Abstract
Background: Modern pharmacological studies have shown that traditional Chinese medicine (TCM)
Introduction
Gastric cancer (GC) is a malignant tumor of digestive tract with high morbidity and mortality. 1 Since the early symptoms of GC are not obvious, it has a low rate of early diagnosis with a 5-year survival rate of less than 20%. 2 Currently, GC is primarily treated by surgery and chemotherapy 3 ; however, it still has a high rate of recurrence or metastasis. Although chemotherapy is the palliative treatment of choice for patients with metastatic GC, 4 the associated side effects and drug resistance impede the treatment of GC. 5 Therefore, it is necessary to explore effective treatments for GC.
Traditional Chinese medicine (TCM) is a commonly used complementary and alternative form of medical treatment.
6
For thousands of years, it has been extensively applied to the clinical treatment of cancer
7
; it effectively alleviates the adverse reactions caused by surgery and chemotherapy, as well as improves the tumor-related symptoms, immune functions, and quality of life.8–10 Previous studies have confirmed that
Network pharmacology is a novel technique used to explore the systematic mechanism of therapeutic compounds in diseases. 15 Here, we used pharmacological analysis to search the mechanism of TRX in the treatment of GC. Additionally, we predicted the targets of TRX against GC based on the public database, followed by creating a protein–protein interaction (PPI) network and performing the Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis. Finally, we conducted in vitro experiments to verify the results.
Materials and methods
Network pharmacology
Pharmacophore targets for TRX
The pharmacophore targets were predicted using PubChem (https://pubchem.ncbi.nlm.nih.gov/) and PharmMapper (http://www.lilab-ecust.cn/pharmmapper/). The 2D and 3D structures of TRX were obtained by PubChem, and the images were saved in an SDF format (Figure 1(a) and (b)). Then, the 3D SDF format file was uploaded to the PharmMapper server, and the parameters were set based on previous literature, that is, generate conformation: yes; maximum generate conformation: 300; select target set: only human protein target (v2010, 2241); and reserve the number of matching targets: 300. Finally, 220 target proteins were obtained, which were annotated using the PERL software. After eliminating the duplicate data, we obtained 209 target genes and set up a Drug.txt file for further analysis. (a) 2D structure of TRX. (b) 3D structure of TRX.
Targets collection of GC
We identified 11,345 disease targets from Gene Cards (https://www.genecards.org/) and 1410 disease targets from the Online Mendelian Inheritance in Man (OMIM) database (http://omim.org/). After removing the duplicate data, we obtained 11,685 disease targets and set up a Disease.txt file for further analysis.
Venn diagram
We used the “VennDiagram” package of R software to identify the intersection proteins from the TRX potential targets and GC targets. We obtained 186 common targets (Figure 2(a)) and set up the Drug-Disease.txt file. (a) Venn diagram. The green represents the GC targets, the red represents TRX targets, and the shaded part represents the intersection targets of TRX and GC. (b) Top 30 target proteins of TRX for the treatment of GC. (c) Target proteins interaction network. In the network, nodes represent proteins, lines represent functional associations between proteins, and line thickness corresponds to the confidence level of the reported association.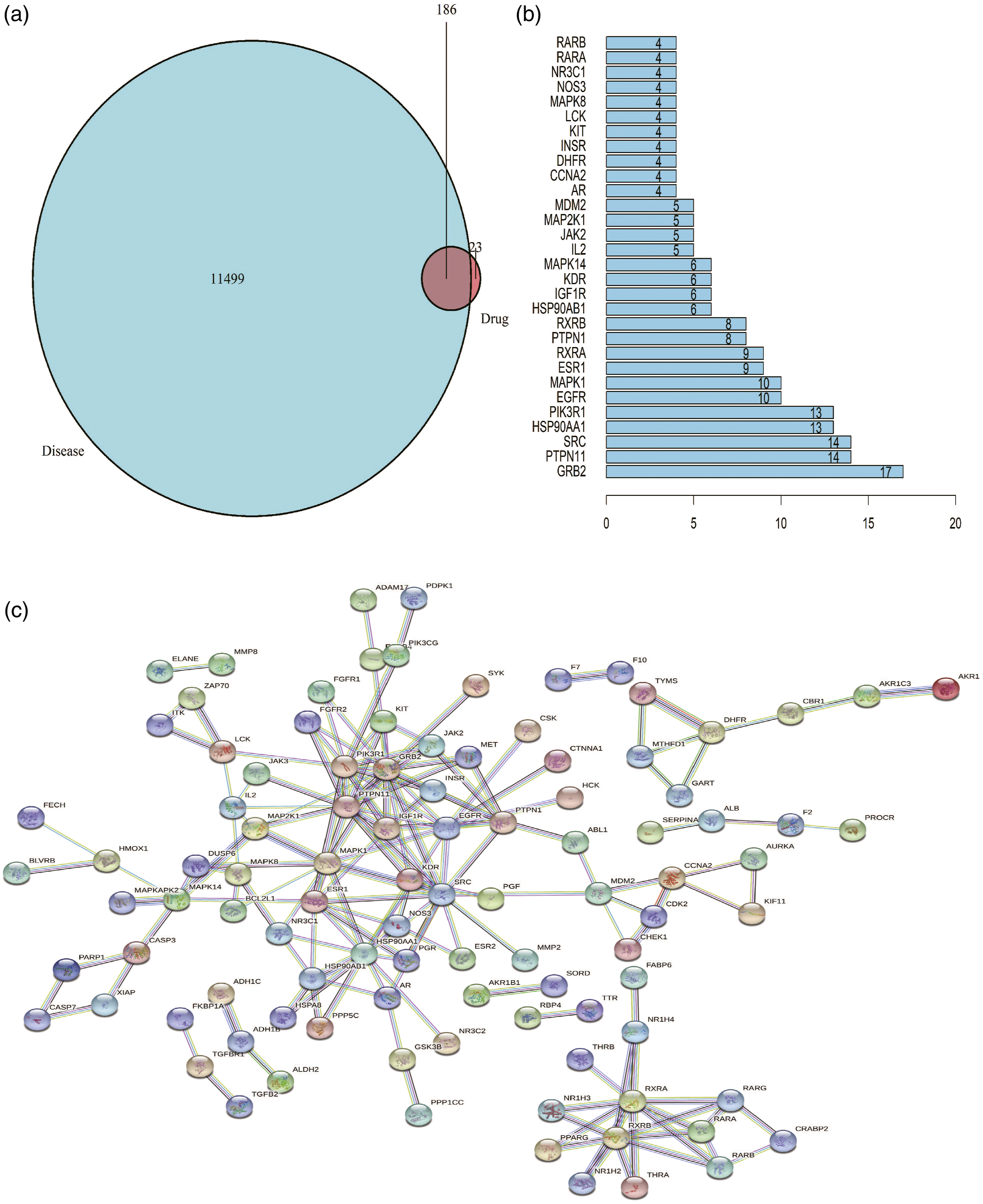
PPI network
The common targets were imported into the STRING database (https://string-db.org) to acquire the drug-disease PPI network. The following parameters were used: species: “
Gene function and KEGG enrichment analysis
We performed the GO and KEGG enrichment analyses using R3.6.2 software. We prepared the common target file “drug-disease.txt,” and then ran the BioConductor package to convert the common drug-disease target, and analyzed the enrichment of GO and KEGG of the key target genes based on the transformed “entrezID,”
Experimental analysis
Drug and main reagents
Taraxerol (purity > 96%) was gained from the New Drug Research and Development Center of North China Pharmaceutical Group Corporation (Hebei, China). The Roswell Park Memorial Institute (RPMI)-1640 medium and fetal bovine serum (FBS) were purchased from Gibco-BRL (Invitrogen Life Technologies, USA). 3-(4,5)-dimethylthiahiazo (-z-y1)-3,5-di- phenytetrazoliumromide (MTT), dimethyl sulfoxide (DMSO), the Annexin V-fluorescein isothiocyanate (FITC) kit, and the cell cycle detection kit were purchased from Sigma (St. Louis, USA).
Cell culture
Gastric cancer cell lines HGC-27 and NCI-N87 were purchased from the Cell Bank of the Chinese Academy of Sciences, Shanghai Institute of Cell Biology (Shanghai, China). The cells were cultured in RPMI-1640 medium containing 10% FBS and 1% antibiotics mixture (streptomycin 100 μg/mL and penicillin 100U/mL) at 37°C in a 5% CO2 incubator. TRX was diluted with RPMI-1640 to the final concentration of 0 μ M, 0.5 μ M, 1 μ M, and 2 μ M, then cells were cultured for 24 h for 48 h or 72 h. Furthermore, cells were pretreated with 10 μM IGF-1 for 30 min to explore the mechanisms of TRX induced apoptosis.
Cell viability detection
The viability of treated GC cells was tested by the MTT assay. The GC cells (5 × 104 cells/well) were seeded in a 96-well plate, followed by the addition of TRX (100 μL) at various concentrations (0 μM, 0.5 μM, 1 μM, and 2 μM). The plate was incubated for 24 h, 48 h, and 72 h, and then 10 µL of 10 mg/mL MTT was added to each well and incubated for an additional 3 h at 37°C. Next, the supernatant was removed, and 150 µL of DMSO was added and allowed to react for 15–20 min. We adjusted the blank control hole to zero and measured the OD value at 490 nm using an automatic enzyme labeling instrument.
Cell apoptosis assay
We applied the Annexin V-FITC/propidium iodide (PI) dual staining assay to examine cellular apoptosis. The GC cells in the logarithmic growth phase were seeded in a 6-well plate, followed by treatment with TRX (0 μM and 1 μM) for 24 h. Next, the cells were digested with 0.25% trypsin, suspended in phosphate-buffered saline (PBS), and centrifuged (5–min, 1000 r/min). After removing the supernatant, we added Annexin V-FITC (5 μL) and PI (5 μL) to 500 μL of cell suspension, mixed, and allowed to react for 15 min in the dark at room temperature. Apoptosis was detected by flow cytometry.
Cell cycle detection
The GC cells (5 × 104 cells/well) were seeded in 6-well plates, treated with TRX (0 μM and 1 μM) for 24 h. The cells were collected, washed with pre-cooled PBS, centrifuged (1500 r/min, 5 min), and then re-suspended in pre-cooled PBS. The cell suspension was added to pre-cooled 70% ethanol and fixed at 4°C for more than 4 h. The supernatant was washed twice with PBS, and then PI (400 μL) and RNase A (100 μL) were added, followed by incubation for 30 min at 4°C. Finally, the cell cycle was analyzed by flow cytometry.
Cell migration assay
Gastric cancer cells (5 × 104 cells) were inoculated in a 6-well plate and cultured till 80% confluent. The two groups of cells were treated with TRX (0 μM and 1 μM), respectively, and cultured in a serum-free medium for 24 h. The cell layer was scraped on each culture plate with the tip of a straw to form a scratch wound, and then gently the culture was gently rinsed with PBS to remove the loose cells. The scratch width was recorded by an inverted microscope at 0 h and 24 h, respectively.
Cell invasion assay
We used the Transwell (Corning Costar, USA) assay to measure the invasion ability of the cells. After treatment withTRX (0 μM and 1 μM) for 24 h, HGC-27 and NCI-N87 cells (5 × 104 each) were seeded in the uncoated membrane of the upper chamber (24-well insert; 8-μm pore size). After 24 h of incubation, the non-migratory cells in the upper surface of the membrane were removed with cotton swabs, and the cells penetrating the lower surface of the membrane were stained with crystal violet. Finally, the cells were randomly observed in five visual fields under ×400 magnification, and the cell count of the migrating cells was obtained.
Western blotting
After treatment, the cells of each group were washed thrice with PBS and centrifuged (15 min, 4°C, 12,000 r/min). The supernatant was collected, and the protein content was quantified. The protein samples (20 μg) were separated on an SDS-PAGE gel and transferred to a Polyvinylidene-Fluoride membrane. The membrane was incubated overnight with the primary antibody in 5% skimmed milk at room temperature for 2h and 4°C, followed by incubation with the HRP-conjugated secondary antibody at room temperature for 1.5 h. Finally, the bands were visualized by ECL kit and X-ray film exposure and quantified using ImageJ software.
Statistical analysis
All data were analyzed by one-way ANOVA for variable groups (GraphPadPrism seven software). In some cases, we performed the Student’s
Results
PPI network analysis
The common targets were imported into the STRING database to understand their interaction relationship, and the scoring value > 0.96 was selected as the “high confidence” basis for protein interactions. The interaction network has 186 nodes and 168 edges (Figure 2(c)). Based on the number of nodes, the first 30 key targets of the interaction were calculated using the Count. R package of R software. The longer the line, the more important was the target (Figure 2(b)).
GO and KEGG enrichment analysis
The key proteins could regulate 170 biological processes, primarily containing steroid hormone receptor activity, nuclear receptor activity, transcription factor activity, direct ligand regulated sequence-specific DNA binding, protein tyrosine kinase activity, endopeptidase activity, carboxylic acid binding, and organic acid binding. Figure 3(a) shows the top 20 biological processes. Regarding pathway analysis, the key proteins were involved in 126 signaling pathways, primarily the PI3K-Akt signal pathway, MAPK signaling pathway, Ras signaling pathway, proteoglycans in cancer, prostate cancer, Th17 cell differentiation, and hepatocellular carcinoma. Figure 3(b) shows the top 20 pathways. (a) GO enrichment analysis. The larger the bar chart is, the higher the enrichment degree of each biological processes, and the color of the bar plot map represents the gene enrichment of each biological processes. (b) KEGG enrichment analysis. The larger the bar plot diagram, the higher the enrichment degree of each KEGG pathway, and the color of the bar plot map represents the gene enrichment of each KEGG pathway.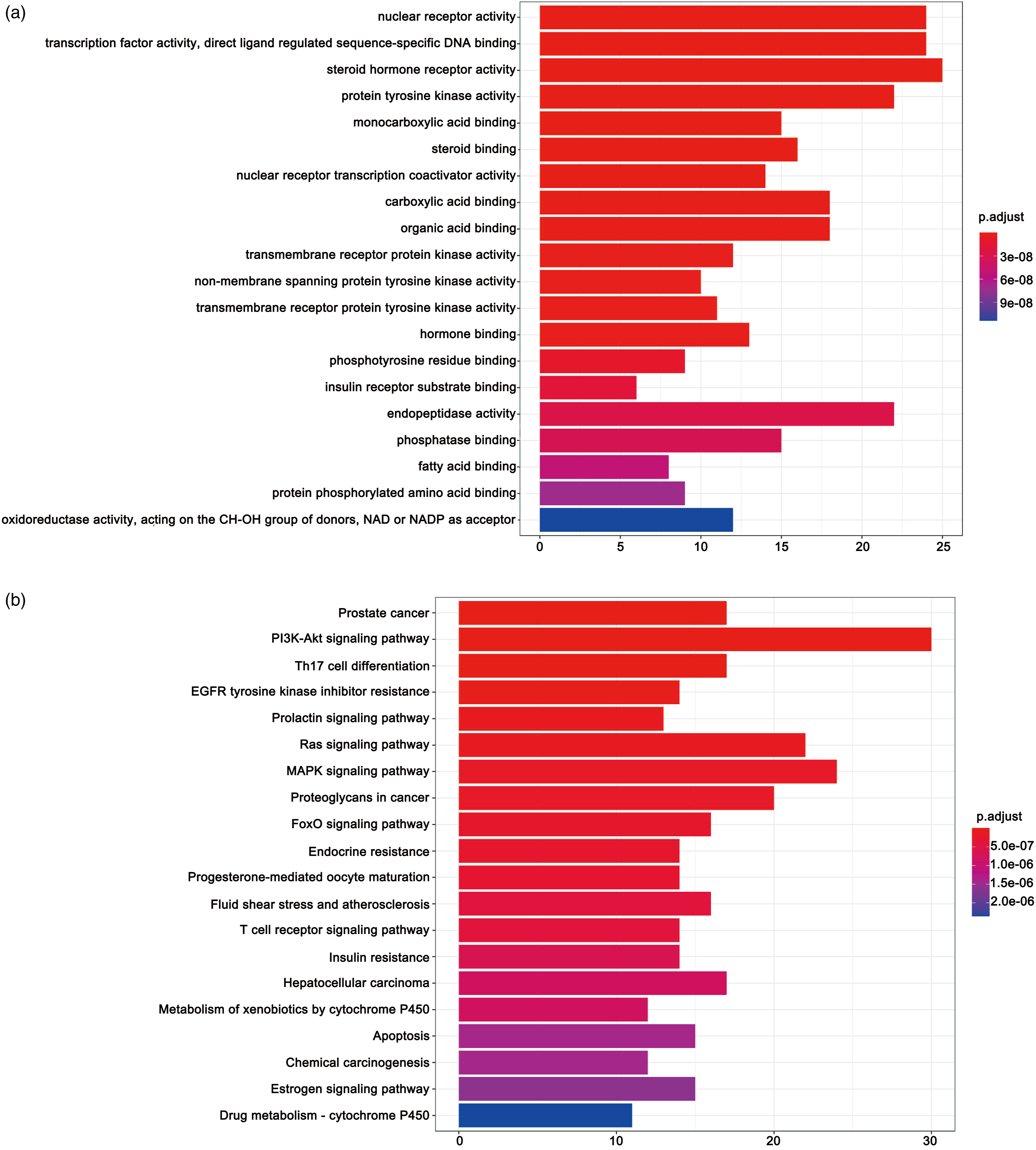
Experimental validation
TRX suppresses GC cells viability
The effect of TRX on the viability of GC cells was detected by MTT assay. HGC-27 cells and NCI-N87 cells were treated with various concentrations of TRX for 24 h, 48 h, and 72 h, respectively. We observed a time-dependent and concentration-dependent decrease in cell viability. Additionally, the cell survival rate in the TRX-treated group was significantly lower than that in the control group (Figure 4). These results indicated that TRX suppressed GC cell viability. Effect of TRX on cell proliferation. (a) HGC-27 and (B) NCI-N87 cells were treated with various concentrations of TRX (0, 0.5, 1, and 2 µM) for 24 h, 48 h, and 72 h, and cell viability was measured by MTT assay. (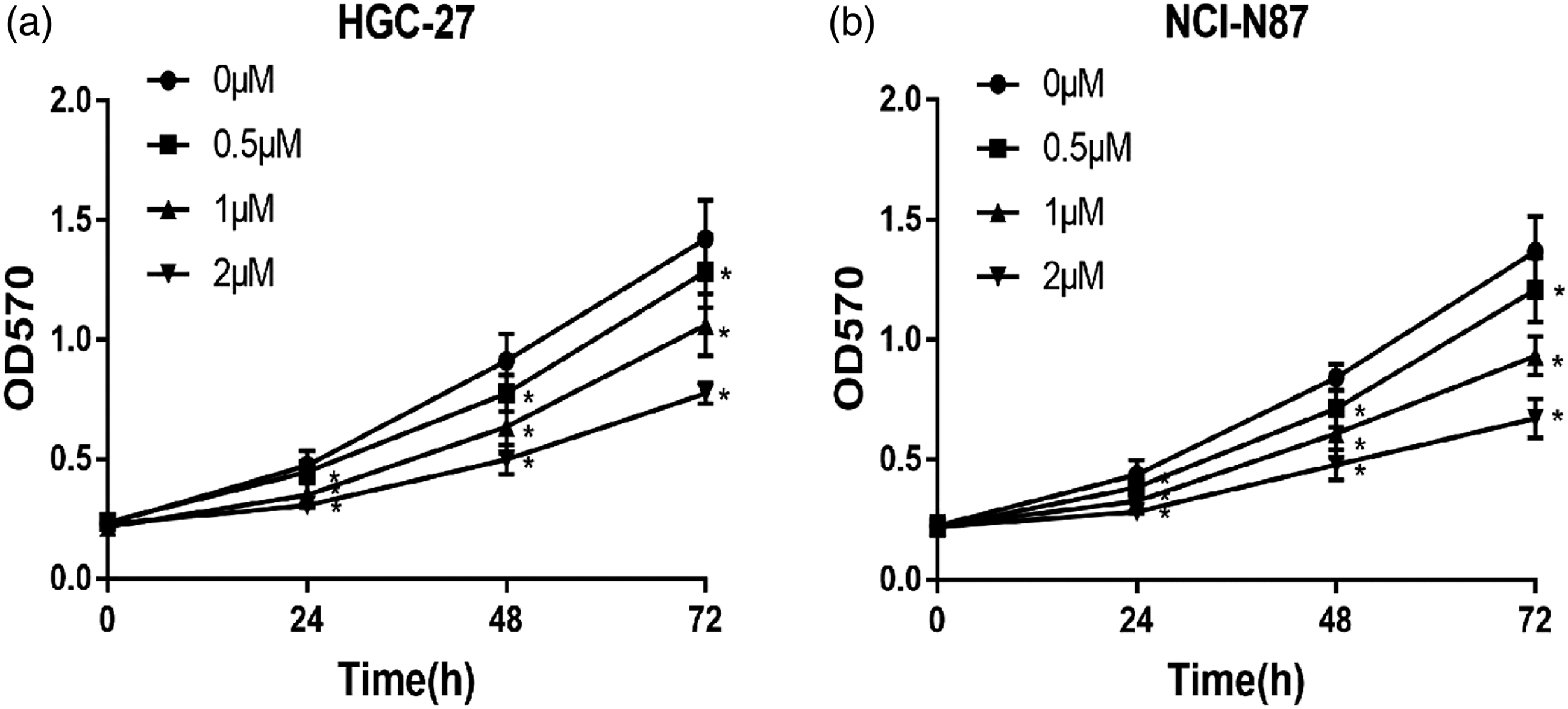
TRX induces apoptosis in GC cells
The above results showed that 1 μM TRX inhibited GC cell proliferation. Therefore, we used a 1 μM dose of TRX in subsequent experiments. Next, we studied the influence of TRX on apoptosis of GC cells by flow cytometry. These results indicated that the apoptosis rates of TRX-treated HGC-27 and NCI-N87 cells were significantly higher compared with the control group (Figure 5(a)–(b), (e)-(f)). Next, we measured the expression of apoptosis-related proteins in GC cells treated with TRX for 24 h by Western blot to explore the molecular mechanism of TRX-treated GC cell apoptosis (Figure 5(c) and (d), (g)–(h)). The results indicated that the levels of Bcl2 and survivin in the TRX group were lower than those in the control group. The expression of caspase-3, cleaved caspase-3, and Bax in the TRX group was significantly higher compared with the control group. These results suggested that TRX induced GC cell apoptosis. Cell apoptosis by TRX. Flow cytometry analysis of (a) HGC-27 cells and (E) NCI-N87 cells. The overall rates of apoptosis increased significantly after TRX treatment (b and f). HGC-27 (C and D) and NCI-N87 (g and h) cells were treated with TRX for 24 h and subjected to Western blot with Bcl-2, Bax, cleaved caspase3, caspase-3, and survivin antibodies. *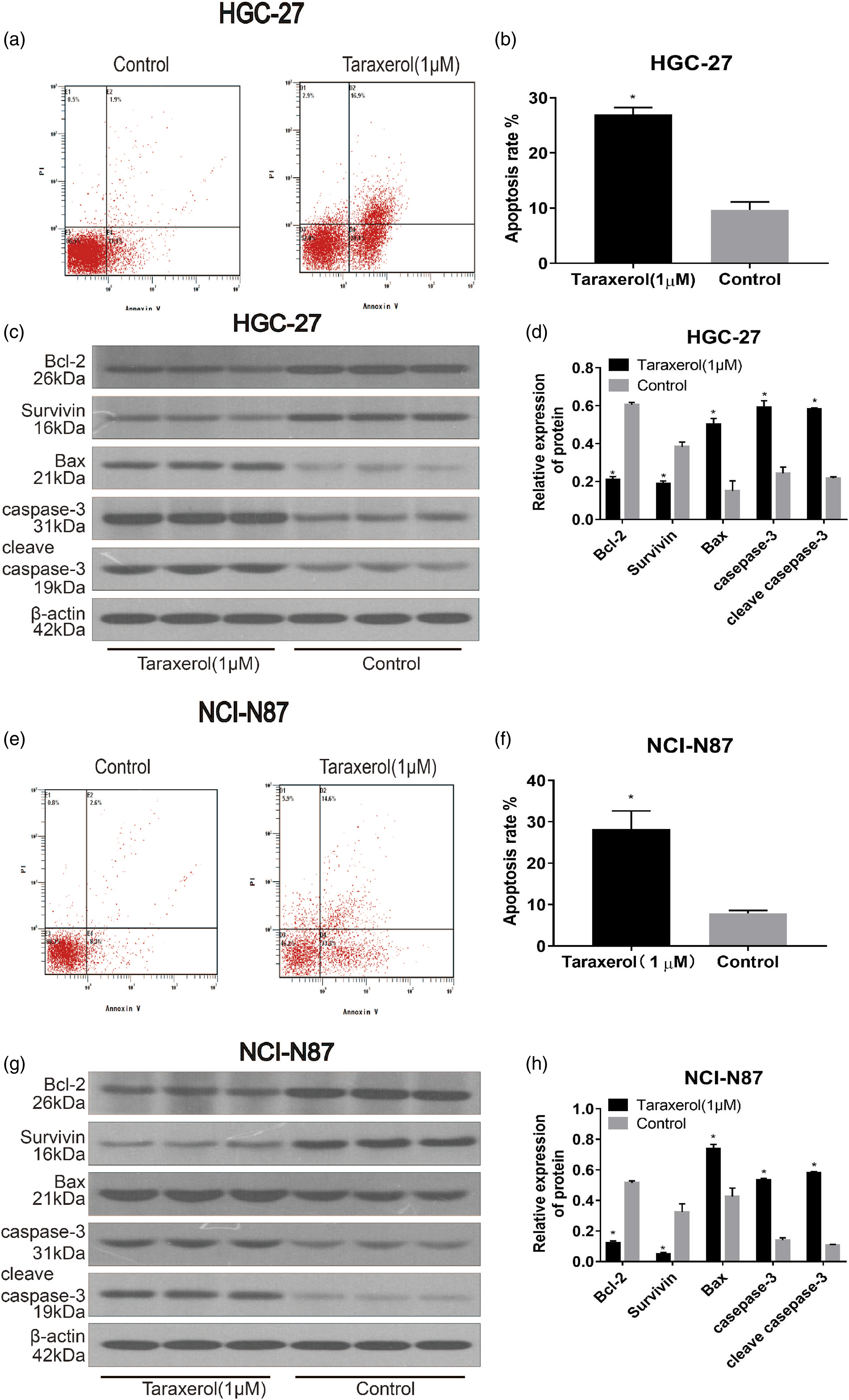
TRX induces cell cycle arrest at G0/G1
Flow cytometry was applied to investigate the number of GC cells in each phase of the cell cycle to identify the ability of TRX to induce cell cycle arrest. We observed that the number of cells in the G0/G1 phase in the TRX group was significantly higher than that in the control group (Figure 6(a)–(b), (e)–(f)). Additionally, we also used Western blot to detect the changes in the expression of related proteins after 24 h of treatment with TRX. The results showed significantly downregulated levels of Proliferating Cell Nuclear Antigen (PCNA) and CyclinD1 and significantly upregulated levels of p21 in the TRX group (Figure 6(c) and (d), (g)–(h)). These data suggested that TRX induced cell cycle arrest in G0/G1 phase, which affected the proliferation of GC cells. Cell cycle analysis revealed that TRX arrested cell growth at the G0/G1 phase. (a) HGC-27 and (e) NCI-N87 cells were incubated with the indicated dose of TRX for 24 h, stained with Propidium iodide, and DNA content of (b) HGC-27 and (f) NCI-N87 cells was detected via flow cytometry. HGC-27 (c–d) and NCI-N87 (g–h) cells were treated with TRX for 24 h and subjected to Western blot with PCNA, p21, and cyclin D1 antibodies.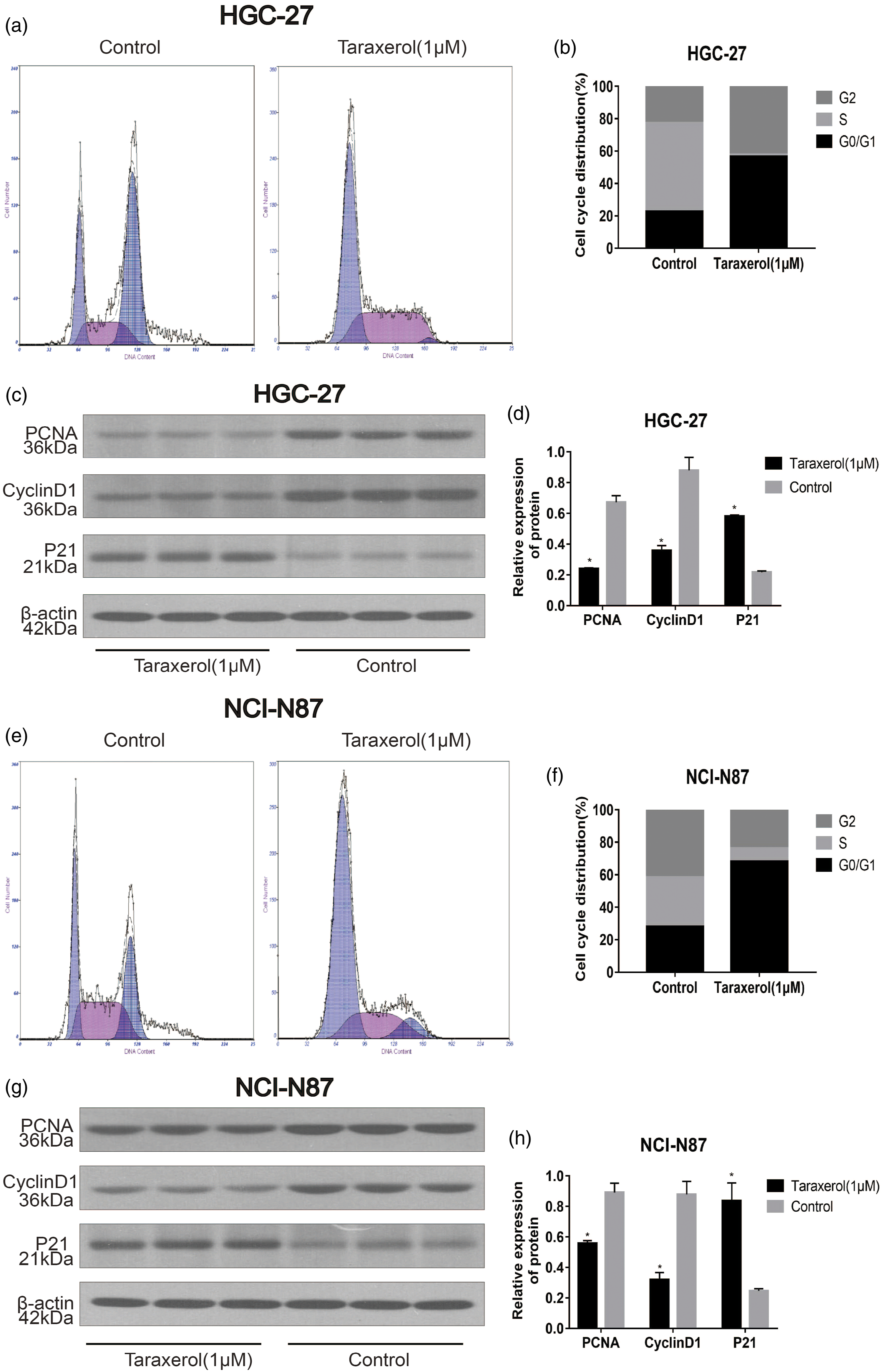
TRX inhibits the migration and invasion in GC cells
The anti-metastatic effect of TRX on GC cells was observed by wound healing assay. We observed significantly lower cell migration in the TRX group compared with the control group (Figure 7(c) and (d)). The effects of the Transwell assay indicated that the number of transmembrane cells in the TRX group was significantly less than that in the control group (Figure 7(a) and (b)). Additionally, we applied Western blot to study the expression of related proteins after 24 h of treatment with TRX. The results indicated significantly downregulated levels of MMP2 and MMP9 in the TRX group (Figure 7(e)–(h)). Thus, TRX inhibited the migration and invasion of GC cells. TRX inhibited the migration and invasion of HGC-27 and NCI-N87 cells. (a–b) Transwell assay was applied to determine the effect of TRX on cell invasion. (c–d) Effects of TRX on the migration of HGC-27 and NCI-N87 cells via wound healing assay. Western blot analysis of MMP2 and MMP9 in HGC-27 (e–f) and NCI-N87 (g–h) cells.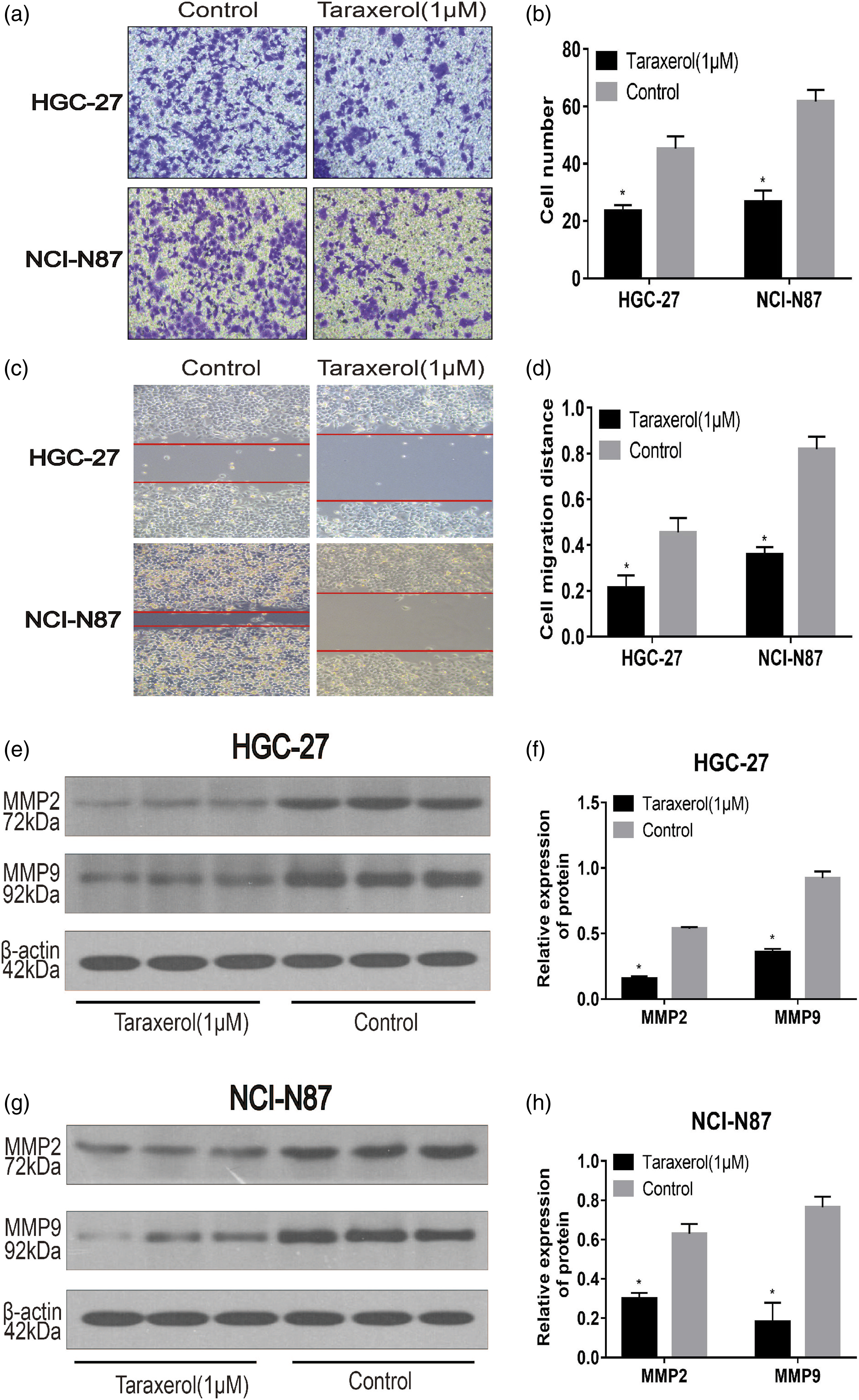
Effect of TRX on PI3K/Akt signaling pathway
We analyzed the effect of TRX on PI3K-AKT pathway in GC cells. The results of Western blot analysis showed that TRX treatment resulted in significantly decreased protein expression of PI3K, p-PI3K, and p-AKT, while there was no effective change in the protein expression of AKT (Figure 8). It is suggested that PI3K/Akt pathway is involved in the process of apoptosis induced by TRX in GC cells. After further intervention with PI3K/Akt agonist IGF-1, Western bolt results (Figure 9) showed that the pro-apoptotic effect of TRX was inhibited. Effects of TRX on the expression of PI3K/AKT pathway proteins in HGC-27 (a and c) and NCI-N87 (b and d) cells. Western blot analysis of Bcl-2, Bax, cleaved caspase3, and caspase-3 expression in HGC-27 (a and c) and NCI-N87 (b and d) cells treated with TRX in the presence or absence of IGF-1.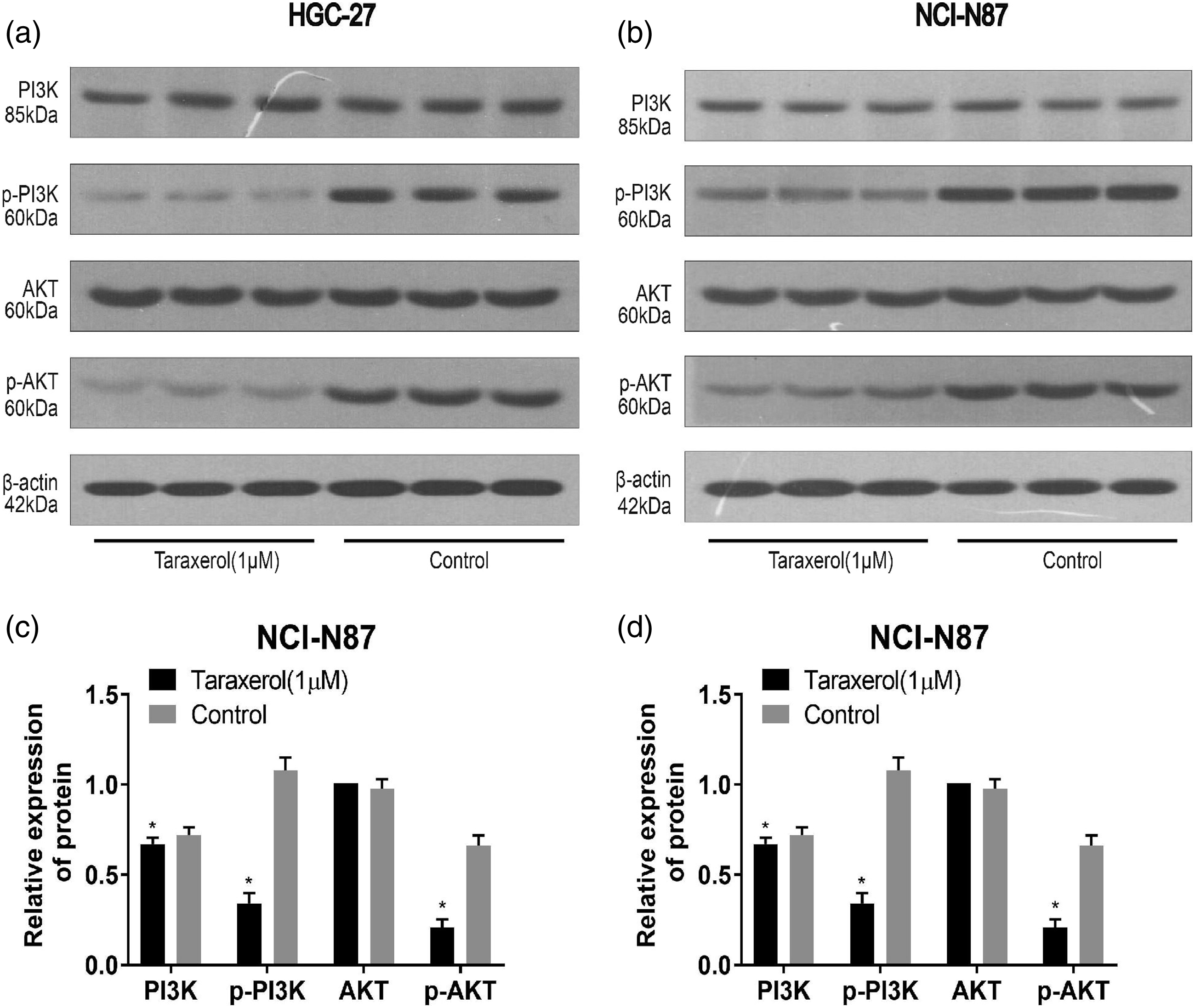
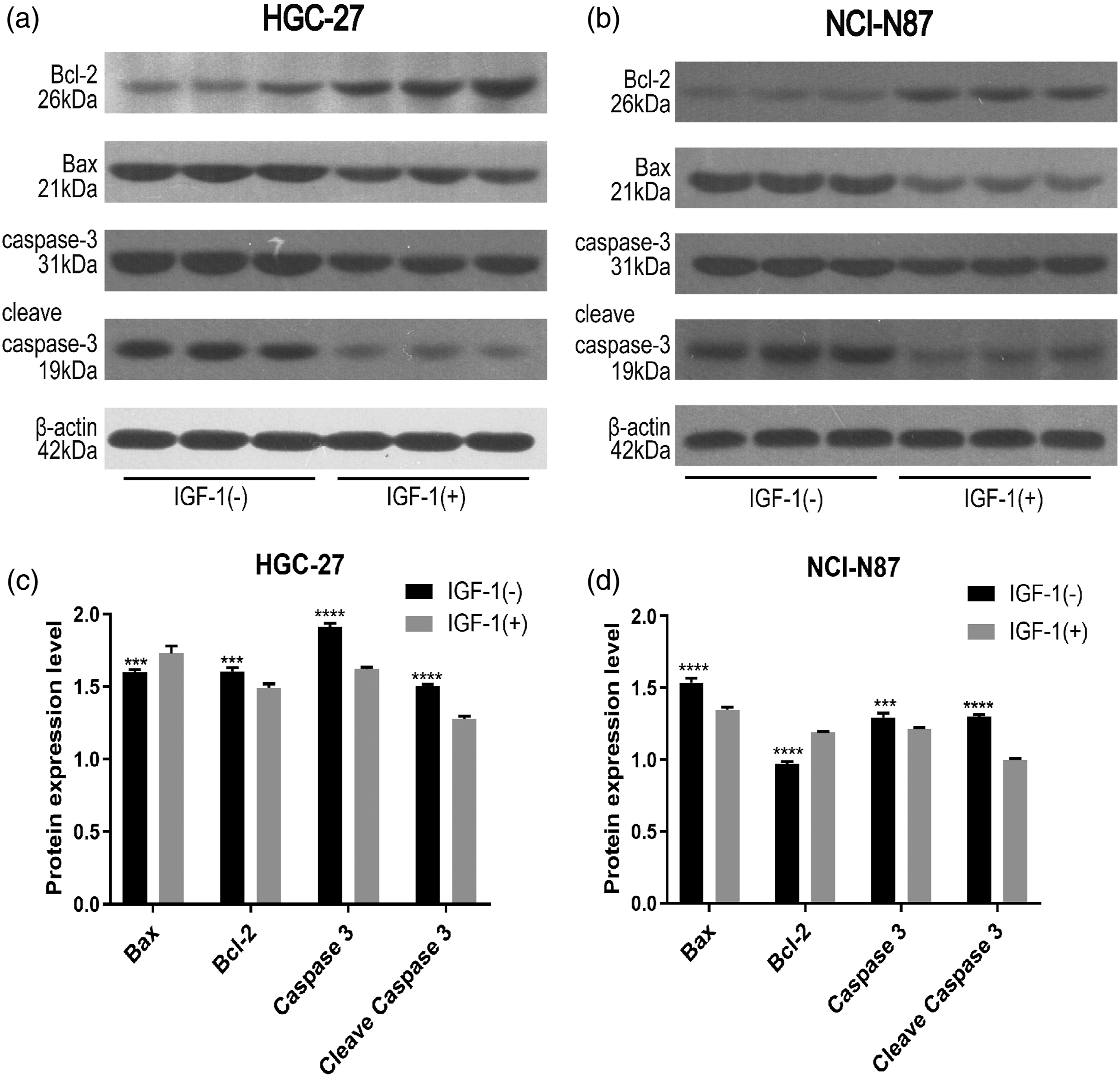
Discussion
In recent years, TCM has been widely accepted as a supplement or substitute for the treatment of diseases because of its good curative effect and low toxicity. 16 However, since TCM exert their effects through multi-target, multi-pathway, and multi-action mechanisms, it is difficult to detect their mechanisms. 17 As a new method to explore the systematic mechanism of therapeutic compounds in diseases, network pharmacology has been used to elucidate the mechanism of TCM.18,19 Here, we applied network pharmacology to research the regulatory mechanism of TRX in GC and performed in vitro experiments to verify those results to scientifically and systematically illustrate the mechanism of TRX in the treatment of GC.
Here, PPI analysis indicated that several key genes of TRX showed anti-tumor effects in the treatment of GC. For example, GRB2 played a critical role in tumor transformation and proliferation. 20 Aberrant expression of PIK3R1 has been shown in several tumors and is known to be related to elevated cell proliferation and invasion and reduced apoptosis. 21 Epidermal growth factor receptor signal is known to regulate various cell functions and promotes cell proliferation, differentiation, migration, growth, and survival. 22 The GO and KEGG analyses showed that TRX exhibited an anti-GC effect via multiple pathways, the most important one being the PI3K-AKT pathway. Previous studies have shown that PI3K/AKT signal pathway is one of the most common signaling pathways in tumors, participating in cell proliferation, cell cycle, migration, angiogenesis, and apoptosis. 23
Here, we discovered that TRX inhibited the proliferation of GC cells in dose- and time-dependent. We also found that TRX induced GC cell cycle arrest in GO/G1 phase. The abnormal regulation of the cell cycle has been shown to be involved in the occurrence and development of tumors. 24 Unregulated growth is not only a unique feature of cancer cells but also a major requirement in carcinogenesis. 25 Cell proliferation is controlled by cytoplasmic proteins involved in the cell cycle. 26 Therefore, we studied the expression of cell cycle-related proteins. Western blot analysis suggested that treatment with TRX resulted in a downregulated expression of cyclin D1, PCNA, and upregulated expression of p21. Some studies have shown that cyclin D1 positively regulates the cell cycle, and its overexpression can shorten the G1 phase of cells, resulting in excessive cell proliferation, and thus, the occurrence and development of tumors. 27 Also, p21 is a mature inhibitor of cyclin-dependent kinase (CDK), which plays a significant effect in controlling the cell cycle progression. 28 PCNA is an important gene that marks the state of cell proliferation. 29 It has been found that the expression of p21 is related to the expression ofcyclin D1 and PCNA; it inhibits DNA replication by interacting with PCNA, resulting in cell cycle arrest in the G1 phase. 30 All these results suggested that TRX could block the GC cell cycle in GO/G1 phase by reducing the expression of cyclin D1 and PCNA and increasing the expression of p21, thus inhibiting the proliferation of GC cells.
Apoptosis or programmed cell death is known to be related to various biological processes related to tumorigenesis. 31 The results of flow cytometry indicated that TRX induced apoptosis of GC cells. Additionally, we found that TRX upregulated the expression of pro-apoptotic proteins Bax, caspase-3, and cleaved caspase-3 and downregulated the expression of anti-apoptotic proteins, Bcl-2, and survivin. Studies have shown that the ratio of Bax/Bcl-2 is vital for determining the entry of cells into the apoptosis pathway. 32 Bcl-2, combined with Bax, is known to inhibit apoptosis. Also, caspase-3 and its active form, cleaved caspase-3, are known to play a significant effect in the apoptosis pathway. 33 Caspase family proteins can activate each other as well as themselves, which amplifies their effect and leads to apoptosis. 34 Previous research studies have indicated that the formation of Bax channels promotes the release of cytochrome c into the cytoplasm, separating Bcl-2 and Apaf1, leading to the activation of caspase to induce apoptosis. 35 Activated caspase-3 can cut off Bcl-2 at Asp34, resulting in the loss of the anti-apoptotic effect of Bcl-2. 34 Survivin is an anti-apoptotic protein, which can both promote cell cycle as well as inhibit apoptosis. 36 Some studies have suggested that survivin regulates apoptosis by directly targeting caspase-3 in the apoptotic pathway, 37 and it has been proved that the anti-apoptotic mechanism of survivin and Bcl-2 might act synergistically, resulting in a stronger anti-apoptotic effect and promoting the occurrence of squamous cell carcinoma of the nasal cavity and paranasal sinuses. 38 Therefore, our research suggested that TRX induced apoptosis of GC cells through the internal regulation of several key proteins.
Metastasis based on the migration of cancer cells and their invasion of surrounding tissues and blood vessels is regarded as the main cause of cancer-related death. 39 The results of the Transwell assay and the wound healing assay indicated that TRX inhibited the invasion and migration of GC cells and downregulated the expression of MMP2 and MMP9. It has been confirmed that GC, especially metastatic tumors, exhibit a high expression of MMP-2 and MMP-9 compared with normal gastric mucosa.40,41 Therefore, TRX exhibited not only an anti-proliferative and pro-apoptosis effect but also an anti-metastatic effect by downregulating the expression of MMP2 and MMP9.
PI3K/AKT signaling pathway has been widely studied in tumor progression and is known to play a crucial effect in cell proliferation, apoptosis, cell cycle regulation, and metastasis. 42 Additionally, PI3K/AKT has been shown to be a common downregulated signaling pathway in human tumors. 43 In this study, TRX effectively inhibited the expression of PI3K, p-AKT, and p-PI3K in GC cells, and taken together with the research results, these results suggested that TRX inhibited the activation of PI3K/AKT signal pathway. In addition, we also found that IGF-1, a PI3K/Akt agonist, significantly inhibited TRX-induced apoptosis. Therefore, we further confirmed that TRX induces apoptosis of GC cells by PI3K/AKT pathway.
As far as we know, this is the first time to report the anti-cancer effect of TRX on GC. This study reported the multi-target and multi-pathway regulation of TRX on GC through network pharmacology, and confirmed the anti-GC effect of TRX, but the limitation of this study is that the molecular mechanism is simple, only a simple in vitro experimental verification. Next, it needs to be verified in vivo, and future studies need to continue to explore the molecular mechanism of TRX against GC.
Conclusions
Thus, our results indicated that TRX inhibited the proliferation, metastasis and invasion of GC cells, and induced cell cycle arrest and apoptosis. Additionally, our study also showed that TRX exhibited an anti-GC effect by downregulating the PI3K/AKT signaling pathway. Therefore, TRX could be used as a potential drug candidate for the treatment of GC.
Supplemental Material
sj-xlsx-1-iji-10.1177_20587384211063962 – Supplemental Material for Research on the mechanisms of taraxerol for the treatment of gastric cancer effect based on network pharmacology
Supplemental Material, sj-xlsx-1-iji-10.1177_20587384211063962 for Research on the mechanisms of taraxerol for the treatment of gastric cancer effect based on network pharmacology by Bingjie Huo, Yanru Song, Bibo Tan, Jianbo Li, Jie Zhang, Fengbin Zhang, Lianmei Zhao and Liang Chang in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-xlsx-2-iji-10.1177_20587384211063962 – Supplemental Material for Research on the mechanisms of taraxerol for the treatment of gastric cancer effect based on network pharmacology
Supplemental Material, sj-xlsx-2-iji-10.1177_20587384211063962 for Research on the mechanisms of taraxerol for the treatment of gastric cancer effect based on network pharmacology by Bingjie Huo, Yanru Song, Bibo Tan, Jianbo Li, Jie Zhang, Fengbin Zhang, Lianmei Zhao and Liang Chang in International Journal of Immunopathology and Pharmacology
Footnotes
Author contributions
Bingjie Huo and Yanru Song designed the research, performed the experiments, analyzed data, and wrote the paper; Yanru Song, Jianbo Li, and Jie Zhang performed the experiments; Bibo Tan, Fengbin Zhang and Liang Chang provided technical support and all of the reagents and chemical; Liang Chang and Bibo Tan reviewed the paper for intellectual content. Bingjie Huo and Yanru Song contributed equally to this work and should be considered as co-first authors. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the National Natural Science Foundation of China (81202679) and the Scientific Research Program of Hebei Provincial Administration of traditional Chinese Medicine (2016053).
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for the article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
