Abstract
Introduction: To investigate antitumor activity and mechanism of flavonoids from
Keywords
Introduction
Gastric cancer (GC), the third most common cause of cancer-related deaths worldwide, is an aggressive disease that continues to have a daunting impact on global health. 1 The treatment of GC is mainly depending on surgery, chemotherapy, radiotherapy, target therapy, and immunotherapy. Nowadays, most GC patients undergo chemotherapy that is mainly assisted by adjuvant therapy including radiation, target therapy, and immunotherapy. 2 Unfortunately, many patients fail to show responses after chemotherapy due to multidrug resistance. Therefore, it is urgent to develop new agents against GC with satisfactory efficiency.
Natural agents, featured by multi-components, multi-targets, and corporation of multiple signaling pathways, show superior advantages in treating complicated diseases involving multi-targets and multiple signaling pathways.
3
The plant
STAT3, a member of signal transducer and activator of transcription proteins (STATs), can be activated by several cytokines in an indirect or direct manner including interleukin-6 (IL-6), IL-1, IL-17, and vascular endothelial growth factor (VEGF). It played a pivotal role in the regulation of the pathogenesis and development of many malignancies. The downstream target gene of STAT3 was closely related to the proliferation, apoptosis, and the cell cycle. Upon activation, it could lead to upregulation of cellular modulin and surviving, resulting in the malignant proliferation of cancer cells. Besides, STAT3 could promote the expression of VEGF and the angiogenesis. Moreover, it could upregulate the expression of MMP-9, which then triggered the invasion and metastasis of cancer cells.8, 9
In this study, we aim to investigate the effects of flavonoids against GC in the mouse fore-stomach carcinoma (MFC)–challenged mice and human BGC-823 cells. Meanwhile, we analyzed its molecular mechanism in terms of cellular apoptosis and cell cycle signaling pathway.
Materials and methods
Reagents
The fetal bovine serum (FBS), RPMI 1640 medium, and penicillin-streptomycin antibody were purchased from Life Tech (MI, USA). Rabbit Caspase-3, Caspase-8, Caspase-9, CyclinD1, CyclinB1, Bax, Bcl-2, and STAT3 were purchased from Abcam (CA, USA). The β-actin, HRP-labeled goat anti-mice secondary antibody, and goat anti-rabbit antibody were purchased from Boster Corporation (Wuhan, China). The commercial kits for the determination of proliferating cell nuclear antigen (PCNA), matrix metalloproteinase-9 (MMP-9), VEGF, IL-6, IL-1β, and IL-17 were purchased from Andygene (Beijing, China).
Compound extraction and isolation
The dried rhizomes of
Determination of content and compound structure
The content of total flavonoid (TFAO) of the fraction was determined by ultraviolet spectroscopy (UV) in NaNO2-Al(NO3)3-NaOH system at 510 nm and high efficiency liquid phase chromatography (HPLC). In the UV spectrum study, the results showed that content of total flavonoid was 52.2%. In the HPLC study, galangin, pinocembrin and kampheride, the top three components in the compound, were used as standards which isolated from TFAO in our previous study. 10 The relative amount of galangin was 32.13% according to the HPLC.
Galangin (3,5,7-Trihydroxyflavone) was purified and appeared as a yellow powder. The parameters of H-NMR and C-NMR were as follows: 1H-NMR (CD3OD, 400 MHz), δ6.22 (1H, d,
13C-NMR (CD3OD, 100 MHz): δC176.5 (C-4), δC164.7 (C-7), δ161.4 (C-5), δ157.0 (C-9), δ131.4 (C-1′), δ129.4 (C-2′), δ129.5 (C-6′), δ128.2 (C-3′), δ128.2 (C-5′), δ127.6 (C-4′), δ103.5 (C-10), δ97.9 (C-6), δ93.0 (C-8), δC145.6 (C-2), and δ137.0 (C-3). HR-MS(ESI)m/z:Calcd forC15H10O5 [M + H]+270.24 found 270.24.
Animals
ICR mice (male: 30; female: 30; 4–6 weeks, 20 ± 2 g) were purchased from the SLAC Laboratory Animal Center [SCXK (Hu) 2012-0002, Shanghai, China]. All the mice were fed in the animal chamber of SPF grade in the Shanghai University of Traditional Chinese Medicine (Shanghai, China) under a persistent temperature and humidity. All the animals had free access to food and water.
Induction of MFC tumor-bearing mice model
MFCs were purchased from the Cell Center, Shanghai Institutes for Biological Sciences (Shanghai, China). Cells at a logarithmic phase were prepared into solutions with a concentration of 1.2 × 107/ml under aseptic conditions. Tumor model was established by subcutaneous inoculation of MFC cells (1.8 × 106) to the right armpit. Sixty mice were randomly divided into control group (received normal saline, 20 mL/kg,
Determination of tumor weight, tumor inhibition rate, and organ indices
About 24 h after the last treatment, the body weight was determined, followed by animal sacrifice through cervical dislocation. The tumor, thymus, and spleen were obtained and weighted. Then, the tumor inhibition rate, thymus, and splenic indices were determined according to the previous description. 11
The indices of immune organs including thymuses and spleens were calculated according to the following formula: immune organ indices = 10 × A1/A2 (A1: average weight of thymus or spleen; A2: body weight). The tumor inhibitory ratio was evaluated using the following formula: inhibitory ratio (%) = (M1–M2)/M1 × 100, where M1 represented the average tumor weights of model group and M2 represented the average tumor weights of treated groups, respectively.
H&E staining
The tissues from heart, liver, spleen, lung, and kidneys were fixed using 4% paraform, and were embedded using paraffin. The sections were stained using HE method. Then, the pathological changes of the tissues were determined using H500S microscope (Nikon, Tokyo, Japan) under a magnification of ×200.
ELISA
Before animal sacrifice, the blood was obtained from the eyeballs of each mouse. Subsequently, the serum was separated, and then ELISA was performed to determine the serum VEGF, PCNA, MMP-9, IL-6, IL-17, and IL-1β (AndyGene Biotechnology, Beijing, China). All the tests were conducted at least in triplicate.
Cell culture and reagents
Human gastric cancer (BGC-823) cells (KeyGen Biotech Nanjing, China) were maintained in DMEM medium (Life technologies, USA). All types of medium were supplemented with 10% fetal bovine serum (Gibco, Grand Island, NY, USA). All the cells were cultured at 37°C under a humidified atmosphere incubator with 5% CO2. Cells (5 × 104/mL) were inoculated on 96-well plates. The grouping was as follows: control group, low (13 μg/mL), moderate (21 μg/mL), and high galangin groups (33 μg/mL), respectively.
Cytotoxic activities assay
The sensitivity of the human gastric cancer (BGC-823) cell line to galangin compound was evaluated by the CCK-8 assay. For experimental procedures, human gastric cancer (BGC-823) cell line was seeded in 96-well plate. After 24 h incubation, the medium was changed and galangin was added to cell culture medium with final concentrations ranging from 10 to 80 μM. Then, the cells were cultured for 72 h, followed by addition of 100 μL 10% CCK-8 solution (Beibo Biotech, Shanghai, China) solution for 1.5 h. Then, the medium was removed and 200 μL DMSO was added to the cells. Finally, absorbance was measured at 450 nm using the Multiskan MK3 microplate reader (Thermo Labsystem, MA, USA). IC50 values were calculated using SPSS software version 19.0 (IBM Corp., Armonk, NY, USA).
Cell counting kit-8 (CCK-8) assay
The cellular mixture in each group was incubated for 24 h, and then the medium was removed. Afterward, 10% CCK-8 solution (100 μL/well) was added and incubated for 1.5 h. Finally, the test was conducted using an ELISA reader at 450 nm. The IC10, IC25, and IC50 were selected for the subsequent analysis.
Apoptosis assay by flow cytometry
The effects of galangin on cell apoptosis were evaluated by Annexin V-FITC Apoptosis Detection Kit (Calbiochem, Darmstadt, GER). BGC-823 cells (4 × 105) were seeded in 96-well plates, and then were exposed to galangin of a concentration of 13 μM, 21 μM, and 33 μM, respectively. After culture for 24 h, cells (1 × 106/mL) were digested and harvested, and each cell suspension (500 μL) was incubated with 10 μL Media Binding Reagent and 5 μL FITC conjugated Annexin-V for 15 min at room temperature. The supernatant was discarded and cells were resuspended in 400 μL pre-cooled Binding buffer. Then, 10 μL PI staining solution was added. The fluorescence intensity was detected by flow cytometry.
Cell cycle assay
For the determination of cell cycle, trypsogen-digested cells and collected cell culture were centrifuged at 1200 rpm for 5 min and were treated using ice-pretreated ethanol (75%). Upon fixation for 12 h at 4°C, the mixture was centrifuged at 2500 rpm for 5 min. The supernatant was discarded, and the pellet was re-suspended using PBS. After filtration using a 200 mesh, the single cell suspension was prepared. RNAase and PI were added, and the mixture was incubated at 4°C for 30 min. The cell cycle was determined using flow cytometry at a wavelength of 488 nm.
Determination of sliced Caspase-3, non-sliced Caspase-8, and Caspase-9
The concentration of sliced Caspase-3, non-sliced Caspase-8, and Caspase-9 in each group under in vitro conditions was determined using commercial kits, strictly adhered to the manufacturer’s instructions (Beyotime, Shanghai, China). All the tests were performed at least in triplicate.
Cells were lysed in radioactive immunoprecipitation assay (RIPA) lysis buffer containing 1 mM PMSF (Solarbio), and then quantified using an Enhanced BCA Protein Assay Kit (BiyunBio, Shanghai, China). Equal amounts of proteins were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) analysis and electro-transferred onto polyvinylidene fluoride membranes (Millipore, Germany). The membranes were incubated with primary antibodies at 4°C overnight. This was followed by incubation with corresponding secondary antibodies (Abcam, MA, USA) for 2 h at room temperature. The immunoblots were scanned with an Ultra-Sensitive Enhanced Chemiluminescent Substrate Kit (Thermo Fisher Scientific, MA, USA) and a FluorChem E System (ProteinSimple, San Jose, CA, USA). The bands of interest were quantified using BandScan software (Glyco, Inc., Madison, WI, USA).
Preclinical evaluation of acute toxicity
The evaluation of acute toxicity for TFAO was performed in Kunming mice. All procedures were approved by the Ethical Committee of Xinjiang Medical University. A single dose of 1.6 g/kg TFAO dissolved in 0.5% Tween 80 in PBS via gavage was administrated to drug group mice. Vehicle group (0.5% Tween 80 in PBS) is as control. Each group has 4 mice. After administration, the animals were observed carefully for any gross effects or mortality. Weights, symptoms, and deaths were recorded. After 14 days, all live mice were sacrificed for autopsy to observe whether all organs were normal.
Statistical analysis
Data analyses were performed using SPSS software version 19.0 (IBM Corp., Armonk, NY, USA). Values are expressed as the mean ± standard deviation and were determined to follow a normal distribution. Therefore, the differences between two groups were analyzed using Student’s t-test, while differences between multiple groups were assessed by one-way analysis of variance followed by the Dunnett’s post hoc test.
Results
Toxicity assay
To evaluate TFAO as candidate compound with antitumor activity, toxicity was assessed. Mice treated with TFAO show no changes in body weight, consumption of water, and feed on day 7, 10, and 14 after administration of drug. No apparent changes were found in all examined organs.
Effects of TFAO on the tumor weight in the MFC tumor-bearing mice
At the end of treatment, mice in each group were sacrificed, followed by tumor photography (Figure 1) and measurement of weight. Then, the inhibition rate of tumor growth was calculated. The tumor weight in the positive control group, low, moderate, and high TFAO groups was 0.46 ± 0.26 g, 0.58 ± 0.39 g, 0.55 ± 0.31 g, and 0.33 ± 0.20 g, respectively. The tumor inhibition rates were 57.80% (positive control), 46.79% (low TFAO group), 49.54% (moderate TFAO group), and 69.72% (high TFAO group), respectively (Table 1). Effect of TFAO on the tumor weight of the MFC tumor bearing mice. Tumor weight was significantly reduced after TFAO treatment. Effects of TFAO on the MFC tumor-bearing mice. #

The growth of MFC tumor xenograft was effectively suppressed by 46.79, 49.54%, and 69.72% (IRT) after treatment for 2 weeks in the MFC tumor model, respectively. Notably, TFAO displayed significant inhibitory activity against tumor growth in vivo.
Effects of TFAO on thymus index and splenic index in the MFC tumor-bearing mice.
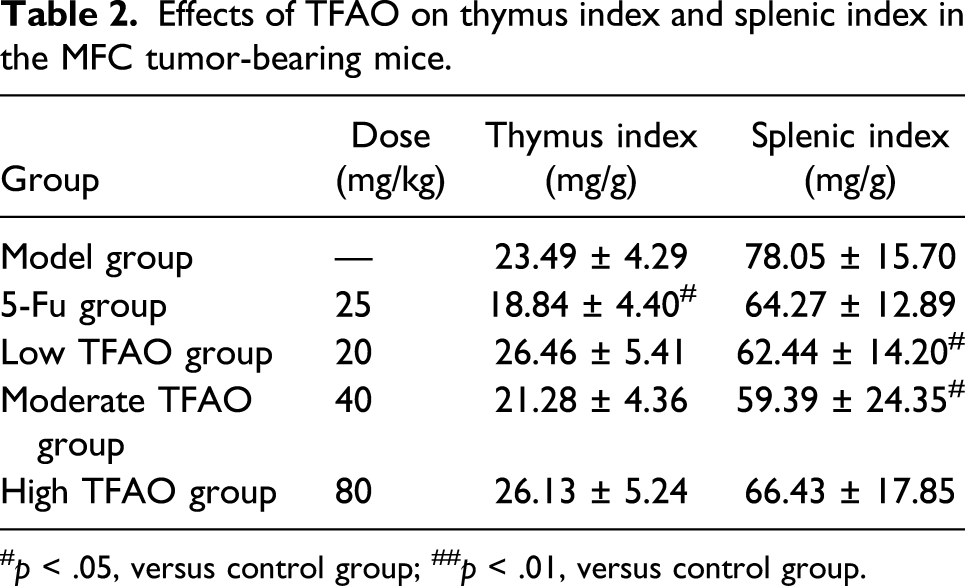
#
The thymus/spleen indices of MFC tumor-bearing mice in different groups are displayed in Table 2. The thymuses in positive control group were obviously degenerated (
Effects of TFAO on the organs in MFC tumor-bearing mice
Compared with the model group, no statistical differences were noticed in the pathological changes of the lung, spleen, and heart in the high TFAO group. There was increase of inflammatory cell infiltration in liver and kidneys sporadically in four mice (Figure 2). Histological analysis of the heart (a), lung (b), liver (c), kidney (d) and spleen (e) tissues taken from the mice in the model group (A) and high TFAO group (B).
TFAO regulated the production of cytokines in MFC tumor-bearing mice
The levels of serum cytokines are summarized in Figure 3. The expression of PCNA in the 5-Fu group and TFAO groups showed significant decline compared with model group ( Effects of TFAO on the secretion of tumor associated cytokines in MFC tumor-bearing mice. TFAO inhibits the secretion of serum PCNA (a), MMP-9 (b), VEGF (c), IL-6 (d), IL-1β (e), and IL-17 (f). *
Effects of galangin on the proliferation of BGC-823 cells
In this section, we determined the effects of galangin on the proliferation of BGC-823 cells under different concentrations (13 μg/mL, 21 μg/mL and 33 μg/mL) using CCK8 method. As shown in Figure 4, galangin could significantly inhibit the proliferation of BGC-823 cells compared with the control group ( Effects of galangin on the proliferation, apoptosis and cell cycle of the BGC-823 cells. (a, d) Relative growth of BGC-823 cells indicating dose-dependent antitumor effect of galangin. (b) Flow cytometry analysis of BGC-823 cells and apoptotic rate (e) following galangin treatment. (c) Flow cytometry profiles representing the differences in cell cycle of BGC-823 cells and (f) percentage of cells in each stage following treatment with galangin. *
Effects of galangin on the apoptosis of BGC-823 cells
Flow cytometry was carried out to determine the apoptosis of BGC-823 cells in the presence of various concentrations of galangin. As shown in Figure 4, galangin promoted the apoptosis of BGC-823 cells compared with the control group, which presented in a dose-dependent manner.
Effects of galangin on the distribution of cell cycle of BGC-823 cells
Compared with the control group, galangin triggered the accumulation of cells arrested in the G0/G1 phase significantly, which presented in a dose-dependent manner. Meanwhile, the number of cells arrested in S phage and G2/M phase showed significant decline in the galangin groups compared with the control group.
Galangin modulated the activities of Caspase-3, Caspase-8, and Caspase-9 in BGC-823 cells
The Caspase-3, Caspase-8, and Caspase-9 were activated upon treating with galangin for 24 h compared with the control group (Figures 5(a)–(c)). The activation of Caspase-3, Caspase-8, and Caspase-9 by galangin was in a dose-dependent manner. Effects of Galangin on the Caspases activity and the expression levels of STAT3, Bcl-2, Bax, Caspase-3, Caspase-8, Caspase-9, CyclinB1 and CyclinD1 protein in BGC-823 cells. Activities of (a) Caspase-3, (b) Caspase-8, (c) Caspase-9 in BGC-823 cells were measured using ELISA. (d) Western blotting assays showing the protein levels of STAT3, Bcl-2, Bax, Caspase-3, Caspase-8, Caspase-9, CyclinB1 and CyclinD1 following the treatment of BGC-823 cells with galangin. Statistical analysis of (e) Caspase-3, Caspase-8, Caspase-9, (f) Bcl-2/Bax ratio, (g) CyclinB1, CyclinD1, and (H) STATE3 expression. *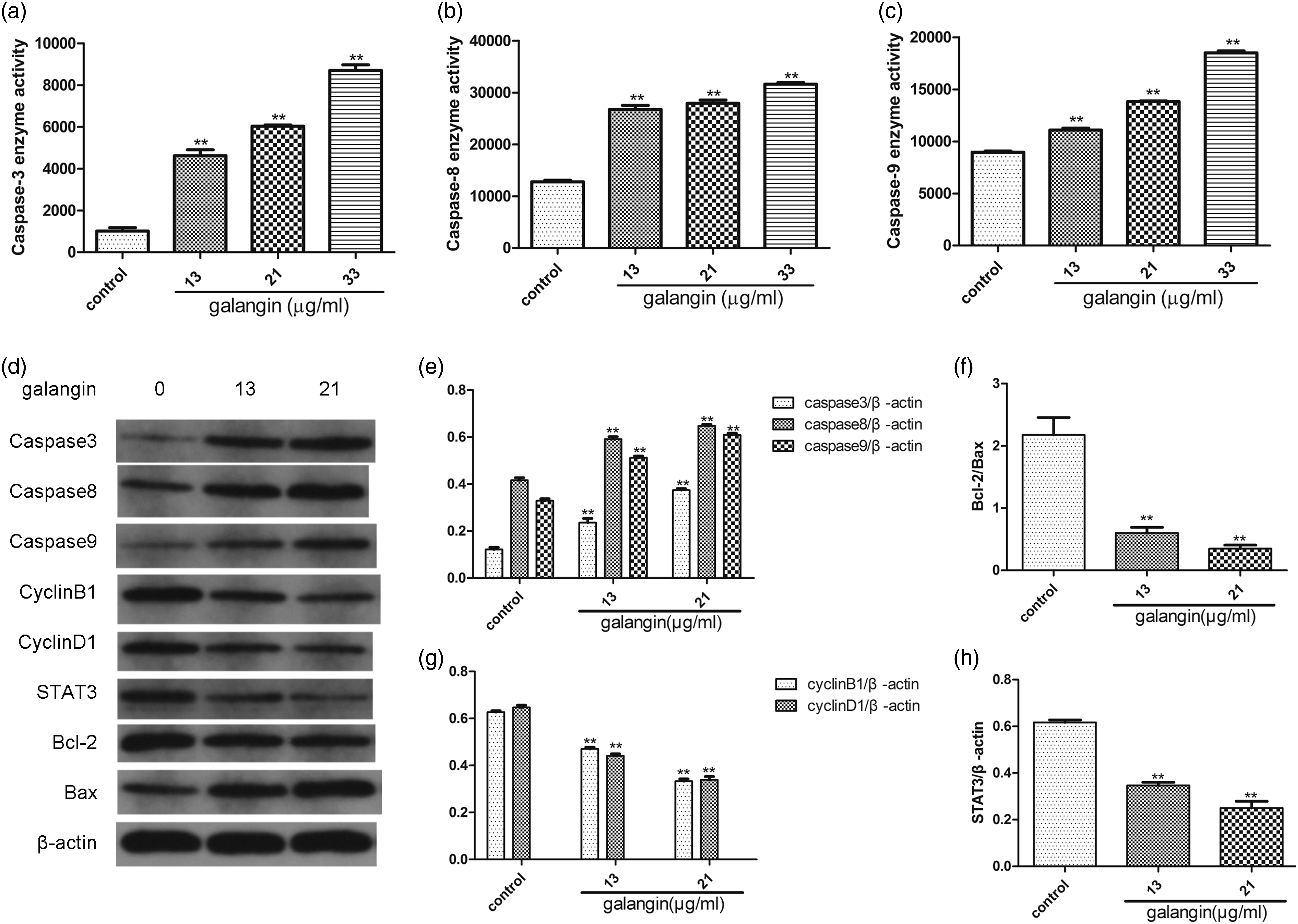
Effects of galangin on the protein expression of STAT3, Bcl-2, Bax, Caspase-3, Caspase-8, Caspase-9, CyclinB1, and CyclinD1 in BGC-823 cells
Western blot analysis indicated that the expression of STAT3, Bcl-2, CyclinB1, and CyclinD1 showed gradual reduction when treating with galangin, which was in a dose-dependent manner compared with control. The expression of Bax, Caspase-3, Caspase-8, and Caspase-9 in the galangin-treated BGC-823 cells showed gradual increase compared with the control group. In the cells treated with 13 μg/mL and 21 μg/mL galangin, the expression of STAT3, CyclinB1, CyclinD1, and Bcl-2/Bax protein showed significant reduction compared with the control group (
Discussion
Surgery has been commonly utilized for treating gastric cancer; however, a large number of patients would present recurrence and distal metastasis within 2 years with lung as the mostly involved organ. The incidence of pulmonary metastasis was in a range of 2.27%–3.84%, especially those with cancer in the upper 1/3 position.12, 13 For the patients with lung metastasis, the majority (80.9%) showed highly differentiated carcinoma. To date, rare studies have been focused on the prognostic analysis after pulmonary metastasis in gastric cancer patients. Therefore, the MFC model was utilized in this study. The MFC cells were obtained after in vitro culture as previously described. 14
In vivo experiments demonstrated that TFAO could significantly inhibit the growth of tumor. Interestingly, the thymus index in the MFC tumor-bearing mice showed obvious decline after treating with 5-Fu. The splenic index in the MFC tumor-bearing mice showed obvious decline after TFAO treatment. The thymus and splenic indices could reflect immune function. 16 This demonstrated that 5-Fu and TFAO might attenuate the immune function. Treg, a type of T lymphocyte subset with immunosuppressive activities, plays important roles in the immune balance. DCs with strong capacity of antigen presentation could regulate the activation and differentiation of T lymphocytes, which showed strong immunologic enhancement. 17 In future, more studies are required to explain this phenomenon.
Besides, the levels of serum PCNA, MMP-9, VEGF, IL-6, IL-1β, and IL-17 showed significant decline after treating with TFAO compared with that of the control. To our best knowledge, PCNA, serving as an important accessory protein of DNAase, was involved in regulating the synthesis of DNA. It has been considered as an important index for evaluating the proliferation of cancer cells. MMP-9 was reported to contribute to the invasion and metastasis of cancer cells through degrading the extracellular matrix. 11 VEGF could trigger the proliferation and migration of vascular endothelial cells, which then promoted the angiogenesis of tumor vessels. 18 Our data showed that TFAO could significantly downregulate the expression of serum PCNA, MMP-9, and VEGF in MFC tumor-bearing mice. This implied that TFAO could inhibit the proliferation, invasion, and metastasis of cancer cells, together with inhibiting the angiogenesis of tumor vessels. The complicated cytokine network formed by IL-6, IL1β, IL-17, and TNF-α could recruit inflammatory cells, which then deteriorated the inflammation. In addition, these cytokines could regulate the cancer-related inflammations through promoting the growth and metastasis of cancer cells, and the formation of tumor vessels and lymphatic vessels. 19 In this study, TFAO could downregulate the levels of serum IL-6, IL1β, and IL-17 in the MFC tumor-bearing mice, which implied that TFAO could attenuate the cancer-related inflammation.
IL-6 family could activate the downstream STAT3 through binding with the receptor coupling protein. VEGF could activate the STAT3 through tyrosine kinase receptors–mediated cascade reactions. As is known to all, IL-17 and IL-1β trigger secretion of IL-6, which then indirectly activate the STAT3 that played key roles in the cytokine networks formed by IL-6, IL-1β, IL-17, and VEGF. 20 In this study, based on the in vivo experiments, we speculated that TFAO may involve in the inhibition of the STAT3 activity that was closely related to these networks. Galangin ranked the highest in flavonoids in the TFAO showing definite effects against GC. Therefore, based on the in vitro experiments, we investigated the potential molecular mechanisms of galangin against GC through analyzing the STAT3-mediated cellular apoptosis and cell cycle pathways.
STAT3 played important roles in the pathogenesis, invasion, and metastasis of malignancies. The activation of STAT3 could attenuate the protective effects of P53 on the cytogene stability, which then resulted in increased risk of gene mutation and DNA damages and the subsequent pathogenesis of cancer. The downstream target gene of STAT3 was closely related to the cellular proliferation, apoptosis, and cell cycle. Its activation could upregulate the expression of cyclin and apoptosis-regulating factors, which then triggered the proliferation of cancer cells. STAT3 could upregulate the twist expression and the epithelial-mesenchymal transition (EMT) of cancer cells, leading to invasion and metastasis of cancer cells. In addition, its activation was crucial for decreasing the immunity of the natural killer T (NKT) cells, dendritic cells (DCs), and T lymphocytes. Also, it could indirectly contribute to the immunosuppression and immunological tolerance mediated by regulatory T cells (Treg). Moreover, STAT3 led to elevation of inflammatory factors such as IL-17, while the IL-17 could increase the secretion of IL-6, forming a positive feedback mechanism. Such process may further activate the STAT3, resulting in the malignant proliferation of cancer cells. 21
In vitro experiments indicated that galangin could inhibit the proliferation of BGC-823 cells and induce the apoptosis of BGC-823 cells in a dose-dependent manner. On this basis, we determined the effects of galangin on the distribution of cell cycle of the BGC-823 cells. Our data showed that the number of cells arrested in G0/G1 phase increased after treating with galangin for 24 h, whereas the number of cells arrested in S phase and G2/M phase showed significant decline. These implied that after treating with galangin, more cells were arrested in G0/G1 phase. Galangin could obviously inhibit the activation of STAT3 and downregulate the expression of downstream CyclinB1 and CyclinD1. Besides, it led to decline of the Bcl-2/Bax ratio and increased the activity and expression of Caspase-3, Caspase-8, and Caspase-9. These results indicated that the reduction of BGC-823 cell proliferation, induction of BGC-823 cell apoptosis, and arresting of cell cycles mediated by galangin may be related to the inhibition of STAT3 signaling pathway. Besides, it could further regulate the downstream proteins such as downregulation of CyclinB1, CyclinD1, and Bcl-2, as well as the upregulation of Bax, Capase-3, Capase-8, and Caspase-9. Taken together, the data confirmed that the STAT3-mediated apoptosis and the cell cycle–related signaling pathway were important targets for the galangin against GC.22-26
There are really some limitations in this study. For the sample size, as this study was not a random control trial, we did not calculate the required sample size according to the power analysis and justification of the size, but only chose 10 animals in each group. Pathological findings indicated that TFAO may lead to hepatic and renal injuries. It may be related to the establishing of the animal model, high dose of TFAO agents, and the potential pharmaceutical effects of TFAO on liver and renal injuries. In future, we will focus on this by deep investigations.
Conclusion
In summary, our findings implicated a potential application of flavonoids from the
Footnotes
Acknowledgments
We thank Professor Lili Ji, TCM Institution, Shanghai TCM University, for the kind help for the technical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation (No. 81460616), and Key Medical Discipline Year Plan Period (Pharmacy-Plateau Discipline) Construction Project of Xinjiang Uygur Autonomous Region during the 13th Five-Year Plan Period (Xin Educational Research 2016–7).
Ethical approval
Ethical approval for this study was obtained from the Ethical Committee of Xinjiang Medical University (K20160920-02).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Data availability statement
All the data were available upon appropriate request.
Contributorship
YM drafted the article and revised it critically for important intellectual content; HR, CYY made a substantial contribution to the concept and design of the work; HA, WXM made a contribution to the acquisition, analysis and interpretation of data; AP approved the version to be published.
