Abstract
Introduction:
Patients with locally advanced gastric cancer often face postoperative complications and insufficient short-term outcomes. Understanding the changes in peripheral lymphocyte subsets following laparoscopic D2 gastrectomy is critical to addressing these challenges and enhancing postoperative recovery.
Objective:
This study investigates the dynamics of peripheral lymphocyte subsets in gastric cancer patients post-laparoscopic D2 gastrectomy. Our goal is to identify factors contributing to postoperative reductions in these immune cells, thereby improving management strategies.
Methods:
We retrospectively analyzed clinicopathological data from 169 gastric cancer patients, focusing on perioperative lymphocyte subset variations. Utilizing univariate and multivariate analyses, we identified factors significantly influencing lymphocyte reductions after surgery.
Results and Conclusion:
By postoperative day 7, we observed median decreases in T cells, B cells, NK cells, and memory T cells of −26.1%, −30.8%, −44.8%, and −2.3%, respectively. In contrast, naive T cells and regulatory T cells increased by 6.0% and 15.0%. Thymosin alpha 1 (Tα1) treatment proved to be a protective factor, significantly reducing the decline in T and B cell counts (p = 0.05). Multivariate analysis identified higher Interleukin-1β levels (HR = 3.66, p = 0.01), longer operation times (HR = 2.98, p = 0.02), and Tα1 therapy (HR = 0.15, p = 0.01) as independent predictors of T cell reduction. These findings highlight Tα1’s potential as a therapeutic intervention to mitigate lymphocyte depletion, suggesting its incorporation into postoperative care could enhance immune recovery and patient outcomes. This study illuminates key immunological changes following gastric cancer surgery, offering pathways to improve postoperative management and patient health.
Keywords
Introduction
The progression of cancer is governed by a myriad of genetic and epigenetic alterations that induce significant functional and compositional disturbances within the immune system. 1 These disruptions are further exacerbated by conventional cancer therapies, including surgery, chemotherapy, and radiotherapy, each exerting profound effects on immune function. Such perturbations within the immune milieu may not only predict poor prognostic outcomes but also hinder the effectiveness of anticancer treatments. Consequently, the identification of novel markers that evaluate immune function is imperative for predicting therapeutic responses. 2
Gastric cancer (GC), the fifth most prevalent malignancy, remains one of the leading causes of cancer-related mortality globally. 3 Despite contemporary advancements in multidisciplinary treatment modalities, surgical resection accompanied by lymphadenectomy continues to be the cornerstone for curative treatment of gastric cancer.4–6 However, patients undergoing gastrectomy with lymphadenectomy often face high rates of postoperative complications, which are known to compromise immune function and may correlate with adverse long-term outcomes. 7
The evolving landscape of systemic therapy for gastric cancer demands a nuanced understanding of both therapeutic innovations and host-specific prognostic determinants. Recent advances have expanded beyond traditional chemotherapy to incorporate molecularly targeted agents and immune checkpoint inhibitors (ICIs). The DESTINY-Gastric01 trial established trastuzumab deruxtecan (T-Dxd) as a breakthrough for HER2-positive advanced gastric cancer, achieving durable responses even in trastuzumab-pretreated patients. 8 Concurrently, adjuvant PD-1/PD-L1 inhibitors demonstrate significant relapse-free survival benefits across diverse populations, with pooled analyses revealing a 28% risk reduction (HR = 0.72, 95% CI: 0.67–0.78) in phase III trials. 9 However, the efficacy of these therapies hinges on preserved immune competence—a factor potentially compromised by surgical stress. Emerging prognostic biomarkers like the Cachexia Index (CXI) and Neutrophil-to-Eosinophil Ratio (NER) further illuminate this interplay: lower CXI (HR = 2.03 for OS) and elevated NER (HR = 1.74 for OS) correlate with poor survival, reflecting malnutrition-inflammation cascades that may exacerbate treatment resistance.10,11 Notably, DNA damage response alterations in gastric tumors may underpin both chemoresistance and impaired lymphocyte recovery post-resection. 12 These findings collectively underscore the paradox of modern gastric cancer management: while systemic therapies increasingly rely on intact antitumor immunity, radical surgery—the cornerstone of curative intent—induces transient lymphopenia and immunosuppression. Quantifying postoperative lymphocyte dynamics thus becomes critical for optimizing therapeutic sequencing, particularly as adjuvant ICIs gain clinical traction.
Peripheral lymphocyte subsets represent critical components of the immune response and serve as valuable indicators of immune status. Our prior investigations have underscored the pivotal role of immune function in postoperative recovery following laparoscopic gastrectomy. 12 Yet, the determinants influencing postoperative peripheral lymphocyte subset counts remain inadequately understood. This study endeavors to delineate the postoperative dynamics of peripheral lymphocyte subsets in gastric cancer patients’ post-laparoscopic gastrectomy and to explore the factors impacting their absolute counts, aiming to enhance prognostic accuracy and therapeutic efficacy in this patient population.
Methods
Study design and participants
We retrospectively collected data from patients with gastric cancer who underwent laparoscopic gastrectomy with D2 lymphadenectomy according to the Japanese Gastric Cancer Treatment Guidelines 13 in hospital between April 2022 and July 2024. Patients who met the following inclusion criteria were recruited: (1) histopathologically confirmed diagnosis of gastric cancer and (2) patients underwent operation as laparoscopic gastrectomy with D2 lymph node dissection, (3) no history of neoadjuvant chemotherapy, (4) no history of autoimmune disease, (5) no evidence of distant metastasis. The exclusion criteria were (1) incomplete records of important data, (2) synchronous diagnosis of other malignancies.
This study was approved by the institutional medical ethics committee (TJH20200315), and all aspects of this study complied with the 1964 Helsinki Declaration and its later versions. Informed consent was obtained from all patients.
Data collection and blood sample analysis
We collected demographic, laboratory, and clinical data from the electronic medical records. All participants underwent laparoscopic gastrectomy with D2 lymph node dissection. The brief procedure was described in previous publication. 14 Blood samples were collected preoperatively and on postoperative day 3 and day 7. The absolute counts of different lymphocyte subsets were detected by flow cytometry as previous described. 12
Statistical analyses
Quantitative variables were described as mean and standard deviation (SD) or median and range. Comparisons between different groups were performed with student t-test or Mann-Whitney U test. We analyzed the differences of lymphocytes subset counts before and after surgery by paired t-test. We present qualitative variables as quantity and percentage and compared using chi-square test or Fisher’s exact test. The cut-off values for lymphocyte subsets count were determined using receiver operating characteristic (ROC) curve. The cut-off value of T, B, and NK cell were 730 cells/μL, 205 cells/μL, and 163 cells/μL individually. We applied the univariate logistic regression to evaluate predictive factors for postoperative peripheral lymphocyte decrease. The multivariate logistic regression model was used to identify independent predictive factors. All p-values were two-sided, and statistical significance level was 0.05. All statistical analyses were performed using SPSS version 24.0 (IBM Corp. Armonk, NY, USA) and GraphPad Prism 9.0 (Graphpad, Inc, San Diego, CA, USA).
Results
Baseline characteristics
We retrospectively included 169 gastric cancer patients who underwent laparoscopic gastrectomy between April 2022 and July 2024. A flow chart of patients’ inclusion is shown in Figure 1. Table 1 summarized the clinical and pathological data of all patients in detail. The mean age of the included patients was 57.7 ± 11.6 years. 110 (65.1%) patients were male, and 59 (34.9%) patients were female. The mean absolute lymphocyte count was 1.7 ± 0.6*109/L. 29 (17.2%), 61 (36.1%), 66 (39.1%), and 13 (7.7%) patients had stage I, II, III, and IV disease, respectively. Other information is summarized in Table 1.

Flow chart of patient inclusion.
The clinical information of all patients (n = 169).

CEA: carcinoembryonic antigen; IQR: interquartile range.
Perioperative peripheral lymphocyte subsets count dynamics
We observed a decline in peripheral T cells, CD4+ T cells, CD8+ T cells, B cells and NK cells count on postoperative day 7. (Figure 2(a)–(e)) Memory T (CD4+CD45RO+) cells frequency decrease from 67.3 ± 12.1% to 65.3 ± 13.2%. (Figure 2(g)) A slight increase in Naïve T (CD4+CD45RA+) cells and regulatory T (CD4+CD25+CD127low+) cells frequency was found on postoperative day 7 (Figure 2(f) and (h)). The absolute changes of peripheral lymphocyte subsets were shown in Supplemental Table 1 in details. Compared to preoperative level, the median percentage of T cell, B cell, NK cell, and memory T cell change was −26.1%, −30.8%, −44.8%, and −2.3% respectively. The median percentage of naive T cell and regulatory T cell change was 6.0% and 15.0% respectively. (Supplemental Table 2)

The postoperative dynamics of peripheral lymphocyte subsets counts. (a) Total T cell. (b) CD4+ T cell. (c) CD8+ T cell. (d) B cell. (e) NK cell. (f) Naïve T cell. (g) Memory T cell. (h) Regulatory T cell.
Correlation between lymphocyte subsets change and clinical parameters
We investigated the correlation between peripheral lymphocyte subsets change and different clinical parameters. No significant differences in the change of T cells, B cells, NK cells, and regulatory T cells between age (<60 years vs ⩾60 years), gender (male vs female), CA19-9 (<34 ng/mL vs ⩾34 ng/mL), operation time (⩽240 min vs >240 min), T stage and lymph node metastasis (No vs Yes). The change of regulatory T cells is significantly higher in patients with elevated CEA level (>5 ng/mL) then those with normal CEA level (15.6% vs 14.9%, p = 0.05). The change of regulatory T cells was significant lower in patients with postoperative complication than patients with no complication (1.0% vs 17.7%, p = 0.01). (Table 2)
The change of perioperative dynamics of peripheral lymphocyte subsets absolute count between different clinical variables.
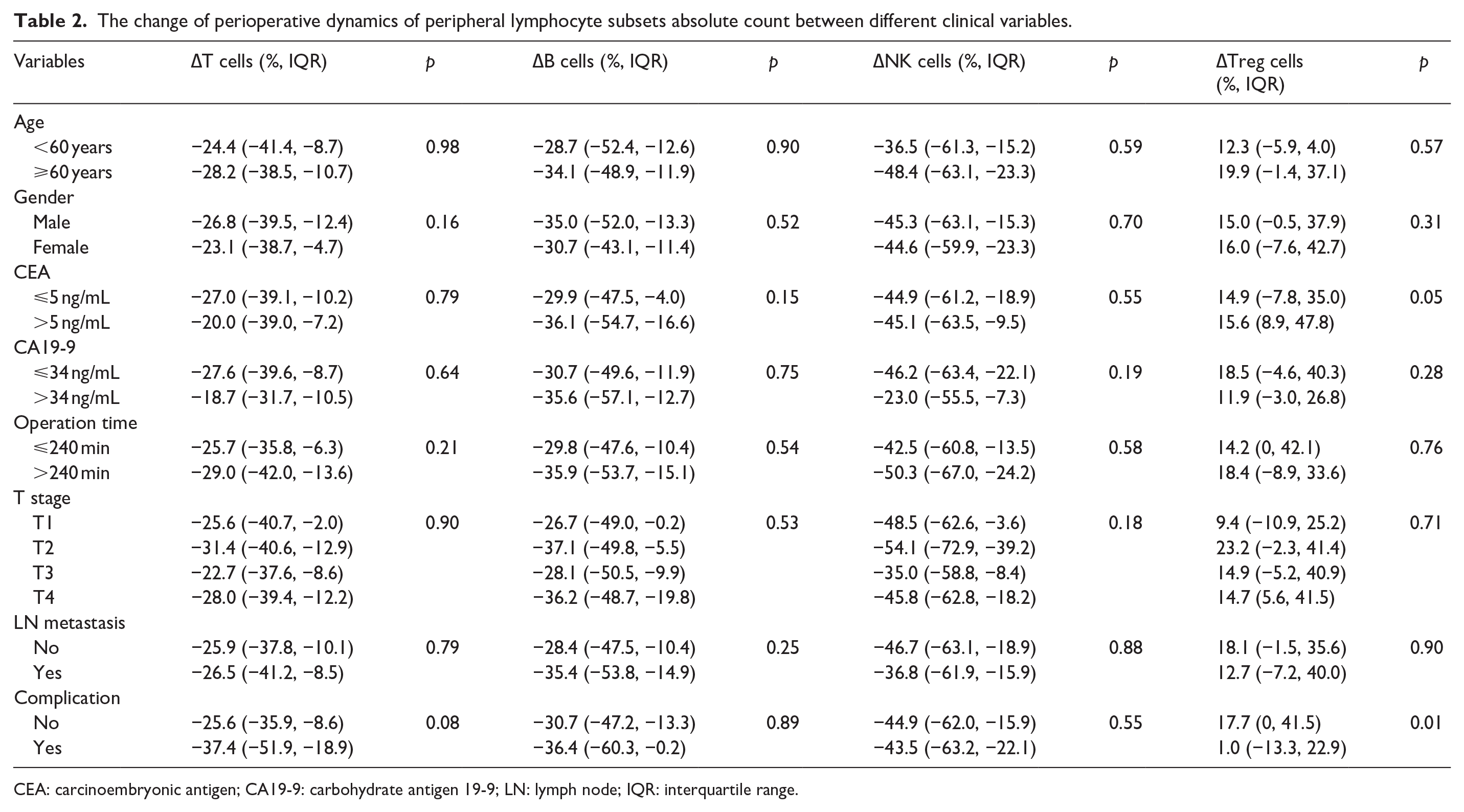
CEA: carcinoembryonic antigen; CA19-9: carbohydrate antigen 19-9; LN: lymph node; IQR: interquartile range.
Effect of Thymosin alpha 1 (Tα1) on the perioperative dynamics of lymphocyte subsets
According to the drug instruction, 1.6 mg Tα1 (Sciclone Phrmaceuticals, Hong Kong) was injected subcutaneously twice a week postoperatively. No adverse reaction was recorded. We retrospectively assessed the lymphocyte subsets change on postoperative day 7 in 169 patients, among whom 73 (43.2%) received postoperative Tα1 therapy and 96 (56.8%) did not. Compared to patients with Tα1 therapy, patients without Ta1 therapy demonstrated a significant profound decrease in T cell, CD4+T cell and B cell (p = 0.05; Figure 3(a), (b), (d)). There was no significant statistical difference in the change in CD8+ (p = 0.25), NK cell (p = 0.48), and regulatory T cell (p = 0.09) between two groups (Figure 3(c), (e), (h)). Interestingly, patients with Tα1 therapy showed a significant increase in Naïve T cell than those without Tα1 therapy. (p = 0.05; Figure 3(f))
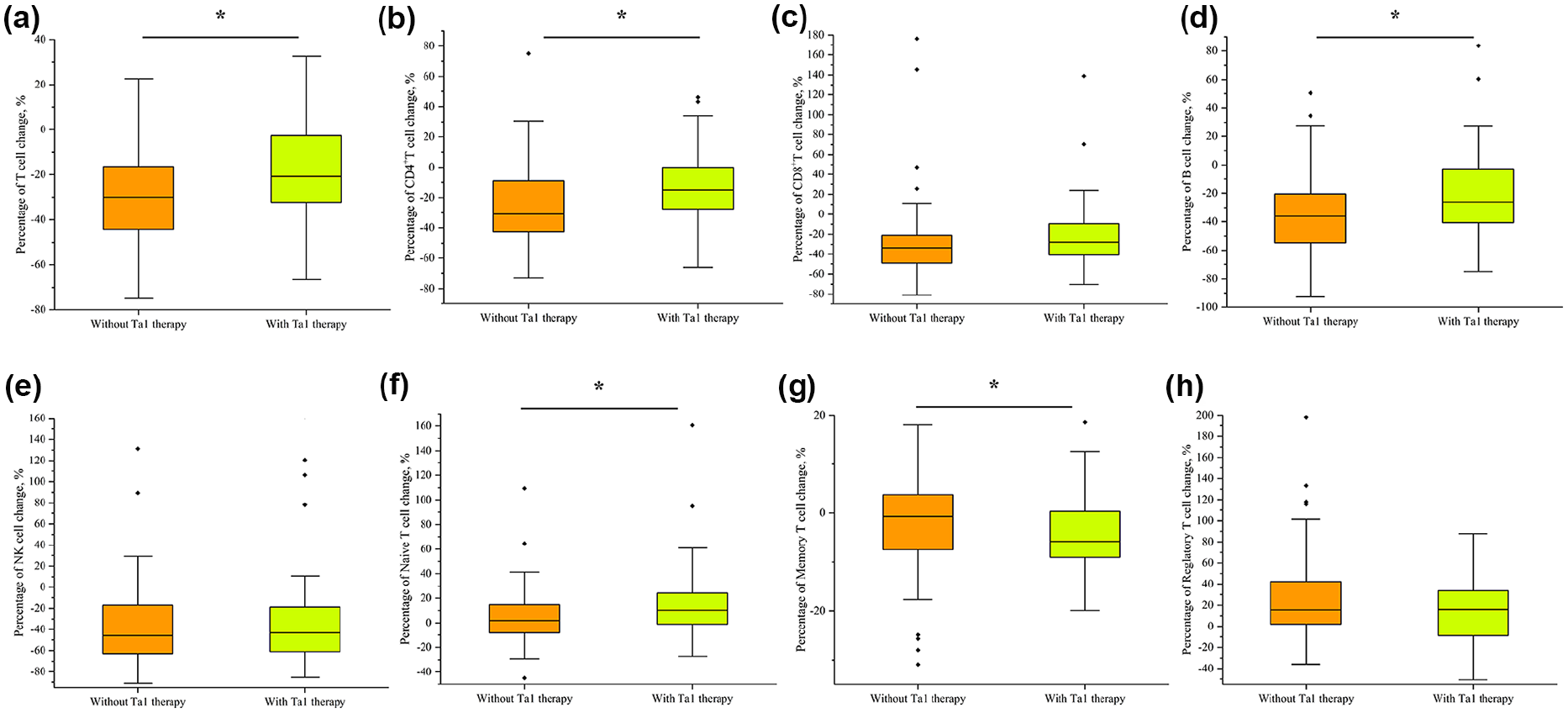
The change of peripheral lymphocyte subsets counts among gastric cancer with or without Tα1 therapy. (a) Total T cell. (b) CD4+ T cell. (c) CD8+ T cell. (d) B cell. (e) NK cell. (f) Naïve T cell. (g) Memory T cell. (h) Regulatory T cell.
Factors associated with postoperative lymphocyte subset change
The predictive factors for postoperative T cell decrease (⩽730 cells/μL) were identified using univariate analysis (Table 3). The results revealed that postoperative T cell decrease was related to serum Interleukin-1β level (>5 pg/mL vs ⩽5 pg/mL, hazard ratio (HR) = 2.40, p = 0.04), operation time (>240 min vs ⩽240 min, HR = 1.88, p = 0.10), complication (Yes vs No, HR = 3.01, p = 0.02), and Tα1 therapy (Yes vs No, HR = 0.26, p = 0.01). Multivariate logistic regression analysis identified that Interleukin-1β level (HR = 3.66, p = 0.01), operation time (HR = 2.98, p = 0.02), and Tα1 therapy (HR = 0.15, p = 0.01) were independent predictive factors associated with postoperative T cell decrease.
Cox hazard model for postoperative peripheral T cell decrease in gastric cancer patients (n = 169).

CEA: carcinoembryonic antigen; HR: hazard ratio; CI: confidence interval; CA19-9: carbohydrate antigen 19-9; TNFα: tumor necrosis factor α; LN: lymph node metastasis; Tα1: thymosin alpha-1.
We also investigate the independent factors associated with postoperative B cell decrease. Multivariate analysis showed that gender (female vs male, HR = 0.31, p = 0.03), serum Interleukin-1β level (>5 pg/mL vs ⩽5 pg/mL, HR = 2.90, p = 0.02), complication (Yes vs No, HR = 4.23, p = 0.01), and Tα1 therapy (Yes vs No, HR = 0.22, p = 0.01) were independent factors associated with B cell decrease. (Supplemental Table 3)
Discussion
For gastric cancer patients without distant metastasis, surgical resection complemented by adequate lymphadenectomy and chemotherapy—whether perioperative or adjuvant—remains the cornerstone treatment strategy.4–6 Large-scale studies have demonstrated that laparoscopic gastrectomy with D2 lymphadenectomy offers surgical and oncological outcomes comparable to those of open surgery in patients with early or locally advanced gastric cancer.15–17 The surgical excision of tumors presents a double-edged sword, impacting both the immune system and the immunological management of cancer recurrence post-surgery.
This study aims to investigate changes in postoperative peripheral lymphocyte subsets and their influencing factors in gastric cancer patients who undergo laparoscopic gastrectomy with lymphadenectomy. Our findings indicate that the counts of peripheral T cells, B cells, and NK cells decrease, while the counts of naïve T cells and regulatory T cells increase 1 week following surgery. Notably, a decrease in postoperative T cell levels was associated with elevated preoperative interleukin-1β levels and longer operation times. Importantly, postoperative therapy with thymosin alpha 1 (Tα1) emerged as a protective factor against the reduction of postoperative T and B cells. This study highlights the nuanced interplay between surgical intervention, immune function, and potential therapeutic strategies in optimizing postoperative outcomes for gastric cancer patients.
Postoperative impairment of immune function is a common occurrence following major surgical procedures, and it often correlates with unfavorable short-term and long-term outcomes. 18 A decline in peripheral lymphocyte numbers is frequently observed after major abdominal surgeries, which may be attributed to reduced lymphocyte proliferation, apoptosis-induced lymphocyte death, and lymphocyte redistribution under surgical stress.19–23 Functional deficiencies in cellular immunity are associated with decreased circulating T cells post-surgery. Previous research has identified a prolonged activation period of T lymphocytes in response to mitogens, lasting up to a week postoperatively. The immunological consequences of D2 lymphadenectomy may partially explain the observed T cell depletion. As an anatomically extensive procedure involving resection of perigastric and celiac axis lymph nodes, 13 D2 dissection inevitably disrupts lymphatic circulation and removes secondary lymphoid organs critical for T cell trafficking and homeostasis. 20 Experimental studies demonstrate that surgical removal of lymph nodes reduces lymphocyte recirculation by 40%–60%, creating a “lymphocyte sink” effect that depletes peripheral T cell pools. 23 Additionally, the prolonged operative time (median 240 min in our cohort) and increased surgical trauma associated with D2 procedures amplify stress hormone release (e.g. cortisol and catecholamines), which directly induce T cell apoptosis via Fas/FasL pathways.21,24,25 This dual mechanism—mechanical disruption of lymphoid architecture combined with neuroendocrine-mediated immunosuppression—likely underlies the correlation between D2 lymphadenectomy and T cell decline.
The immunological perturbations observed in gastric cancer patients post-D2 lymphadenectomy mirror patterns seen in breast cancer populations undergoing lymph node dissection and chemotherapy, though mechanistic distinctions exist. In breast cancer, longitudinal studies reveal that chemotherapy induces profound Th1-to-Th2 immune polarization (reduced IL-12, TNF-α, IFN-γ with Th2 elevation) alongside cytotoxic T cell expansion. 26 This contrasts with our findings of pan-lymphocyte suppression post-gastrectomy, suggesting that surgical resection and chemotherapy differentially remodel immune landscapes. Notably, both cohorts demonstrate sustained lymphocyte depletion—breast cancer patients exhibit a CD4+ T cell loss after systematic treatment, 27 paralleling our observation of 26.1% T cell reduction in gastric cancer. However, chemotherapy amplifies this effect, with premenopausal breast cancer patients showing persistent lymphopenia at 12 months post-treatment, 27 likely due to myelosuppressive drug toxicity. Intriguingly, Treg dynamics diverge between malignancies. While gastric surgery triggered a 15% Treg increase (Figure 2(h)), breast cancer patients exhibited elevated Tregs pre-chemotherapy that paradoxically rose further post-treatment. 28 This suggests that surgical trauma and chemotherapy both amplify immunosuppressive niches, albeit through distinct pathways—surgery via acute stress responses versus chemotherapy through direct hematopoietic disruption. Furthermore, breast cancer studies highlight stage-dependent immune dysfunction, with advanced tumors showing greater Treg infiltration and CD8+ T cell exhaustion, a pattern warranting investigation in gastric cancer progression. 28
The postoperative decline in lymphocyte subsets—encompassing T cells (CD4+ and CD8+), B cells, and NK cells—reflects a multifaceted immunosuppressive state driven by surgical stress and inflammatory cascades. Experimental models reveal that abdominal surgery significantly impairs NK cell frequency and function, facilitating metastatic dissemination, while tumor-specific T cell responses remain compromised for 7–10 days postoperatively.29,30 Clinically, colorectal cancer patients exhibit prolonged NK cell dysfunction, evidenced by suppressed IFN-γ production for up to 2 months, 31 mirroring our findings of a 44.8% NK cell reduction in gastric cancer patients. Similarly, T cell populations—critical for adaptive immunity—declined by 26.1%, with CD4+ T cells (mediating immune memory) and CD8+ T cells (executing cytotoxicity) both affected. This aligns with studies linking T cell depletion to postoperative sepsis and recurrence,19,29 while the 30.8% B cell reduction suggests impaired humoral defenses against bacterial pathogens. Mechanistically, prolonged operative time (associated with cortisol-driven apoptosis),21,24 elevated preoperative IL-1β (HR = 3.66, p = 0.01), and radical lymph node dissection (disrupting lymphoid trafficking) synergistically drive this “immunological void.” Additional contributors include neoadjuvant chemotherapy, glucocorticoid use, and open surgical approaches,20,32–34 all exacerbating lymphocyte loss. The coordinated depletion of innate (NK cells) and adaptive (T/B cells) immunity creates a vulnerable window for infections and micrometastasis, underscoring the imperative for immunoprotective strategies during this critical phase. 35
Elevated levels of regulatory T cells and pro-inflammatory cytokines also appear to play a significant role in compromising cell-mediated immunity.35–37 Our study demonstrated that within the first week following laparoscopic gastrectomy, gastric cancer patients experienced a reduction of up to 30%–40% in peripheral T, B, and NK cell counts. Conversely, the regulatory T cell count showed an approximate increase of 15% (−4.0%, 38.7%) postoperatively. Pro-tumorigenic mechanisms, such as impaired immune function, the release of tumor cells during surgical procedures, and the activation of pro-inflammatory pathways post-surgery, have been implicated in various cancer types and may contribute to cancer recurrence following radical tumor resection. 38
Recurrence after radical resection is prevalent and correlates with poor outcomes in gastric cancer.39–41 Previous findings from our research indicated that lower preoperative peripheral T or B cell counts were independently associated with adverse short-term surgical outcomes. 12 Taken together, these observations suggest that surgical resection can induce a temporary state of immunological suppression, and disruptions in systemic immunity may correlate with unfavorable clinical outcomes. This underscores the need for strategies to mitigate immune suppression and improve long-term prognoses in patients undergoing surgery for gastric cancer.
In addition to the transient suppression of the immune system, successful surgical resection of the primary tumor may contribute to the restoration of systemic immune function. A landmark study by Breanna et al. demonstrated that tumor progression dynamically altered the composition and functionality of systemic immunity in mouse tumor models, leading to attenuated responses to immune challenges. Interestingly, these immunosuppressive effects were largely reversed following surgical removal of the primary tumor, highlighting the remarkable plasticity of the immune system. 42 In alignment with these findings, our study observed a postoperative increase of 6.0% in naïve T cell counts, with a more pronounced increase among patients who received Tα1 therapy. Naïve T cells serve as a critical reservoir for effector immune cells, characterized by their diverse surface phenotypes and cytokine production capabilities. 43 Our findings suggest that systemic immunity undergoes a process of recovery following surgical resection. Such restoration of immune function may hold potential benefits in reducing the risk of long-term cancer recurrence by enhancing the immune system’s capacity to combat residual tumor cells and suppress metastatic progression.
Postoperative immunosuppression in gastric cancer patients has been linked to early recurrence, highlighting the need for strategies that enhance immune recovery to potentially improve both short-term and long-term outcomes. One such strategy is immune augmentation through the administration of thymosin alpha 1 (Tα1), a polypeptide hormone produced by thymic epithelial cells that plays a pivotal role in modulating both innate and adaptive immunity. Tα1 supports T cell differentiation, maturation, and proliferation, thereby enhancing the overall immune response.44–48 Previous studies have demonstrated the immunomodulatory effects of Tα1 in various clinical settings. Ding et al. investigated the impact of Tα1 administration in recipients of stem cell transplants for hematologic malignancies. While no significant differences in CD4+ or CD8+ T cell levels were observed between the Tα1-treated and control groups, the Tα1 group exhibited elevated levels of activated T cell-derived cytokines, including IFN-γ, IL-2, IL-10, and IL-12, indicative of enhanced immune activation. 49 Similarly, Ji et al. 50 found that Tα1 treatment significantly increased CD4+ T lymphocyte counts in renal transplant recipients, thereby improving resistance to cytomegalovirus infection. These findings underscore the potential of Tα1 as an adjuvant therapy to bolster immune recovery, which may also have translational implications for improving postoperative outcomes in gastric cancer patients.
Given its multifaceted role as an immune modulator, Tα1 holds promise for clinical application in cancer patients. Both in vitro and in vivo studies have indicated that the combination of Tα1 with chemotherapy enhances anti-tumor efficacy, particularly in nasal natural killer/T-cell lymphoma (NNKTL). 51 Clinical evaluations further suggest that Tα1, when used alongside conventional therapies, may synergistically improve outcomes in non-small cell lung cancer (NSCLC) patients following curative resection, as well as in those with metastatic melanoma.52,53 In prior research, we demonstrated that the preoperative immune status significantly influences the recovery trajectory of gastric cancer patients. 12 In the current study, we observed that short-term administration of Tα1 leads to an increase in postoperative peripheral lymphocyte counts. Additional studies corroborate Tα1’s role as a toll-like receptor agonist, which enhances antigen presentation and stimulates antibody production via B cell-dependent pathways.54–56 While our findings indicate that Tα1 treatment can attenuate the postoperative decline in peripheral T and B lymphocytes, its protective effects in gastric cancer surgery patients appear to be multifaceted. This suggests that Tα1 not only aids in immune recovery but also offers a potential therapeutic strategy for enhancing short-term postoperative outcomes in gastric cancer treatment.
Future directions
This study illuminates the dynamic interplay between surgical intervention and immune reconstitution in gastric cancer, yet it opens several transformative avenues for future investigation. First, our findings on Tα1’s protective effects hold immediate translational potential: we envision perioperative Tα1 administration may become the next research topic to optimize patients for adjuvant immunotherapy. Second, critical knowledge gaps persist: (i) How do lymphocyte kinetics influence checkpoint inhibitor efficacy? (ii) Can lymphocyte recovery patterns predict metastasis risk? (iii) Does microbiome restoration accelerate immune reconstitution? To tackle these, we propose the following research area: (i) Multi-omics profiling (single-cell RNA-seq + TCR clonotyping) to map functional lymphocyte recovery trajectories; (ii) Clinical trials combining Tα1 with PD-1 inhibitors (e.g. nivolumab) in the adjuvant setting; (iii) Longitudinal liquid biopsies tracking ctDNA clearance and lymphocyte expansion correlation.
In the future, gastric cancer management will undergo a transformative shift toward precision immunomodulation, redefining curative intent through three integrated innovations: (1) Preoperative “immune fitness” scoring synthesizing lymphocyte subsets, inflammatory cytokines, and cachexia indices (CXI) to stratify surgical risk; (2) Real-time immune monitoring via wearable biosensors detecting lymphocyte-derived exosomes for dynamic intervention; and (3) Surgery-immunotherapy sequencing algorithms continuously calibrated to postoperative lymphocyte thresholds. This paradigm evolution—from traditional cytotoxic bombardment to proactive immune stewardship—will fundamentally reconfigure therapeutic success in gastrointestinal oncology by preserving antitumor immunity while maximizing treatment efficacy.
Clinical implications and limitations
Assessing host systemic immunity remains a significant clinical challenge. Disruptions to immune function caused by cancer and surgical interventions can lead to adverse clinical outcomes. A major clinical concern is the absence of precise methodologies to evaluate patients’ immune status. Previous research established an immune scoring system that integrates peripheral lymphocyte number, function, and phenotype, demonstrating promising potential for quantitatively assessing host immunity. 57 We acknowledge several limitations that warrant cautious interpretation of our findings: (1) Functional Immune Assessment Gap: While we quantified lymphocyte subset counts, functional assays (e.g. IFN-γ secretion, cytotoxic activity) were not performed, potentially overlooking critical immune competence metrics beyond numerical changes. (2) Sample Size Constraints: Our cohort (n = 169) limits subgroup analyses—particularly for stage-specific or Tα1 dose-response relationships—reducing statistical power to detect subtle immunological shifts. (3) Unmeasured Confounders: Key factors influencing lymphocyte kinetics (e.g. nutritional status, microbiome composition, stress hormone fluctuations) were not systematically captured, possibly obscuring mechanistic insights. (4) Treatment Heterogeneity: Variability in adjuvant therapies (initiated in 37% of patients during follow-up) may confound long-term immune recovery patterns. (5) Short-Term Focus: The 7-day observation window cannot capture later immune reconstitution phases critical for adjuvant therapy readiness (typically 4–8 weeks post-surgery). To address these issues, future studies should incorporate a broader range of clinical parameters to better evaluate postoperative immune function in gastric cancer patients. Furthermore, extending the observation period would be valuable for exploring the dynamics of peripheral lymphocyte subset counts and their relationship with clinical outcomes over time. These efforts could enhance our understanding of postoperative immune recovery and inform strategies to improve long-term patient outcomes.
Conclusions
In conclusion, our study elucidates the postoperative dynamics of peripheral lymphocyte subset counts in gastric cancer patients undergoing laparoscopic gastrectomy. We demonstrated that postoperative administration of Tα1 effectively mitigates the reduction in peripheral lymphocyte subsets following surgery. The use of Tα1 postoperatively may offer significant benefits, enhancing recovery and improving short-term outcomes for gastric cancer patients.
Supplemental Material
sj-docx-1-iji-10.1177_03946320251352344 – Supplemental material for Unveiling lymphocyte dynamics: Navigating postoperative immune landscapes in gastric cancer patients undergoing laparoscopic D2 gastrectomy
Supplemental material, sj-docx-1-iji-10.1177_03946320251352344 for Unveiling lymphocyte dynamics: Navigating postoperative immune landscapes in gastric cancer patients undergoing laparoscopic D2 gastrectomy by Chun Gao, Li Zhu, Yi Xin Tong and Sheng Zhang in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-docx-2-iji-10.1177_03946320251352344 – Supplemental material for Unveiling lymphocyte dynamics: Navigating postoperative immune landscapes in gastric cancer patients undergoing laparoscopic D2 gastrectomy
Supplemental material, sj-docx-2-iji-10.1177_03946320251352344 for Unveiling lymphocyte dynamics: Navigating postoperative immune landscapes in gastric cancer patients undergoing laparoscopic D2 gastrectomy by Chun Gao, Li Zhu, Yi Xin Tong and Sheng Zhang in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-docx-3-iji-10.1177_03946320251352344 – Supplemental material for Unveiling lymphocyte dynamics: Navigating postoperative immune landscapes in gastric cancer patients undergoing laparoscopic D2 gastrectomy
Supplemental material, sj-docx-3-iji-10.1177_03946320251352344 for Unveiling lymphocyte dynamics: Navigating postoperative immune landscapes in gastric cancer patients undergoing laparoscopic D2 gastrectomy by Chun Gao, Li Zhu, Yi Xin Tong and Sheng Zhang in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
We thank Ms. Cheng Chen for the English grammar correction of this manuscript.
Author contributions
All the authors participated in the study design. Sheng Zhang: Conceptualization and writing the original draft. Li Zhu: Formal analysis and project administration. Yi Xin Tong: Data curation, methodology, and software. Chun Gao: Conceptualization and writing – original draft. All authors have agreed to the final version and meet the major criteria recommended by the ICMJE (![]() ).
).
Availability of data and materials
The database used and/or analyzed during the current study is not publicly available (to maintain privacy) but can be made available by the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hainan province, (No. 821MS0855).
Ethics approval
This study was approved by the institutional medical ethics committee (TJH20200315), and all aspects of this study complied with the 1964 Helsinki Declaration and its later versions.
Informed consent
Written informed consent was obtained from all patients.
Consent for publication
All authors agreed the publication of this study.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
