Abstract
Background
Lactate dehydrogenase (LDH) is a predictor of clinical outcome in hepatocellular carcinoma (HCC) patients. However, its predictive role in the clinical outcomes of sorafenib treatment has been poorly documented. The correlation between LDH levels and clinical outcomes in HCC patients treated with sorafenib and included in the nationwide Italian database ITA.LI.CA was investigated here.
Patients and Methods
The ITA.LI.CA database contains data for 5,136 HCC patients. All patients treated with sorafenib treatment and with available LDH values were considered. Overall survival (OS) and time to progression (TTP) were compared in patients with LDH levels above and below a defined threshold, determined through an ROC analysis. An explorative analysis investigated the relationship between the variation of LDH levels during treatment and response to sorafenib.
Results
Baseline LDH levels were available for 97 patients. The most accurate cutoff value for LDH concentration was 297 U/L. Patients with LDH values above (n=45) and below (n=52) this threshold showed equal OS (12.0 months) and TTP (4.0 months) values. Data on LDH levels during sorafenib treatment were reported for 10 patients. LDH values decreased in 3 patients (mean difference = -219 U/L) who also reported a prolonged OS and TTP versus those with unmodified/increased LDH (OS: NE (not evaluated) vs. 8.0 months, p=0.0083; TTP: 19.0 vs. 3.0 months, p=0.008).
Conclusions
The clinical benefits of sorafenib do not seem to be influenced by baseline LDH. According to the results of an explorative analysis, however, a decreased LDH concentration during sorafenib might be associated with improved clinical outcomes.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is one of the most frequent cancers worldwide, and its incidence is continuously increasing (1). In most cases, HCC develops in patients who have a history of chronic hepatitis or cirrhosis, because these conditions lead to continuous inflammation and regeneration of hepatocytes (2). The coexistence of inflammation and cirrhosis makes the early diagnosis and prognostic evaluation of HCC quite challenging. Therefore, the identification of valuable biomarkers for the diagnosis and treatment of HCC is a matter of research (2). In recent years, the prognostic value of several tumor markers has been extensively investigated (2–3–4–5–6–7–8–9–10–11–12).
Among these, lactate dehydrogenase (LDH) has recently attracted particular interest (11, 12). In many tumor types, such as colorectal and gastric cancer (13–14–15), serum LDH levels may represent an indirect marker of tumor hypoxia, a key biological mechanism for the development of treatment resistance in cancer cells via the formation of new blood vessels (11). Furthermore, mounting evidence indicates that hypoxia might actually promote cancer development. For instance, a recent study documented that pretreatment LDH levels can predict the clinical outcome for HCC patients who undergo transarterial chemoembolization (TACE), the most established treatment for intermediate-stage HCC (BCLC-B according to the Barcelona Clinic Liver Cancer classification) (11). On the other hand, the potential predictive role of LDH levels in the clinical outcomes of sorafenib treatment – the mainstay therapy for advanced-stage HCC (BCLC-C) – has been only poorly investigated. Since sorafenib acts mainly on the angiogenetic pathway, the correlation between the administration of this drug and markers of hypoxia such as LDH could be an important tool in patient management. To our knowledge, this issue has only been explored in a multicenter observational study recently published by Faloppi et al (12). They suggest that patients with higher LDH values before treatment (as well as patients with increasing LDH values during treatment) experience a significantly lower benefit from sorafenib in terms of both progression-free survival (PFS) and overall survival (OS) (12). Additional evidence on this topic is eagerly awaited.
We investigated here the correlation between baseline LDH levels and the clinical outcomes in HCC patients treated with sorafenib who were included in the large, nationwide Italian database ITA.LI.CA (Italian Liver Cancer). An explorative analysis was also conducted to investigate the potential correlations between the variation of LDH values during treatment and the response to sorafenib.
Patients and Methods
ITA.LI.CA database
The ITA.LI.CA database contains data for 5,136 HCC patients consecutively histologically diagnosed with HCC from January 1987 to date, at 18 Italian medical institutions (16, 17). These data have been prospectively collected and are updated every 2 years (last updated in April 2013). After data entry by any single center, the consistency of the data set is checked by the coordinating group, and when clarifications or additional information is needed, it is resubmitted to each center before statistical evaluation.
Patients provided their informed written consent before any therapeutic decision and consented to data collection and analysis. The study was conducted in accordance with the Declaration of Helsinki. The ethics committee of the ITA.LI.CA group approved this retrospective study. Patient records/information was anonymized and deidentified prior to analysis.
Data collection
We included in the study all patients who received sorafenib treatment and for whom LDH values were collected prior to the initiation of the treatment, during sorafenib therapy, at different time points according to the standard practice of each center, or both. LDH serum levels were determined at each participating center according to the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) method. In particular, we analyzed the LDH isoenzymes specific for the liver: LDH 4 and LDH 5. For the quantitative determination of LDH levels, we used a commercial test (LDH Pyruvate, Kinetic UV, DGKC; InterMedical, Villaricca, Italy) based on the photometrical measurement of nicotinamide adenine dinucleotide phosphate (NADPH), a substrate of LDH. The rate of decrease in concentration of NADPH is proportional to the catalytic concentration of LDH present in the sample, thus enabling its quantification.
Study end points
The following study end points were considered: OS, defined as time from the initiation of sorafenib to death for any cause (primary end point) or the last visit; time to progression (TTP), i.e., the time from the initiation of sorafenib to the first occurrence of metastasis or intrahepatic tumor progression; disease control (DC), defined as the sum of patients who achieved a complete response (CR) or a partial response (PR) or a stable disease (SD). These were established according to the Response Evaluation Criteria in Solid Tumors (RECIST) criteria (1.0, 1.1 or mRECIST according to the standard practice of each single center at the time of data entry).
Data analysis
All data were analyzed by descriptive statistics. Because studies of the evaluation of LDH as a biomarker for the diagnosis and treatment of HCC are lacking, a receiver operating characteristic (ROC) analysis was performed to measure the discrimination accuracy of LDH as a predictor of survival, and to find a suitable threshold for baseline LDH levels to be used in subsequent analyses. ROC analysis was performed at the overall median survival time (t=12 months) by means of the nearest neighbor estimator method of Heagerty et al for time-dependent ROC curves (18). OS and TTP differences in the 2 identified subgroups were then analyzed by means of Kaplan-Meier estimators, whereas association between LDH class and DC rate was evaluated with a contingency table of frequencies.
In an explorative analysis, differences in the study end points were also statistically tested according to different patterns of LDH levels which occurred during treatment with sorafenib (increased vs. decreased LDH levels as assessed at the initiation and the end of the treatment): due to the small sample size, OS and TTP were tested by means of an exact log-rank permutation test with Sun's scores (19, 20), whereas DC rate was tested by means of Fisher's exact test on a contingency table. All analyses and graphics were performed using the R statistical computing environment (21).
Results
Patient population
In total, data for 5,136 patients were available at the time of this analysis (July 2013). Of these, 322 had received sorafenib, and data on LDH levels prior to the initiation of treatment were available for 97 patients (treated between 2008 and 2013). Data on LDH levels during sorafenib therapy were reported for 10 patients.
Patients’ characteristics are depicted in Table I. The main cause of liver disease was HCV infection (n=45; 46%). Most patients were males (n=85; 88%) and were in good condition (performance status according to the Eastern Cooperative Oncology Group [PS ECOG] of 0-1) (n=87; 90%). The Child-Pugh class was A in 62 patients (64%), and the BCLC stage was B in 24 patients (25%) and C in 61 (63%). Patients classified as BCLC-B and BCLC-C presented with clinical characteristics typical of stage C and B according to the Barcelona Clinic Liver Cancer staging system. Median LDH concentration before the initiation of sorafenib was 291 U/L (range 129-730 U/L).
Baseline Characteristics Of Patients Evaluated
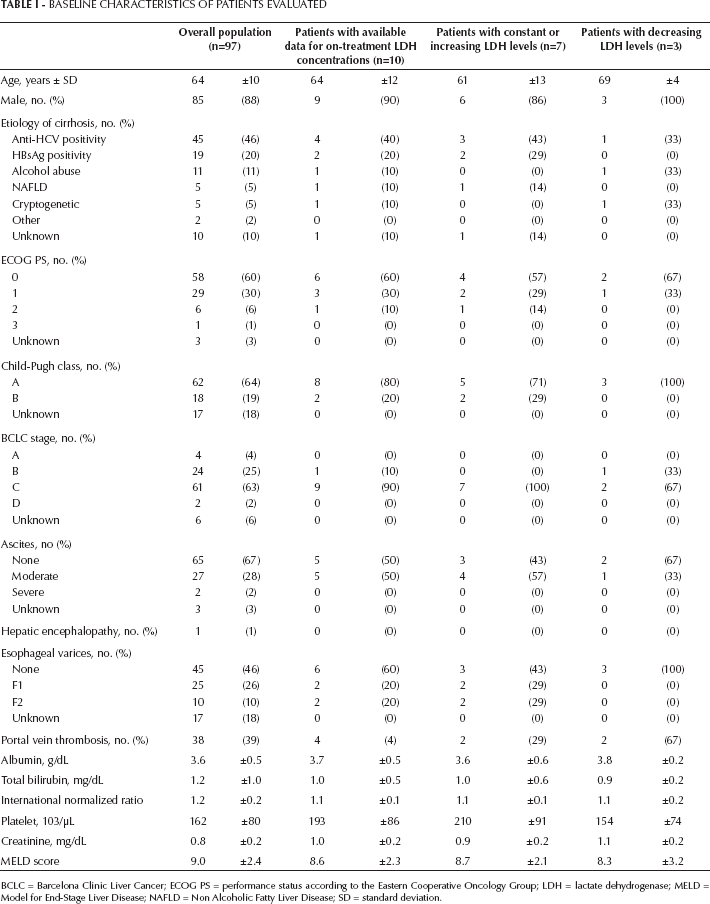
BCLC = Barcelona Clinic Liver Cancer; ECOG PS = performance status according to the Eastern Cooperative Oncology Group; LDH = lactate dehydrogenase; MELD = Model for End-Stage Liver Disease; NAFLD = Non Alcoholic Fatty Liver Disease; SD = standard deviation.
Analysis of pretreatment data
At the time of the analysis (September 2013), 50 patients (52%) had died, and median OS was 12.0 months (95% confidence interval [95% CI], 11.0-25.0 months). ROC analysis overall revealed a poor performance of basal LDH levels in predicting OS (area under the curve [AUC] = 0.531). The most accurate cutoff value was identified for a concentration of 297 U/L (Fig. 1): 45 patients were above this threshold, while 52 patients presented with baseline LDH levels lower than 297 U/L. This cutoff was associated with a sensitivity of 0.58 and a specificity of 0.67. No differences in OS were reported between patients with LDH values ≥297 and those with lower LDH concentrations (12.0 months in each population; p=0.922, 95% CI, 10.0-26.0 vs. 9.0-36.0 months, respectively) (Fig. 2A).
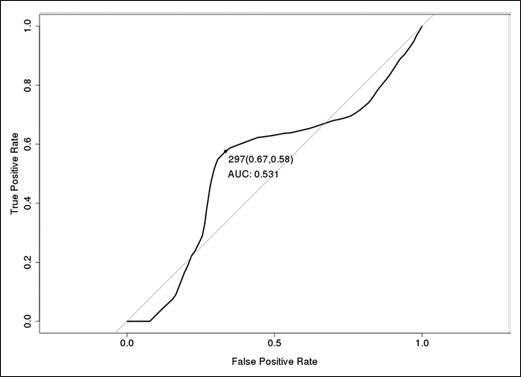
Receiver operating characteristic (ROC) curve for lactate dehydrogenase (LDH) as a predictor of overall survival (OS).
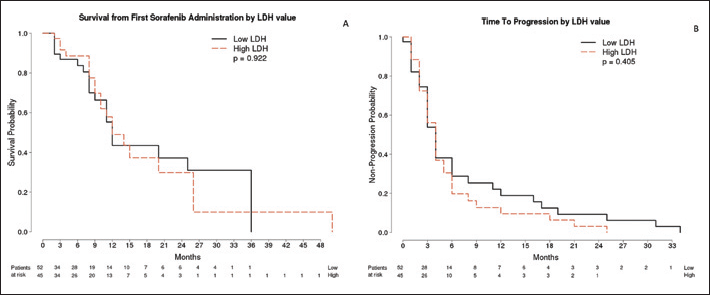
Kaplan-Meier analysis for overall survival (OS) (A) and time to progression (TTP) (B).
Median TTP in the overall population was 4.0 months (95% CI, 3.0-5.0 months). No differences between patients with LDH values ≥297 and those with lower concentrations were reported in terms of TTP (4.1 months, 95% CI, 3.0-5.0; vs. 3.9 months, 95% CI, 3.0-6.0 months; p=0.405) (Fig. 2B).
DC was achieved in 23 patients (24%; 11 patients with LDH values ≥297 [7 PR] and 12 [1 CR, 3 PR] among those with lower LDH), without differences between patients with LDH values ≥297 and those with lower concentrations (odds ratio = 1.13, 95% CI, 0.42-3.04, p=0.23).
Analysis of on-treatment data
During treatment, LDH values decreased in 3 patients (mean LDH difference = -219, range -20 to -400), whereas they increased or did not vary in 7 subjects (mean LDH difference = 182, range 0-806). Overall, the baseline characteristics of patients with decreased values were similar to those reported in patients with increased or unmodified values (Tab. I).
However, patients with decreasing LDH during treatment had a better OS, compared with those with unmodified or increased values (Fig. 3; p=0.0083, exact log-rank test). Median OS for patients who experienced decreasing LDH values was 10.0 months (95% CI, 8.0 to not applicable [NA] months [some patients were still alive at the time of analysis]). Median OS for patients who experienced increasing LDH values was 8.0 months (95% CI, 8.0-NA months). Median TTP was 19.0 months (95% CI, 16.0-NA months) in patients with decreasing LDH concentrations, and 3.0 months (95% CI, 2.0-NA months) in those with increasing values (p=0.008). In addition, they showed a longer exposure to sorafenib treatment. Median treatment duration was 3.0 months (95% CI, 2.0-NA months) for the increasing LDH group and 22.0 months (95% CI, 20.0-NA months) for the decreasing LDH group (p=0.017). None of the 7 patients with increasing LDH values achieved DC, whereas 2 patients with decreasing LDH values did (1 PR and 1 CR). However, Fisher's exact test did not show statistical significance in this respect (p=0.083).
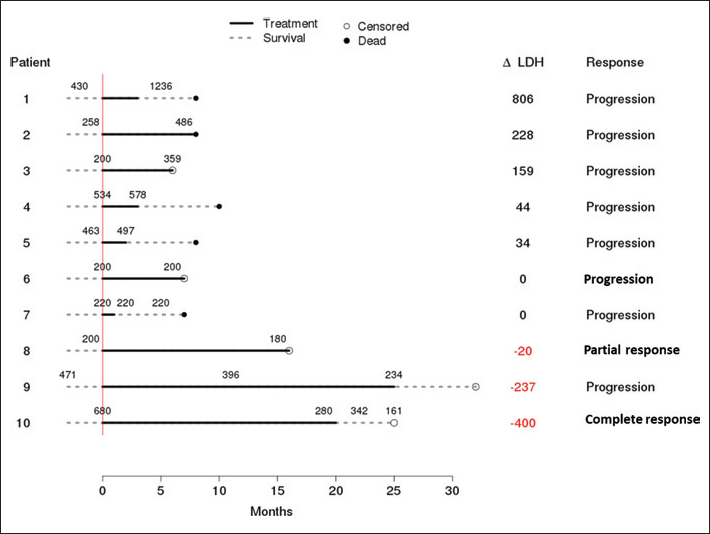
Survival and treatment outcomes in the 10 patients with available on-treatment lactate dehydrogenase (LDH) values.
Discussion
The results of our study suggest that LDH concentration prior to sorafenib treatment is not a predictor of clinical outcomes in HCC patients. In the clinical practice population we observed, the clinical benefit associated with sorafenib was in fact similar in patients with high and low baseline values of LDH (as defined by ROC analysis). In addition, it was overall in line with current clinical experience of the drug (22, 23). The observational nature of our analysis and the fact that data were collected in different centers according to the standard practice of each Institute should be taken into account for a complete evaluation of our findings.
This finding differs from what was reported in a recent study by Scartozzi et al, which showed that LDH seemed able to predict clinical outcomes for HCC patients undergoing another established treatment for HCC – i.e., TACE (11). However, differences between study populations may have contributed to these discrepancies: indeed, our series evaluated patients no longer suitable for a locoregional approach, when the tumor has arguably developed a more complex angiogenic network. Moreover, the modulation of tumor angiogenesis by TACE is relatively less complex than that achieved with a multitargeted agent such as sorafenib. In addition, a very recent study by the same group investigated the correlations between LDH values and the clinical outcomes associated with sorafenib (12). Overall, 78 subjects were analyzed: in patients with LDH values under 407 U/L, median PFS was 6.7 months, whereas it was significantly shorter (1.9 months; p=0.0002) in patients above this cutoff. Accordingly, median OS was 13.2 months and 4.9 months, respectively (p=0.0006). Although the sample size and baseline characteristics of the 2 populations are comparable, data from this study are in contrast with our analysis, as they would suggest that pretreatment LDH concentration can predict treatment benefit for HCC patients treated with sorafenib. The reasons for these different conclusions may lie in some differences in patient characteristics or in the employment of different cutoff values for LDH (408 vs. 291 U/L). New studies are needed to better clarify the relation between LDH levels at baseline and response to sorafenib. On the other hand, agreement can be seen with regard to the variation of LDH during treatment: both analyses reported that patients with decreased LDH values after treatment showed a better outcome – namely, they reported a median PFS of 6.8 months and median OS of 21.0 months, versus 3.9 months (p=0.0087) and 8.6 months (p=0.0035) observed in patients with increased LDH levels, respectively.
Taken together, these 2 reports suggest that LDH dynamics during treatment may be a better predictor of sorafenib benefit than a single value evaluated before treatment initiation. This finding may reflect the complexity of tumor angiogenesis, which, being a dynamic process, cannot be entirely captured by the single measurement of a unique biomarker. On the other hand, even though variations in LDH concentration do not depict the modulation made by all the actors involved in tumor angiogenesis, they may be a more reliable epiphenomenon of such changes.
The small sample size of the population with available on-treatment LDH data, the different durations of sorafenib treatment in single patients and the lack of a standardized timing for LDH measurements hampered our analysis, which should be considered explorative. However, the clinical characteristics of this subgroup were not different from those of the entire population analyzed, because most of the baseline characteristics showed very similar distributions, and all continuous variables differed by less than a standard deviation. In this respect, it is worth noting that our study analyzed the data collected from a large, nationwide Italian database, including a very large number of patients (5,136, of whom 322 were treated with sorafenib). However, data on LDH concentrations were available only for 97 patients (2% of the total and 31% of those treated with sorafenib). Given the mounting importance of investigations of biomarkers in the scientific debate on HCC management, and the inherent difficulty in having complete data in any analysis of large, clinical practice databases, we advocate that new targeted studies should be planned to explore the potential usefulness of LDH as a predictor of treatment outcome in HCC patients.
Of note, our study and that by Faloppi et al do not help in deciding the optimal schedule of LDH evaluation during sorafenib treatment: Indeed, in both these retrospective investigations, the timing of LDH sampling was not predetermined (in our study it was at investigator's discretion, while in the report by Faloppi et al, basal levels were compared with levels at the end of treatment). It will be of great interest to explore in future trials if changes in LDH values may anticipate radiological disease progression, thus further exploring their potential relevance as a predictive factor in treatment benefit. Moreover, sequential evaluation of LDH during treatment will also be important to define a threshold of LDH changes which is associated with treatment outcome and therefore may help in identifying subgroups of patients with different outcomes.
While such studies appear eagerly awaited, our analysis – even with the limitations of any retrospective database investigation – suggests that the clinical benefits associated with sorafenib are not influenced by pretreatment LDH values. In addition, according to the results of an explorative analysis, a decrease of LDH concentration during treatment might be associated with improved clinical outcomes.
Footnotes
*Other members of the ITA.LI.CA group
Dipartimento di Scienze Mediche e Chirurgiche, Alma Mater Studiorum - Università di Bologna: Franco Trevisani, Mauro Bernardi, Luigi Bolondi, Fabio Piscaglia, Marco Zoli, Maurizio Biselli, Paolo Caraceni, Alessandro Cucchetti, Marco Domenicali, Marta Frigerio, Virginia Erroi, Francesca Garuti, Annagiulia Gramenzi, Barbara Lenzi, Donatella Magalotti, Anna Pecorelli, Laura Venerandi; Dipartimento di Scienze Chirurgiche e Gastroenterologiche, Università di Padova: Fabio Farinati, Anna Giacomin, Veronica Vanin, Caterina Pozzan, Gemma Maddalo;Unità Operativa di Chirurgia, Policlinico S. Marco, Zingonia: Francesca Ciccarese, Paolo Del Poggio, Stefano Olmi; Unità Operativa di Medicina, Azienda Ospedaliera Bolognini, Seriate, Italia: Mariella Di Marco, Claudia Balsamo, Maria Anna Di Nolfo, Elena Vavassori; Dipartimento di Medicina Clinica e Sperimentale, Università di Padova: Alfredo Alberti, Luisa Benvegnù, Angelo Gatta, Maurizio Gios; Dipartimento di Malattie Apparato Digerente e Medicina Interna, Azienda Ospedaliero-Universitaria di Bologna, Unità Operativa di Radiologia: Rita Golfieri, Emanuela Giampalma, Cristina Mosconi, Matteo Renzulli; Unità di Medicina Interna e Gastroenterologia, Complesso Integrato Columbus, Università Cattolica di Roma, Roma: Gian Lodovico Rapaccini, Giulia Bosco; Unità Operativa di Gastroenterologia, Ospedale Belcolle, Viterbo: Eugenio Caturelli, Paola Roselli; Unità Operativa di Medicina Protetta, Ospedale Belcolle, Viterbo: Serena Dell'Isola, Anna Maria Ialungo; Dipartimento di Medicina Interna, Unità di Gastroenterologia, Università di Genova: Edoardo G. Giannini, Domenico Risso, Simona Marenco, Linda Bruzzone, Vincenzo Savarino, Antonino Picciotto; Unità di Gastroenterologia, Ospedale Sacro Cuore Don Calabria, Negrar: Maria Chiaramonte; Dipartimento Biomedico di Medicina Interna e Specialistica, Unità di Gastroenterologia, Università di Palermo: Giuseppe Cabibbo, Calogero Cammà, Marcello Maida, Arezia Di Martino, Maria Rosa Barcellona; Ospedale Regionale di Bolzano, Unità di Gastroenterologia, Bolzano: Andrea Mega; Unità di Medicina Interna e Gastroenterologia, Policlinico Gemelli, Università Cattolica di Roma, Roma: Antonio Gasbarrini, Emanuele Rinninella; Dipartimento di Gastroenterologia, Azienda Ospedaliero-Universitaria Pisana, Pisa: Virginia Rotella, Barbara Ginanni, Caterina Vivaldi; Dipartimento di Medicina Interna; Ospedale per gli Infermi di Faenza, Faenza: Francesco Giuseppe Foschi, Arianna Lanzi, Giuseppe Francesco Stefanini, Anna Chiara Dall'Aglio, Federica Mirici Cappa, Elga Neri, Paolo Bassi, Miriam Zanotti; Unità di Malattie Infettive ed Epatologia, Azienda Ospedaliero-Universitaria di Parma: Gabriele Missale, Elisabetta Biasini, Emanuela Porro; Dipartimento di Medicina Clinica e Chirurgia, Unità di Gastroenterologia, Università di Napoli “Federico II”, Napoli: Filomena Morisco, Maria Guarino; Clinica di Gastroenterologia, Università Politecnica delle Marche, Ancona: Gianluca Svegliati Baroni, Laura Schiadà, Stefano Gemini. Dipartimento di Medicina, Unità di Radiologia, Ospedale Fatebenefratelli, Milano: Francesco Borzio. Dipartimento Biomedico di Medicina Interna e Specialistica, Unità di Medicina Interna 2, Azienda Ospedaliera Ospedali Riuniti Villa Sofia-Cervello, Palermo: Roberto Virdone.
Acknowledgements
Scientific and statistical assistance was provided by Luca Giacomelli, PhD, Ambra Corti and Marco Barbara; this assistance was funded by Bayer Italy.
