. The role of microfossils through ages has helped in delineating the past climatic manifestations and global changes and amongst them diatoms also provide a valuable tool for inferring past climate and ecological variability in the dynamic coastal system. In the study, a modern set of 77 samples from different climatic zones along the west coast of India has been studied to gain knowledge about the present-day climate status and record environmentally susceptible diatoms to form a modern analogue for coastal settings. The study shows various associations of planktic and benthic diatoms that hitherto provide precious information about the diatoms and their relation to the climatic regions and ecological status. The change in the frequency of planktic and benthic diatoms inculcate the water levels, pH, salinity changes, tidal variability and trophic status. Hence, the study aims to provide a robust database on diatoms for the various climatic zones from the west coast of India and would inevitably fill knowledge gap in terms of primary productivity for broad implications. The study also aims to provide background for transfer-based function for the reconstruction of long-term palaeoclimatic records for the transition zone of terrestrial and oceanic realms.
Introduction
In the present global climate change scenario, the primary focus is to understand the climatic processes through time and its probable effects on ecology and human culture. In recent years, interest in climate change has increased due to multi-facet reasons. The amount of scientific literature on climate change has increased in tandem, and the rate at which significant articles presenting the findings of climate study are published now in an unimaginable pace than what was done few years back. Presently, the climate manifestations are explored using a broad variety of data types, using multi-proxy records such as ice, lake sediment, pollen/spore, diatoms, dinoflagellate cysts, nannofossils, foraminifera, tree rings, palaeosol, geomorphology and archaeological evidence (Bradley, 1999). Recently, the public interest in the topic has increased, as scientific understanding of the causes of climate change has expanded. The people living today may witness notable increases in the earth’s temperature and sea level, along with decreases in snow cover, sea ice, land ice sheets, glaciers, permafrost, rise of tropical storms and changes in precipitation patterns in many parts of the world at various latitudes and longitudes (IPCC, 2007; Wiltshire et al., 2015). In case the effect of greenhouse gases stabilise in the coming decades, the alterations at current levels would last for centuries and accelerate (Anderson et al., 2007). Climate change can force plant and animal species beyond their geographically feasible ranges, and cause their localised or global destruction, and in the coming future many species are anticipated to suffer this fate as the temperature warms, forcing cold-tolerant species to go to refugia at ever-higher altitudes until they are extinct (Delcourt & Delcourt, 2004; Montoya & Raffaelli, 2010).
Understanding the types of proxy data and the techniques applied in their analysis is the initial step to study previous climates (Bradley, 1999). It is important to recognise the challenges and presumptions with the proxy so that it can be utilised effectively. With this context, it could potentially be able to combine many lines of data into a comprehensive picture of past climatic changes and evaluate ideas regarding the origins of climate change (Bradley, 1999).
It is often observed that the response of vegetation either macro or micro communities (trees to algae) vary, and may possibly represent either critically dependent or least responsive to changes in rainfall and temperature. Changes in climate and resource structure are frequently time transgressive, and reflect lags between climate data and biotic response. It is possible that the local conditions and changes may not always adhere or be closely related to global patterns (Anderson et al., 2007; Davis & Botkin, 1985; Singhvi & Kale, 2010). The lack of long-term data on palaeoclimatic conditions continues to make it obscure to anticipate future climatic change, and the instrumental records are insufficiently low (usually less than 100 years) to make regional predictions of the size and pace of upcoming climatic manifestations (Weckström et al., 2017). The climate change scenario is witnessed in response to biotic community, terrestrial, coastal or marine realms. However, the coastal dynamic to climate change is manifold.
Coastal ecosystems are dynamic systems lying between the land and the ocean and have wide effect in terms of natural disasters and human activities, for example, climate adversities, sea level changes, subsidence, sediment discharge, eutrophication and acidification (Li et al., 2018). The typical examples of coastal environments include mangroves, salt marshes, coral reefs, beaches, estuaries and coastal wetlands and these were all included in the Ramsar Convention’s definition of coastal ecology (Ramsar Convention Secretariat, 2010) as an integral part of sea and land mixed habitats. The coastal ecosystem offers vital benefits that include direct supplies like food and biomaterials, wildlife conservation, carbon sequestration, protection from storm surges and many more to count (Li et al., 2018). In comparison to the terrestrial ecosystems, coastal wetlands have a relatively modest surface area, but they are among the most prolific ecosystems in terms of productivity (Li et al., 2018). Vegetated zones and tidal streams serve as important habitats for many terrestrial and marine species, offering a wide range of wild animals refuge and food supplies, resulting in high biodiversity and distinctive food webs. In order to precisely measure past climatic changes and biotic responses, palaeoecological records of terrestrial such as pollen and tree rings, plant macrofossils (Birks & Birks, 2006) and aquatic (such as diatoms) biota are becoming more and more relevant (Smol et al., 1991; Kilham et al., 1996; Thakur et al., 2019).
Diatoms have been recognised as one of the useful proxies for limnological fluctuations and climate change in the food web (Kilham et al., 1996; Smol & Cumming, 2000; Smol & Stoemer, 2010; Smol et al., 1991). They are microscopic, unicellular algae that have a siliceous cell wall (frustule), making them unique for a variety of applications and serve as a unique proxy for climate change since they are sensitive to a wide range of ecological states. The variations in species abundance within the sediment interval can be used to infer past changes in water chemistry, precipitation and temperature (Smol & Stoemer, 2010; Mutshinda et al., 2013b). These species serve as reliable indicators of the lake’s level, nutrient availability and other ecological variables. They respond to a variety of parameters including precipitation, sun light and wind speed along with upwelling and erosion processes (Wiltshire et al., 2015). They respond rapidly to shifting environmental conditions and are sensitive to a number of biogeochemical components. Any aquatic environment, including ponds, rivers, streams, lakes and coastal areas (such as lagoons, deltas and estuaries), is home to them. The distribution of diatoms is one of the helpful markers in environmental reconstruction on eutrophication, extreme climates, lake acidification and climatic manifestations (Smol & Stoemer, 2010). Diatoms are subject to various climate effects. Diatom species and their growing regions are impacted by changes in the frequency and severity of droughts and floods. Climate also affects the circulation patterns and thermal stratification of lakes and oceans, which change the diversity of diatom species (Bigler & Hall, 2002; Bigler et al., 2002). Their distributions in older sediments (fossils) reveal details on the environment, ecology and climate that prevailed during the past time. The physical, chemical and biological characteristics of the aquatic ecosystem are revealed by the diatom assemblages in the surface sediments. Diatoms are utilised in rapid environmental change as they are widely disseminated, immobile, tiny, covered in siliceous material and well preserved in sediments. The effects of freshwater inflows, sea level change and nutrient availability are all particularly subtle on coastal habitats (Finkl & Makowski, 2017). Understanding the present-day diatom response in the coastal ecosystem with varying climatic zones such as humid, sub-humid to semi-arid regions is essentially required for delineating deep time inferences for climate change. The previous studies mostly focussed on sedimentology, tectonics, water quality, elemental studies but the biotic response, that is, primary productivity based on diatom assemblages, their composition and distribution patterns that are preserved in the sediments and the relationship between these assemblages and climatic changes have not yet been discussed. Climatically, India has presently a diverse climate as per the Koppen-Geiger classification. To establish the changes in the diatom assemblage in different climatic zones in surface sediments, the west coast of India is one of the pioneering coastal domains. By comparing the two coastline regions of India, we can see that the east coast is mostly restricted to a tropical savannah climate, whereas the west coast includes tropical monsoon, tropical savannah, arid steppe hot and arid desert hot climatic regions (Figure 1) according to Koppen-Geiger climate classification (Beck et al., 2018).
The different climatic regions along the west coast of India. Source: Beck et al. (2018).
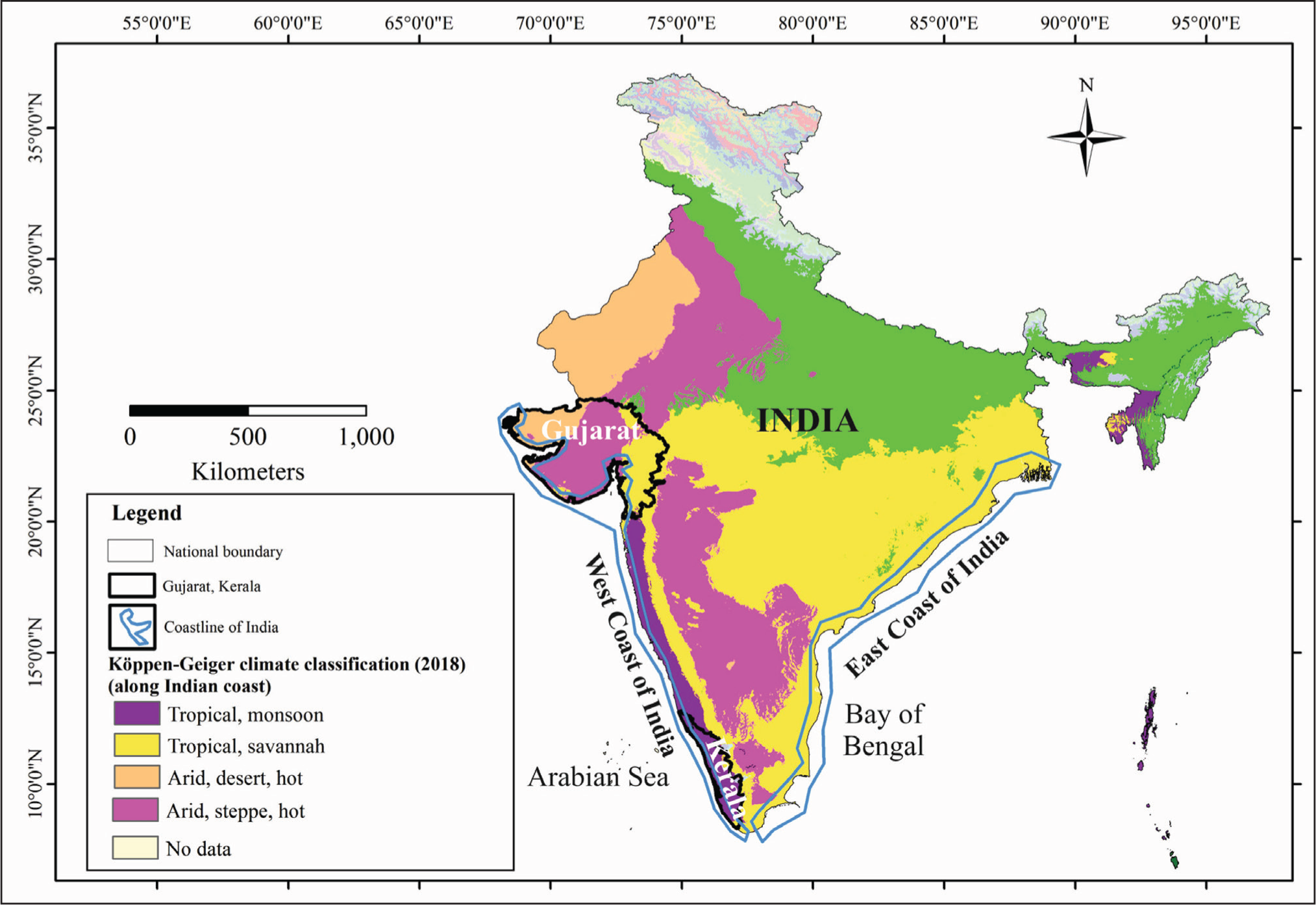
India has more than 7,500 square kilometres of coastline and is very vulnerable to sea-level changes due to climatic manifestations. The Western Coastal Plains span from the Arabian Sea to the Western Ghats between Gujarat and Kerala extending ~1400 km in length, and width ranging from 10 to 80 km. The east coast extends ~1500 km in length. The major features comprise sand beaches, coastal sand dunes, mudflats, lagoons, alluvial tracts, estuaries, lateritic platforms and residual hills.
Surface sediment assessment has received great attention around the world since it is a unique way to assess ecological conditions due to their spatial-temporal accumulations and climatic signatures (Bigler & Hall, 2002; Brush, 2009; Crosta et al., 2012; Costa-Böddeker et al., 2017; Finlayson, 2018; Ip et al., 2004; Manoj et al., 2020; Nodine & Gaiser, 2014). We examined the spatial diatom composition and abundance in surface sediments from various climatic zones of western coastal settings in order to gauge the current ecological status of India’s west coast.
Materials and Methods
For the diatom analysis, 77 surface sediments were processed using HCl (35%) and H
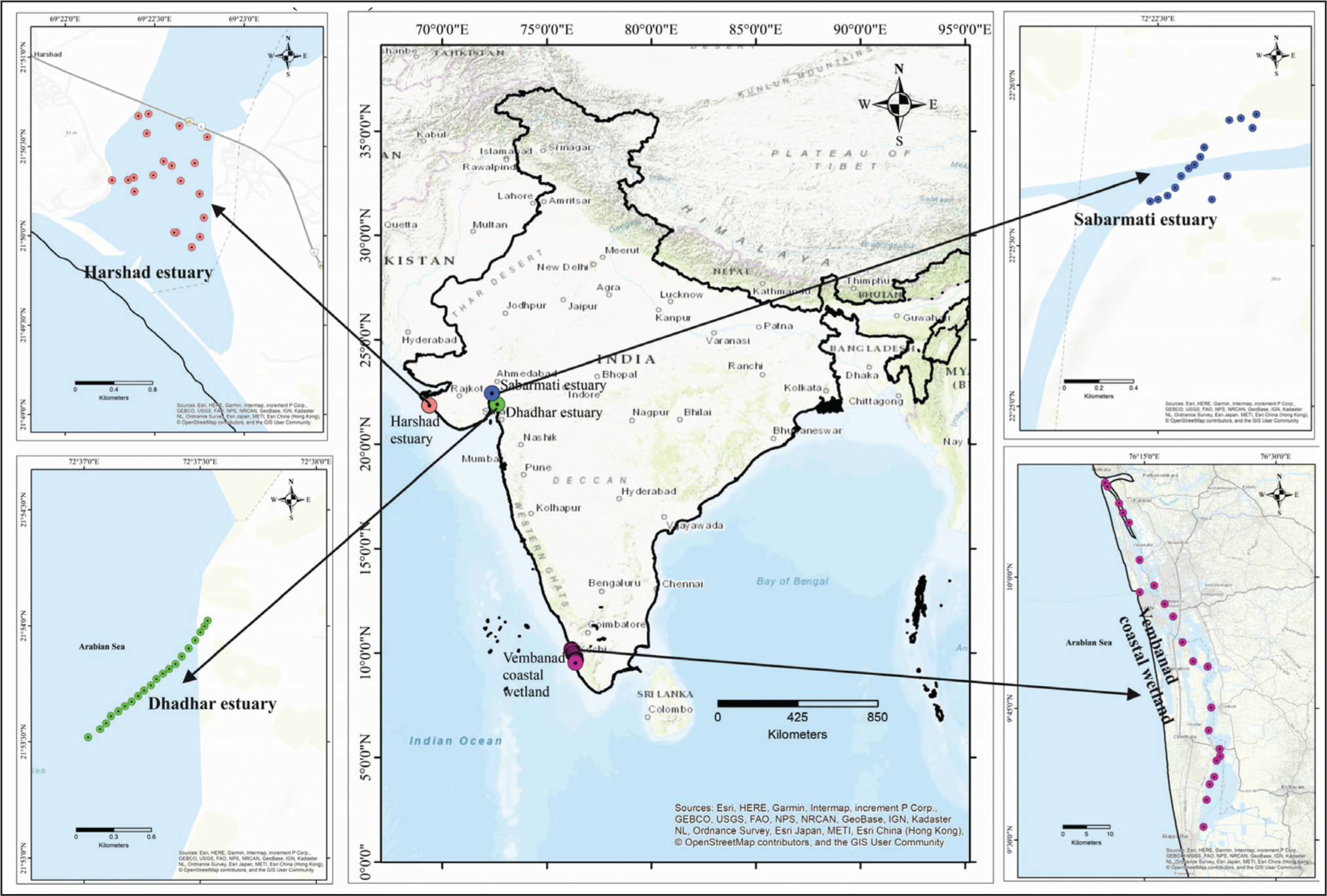
Study Area
Location map of study area along west coast.a
Results
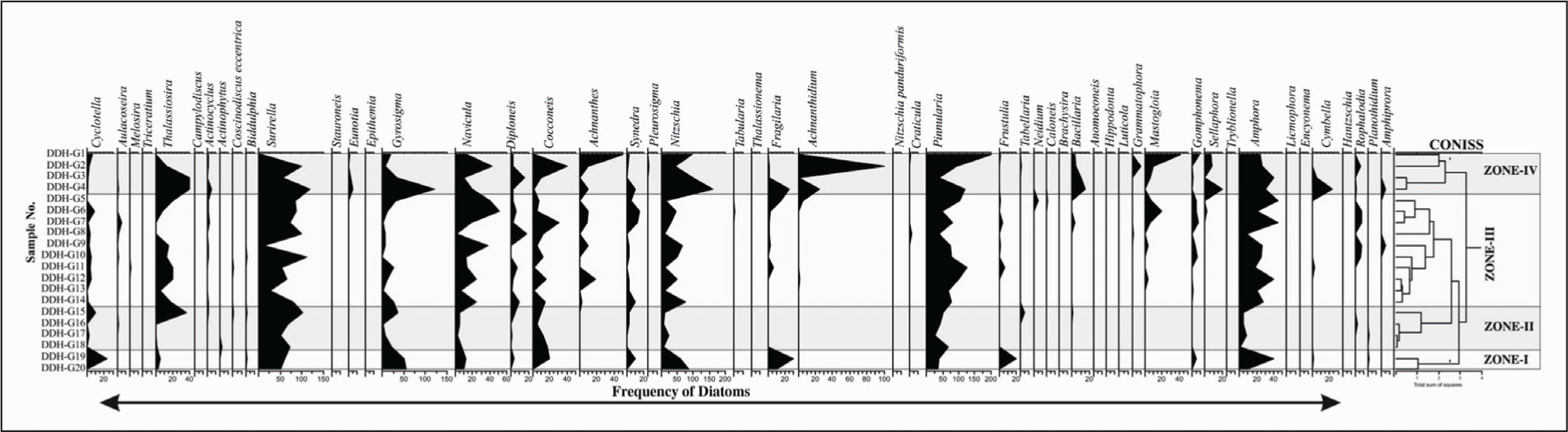
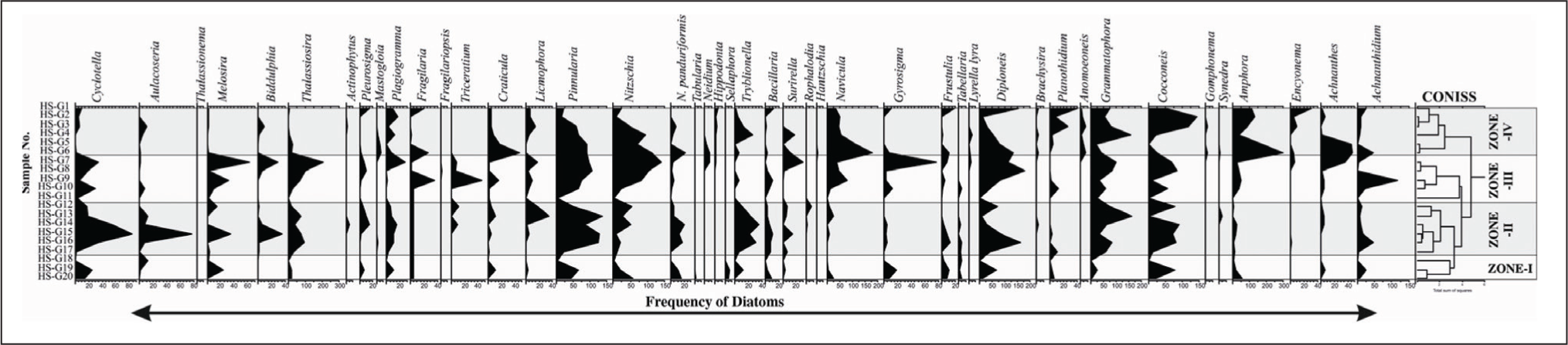
The distribution of diatoms in the various climatic regions of Sabarmati, Harshad, Dhaddhar, and Vembanad along India’s west coast is used as a biological proxy in the present study to ascertain the primary productivity. The frequency of the diatoms was analysed using the TILIA ver.1.7 software that is used to generate the diatom frequency range chart and cluster analysis for each location. For Vembanad coastal wetland (5 zones), Dhadhar estuary (4 zones), Sabarmati estuary (3 zones) and Harshad estuary (4 zones) were identified with CONISS cluster analysis based on dominance and sub-dominance of diatom frequency (Grimm, 1987, 2000).
Vembanad Coastal Wetland
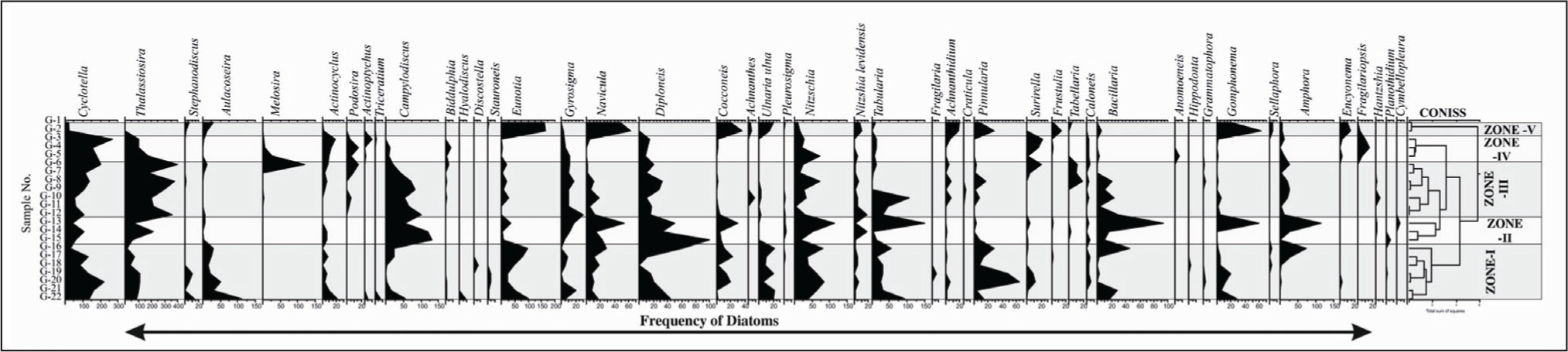
In the present study based on the diatom analysis, five ecological zones are identified (Figure 3 and Plate 1).
Frequency distribution range chart and CONISS cluster analysis of Vembanad coastal wetland.
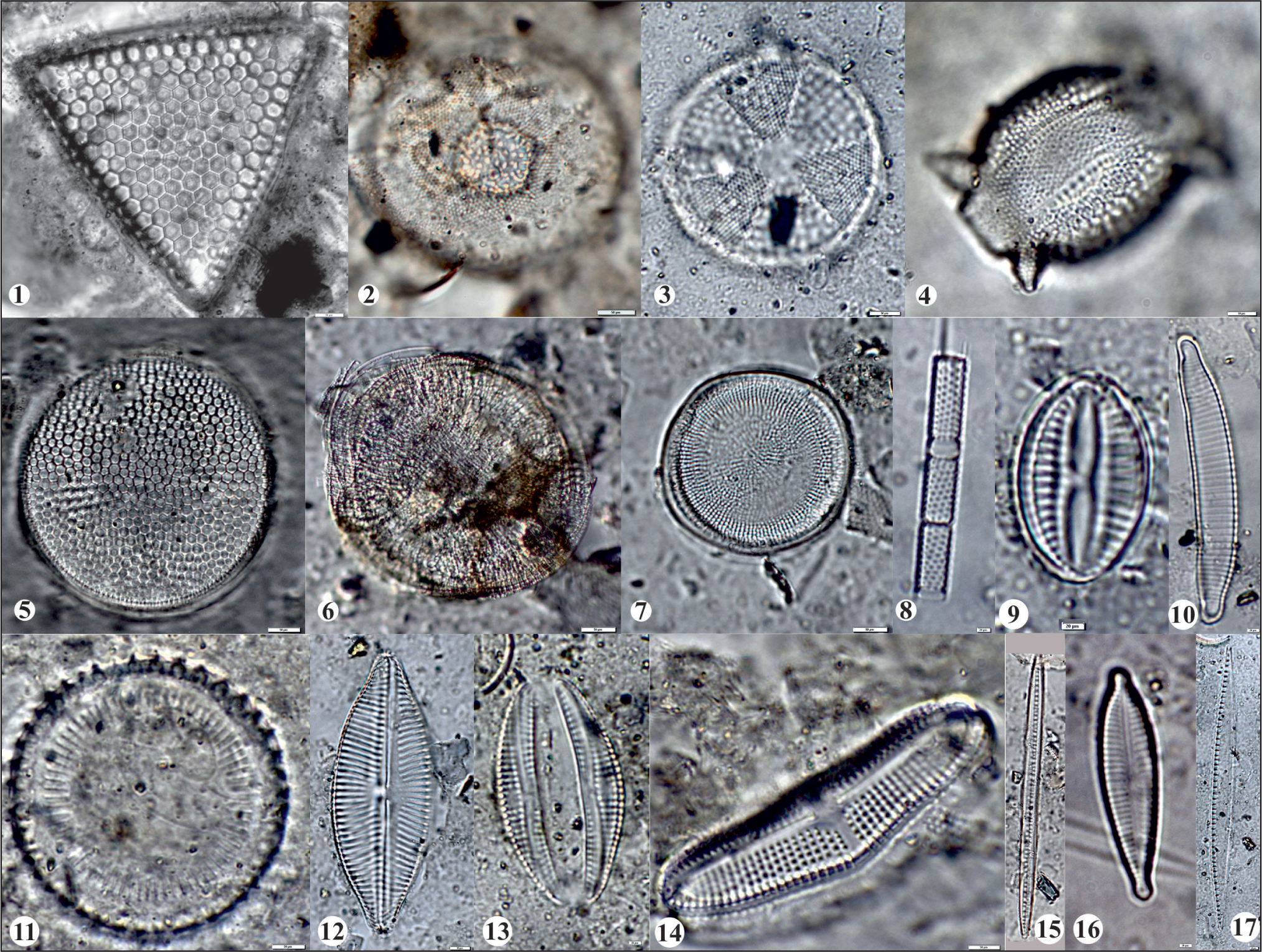
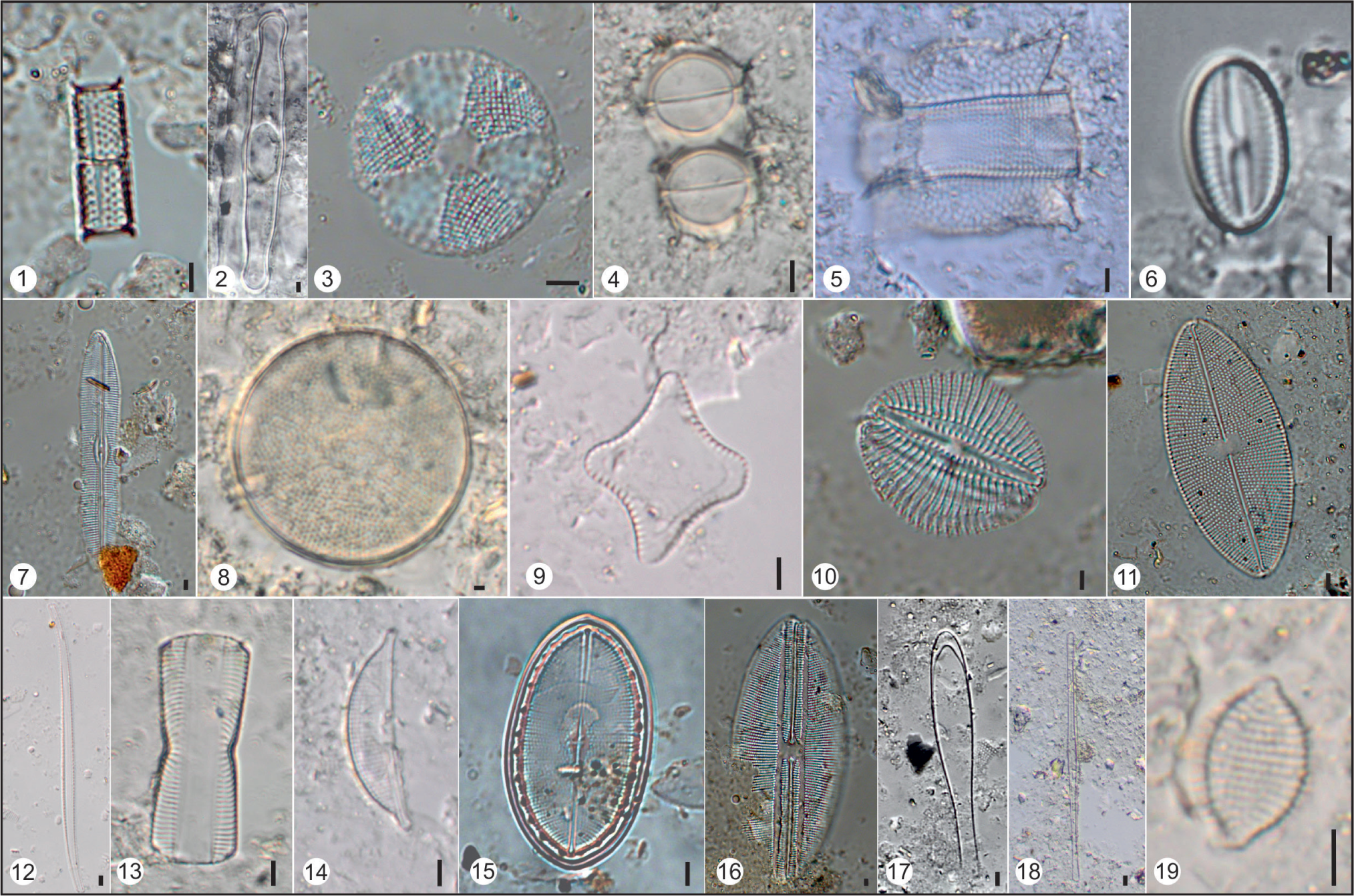
Diatom flora of Vembanad coastal wetland: 1) Triceratium favus; 2) Hyalodiscus sp.; 3) Actinoptychus sp.; 4) Biddulphia sp.; 5) Thalassiosira devious; 6) Campylodiscus clypeus; 7) Actinocyclus normanii; 8) Aulacoseira granulata; 9) Diploneis smithii; 10) Eunotia incisa; 11) Cyclotella striata; 12) Navicula cryptocephala;a 13) Amphora veneta; 14) Achnanthes brevipes; 15) Bacillaria paradoxa; 16) Gomphonema parvulum; 17) Nitzschia amphibian; Scale bar = 50 micron.
Dhadhar Estuary (Figure 4 and Plate 2)
Frequency distribution range chart and CONISS cluster analysis of Dhadhar estuary.
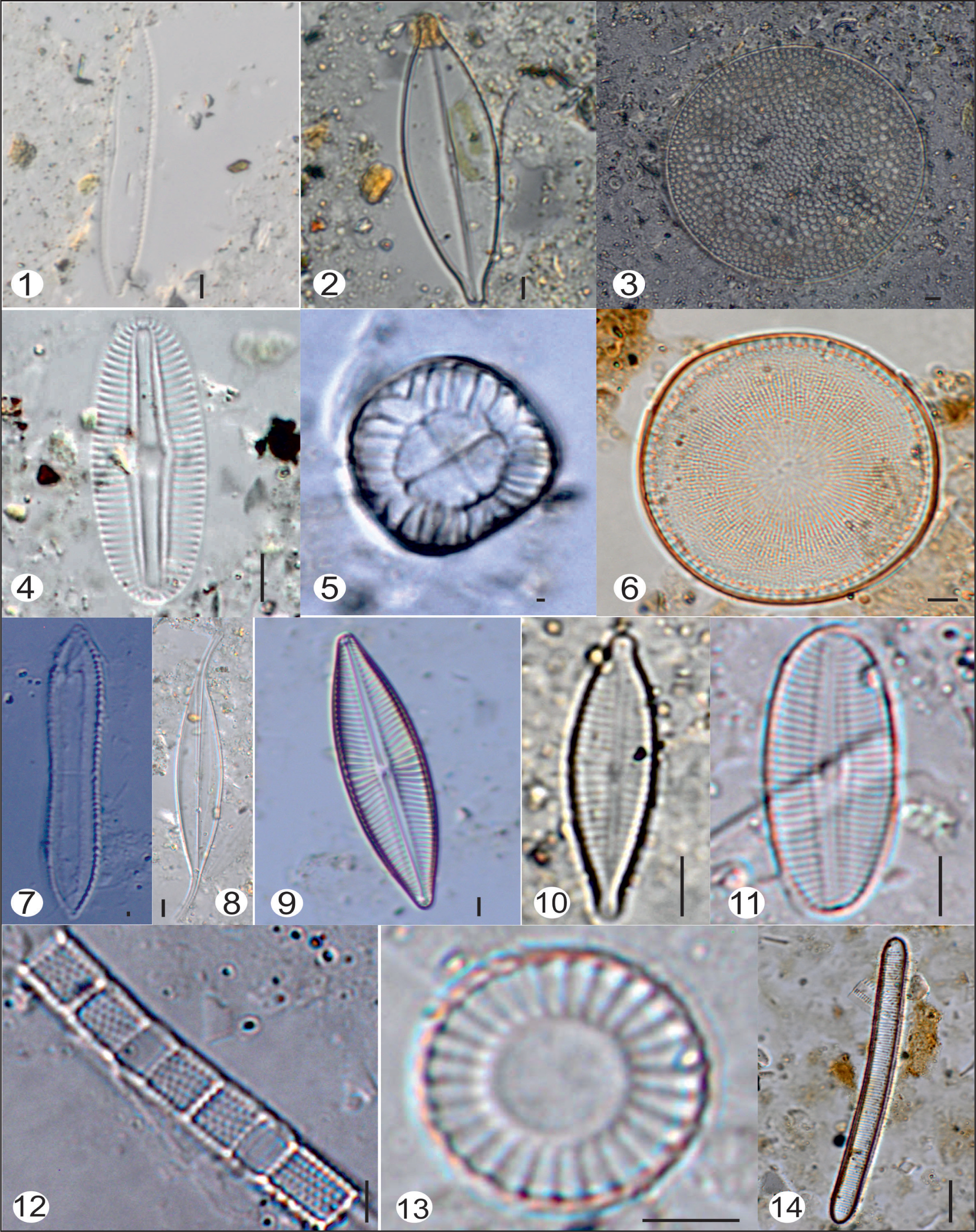
Diatom flora of Dhaddhar: 1) Nitzschia reversa; 2) Navicula sp.; 3) Pinnularia sp.; 4) Surirela tenera; 5) Nitzschia agnita; 6) Amphora veneta; 7) Planothidium sp.; 8) Gyrosigma eximium; 9) Nitzschia linearis; 10) Thalassiosira baltica; 11) Thalassiosira sp.; 12) Cyclotella ocellata; 13) Diploneis crabo; 14) Nitzschia levidensis; 15) Asteromphalus sp.; Scale bar = 5 micron.
Sabarmati Estuary
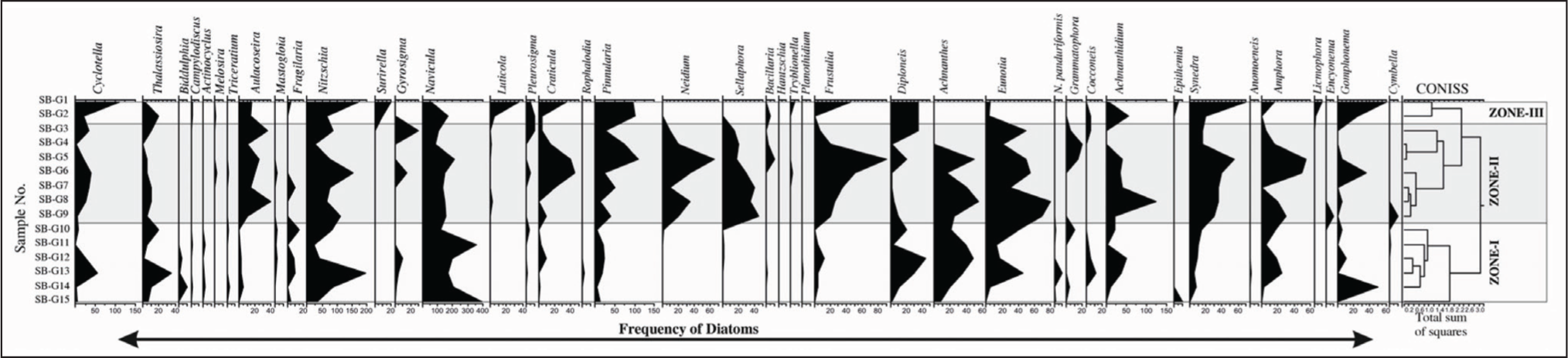
In the Sabarmati coastal region, three zones were identified based on the CONISS cluster analysis (Figure 5 and Plate 3).
Frequency distribution range chart and CONISS cluster analysis of Sabarmati estuary.
Diatom flora of Sabarmati: 1) Nitzschiz palea; 2) Craticula cuspidate; 3) Thalassiosira sp.; 4) & 5) Diploneis litoralis; 6) Campylodiscus sp.; 7) Actinocyclus normanii; 8) Tryblionella gracilis; 9) Nitzschia reversa; 10) Navicula viridula; 11) Gomphonema parvulum; 12) Aulacoseira granulate; 13) Cyclotella meneghiniana; 14) Eunotia bilunaris; Scale bar = 5 micron.
Harshad Estuary
Frequency distribution range chart and CONISS cluster analysis of Harshad estuary.

Diatom flora of Harshad: 1) Aulacoseira granulata; 2) Grammatophora sp.; 3) Actinoptychus senarius; 4) Melosira varians;5) Biddulphia pulchella; 6) Diploneis ovalis; 7) Caloneis alpestris; 8) Actinocyclus normanii; 9) Biddulphia penitens; 10) Rhopalodia gibberula; 11) Aneumastus sp.; 12) Nitzschia reversa; 13) Pinnularia (girdle view); 14) Amphora sp.; 15) Cocconeis placentula; 16) Lyrella lyra; 17) Licmophora sp.; 18) Nitzschia sp.; 19) Raphoneis sp.; Scale bar = 5 micron.
Discussions
Climate change has brought far-reaching consequences due to present day global warming and posed worldwide threat to terrestrial as well as coastal environments. The wetlands, lakes, coasts, estuaries, deltas have divulged into variety of threats due to climate manifestations and affected the ecological settings to a great extent. The west coast of India with different climatic regimes from tropical monsoon to semi-arid and arid is also strongly affected by climate change scenario since past to present and will be continuing in future also. Hence, the coastal margin along the west can play a key role in understanding the dynamics of continental and marine climatic trends. The west coast of India represents highly dynamic environment and ecosystem that is continuously undergoing pronounced changes through the Holocene to the present time (Babeesh et al., 2016; Chamyal & Merh, 1995; Lakhmapurkar & Bhatt, 2010; Pant & Juyal, 1993; Raj, 2007; Sridhar et al., 2014; Thakur et al., 2019).
In this article, the diatom groups and their zonations reflect changes in water salinity, sea water intrusion, limnic character, water depth and trophic status, which can be related to climatic changes, sea-level fluctuations and anthropogenic responses (Bigler & Hall, 2002; Birks et al., 2001, Li et al., 2018). The diatoms indicate alteration between fresh, brackish and marine environments, reflecting the interaction of rainfall and sea level (Smol et al., 2010).
The Vembanad wetland, Kerala coast shows dominance of Thalassiosira and Cyclotella with moderate to low abundance of Campylodiscus, Eunotia, Diploneis, Nitzschia, Navicula, Tabularia, Bacillaria, Gomphonema, Pinnularia and Amphora. The high abundance of the planktic diatoms indicates that the region is confined with high water columns. This may be because of the high monsoonal periods existing in this tropical region. The frequent tidal influence in the region also shows mixing of salt and freshwater regimes creating variable micro-environments in the wetland with assortment of fresh, brackish and marine settings. Along with these taxa there are numerous diatom genera whose occasional presence is also marked in the coastal wetland. It is found that the dominance of Cyclotella and Thalassiosira reflect sites with more salt-tolerant environment and indicate pelagic, marine phytoplankton ecology. These results have also been confirmed from the Congo basin deep sea sediments core reflecting various climatic shifts for 190 ka (Hatin et al., 2017). It is found that Aulacoseira spp. requires heavy silicified cells to attain high frequencies and a high sinking rate, and turbulence to maintain their presence in the water column (Bradbury, 1975; Chen et al., 2014) and are more competitive in the low light circumstances brought by turbulent waters. The abundance of Tabellaria spp. flourishes in electrolyte-poor, oligotrophic, circumneutral or slightly acidic waters (Taylor et al., 2007). It is commonly observed that species from genera like Diploneis, Amphora and Campylodiscus are generally more diverse in marine, coastal, or brackish habitats and this has also been confirmed from the present study. These findings are also evidenced from the findings of Lake Ohrid and Lake Prespa (Levkov & Williams, 2012).
It can be observed that in Dhadhar estuary again the benthic community is dominated by representatives of the genera Pinnularia, Surirella, Nitzschia, followed by Gyrosigma, Amphora, Navicula, Cocconeis and Thalassiosira (planktic). Many other sporadic forms with very low counts also exist in the region. The Dhadhar estuary along the Gujarat coast shows one of the oldest mangrove deposit in this regions. The well-preserved diatom in the mangrove region is believed to be one of the key factors in the sediments. This may be due to the humic acids accumulating in the submerged leaf litter along with pelagic sediments. Many times, it is observed that high tannin concentrations may prevent benthic diatoms from colonising the surface sediments, even though leaf litter does not always have a beneficial enrichment effect on them (Lee, 1999). The association of benthic diatoms recorded in Mecherchar Jellyfish Lake corroborates the present findings with the dominance of raphid pennate genera like Pinnularia, Surirella, Amphora, Nitzschia, Caloneis, Diploneis, Lyrella and Navicula (Smol & Stoemer, 2010).
In the Sabarmati estuarine complex, the highest record of diatoms comprise Navicula spp., and Nitschia spp., with low-to-moderate turnout of Pinnularia, Eunotia, Achanathidium, Synedra, Amphora, Frustulia, Diploneis and Cyclotella (planktic). The study suggests that the high dominance of benthic forms suggests shallow water levels in the estuarine complex. The coastal setting of Sabarmati is well known for its archaeological significance due to climatically-driven past. The diatoms from the present analogue may hitherto throw light on the propounding rich diatom diversity from the region. The past climatic manifestations in this region may be derived from the diatoms in terms of salinity changes, water levels, river influx, run-off related changes and many more to account. It has been found in studies that eutrophication was primarily evidenced by the appearance of Stephanodiscus species, in tracking the ecological history of the Thames archaeological sites as far back as the Middle Ages. The reconstruction of paleosalinity were produced from archaeological assemblages, and the presence of freshwater and marine species in historical sediments revealed that the Thames was tidal in downtown London throughout the Roman period (Smol & Stoemer, 2010). The changing pH because of climate change such as warming has been recorded in many parts of the world such as Austrian Tyrol, Baffin Island in the Canadian Arctic and many other cores bear direct relationship to changing climate with diatom assemblage. The species of Navicula, Eunotia, Fragilaria, Frustulia, Achnanthidium have been associated with changes in pH either as acidic or alkaline environments (Smol & Stoemer, 2010; Van Dam et al., 1994). The high abundance and diversity of Nitzschia taxa indicate varying ranges of subsaline and hyposaline conditions in the coastal environments.
The Harshad estuary diatoms comprise Pinnularia, Diploneis, Grammatophora, Amphora, Cocconeis, Nitzschia,, Navicula, Achnanthidium and Achnanthes as benthic genera while the planktic forms are dominated by Thalassiosira, Cyclotella and Melosira. The sporadic diatoms also comprise many taxa but with extremely low counts. The Harshad estuary is a typical example of tidal estuary with microenvironments reflecting micro, meso and supra tidal settings. The diatom assemblages clearly show a transition from marine–brackish diatoms along with mixed diatom community dominated by freshwater and salt-tolerant freshwater taxa (Manoj et al., 2020; Nodine & Gaiser, 2014; Smol & Stoemer, 2010).
From the present modern analogue there is no longer any doubt that climatic changes brought on by global warming pose a threat to the environment, especially wetlands (inland and coastal), estuary, lagoon, deltas, lakes, human wellbeing and socio-economic systems globally. Despite an increase in the amount of limnological and ecological studies from various climate regimes globally, there is still insufficient information to adequately describe the spatial and temporal patterns of climatic change at decadal to millennial scales, particularly for periods prior to the late Holocene (Chen et al., 2014; IPCC, 2007; Singhvi & Kale, 2010 ). The mechanisms connecting diatoms, lake hydrochemistry and climate are frequently poorly understood, as are numerous other types of inferences made from diatoms. Fossil diatoms have been utilised in numerous research works to chart past variations in lake water salinity. Few have reconstructed lake-level variations and other climate-related factors using changes in the relative abundance of planktic versus benthic diatoms, as well as other diatom alterations. Presently, multivariate techniques and qualitative assessments are incorporated in contemporary diatom studies to directly or indirectly reconstruct past climate conditions (Smol & Stoemer, 2010). Most of the studies related to climate and palaeoclimatic studies using diatoms have been from European and American barring a few from Indian sector.
The diatom assemblage from surface sediment aims to provide interaction among their ‘chosen’ environmental conditions and can be a valuable tool for calibration set, or ‘training set’ (Birks & Birks, 2006; Bopp, 2005; Hatin et al., 2017; Smol & Stoemer, 2010). These diatoms are valuable tool for reconstruction of long-term climatic changes with the aid of transfer function (Gasse et al., 1995; Zou et al., 2021) although the knowledge on this domain is very limited in Indian context.
Hence, to improve our knowledge on the palaeoclimatic inferences prior to instrumental records, proxy data serve as a major feedback.
Conclusion
The different climatic regimes in the present study provide valuable background information for the long-term geological and ecological status based on the diatom assemblages. The diatom assemblage in various climates reflects the character of present-day environment that inculcates the physiography, chemistry, geological and biogeochemical cycling. The variation in the diatom association combats the environmental and ecological conditions for their growth and silica mitigation to record the freshwater runoff, salt water intrusion, water chemistry, human interferences and more possible reasons to account. Thus, the modern diatoms helps to anticipate our present-day environment as they prefer fresh, brackish and marine environments and in down core studies they can be valuably assessed for climatic reconstruction during the geological past. Hence, understanding the variability of the climate system and its relationship with forcing mechanisms, diatoms can be an optimistic approach.
Footnotes
Acknowledgements
The authors would like to thank Dr Vandana Prasad, Director, BSIP for providing infrastructure facility and permission for the present study. Thanks are due to the Head, Department of Geology, University of Lucknow, Lucknow for his support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed the following financial support for the research, authorship and/or publication of this article: Financial support by DST is duly acknowledged for funding the projects SR/FTP/ES-149/2014 and SB/EMEQ-244/2014 for carrying the research work. This is BSIP contribution No. 60/2022-2023.
