Abstract
Background
Nicotinamide N-methyltransferase (NNMT), a metabolic enzyme in the liver, has been implicated in various biological processes, and its high expression in hepatocellular carcinoma has been linked to tumor metastasis and poor prognosis. However, its potential as a serum biomarker for hepatocellular carcinoma diagnosis remains unexplored.
Methods
A total of 172 subjects were included in this study, consisting of 71 hepatocellular carcinoma patients (64 with hepatitis B virus (HBV)-associated hepatocellular carcinoma and 7 with non-HBV-associated hepatocellular carcinoma), as well as 70 healthy controls and 31 HBV-infected individuals. Serum NNMT levels were measured, and clinical–pathological correlations were analyzed. The diagnostic efficacy of serum NNMT for HBV-related hepatocellular carcinoma was evaluated using receiver operating characteristic (ROC) curve analysis.
Results
Serum NNMT levels were significantly elevated in HBV-infected individuals and correlated with poorer prognosis, including reduced overall survival and shorter disease-free survival. Kaplan–Meier analysis revealed that low NNMT expression was associated with longer overall survival (75 vs. 12 months,
Conclusions
Serum NNMT is a promising biomarker for predicting the risk of hepatocellular carcinoma in HBV-infected individuals and may serve as an indicator for the prognosis of hepatocellular carcinoma patients.
Introduction
Hepatocellular carcinoma (HCC) is the sixth most common cancer worldwide and the third leading cause of cancer-related mortality. 1 Its insidious onset, aggressive nature, and high recurrence rate present significant challenges to global health. The conventional biomarker alpha-fetoprotein (AFP) exhibits limitations in sensitivity and specificity, highlighting the urgent need for novel serum biomarkers to complement existing diagnostic tools.
NNMT is a methyltransferase that directly impacts cellular methylation levels and energy metabolism. 2 Under normal physiological conditions, NNMT is predominantly expressed in the liver, with lower expression levels found in other tissues, including the myocardium, smooth muscle cells, adipocytes, lungs, and bladder. 3 NNMT is primarily localized in the cytoplasm, where it plays a crucial role in the metabolic conversion of nicotinamide (NAM) and related compounds, specifically catalyzing the conversion of NAM to 1-methylnicotinamide (1-MNA). Subsequently, 1-MNA is oxidized by aldehyde oxidase 1 (AOX1) to produce N1-methyl-2-pyridone-5-carboxamide (2PY) and N1-methyl-4-pyridone-3-carboxamide (4PY), both of which are excreted in the urine. 4 Additionally, NNMT indirectly influences methylation levels by depleting S-adenosylmethionine (SAM), a critical methyl donor, thereby affecting epigenetic modifications. Research indicates that aberrant regulation of NNMT and its metabolites in various liver diseases—such as alcoholic liver disease, 5 nonalcoholic fatty liver disease (NAFLD),6,7 cirrhosis, and HCC 8 —is closely linked to the progression of liver conditions, suggesting a significant role for NNMT in liver disease pathogenesis. 4
Moreover, numerous studies have reported elevated NNMT expression in blood, suggesting its potential as a novel early diagnostic marker for lung and colorectal cancers.9–11 Building on our previous investigations into NNMT expression and function in HCC, we found that high NNMT expression correlates with HCC progression. This correlation was evidenced by the relationship between elevated NNMT levels in HCC tissues and factors such as vascular invasion, increased serum hepatitis B virus (HBV)-DNA levels, and distant metastasis. Functionally, NNMT promotes HCC cells invasion and metastasis by modifying the mesenchymal characteristics of tumor cells through alterations in histone H3 methylation patterns and transcriptional activation of CD44. 12 Consequently, we hypothesized that NNMT may serve as a specific and promising prognostic biomarker and therapeutic target.
To date, no studies have investigated the application of NNMT as a serum marker for HCC diagnosis. 13 Thus, we conducted a foundational study on the utility of serum NNMT for the early diagnosis of HCC. Our results demonstrated that serum NNMT levels were significantly elevated in patients with HBV-related liver disease and were positively correlated with poor prognosis. Further analysis of serum NNMT as a diagnostic marker for HCC revealed that it outperformed AFP in diagnosing HBV-related HCC. When serum NNMT and AFP were analyzed together, the area under the receiver operating characteristic (ROC) curve (AUC) reached 0.947. These findings suggest that serum NNMT could serve as a specific risk indicator for HBV-associated HCC and may predict the prognostic survival of HCC patients. Furthermore, the combined diagnostic approach utilizing NNMT and AFP not only offers a novel strategy for diagnostic and prognostic evaluation in HBV–HCC but also enhances the management and therapeutic outcomes for HCC patients.
Materials and methods
Study subjects
Four distinct groups of study participants were selected for this investigation: a healthy control group, a group of HBV-infected patients, a group of patients with non-HBV-related HCC, and an experimental group of patients with HBV-related HCC. The study subjects comprised 71 HCC patients who underwent surgical treatment between August 2017 and August 2018 at the Department of Hepatobiliary Surgery, Zhongshan Hospital, Xiamen University. Among these patients, 64 had HBV-associated HCC, while 7 had non-HBV-associated HCC. Additionally, the study included 70 individuals aged 40 years and older who underwent health checkups at the hospital's medical checkup center during the same period and 31 participants with HBV-infected. Among them, the HCC surgical group underwent hepatectomy and was classified as Barcelona Clinic Liver Cancer Stage A, without macrovascular invasion or extrahepatic metastasis. The HBV-infected patients are newly diagnosed HBV-infected individuals who have not received antiviral therapy and have not been screened for cirrhosis. However, the absence of significant cirrhosis can be inferred from clinical presentation and liver function test results. Thus, the total number of subjects was 172. All participants provided informed consent and relevant medical records prior to their involvement in the study. Furthermore, all HCC patients were followed up until May 2024. For patients who did not receive surgical treatment at Zhongshan Hospital, detailed records of their initial treatment were maintained in the medical records.
Collection of serum samples
Following the acquisition of informed consent from all study participants, 3 mL of venous blood samples were collected using vacuum blood collection tubes from each participant in a fasting state during the morning. Upon collection, the samples were processed by centrifugation at 4°C at 4000 rpm for 10 min to obtain serum. The resulting serum samples were subsequently stored at −80°C until they were utilized for blood biochemical analyses. Then, the samples were measured by the Fujian Provincial Key Laboratory of Chronic Liver Disease and Hepatocellular Carcinoma.
Elisa assay for serum NNMT
In this study, we used the human NNMT ELISA kit (Cat#JL19694, Jianglai Biotechnology Co., Ltd China) for the enzyme-linked immunosorbent assay
Statistical methods
In this study, statistical analyses were performed using SPSS version 21, while data visualization was conducted with GraphPad Prism version 10. The results of the statistical parameters were processed based on the distribution characteristics of the data. If the data followed a normal distribution, results were expressed as means ± standard deviations; if the data did not conform to a normal distribution, the median and interquartile range were used to represent the central tendency and dispersion of the data.
For comparisons involving more than two groups, a one-way analysis of variance (ANOVA) was employed. When conducting multiple comparisons, the least significant difference method was preferred. Detailed descriptions of the statistical methods are provided in the main text. Pearson correlation analysis was utilized for normally distributed data, while Spearman correlation analysis was applied to non-normally distributed data to evaluate the correlation between clinical test indicators and serum NNMT levels. Additionally, a binary logistic regression model was developed to analyze the data and create a diagnostic model for the combined indicators. The Hosmer–Lemeshow test was used to assess the goodness-of-fit of the logistic regression model, with a significance threshold set at
Results
NNMT expression levels in serum of patients with HBV infection and HCC
While our previous research demonstrated a correlation between high NNMT messenger RNA (mRNA) expression in HCC tissues and tumor metastasis, cirrhosis, and serum HBV levels, the primary focus of this study is to investigate whether a similar correlation exists at the serum level.
12
To address this, a total of 172 serum samples were collected for NNMT analysis, including 70 healthy controls, 31 HBV-infected patients, 64 patients with HBV-associated HCC, and 7 patients with non-HBV-associated HCC. ELISA results indicated lower serum NNMT expression levels in the healthy control and non-HBV-associated HCC groups; in contrast, NNMT was significantly upregulated in both the HBV-infected and HBV-associated HCC groups, with no significant difference observed between these two groups (HBV vs. HBV–HCC,
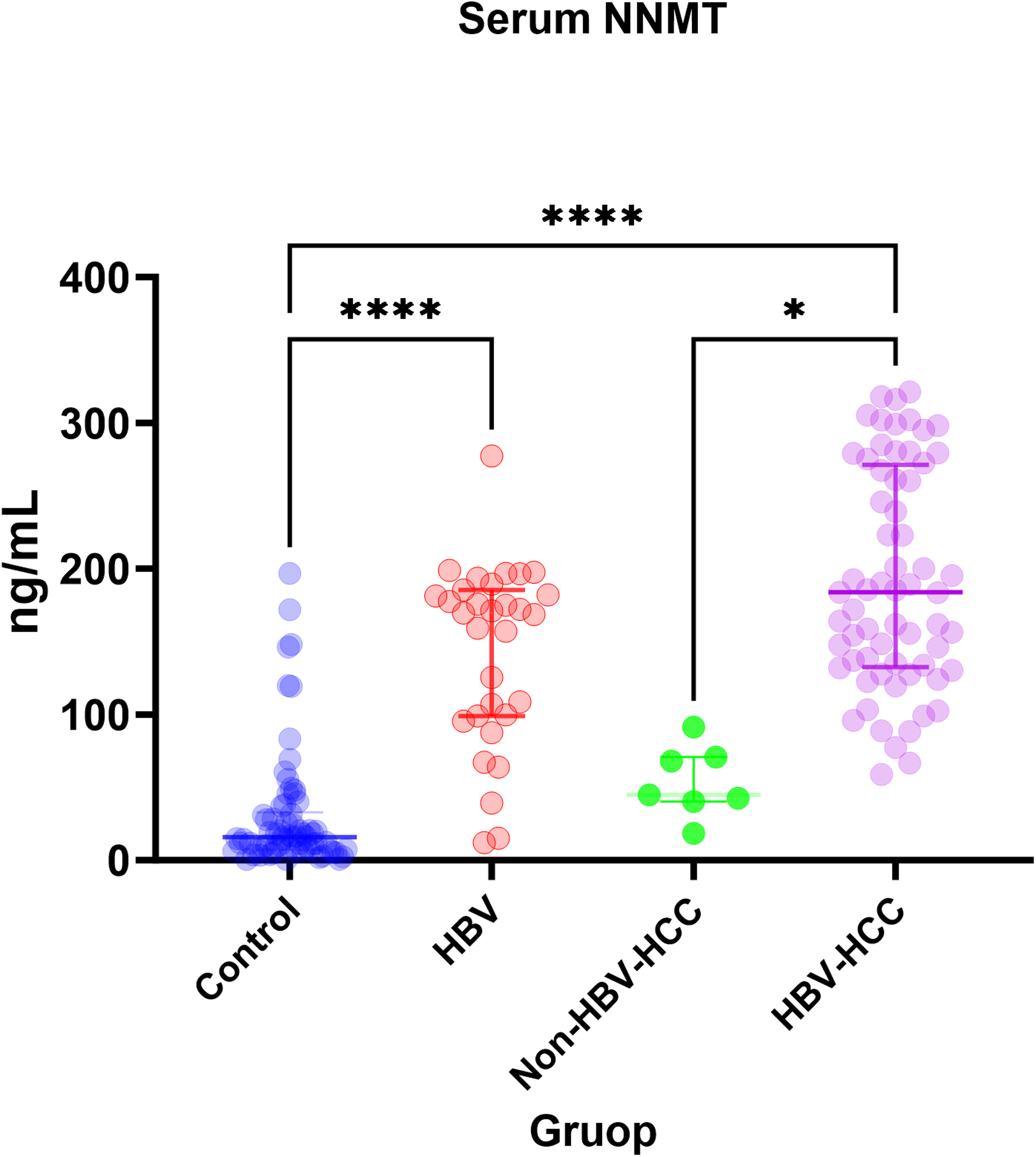
Expression levels of NNMT in the serum of healthy individuals, HBV-infected patients, and those with HCC. In this study, serum NNMT levels were assessed in 70 healthy controls, 31 HBV-infected patients, 64 patients with HBV-related HCC, and 7 patients with non-HBV-related HCC. A pairwise comparison analysis was conducted using one-way ANOVA. * represents
Impact of serum NNMT expression on clinical features and survival of HBV–HCC patients
To further investigate the impact of serum NNMT expression levels on the clinical features and survival of patients with HBV-related HCC, this study conducted a comparative analysis of serum NNMT and related clinical indicators in 64 patients with HBV-associated HCC. First, we divided the 64 cases of HBV-HCC into two groups based on the median serum NNMT value of 183.8 (132.6, 271.3); second, we conducted a comparative analysis. The results, as shown in Table 1, revealed significant differences in several biochemical markers between the high NNMT group and the low NNMT group, including AFP, alanine aminotransferase (ALT), AST, triglycerides, total cholesterol, LDL, apolipoprotein B (ApoB), and albumin. Moreover, Kaplan–Meier survival curve analysis indicated that the overall survival (OS) of patients in the low NNMT expression group was significantly longer than that of the high expression group (
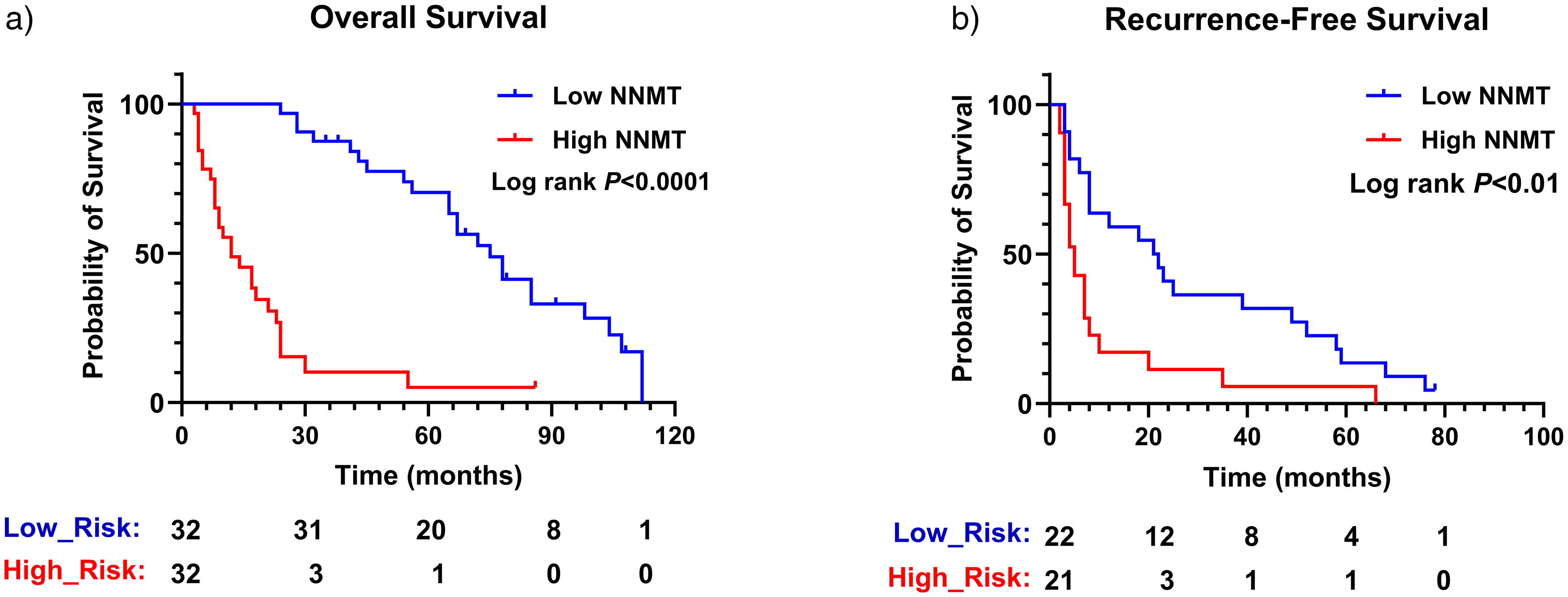
Impact of high and low serum NNMT levels on overall survival and disease-free survival in HBV-related HCC patients. Patients were stratified into high and low groups based on median serum NNMT expression, followed by a comparative analysis of their survival outcomes. (a) Overall survival; (b) Disease-free survival.
Demographic and clinical characteristics of HBV-related HCC in this study.
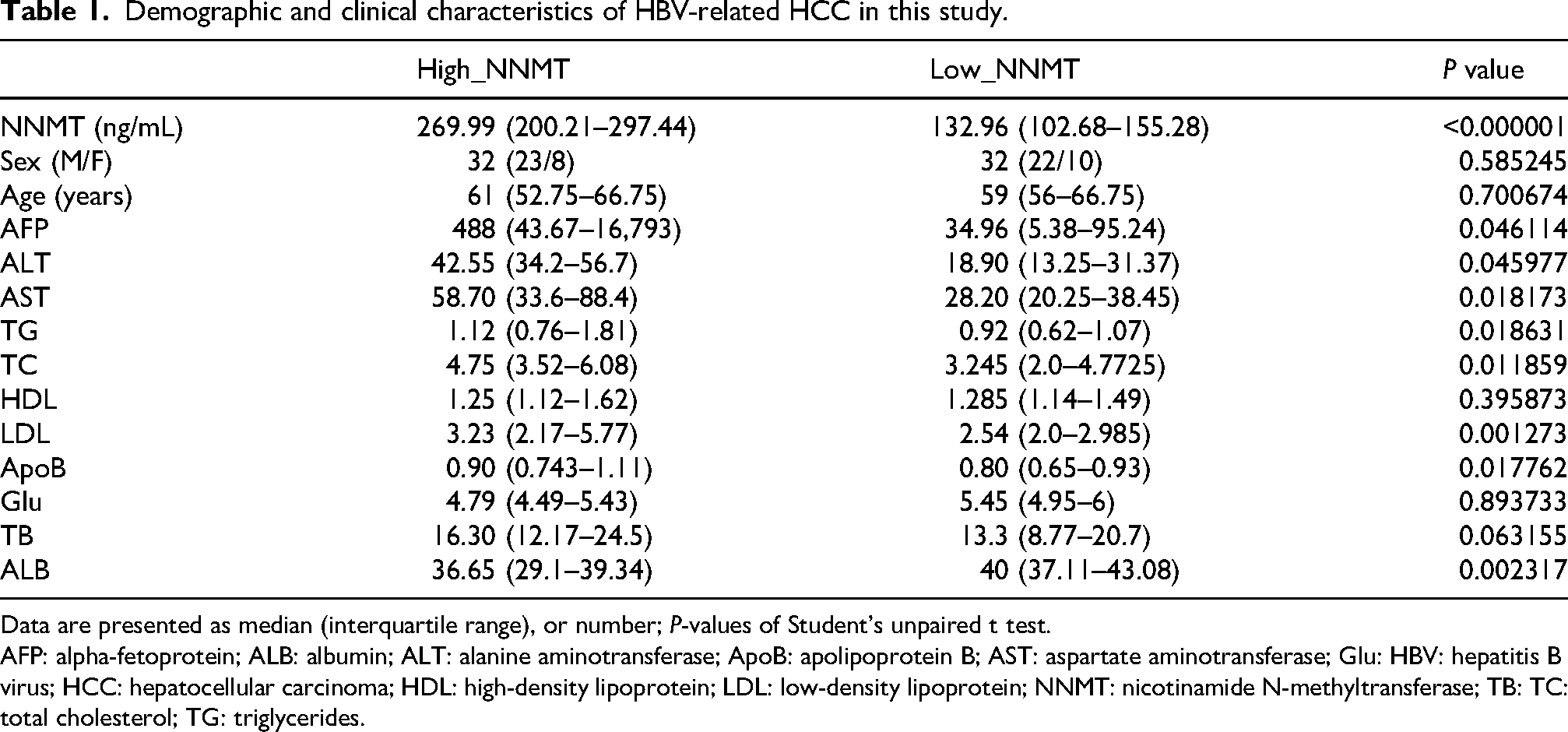
Data are presented as median (interquartile range), or number;
AFP: alpha-fetoprotein; ALB: albumin; ALT: alanine aminotransferase; ApoB: apolipoprotein B; AST: aspartate aminotransferase; Glu: HBV: hepatitis B virus; HCC: hepatocellular carcinoma; HDL: high-density lipoprotein; LDL: low-density lipoprotein; NNMT: nicotinamide N-methyltransferase; TB: TC: total cholesterol; TG: triglycerides.
Serum NNMT as a potential biomarker for risk assessment of HBV–HCC
To investigate whether serum NNMT can serve as a biomarker for assessing the risk of HBV-related HCC, we conducted a correlation analysis between serum NNMT levels and clinical biochemical indicators in HBV–HCC patients. The results of the Pearson correlation analysis revealed that NNMT expression levels were significantly positively correlated with AFP, AST, TG, TC, ApoB, and LDL. In contrast, NNMT expression showed a negative correlation with ALB levels (Figure 3).
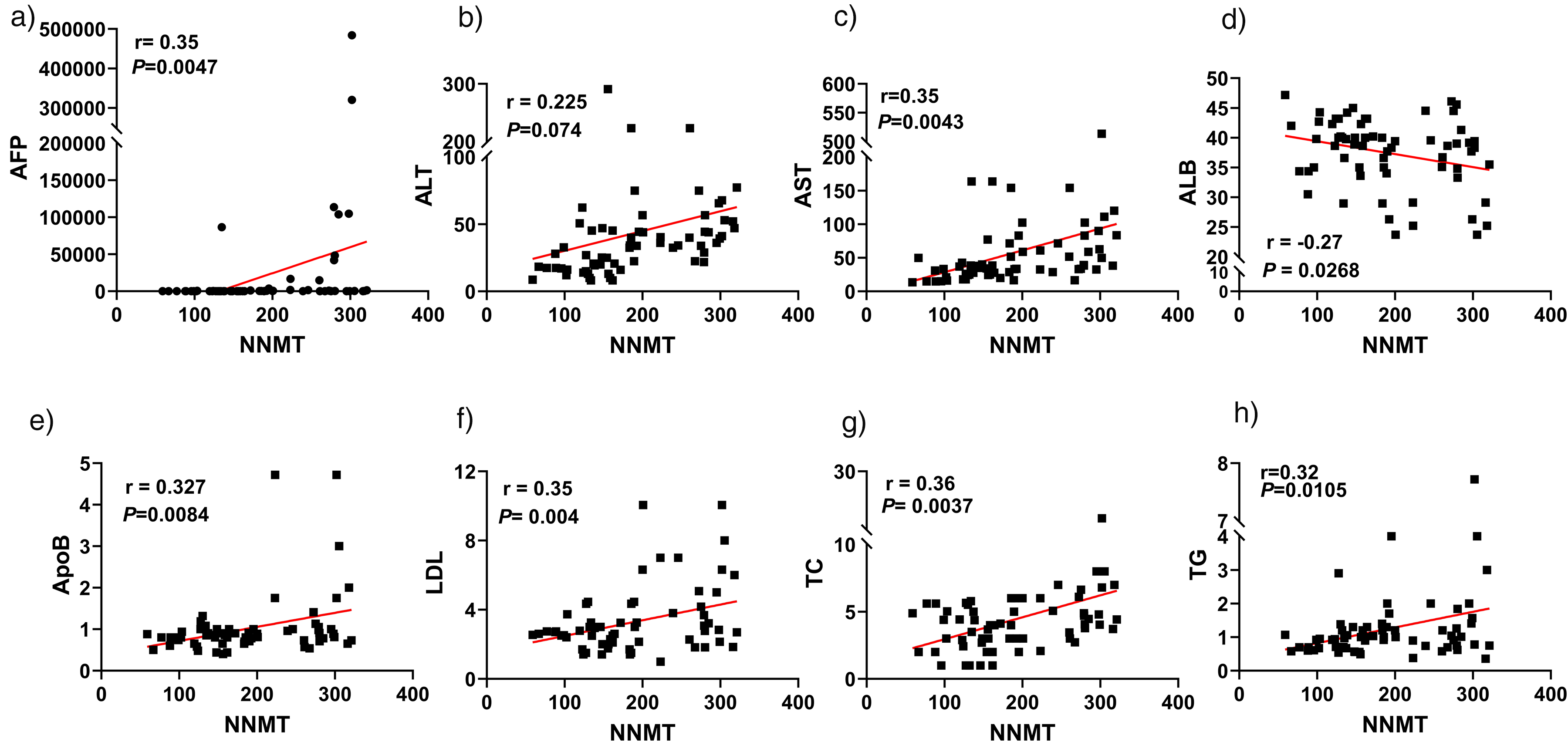
Correlation analysis of serum NNMT with other biochemical parameters in HBV-related HCC patients. (a) Alpha-fetoprotein (AFP); (b) Alanine aminotransferase (ALT); (c) Aspartate aminotransferase (AST); (d) Albumin (ALB); (e) Triglycerides (TG); (f) Total cholesterol (TC); (g) Apolipoprotein B (ApoB); (h) Low-density lipoprotein (LDL). A
To further elucidate the true relationship between serum NNMT and clinical indicators, this study utilized serum NNMT as the dependent variable and selected clinically relevant biochemical indicators as independent variables for multiple linear regression analysis. The results (Table S2 to S4) indicated that although the goodness of fit
Assessment of serum NNMT as a diagnostic biomarker for HBV–HCC
To evaluate the diagnostic efficacy of serum NNMT as a biomarker for HBV-related HCC, we conducted a ROC analysis using serum NNMT as a variable to assess its diagnostic potential for HCC. The results, presented in Figure 4 and Table S5, revealed that the area under the ROC curve for serum NNMT was 0.869, significantly outperforming AFP, which had an area of 0.775. Thus, serum NNMT demonstrated superior diagnostic performance for HBV–HCC compared to AFP.
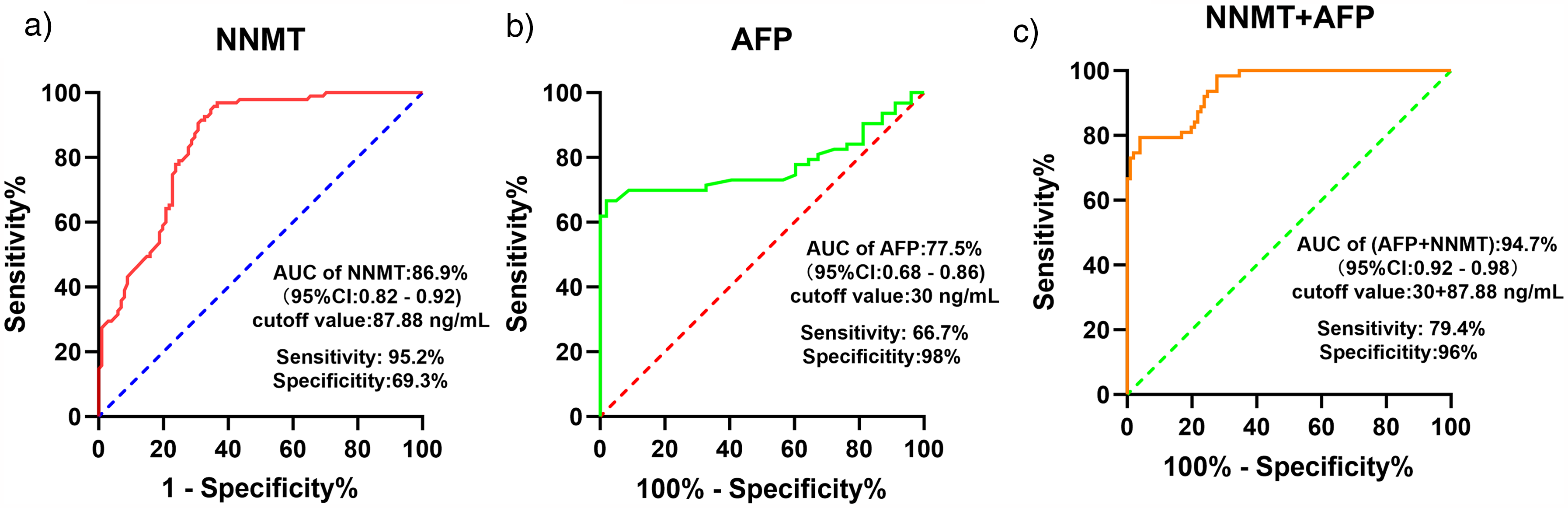
Evaluation of the diagnostic efficacy of serum NNMT alone and in combination with AFP for the diagnosis of HBV-related HCC. (a) represents the diagnostic performance of serum NNMT alone; (b) represents the diagnostic performance of serum AFP alone; (c) represents the diagnostic performance of the combination of serum NNMT and AFP for HBV-HCC.
Further analysis of their sensitivity and specificity revealed that, when the cutoff point for AFP was set at 30 ng/mL, the maximum Yoden index was 0.647, with a sensitivity of 66.7% and specificity of 98.0%. In contrast, when NNMT was set at a cutoff point of 87.88 ng/mL, the maximum Yoden index was 0.645, demonstrating a sensitivity of 95.2% and specificity of 87.8%. When both markers were analyzed jointly, the AUC reached as high as 0.947. Therefore, we believe that NNMT may serve as a promising serum marker for HBV–HCC.
Discussion
HCC is a highly malignant and heterogeneous tumor, making early diagnosis and treatment a critical focus of medical research. Improving patient outcomes and prognosis depends on the implementation of effective diagnostic tools and personalized treatment strategies. 14 Therefore, the development of more sensitive and specific biomarkers, along with comprehensive integration and comparative analysis, is essential to identify the most clinically applicable combinations. This approach not only provides a scientific basis for the early diagnosis of HCC but also opens new avenues for personalized treatment. 15
In this study, we investigated the role of serum NNMT in the diagnosis of HCC, building on previous findings that indicated high NNMT transcript levels in HCC tissues are associated with tumor metastasis, poor prognosis, and a positive correlation with HBV-DNA expression. 12 Our results confirm that serum NNMT is a specific marker for HBV-related HCC, outperforming AFP as a standalone diagnostic tool. When combining serum NNMT and AFP for diagnosis, the AUC reached 0.947, significantly enhancing the diagnostic accuracy for HBV–HCC. Additionally, elevated serum NNMT levels were inversely correlated with liver function markers such as ALB and HDL. These findings suggest that serum NNMT can serve as a specific risk indicator for HBV-associated HCC and may help predict the prognostic survival of HCC patients.
Current studies on NNMT expression in various tumors and its clinical significance have primarily focused on transcriptional levels in tissues. Findings indicate differential NNMT expression across tumor types. 16 For instance, NNMT expression is significantly elevated in gastric cancer, 17 renal clear cell carcinoma, 18 glioblastoma, 19 and diffuse large B-cell lymphoma.20,21 Conversely, NNMT expression is significantly lower in HCC22,23 and cholangiocarcinoma 24 compared to adjacent non-tumorous tissues. Further analysis of the relationship between NNMT expression and OS and DFS in these tumors was conducted. Kaplan–Meier survival analysis revealed that in tumors such as gastric cancer(17), colorectal cancer, 25 clear cell renal cell carcinoma, 18 HCC (12,22), and cholangiocarcinoma, 24 patients with high NNMT expression had significantly lower OS and DFS compared to those with low expression levels. In contrast, patients with high NNMT expression in prostate cancer 26 and diffuse large B-cell lymphoma 27 exhibited longer OS and DFS. These observations align with our previous findings in liver cancer. Additionally, a meta-analysis of 3340 patients with solid tumors from nine published studies suggested that elevated NNMT levels could serve as a biomarker for poor prognosis in solid tumor patients. 28 A substantial body of evidence indicates that NNMT is closely linked to cell proliferation, migration, invasion, progression, and differentiation in certain tumors, making it a crucial clinical reference. 13
Based on these experiments, we investigated the diagnostic potential of NNMT in the serum of HCC patients. By detecting serum NNMT levels in various HCC patients and analyzing their correlation with disease progression and clinical indicators, ELISA results revealed that serum NNMT was significantly elevated in HBV-infected and HBV-associated HCC patients compared to normal controls. In contrast, there was no significant change in other non-HBV-associated HCC patients. Although this result slightly differs from our previous study, which showed lower overall levels of NNMT mRNA in HCC tissues compared to adjacent non-tumor tissues, it aligns with the positive correlation between NNMT expression in HCC tissues and HBV-DNA levels in patients. This suggests that serum NNMT may serve as a specific indicator for predicting HBV infection.
Furthermore, we examined the correlation between serum NNMT levels and prognostic survival in HBV-associated HCC. The results indicated that HCC patients with high serum NNMT expression had significantly shorter OS and disease recurrence-free times compared to those with low serum NNMT expression. This suggests that abnormally elevated serum NNMT is a risk factor for poor prognosis in HCC patients. These findings are consistent with previous studies on serum NNMT in other tumors, such as non-small cell lung cancer and colorectal cancer.11,25
To further analyze the reasons for the discrepancy between serum NNMT expression in HCC and that in HCC tissues, we combined our experimental data with the available literature. We believe that the abnormal elevation of serum NNMT indicates abnormalities in liver function and lipid metabolism. The reasons are:
The difference between serum NNMT level and true liver NNMT level: The serum NNMT level does not objectively reflect the true NNMT level in the liver but represents the overall peripheral NNMT enzymatic level in the tissue. Although NNMT is primarily expressed in the liver, it is also expressed to varying degrees in adipocytes, smooth muscle cells, and other tissues. Therefore, NNMT in the blood represents the overall peripheral expression of the organism. Differences in biochemical effects of NNMT in liver and adipose tissue: The main function of NNMT is to participate in the regulation of nicotinamide (NAM) metabolism. However, the biochemical effects in liver and adipose tissue differ. In liver tissue, when NAM levels are low, NAM is provided to NAMPT to catalyze NAD + synthesis. Only when NAM levels are elevated is NNMT enzyme activity upregulated to remove excess NAM, counteracting the inhibitory effects of NAM on PARPs, sirtuins, or other enzymes. This process inhibits fatty acid and cholesterol synthesis in hepatocytes, thereby lowering intrahepatic triglyceride and cholesterol levels.29,30 Additionally, at acute pharmacological doses, NAM is rapidly converted to NAM-N-oxide primarily through CYP2E1.3,31 In hepatocellular carcinoma (HCC), tumor metabolism is characterized by enhanced gluconeogenesis, increased glucose uptake, lactate production, and augmented fatty acid and cholesterol synthesis, in response to the hypoxic microenvironment.8,32,33 Thus, HCC requires only low levels of NNMT to maintain the corresponding metabolic requirements. Furthermore, impaired detoxification due to tumor dedifferentiation has resulted in significant downregulation of NNMT and CYP2E1 in HCC.
23
The role of NNMT in adipose tissue: The function of NNMT in adipose tissue is not to generate energy via the NAM–NAD + pathway but to deplete S-adenosylmethionine (SAM) through high NNMT expression. NNMT is considered the major methyltransferase in adipocytes, thereby slowing down the spermine pathway and accumulating acetyl coenzyme A for lipid synthesis.
34
This correlates with the fact that high NNMT expression in human adipose tissue is positively associated with obesity and insulin resistance.
35
Serum NNMT originates from hepatic secretion and adipose tissue involvement, with elevated levels indicating poor glucose–lipid metabolism. Pearson correlation analysis revealed a significant positive correlation between NNMT expression and AFP, AST, TG, TC, LDL, APOB, and TB, while showing a negative correlation with ALB. Multiple linear regression analysis further confirmed that changes in HDL and ALB significantly negatively regulate NNMT levels, suggesting that elevated serum NNMT in HCC patients is associated not only with reduced ALB and HDL but also with the patients’ lipid metabolism. This finding aligns with literature trends indicating impaired liver function and disease progression. Thus, serum NNMT could serve as a potential risk assessment marker for malignant progression in HCC.
We also examined the efficacy of serum NNMT as an early diagnostic biomarker for HCC. ROC curve analysis demonstrated that serum NNMT outperformed the traditional marker AFP in diagnosing HBV–HCC. When combining serum NNMT and AFP, both sensitivity and specificity improved significantly, with an AUC reaching 0.94. These results suggest that serum NNMT could serve as a novel biomarker for HBV-associated HCC, aiding in early diagnosis and prognostic evaluation, thereby improving patient management and therapeutic outcomes. However, NNMT is located in the cytoplasm, and the mechanisms by which it is released into bodily fluids remain unclear. Additionally, the regulatory role of the liver on NNMT levels in peripheral organs requires further exploration and in-depth research. Furthermore, it is currently uncertain whether serum NNMT can truly serve as a predictive indicator for liver cancer. Therefore, large-scale clinical studies and prospective research are urgently needed to validate this hypothesis. Only through such studies can we objectively assess the specificity of NNMT as a serum biomarker and provide a more accurate and reliable theoretical basis for clinical diagnostic applications. In conclusion, serum NNMT shows significant potential for clinical application in the early diagnosis and personalized treatment of HBV-related HCC.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155251330664 - Supplemental material for Preliminary investigation of nicotinamide N-methyltransferase as an HBV-specific biomarker for hepatocellular carcinoma diagnosis
Supplemental material, sj-docx-1-jbm-10.1177_03936155251330664 for Preliminary investigation of nicotinamide N-methyltransferase as an HBV-specific biomarker for hepatocellular carcinoma diagnosis by Yi Zhang, Huayang Chen, Song You, Xiaoqin Chi, Yongxi Chen, Zhiyong Wu, Jiepeng Zheng, Xin Cheng, Jianming Liu and Jie Li in The International Journal of Biological Markers
Footnotes
Ethical considerations
The present study was approved by the Ethics Committee of Zhongshan Hospital of Xiamen University.
Author contributions/CRediT
All authors performed the experiments and analyzed the data. Yi Zhang wrote the manuscript. Yi Zhang and Huayang Chen contributed equally to this work. The final manuscript was thoroughly reviewed and endorsed by all authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used during this research are available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
