Abstract
Objective: Rapamycin is a potent inducer of autophagy in podocytes. However, we still understand very little about how autophagy is regulated under podocyte injury conditions. This study aimed to investigate the role of autophagy in podocyte injury and the regulatory mechanism of the PI3K/Akt/mTOR signaling pathway in this process. Methods: The podocytes were cultured in vitro, and the apoptosis rate of each group was determined by flow cytometry. The protein expression and distribution of LC3-II were examined by immunofluorescence. The phosphorylation levels of Akt, LC3-II, mTOR, 4EBP1, and P70S6K were measured using Western Blot. Transmission electron microscopy was used to examine the changes in autophagosomes in each group. Results: Compared with the control group, the puromycin group (PAN) increased podocyte apoptosis, decreased numbers of autophagosomes, and downregulated LC3-II protein expression. Compared with the PAN group, the podocyte apoptosis rate decreased in the Rapamycin group (RAPA), the number of autophagosomes increased, and LC3-II protein expression was upregulated. In addition, PAN evoked an increase in p-Akt expressions, RAPA treatment induced a reversal of PAN-induced p-Akt upregulation, and the phosphorylation levels of mTOR, 4EBP1, and P70S6K were downregulated. Conclusion: PAN can damage podocytes by inhibiting podocyte autophagic activity and promoting apoptosis. Rapamycin can ameliorate PAN-induced podocyte damage by activating autophagy. This effect may be related to rapamycin-mediated PI3K/AKT/mTOR signaling pathway and autophagy.
Introduction
Podocytes are inherent kidney cells, and podocyte damage is an important cause of proteinuria and glomerular sclerosis in nephropathy. 1 Under physiological conditions, podocytes rely on their innately high levels of autophagy to maintain the stability of the intracellular environment. 2 In recent years, studies have shown that autophagy plays an important role in various types of kidney diseases, especially in the maintenance of daily kidney functions, the progression of certain diseases, and the process of kidney aging. In addition, factors such as ischemia, hypoxic injury, toxic substances, and the immune response can induce autophagy in renal epithelial cells. 3 However, the regulation of podocyte autophagy and the related signaling pathways is still poorly understood. The present study investigated the effects of rapamycin-induced autophagy on podocyte injury and the underlying molecular mechanism, providing a basis for the use of mTOR inhibitors to treat podocyte disease. So, this study may have important theoretical implications and clinical significance.
Methods
Podocyte culture and treatment
Conditionally immortalized mouse podocyteclone were cultured as previously described in detail. 4 The control group was cultured with RPMI 1640 medium containing 0.02% DMSO; the PAN group was added with PAN (50 μg/mL, Sigma Company) to cause injury; the RAPA group was added with RAPA (100 ng/mL, Sigma Company) 5 ; PAN + RAPA group was added with PAN (50 μg/mL) and RAPA (100 ng/mL, Sigma Company). And all experiments were repeated 3 times.
Flow cytometry analysis
The podocytes were digested with trypsin and washed twice with PBS. The buffer was prepared according to the instructions of the kit, and the podocytes were incubated with FITC-Annexin V and PI at 4°C for 1 h. Finally, the podocytes were analyzed by flow cytometry.
Immunofluorescence assay
The cell slides were fixed with precooled acetone at −20°C for 10 min, blocked with 0.3% Triton X-100 and 5% BSA for 30 min at room temperature, and incubated with anti-LC3 primary antibodies (Sigma Company) overnight at 4°C. After washing, the sections were incubated with fluorescent secondary antibodies for 1 h at room temperature. Finally, the specimens were stained with DAPI (Sigma Company) to visualize the nuclei. Images were taken under a fluorescent microscope.
Western blotting
Total protein extract and immunoblot analyses were performed as previously described, 4 and the total protein concentration was determined by the BCA method; then, the equal quantities of protein from podocytes were loaded on a 12% gel, after blocking, the membranes were incubated with anti-LC3 antibody overnight at 4°C. Subsequently, membranes were washed, and secondary antibody was added to the membranes for 1 h at room temperature. Finally, the Tanon 5200 chemiluminescence imaging system was used to visualize the proteins.
Transmission electron microscopy
The podocytes were collected by centrifugation and fixed using 2.5% glutaraldehyde. The alcohol was dehydrated and put in 100% acetone. The podocytes were fully soaked at 35°C for approximately 2 h, polymerized, and sliced. The podocytes in each group were double stained with uranyl acetate and lead citrate. The changes in the autophagosomes were carefully observed under an electron microscope.
Statistical analysis
All the data were statistically processed with SPSS version 26.0, and the data are presented as the mean ± standard deviation. Data were analyzed using a Student’s t-test, and p < 0.05 indicated that the difference was statistically significant.
Results
Assessment of podocyte apoptosis
To specifically dissect the role of the RAPA in the regulation of podocyte injury, Flow cytometry was used to determine the apoptotic rates in the different groups. The results showed that the apoptosis increased remarkably in the podocyte treated with PAN (p < 0.05); On the contrary, podocyte pretreated with RAPA got a lower apoptosis compared to the PAN group (p < 0.05), These results suggested that podocyte apoptosis induced by PAN was reversed by RAPA treatment. So, RAPA may protect the podocytes from apoptosis by the activation of autophagy. As shown in Figure 1. The rate of podocyte apoptosis were examined by flow cytometry. Note: Compared with the rate in the Con group, the podocyte apoptotic rate was increased in the PAN group, (p < 0.05). Podocyte pretreated with RAPA got a lower apoptosis compared to the PAN group (p < 0.05). *p < 0.05 vs. PAN; #p < 0.05 vs. Control.
Assessment of podocyte LC3-II protein expression and distribution
The expression of autophagic markers, LC3-II protein, in podocytes was detected by immunofluorescence following treatment with PAN, RAPA, and PAN+RAPA. The results showed that the expression of LC3-II was fairly weak in the control group, while the PAN-injured podocyte had a significantly decrease in LC3-II expression (p < 0.05). After pretreatment of podocyte with RAPA, the expression of LC3-II increased significantly comparing to the PAN group. Autophagy was activated in the podocytes following treatment by RAPA, As shown in Figure 2. The Immunofluorescence staining was utilized to detect LC3-II expression in podocytes. Scale bar, 20 μm.
Assessment of podocyte protein by Western blot analysis
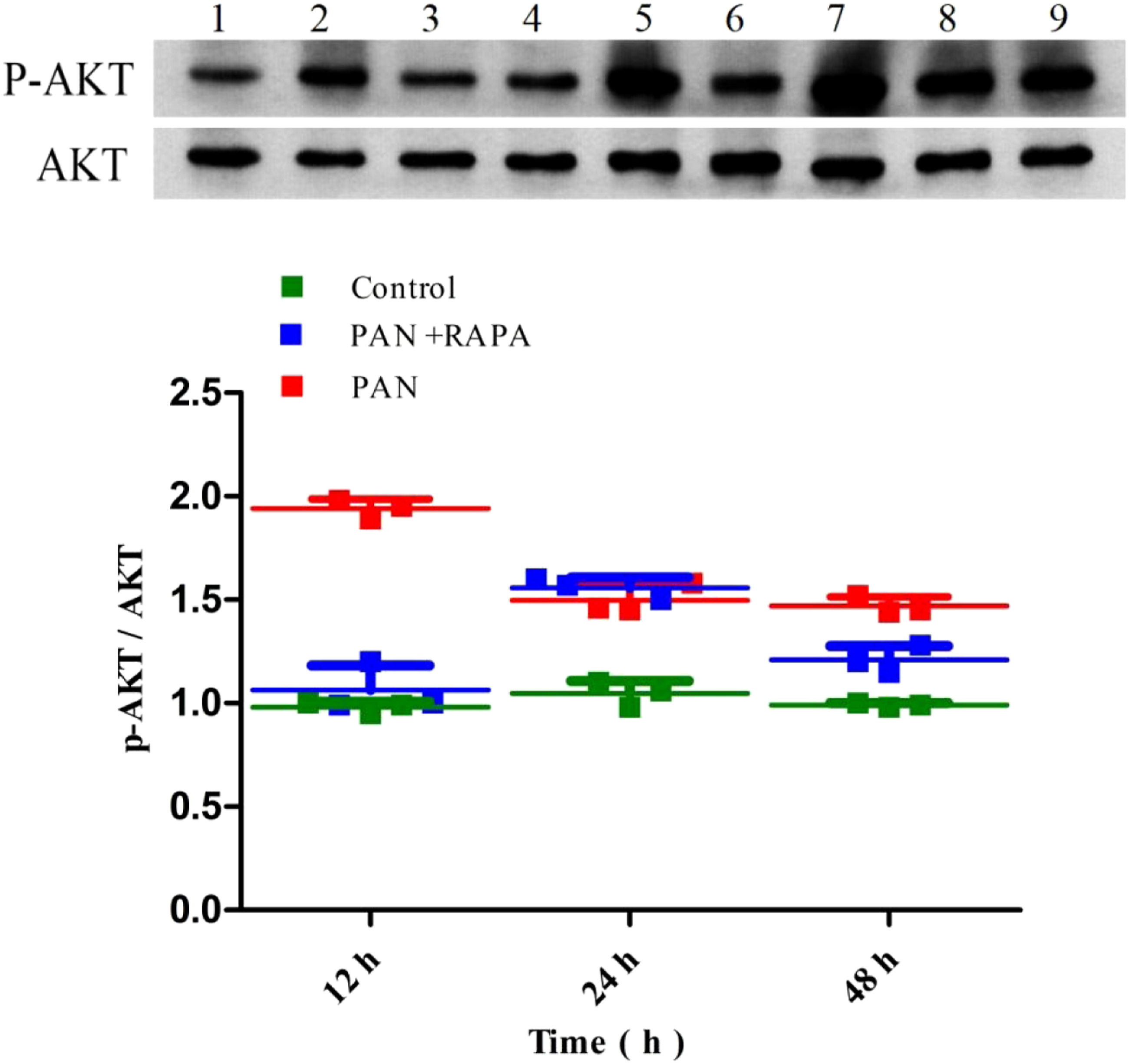
The Western blot analysis was performed to measure the expression of LC3-II, p-Akt/Akt, and mTOR signaling pathway proteins in podocytes. The results showed that compared with the control group, the expression of LC3-II in the PAN group was significantly reduced (p < 0.05), the expression of LC3-II in the PAN + RAPA group was higher than that in the PAN group (p < 0.05). The analysis of p-Akt/Akt showed that the p-Akt/Akt ratio of the PAN group was significantly higher than that of the control group (p < 0.05). In addition, compared with the control group, the phosphorylation levels of mTOR, 4EBP1, and P70S6K in the PAN group were upregulated, and the phosphorylation levels of mTOR, 4EBP1, and P70S6K in the RAPA group were downregulated. Compared with the PAN group, the phosphorylation levels of mTOR, 4EBP1, and P70S6K in the RAPA group were downregulated. The phosphorylation levels of mTOR, 4EBP1, and P70S6K were negatively correlated with podocyte autophagic activity (Figures 3-5). Expression of LC3-||protein detected by Western blot. Note: 1, 2, 3 represent the control at 12 h, 24 h, and 48 h, respectively; 4, 5, 6 represent the PAN+RAPA at 12 h, 24 h, and 48 h, respectively; 7, 8, 9 represent the PAN at 12 h, 24 h, and 48 h, respectively. *p < 0.05 vs. control; #p < 0.05 vs. PAN. Expression of p-AKT, AKT protein detected by Western blot. Note: 1, 2, 3 represent the control at 12 h, 24 h, and 48 h, respectively; 4, 5, 6 represent the PAN+RAPA at 12 h, 24 h, and 48 h, respectively; 7, 8, 9 represent the PAN at 12 h, 24 h, and 48 h, respectively. Expression of mTOR, p-mTOR, 4EBP1, p-4EBP1, P70S6K, and p-P70S6K protein detected by Western blot. Note: *p < 0.05 vs. Control; #p < 0.05 vs. Control.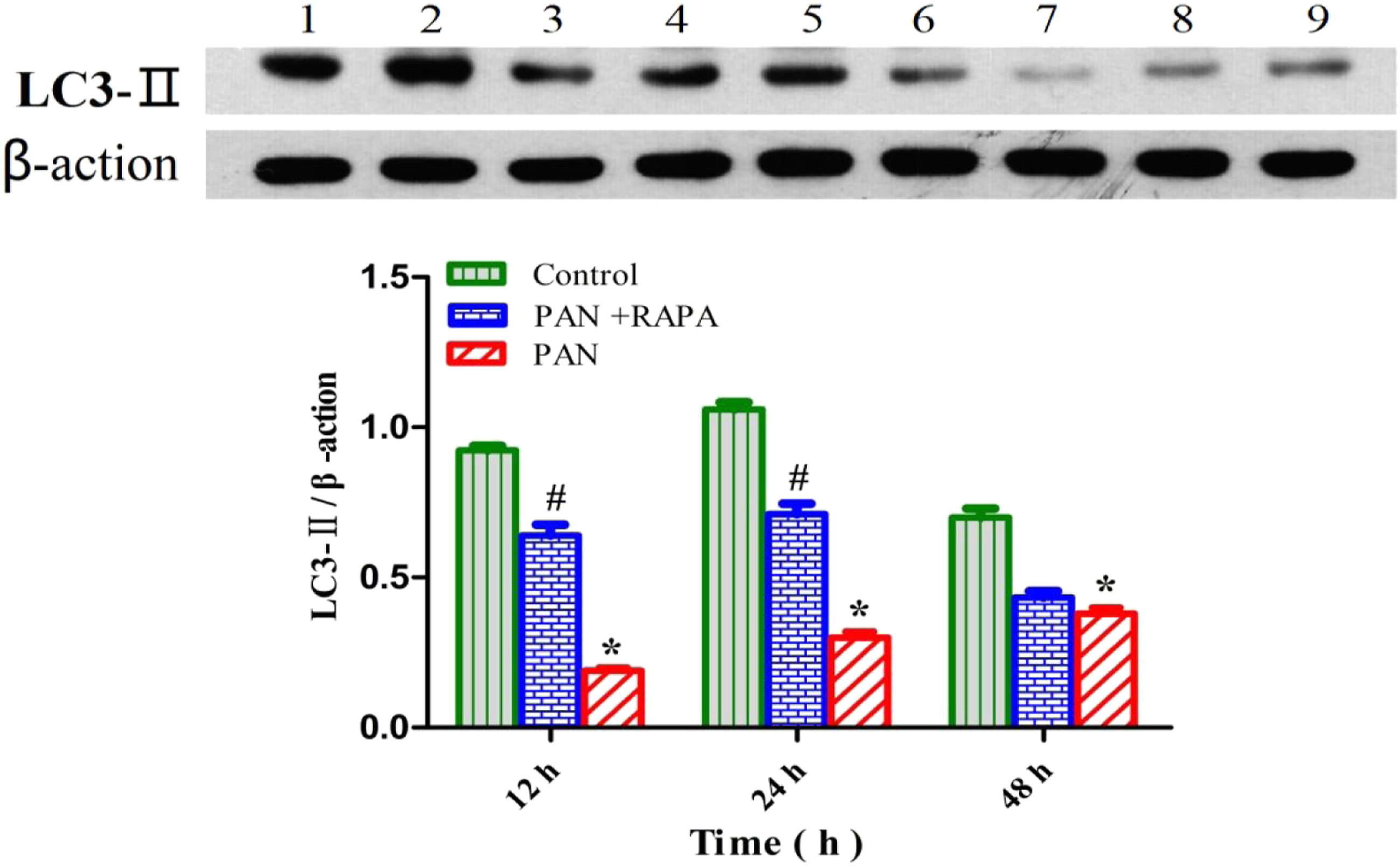

Changes in the number of podocyte autophagosomes in each group
Transmission electronic microscopy was used to observe the effect of PAN, RAPA, and PAN + RAPA on autophagy. The results showed that in the control group, the membrane surface of the podocytes had protrusions, the endoplasmic reticulum structure was clear and autophagosomes were visualized in the cytoplasm. However, in the PAN-treated podocytes, the cytoplasm contained a large number of vacuoles and few autophagosomes. The number of autophagosomes in the cytoplasm of the podocytes increased when treated with RAPA. As Shown in Figure 6. Effects of rapamycin-induced autophagy on the podocyte injury. Electron microscope was utilized to observe autophagosomes in the control group and PAN group with or without RAPA treatment. Scale bar, 1 μm.
Discussion
Autophagy plays an extremely important role in maintaining the integrity of podocytes and the glomerular capillary basement membrane (GBM). Studies have shown that in many glomerular diseases, such as membrane nephropathy, 6 diabetic nephropathy, 7 and focal segmental glomerulosclerosis, 8 podocytes exhibit downregulated phagocytic activity. In addition, podocyte-related molecules also play an important role in maintaining the glomerular filtration barrier via activating the PI3K/Akt signaling pathway and regulating autophagy.9,10 The PI3K/AKT pathway can regulate podocyte autophagy, damage podocytes, and destroy the stability of the SD complex. 11 Studies have confirmed that the PI3K/Akt pathway participates in the development of diabetic nephropathy and septic kidney injury by regulating podocyte autophagy to induce podocyte damage. 12 Moreover, the PI3K/Akt pathway and podocyte autophagy are also involved in damage to other kidney tissues and the occurrence of kidney diseases.13–15 mTOR is an important regulator that balances cell growth and autophagy pathways.16–19 Studies have shown that when mTOR activity is inhibited, the phosphorylation of the two target proteins 4EBP1 and p70S6K downstream of mTOR is also inhibited, thereby promoting the formation of autophagosomes and vice versa. This data is consistent with our findings, the phosphorylation levels of mTOR, 4EBP1, and P70S6K were upregulated in podocyte injury induced by PAN, while the phosphorylation levels of mTOR, 4EBP1, and P70S6K were downregulated in the RAP group. The phosphorylation levels of mTOR, 4EBP1, and P70S6K were negatively correlated with podocyte autophagic activity. In addition, the activity of mTOR depends on the regulation of the PI3K/Akt pathway. Under physiological conditions, the tyrosine kinase receptor is activated by extracellular and intracellular factors to activate PI3K. Activated PI3K catalyzes the transformation of the substrate PIP2 to PIP3 and the formation of phosphoric acid. Inositol-dependent kinase-1 (PDK-1) participates to fully activate Akt. After Akt is activated, the signal is transmitted to mTOR. 20 Activated mTOR activates its downstream factors and inhibits autophagy. 21 Therefore, regulating the PI3K/Akt and mTOR pathways is of great importance for maintaining podocyte autophagy.22,23
Studies have shown that knocking out podocyte mTOR to disrupt the physiological autophagy axis in a mouse model eventually results in the development of proteinuria, which leads to glomerular sclerosis. Excessive inhibition of mTOR can cause podocyte damage. 24 Furthermore, it was found that mTOR activity was enhanced in patients with diabetic nephropathy and in mouse models, accompanied by a decrease in the level of podocyte autophagy. 25 Inhibition or overactivation of mTOR expression can cause imbalances in podocyte autophagy balance and ultimately lead to structural and functional disorders. Our research results also confirmed that the expression of LC3-II in the RAPA group was significantly higher than that in the PAN group.
Our previous studies have also confirmed that there is a certain correlation between podocyte autophagy and apoptosis. 26 In a renal tubular epithelial cell (RTEC) injury model, PAN has also been shown to inhibit autophagy. Phagocytosis can promote RTEC apoptosis; In contrast, increasing the level of autophagy can inhibit RTEC apoptosis. 27 Another study showed that when cells undergo apoptosis, the autophagy-related protein Atg 5, similar to Beclin 1, loses autophagy-inducing activity and interacts with Bcl-2 on mitochondria to accelerate apoptotic death.28,29 Atg 4D, like Beclin 1 and Atg 5, is cytotoxic after caspase 3 cleavage and can also promote apoptosis. 30 This finding is consistent with our research results, that is, PAN can promote podocyte apoptosis by reducing the expression level of autophagy proteins. On the other hand, our findings also suggest that in addition to directly inducing the BAD pathway to initiate apoptosis, PAN can also reduce autophagy. The occurrence of autophagy will affect the survival rate of podocytes, and the synergy of the two processes will cause podocyte damage and eventually cell death.
However, this study is undertaken in vitro with a podocyte cell line, this is a limitation of this study, experimental results need to be further validated using animal experiment experimental and clinical studies.
Conclusion
In summary, this study confirmed the role and significance of autophagy in podocyte injury. Rapamycin can inhibit the PI3K/AKT/mTOR signaling pathway and upregulate autophagy to alleviate podocyte injury. This study provides new ideas for studying the biological functions of podocytes, podocyte damage, and the occurrence and development of proteinuria from the perspective of autophagy.
Footnotes
Author’s contributions
YSY and LR participated in the study design, data interpretation, and manuscript drafting. RQ and CJ performed the experiments. HJ revised the article critically for important intellectual content. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the fund from High Level-Hospital Program, Health Commission of Guangdong Province, China (No.HKUSZH201901018); Guangzhou Planned Project of Science and Technology (No.202102080447); Guangzhou General Science and Technology Project of Health and Family Planning (No.20211A011016); Guangdong Science and Research Project of Traditional Chinese Medicine Bureau (No.20222158); The Science Foundation of Guangzhou First People’s Hospital (No.M2019020);Guangzhou Science and Technology Project of Traditional Chinese Medicine and Combined Chinese and Western Medicine (No.20222A010037).
Ethics approval and consent to participate
This study did not require additional specific approval from an ethics committee as it does not involve research on human subjects.
