Abstract
Introduction
Diagnosis of cholangiocarcinoma (CCA) can be challenging due to unclear imaging criteria and difficulty obtaining adequate tissue biopsy. Although serum cancer antigen 19-9 and carcinoembryonic antigen have been proposed as potential diagnostic aids, their use remains limited by insufficient sensitivity and specificity. This exploratory analysis aimed to identify individual- and combinations of serum biomarkers to distinguish CCA from hepatocellular carcinoma (HCC) and chronic liver disease (CLD) controls using samples from a published study.
Methods
This prospective, multicenter, case-control study included patients aged ≥18 years at high-risk of HCC. Serum and ethylene diamine tetraacetic acid-plasma samples were collected prior to any treatment and confirmed diagnosis of HCC or CCA. Fourteen biomarkers (measured by electrochemiluminescence immunoassays or enzyme-linked immunosorbent assays) were subjected to univariate analysis and 13 included in a multivariate analysis (per selected combinations and exhaustive search).
Results
Overall, 55 CCA, 306 HCC, and 733 CLD control samples were analyzed. For distinguishing CCA from HCC, alpha-fetoprotein and matrix metalloproteinase-2 (MMP-2) showed the best individual performance (area under the curve (AUC) 86.6% and 84.4%, respectively); tissue inhibitor of metalloproteinase-1 (TIMP-1) was most able to distinguish CCA from CLD (AUC 94.5%) and from HCC + CLD (AUC 88.6%). The combination of MMP-2 and TIMP-1 was the best-performing two-marker panel, with AUC >90% for all comparisons.
Conclusion
MMP-2 and TIMP-1 are promising biomarkers that could support differential diagnosis of CCA. Incorporating these assays into the diagnostic algorithm could provide additional diagnostic information in a non-invasive, rapid manner, and could supplement existing diagnostic methods.
Introduction
Cholangiocarcinoma (CCA) describes a diverse cluster of malignancies originating in the bile duct, classified based on location as intrahepatic (iCCA) or extrahepatic (eCCA), which can be further subcategorized as perihilar (pCCA) or distal (dCCA). Accounting for ∼15% of all primary liver tumors, CCA is the second most common liver cancer globally after hepatocellular carcinoma (HCC). 1 Mortality rates for CCA have been increasing worldwide and patients generally have a very poor prognosis, with 5-year survival rates of only 7–20%. 2 Increases in mortality in recent years have largely been driven by iCCA, with mortality from eCCA either plateauing or falling. 3
Given that CCA has long been associated with parasitic liver fluke infections, prevalence is highest in East Asia where these infections are endemic. 4 In the West, risk factors include primary sclerosing cholangitis (PSC), representing 10% of cases, 4 other liver conditions (e.g., bile duct disorders, viral hepatitis, and cirrhosis), and lifestyle factors (e.g., obesity, type II diabetes, alcohol consumption, and smoking).1,5,6 However, the majority of Western cases occur in the absence of any known risk factor. 6 CCA tumors are typically asymptomatic in the early stages and often only diagnosed when the disease becomes advanced, or are found incidentally.5,7,8 Screening patients for early-stage CCA would potentially improve prognosis but is challenging since most of these tumors occur sporadically. 7
Imaging modalities, including endoscopic ultrasound (endoscopic ultrasound and endoscopic retrograde cholangiopancreatography), multiparametric ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI), continue to play a key role in the diagnosis, differential diagnosis, and staging of CCA.9–11 However, obtaining tissue samples via biopsy remains mandatory to confirm the diagnosis. 7 Unfortunately, CCA tumors can be very difficult to access, particularly those in the perihilar region, and as a result cytology sensitivity may be as low as 20–40%. 12
Distinguishing between iCCA, HCC, and other liver conditions can often be challenging but is critical due to differences in their management and treatment. 12 Thus, identifying non-invasive biomarkers such as nucleic acids, proteins, or metabolites in serum, bile, or urine represents a key research focus to avoid reliance on tissue biopsy. 13
Measuring serum tumor markers is an attractive option in patients with suspected CCA due to the ease of sample collection and analysis. Carbohydrate (cancer) antigen 19-9 (CA19-9) and carcinoembryonic antigen (CEA) are both well-established tumor markers, and the National Comprehensive Cancer Network guidelines state that they should be considered if CCA is suspected. 14 However, they are non-diagnostic and have only limited utility; for example, in a European registry study, both markers had low diagnostic sensitivity, but levels did correlate with disease severity. 5 CA19-9 and CEA can be helpful in monitoring the progression of CCA, but there is still an unmet need for accurate and broadly accessible diagnostic, prognostic, and predictive biomarkers to optimize outcomes for patients with CCA.13,15,16
The objective of this exploratory analysis was to identify individual and combinations of serum tumor biomarkers that could distinguish CCA from HCC and benign chronic liver disease (CLD) controls using samples obtained from a previously published study in the early detection of HCC. 17 Thus, in addition to CA19-9 and CEA, a series of biomarkers were selected for evaluation based on their general function as established tumor markers; for example, cytokeratin fraction 21 (CYFRA 21-1), cancer antigen 15-3 (CA15-3), CA125, and ferritin were considered given that they are useful in a variety of cancer diagnoses, as well as the liver cancer-specific markers, alpha-fetoprotein (AFP) and protein induced by vitamin K absence-II (PIVKA-II).18–22 In addition, proteins known to be involved in the rearrangement of extracellular matrix and angiogenesis, (osteopontin (OPN), 23 angiopoietin 2 (ANG2), 24 interleukin-6 (IL-6), 25 matrix metalloprotease-2 (MMP-2), 26 and tissue inhibitor of metalloproteinase-1 (TIMP-1) 26 ), and those that interact with insulin growth factor signaling pathways (insulin-like growth factor-binding protein 3 (IGFBP-3) 27 and OPN 23 ) were also included in this analysis.
Materials and methods
Study cohort
This was a prospective, multicenter, case-control study that has been described previously. 17 In brief, eligible patients were at least 18 years of age and considered to be at high risk of HCC based on a diagnosis of either cirrhotic liver disease (independent of etiology), non-cirrhotic non-alcoholic steatohepatitis, chronic hepatitis B or C virus infection, or liver fibrosis (stage ≥ F3)/cirrhosis (controls). Patients diagnosed with HCC but untreated, or with lesions suspicious for cancer based on imaging, were also eligible (cases). Patients were ineligible if they were receiving cancer treatment, had a history of a non-liver malignancy, had received immunomodulatory treatment within the last 6 months, or did not have sonography information from within the previous 4 weeks.
Samples from patients with a confirmed diagnosis of CCA were obtained from this study, with additional samples provided by Biopartners GmbH (Woodland Hills, CA, USA). Samples from patients with CCA secondary to parasitic infection were excluded from this analysis. Samples from patients with primary biliary cirrhosis (PBC), primary hemochromatosis or PSC were included in the benign CLD control cohort.
Sample preparation, storage and biomarker evaluation
Serum and ethylene diamine tetraacetic acid-plasma samples were collected prior to any treatment. Samples were stored at −70°C until analysis and repeated freeze–thaw cycles were avoided. A diagnosis of HCC or CCA was confirmed using a combination of two radiological methods (ultrasound imaging, CT, or MRI) and/or biopsy and histopathological analysis.
A panel of 14 electrochemiluminescence immunoassays (ECLIA) or enzyme-linked immunosorbent assays (ELISA) for the following in vitro diagnostics biomarkers were used in this analysis (as outlined in the Introduction): CEA, cancer antigen 15-3 (CA15-3), CA125, CA19-9, cytokeratin fraction 21-1 (CYFRA21-1), ferritin, interleukin 6 (IL-6), protein induced by vitamin K absence-II (PIVKA-II), insulin-like growth factor binding protein 3 (IGFBP-3), osteopontin (OPN), angiopoietin-2 (ANG-2), tissue inhibitor of metalloproteinase-1 (TIMP-1), matrix metalloproteinase-2 (MMP-2), and AFP. Measurements were performed on a cobas® e 601 analyzer from Roche Diagnostic Solutions or using a microtiter plate (MTP) ELISA (Total MMP-2 Quantikine ELISA kit and Human TIMP-1 Quantikine ELISA kit (R&D Systems, Minneapolis, MN, USA)) according to the instructions of the manufacturers (Suppl. Table 1). All assays were validated internally for analytical precision, reproducibility, and interfering substances. Calibrators and at least two native quality controls were run in duplicates and monitored across whole studies. Clinical samples were also run in duplicates and measurements with CV >15% were repeated.
Data analyses
Univariate and multivariate analyses were used to determine the area under the curve (AUC) of each individual biomarker for distinguishing between the following: CCA vs. HCC (all, early and late stages); CCA vs. benign CLD controls; and CCA vs. a combined cohort of patients with HCC or benign CLD controls (HCC + benign CLD). Selected biomarkers were analyzed with multivariate models—the aim of which was to identify the best performing biomarkers that could separate CCA from HCC and benign CLD. Multivariate analyses were performed using an exhaustive search including all candidates with logistic regression (fixed panel size of two to four biomarkers). Logistic regression models based on all possible combinations of four biomarkers were evaluated and compared. All possible two to four biomarker combinations were analyzed among 13 biomarkers (CEA, CA15-3, CA125, CA19-9, CYFRA21-1, ferritin, IL-6, PIVKA-II, IGFBP-3, OPN, ANG-2, TIMP-1, and MMP-2).
Estimation of diagnostic accuracy was done by a nested cross-validation, with the data split randomly into training (3/4) and test (1/4) sets. The optimal model was selected using an inner cross-validation of the training set and tested on the remaining data set. This procedure was repeated 100 times. The test data in the outer loop was used to evaluate the clinical performance of the optimal model. The nested cross-validation procedure provided an estimate of the stability and a robust estimate of the performance (to avoid overfitting) for each model. The performance of AUC, sensitivity at 90% specificity, and specificity at 90% sensitivity were reported for each model for detecting CCA.
All biostatistical analyses were performed in-house using R Core Team (2020). 28
Results
Samples were obtained between 2014 and 2016 from 1094 patients at seven centers in four countries (China, Germany, Spain, and Thailand). Overall, biomarkers were analyzed in samples from 55 patients with CCA, 306 patients with HCC, and 733 patients with benign CLD. Of the 306 HCC patients 125 were defined as early (BCLC 0/A) and 169 as late (BCLC B–D) stages. Of the 55 patients with CCA, 3 had confirmed iCCA and 11 had pCCA; no data on biliary tree location were available for the remaining patients. Tumor, node, metastasis staging was available for 35 CCA patients (Tx = 5, T1 = 1, T2 = 6, T3 = 20, T4 = 3).
The demographics and clinical characteristics of patients included in this analysis are shown in Table 1. The mean age of all patients was 57.2 years, with patients with HCC or CCA being slightly older than those with benign CLD (60.8 years and 60.7 years vs. 55.4 years, respectively). Most patients with HCC or CCA were male (78.8% and 63.6%, respectively); however, for patients with benign CLD, male preponderance was lower (56.3%). Most patients with benign CLD or HCC were from Asia (87.6% and 82.0%, respectively) whereas almost all patients with CCA were from Europe (96.4%) and were described as Caucasian (96.4%). Among the benign CLD controls, six patients had PBC and three had PSC. In addition, two patients in the HCC cohort had primary hemochromatosis.
Patient demographics.
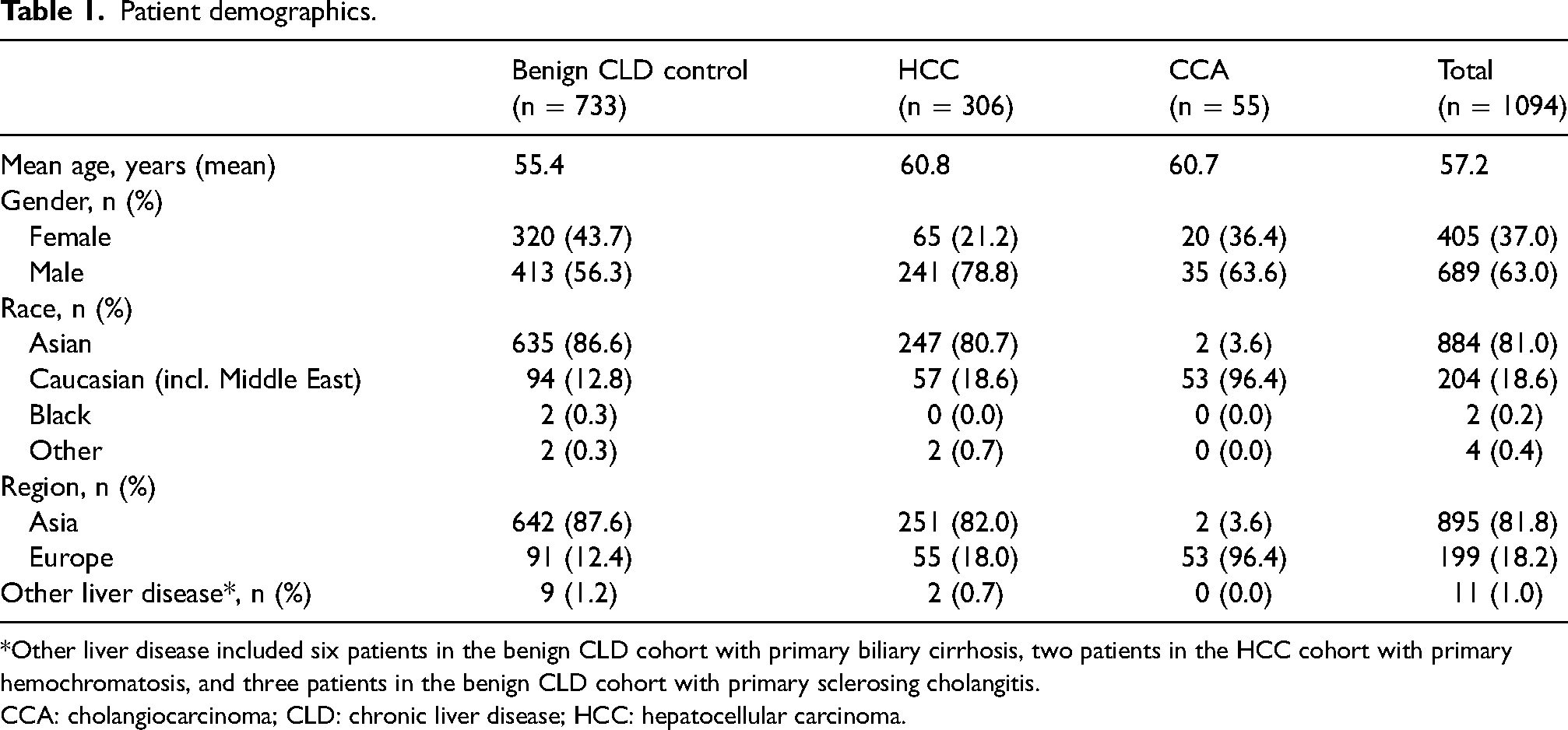
*Other liver disease included six patients in the benign CLD cohort with primary biliary cirrhosis, two patients in the HCC cohort with primary hemochromatosis, and three patients in the benign CLD cohort with primary sclerosing cholangitis.
CCA: cholangiocarcinoma; CLD: chronic liver disease; HCC: hepatocellular carcinoma.
Univariate analysis
For distinguishing CCA from HCC, AFP and MMP-2 were the most promising individual biomarkers with AUCs of 86.6% and 84.4%, respectively (Table 2, Figure 1(a) and (b)). Additionally, for the differential diagnosis CCA vs. HCC, we analyzed performance of the biomarkers for early (BCLC 0/A) and late (BCLC B-D) HCC stages separately. For distinguishing CCA from early HCC, TIMP-1 and MMP-2 were identified as the best performing biomarkers, with AUCs of 87.5% and 86.9%, respectively. As expected, AFP has superior performance for the differentiation of CCA from late-stage HCC, with an AUC of 90.5%, followed by MMP-2 with an AUC of 83.4% (Suppl. Table 2).

Boxplots and ROC curves showing univariate analysis of CCA vs. HCC, CCA vs. benign CLD controls, and CCA vs. HCC + benign CLD controls for (a) AFP; (b) MMP-2; and (c) TIMP-1.
Performance of individual biomarkers in the univariate analysis.
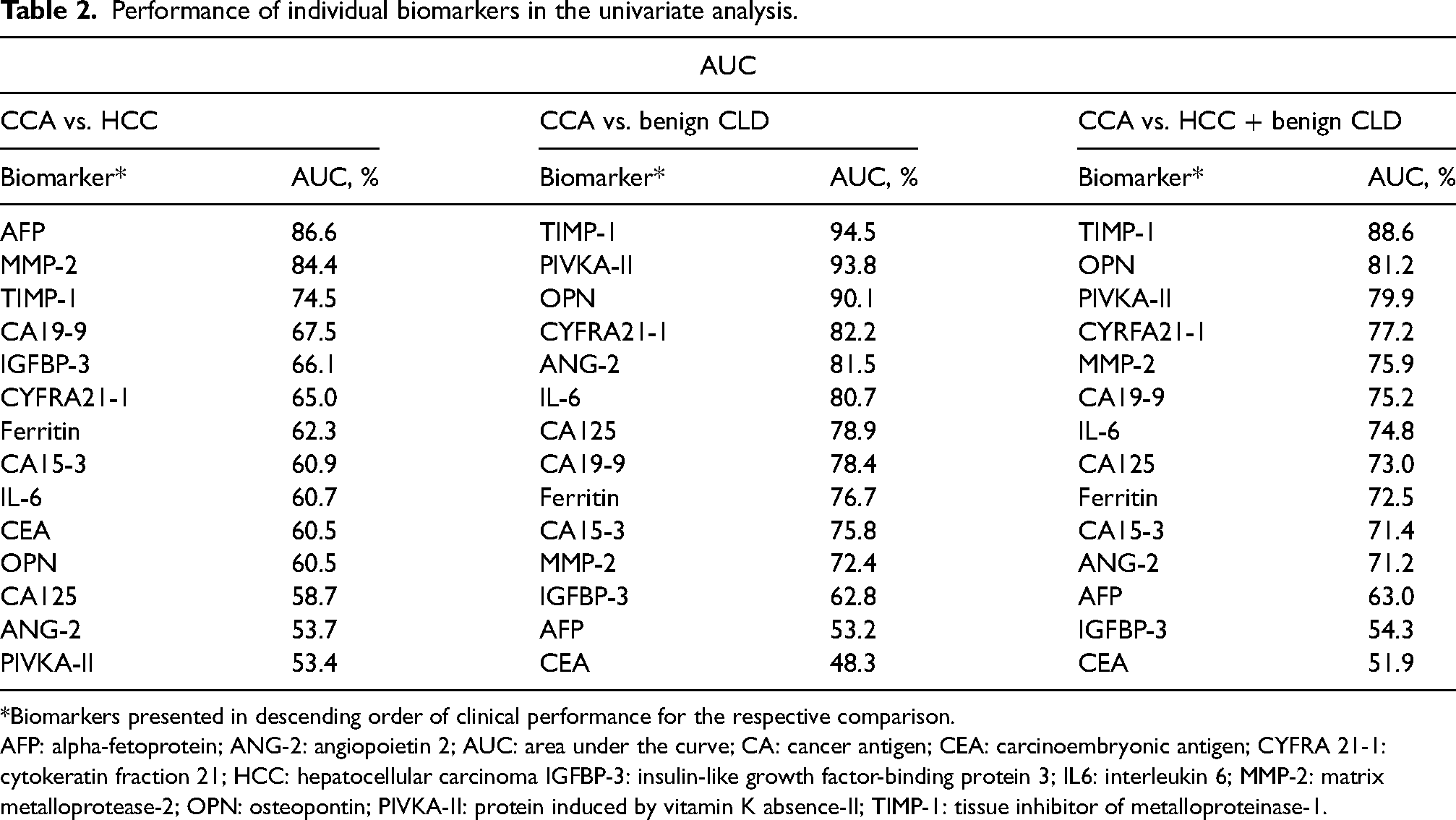
*Biomarkers presented in descending order of clinical performance for the respective comparison.
AFP: alpha-fetoprotein; ANG-2: angiopoietin 2; AUC: area under the curve; CA: cancer antigen; CEA: carcinoembryonic antigen; CYFRA 21-1: cytokeratin fraction 21; HCC: hepatocellular carcinoma IGFBP-3: insulin-like growth factor-binding protein 3; IL6: interleukin 6; MMP-2: matrix metalloprotease-2; OPN: osteopontin; PIVKA-II: protein induced by vitamin K absence-II; TIMP-1: tissue inhibitor of metalloproteinase-1.
For distinguishing CCA from benign CLD or from HCC + benign CLD, TIMP-1 demonstrated the best individual performance, with AUC values of 94.5% and 88.6%, respectively (Table 2; Figure 1(c)). CA19-9 and CEA performed relatively poorly as individual biomarkers, with AUCs ≤78.4% and ≤60.5%, respectively, across all analyses (Table 2; Suppl. Figure 1). Due to the limited information on the CCA location, we could not perform comprehensive data analyses for each type of CCA. However, for the samples with known CCA classification an even distribution of MMP-2 and TIMP-1 values, without a correlation to specific cancer type, was observed (Suppl. Figure 2).
Multivariate analysis—exhaustive search
In a multivariate analysis using an exhaustive search of all combinations of two to four biomarkers, the combination of MMP-2 and TIMP-1 was the best performing two-biomarker panel for distinguishing CCA from HCC (AUC 91.8%, Figure 2(a)). The combination of MMP-2 and TIMP-1 was able to distinguish CCA from both early-stage HCC (AUC 98.3%) and late-stage HCC (AUC 87.8%). Although higher specificity was achieved with a four-biomarker panel (PIVKA-II, IGFBP-3, TIMP-1, MMP-2, (AUC 94.8%)), the sensitivity was lower and the two-biomarker panel was chosen (Table 3(a); Suppl. Figure 3(a)).

Boxplots and ROC curves showing multivariate analysis of TIMP-1 and MMP-2 for (a) CCA vs. HCC; (b) CCA vs. benign CLD controls; and (c) CCA vs. HCC + benign CLD controls.
Optimal marker panels and their classification performance for (a) CCA vs. HCC, (b) CCA vs. benign CLD controls, and (c) CCA vs. HCC + benign CLD controls.
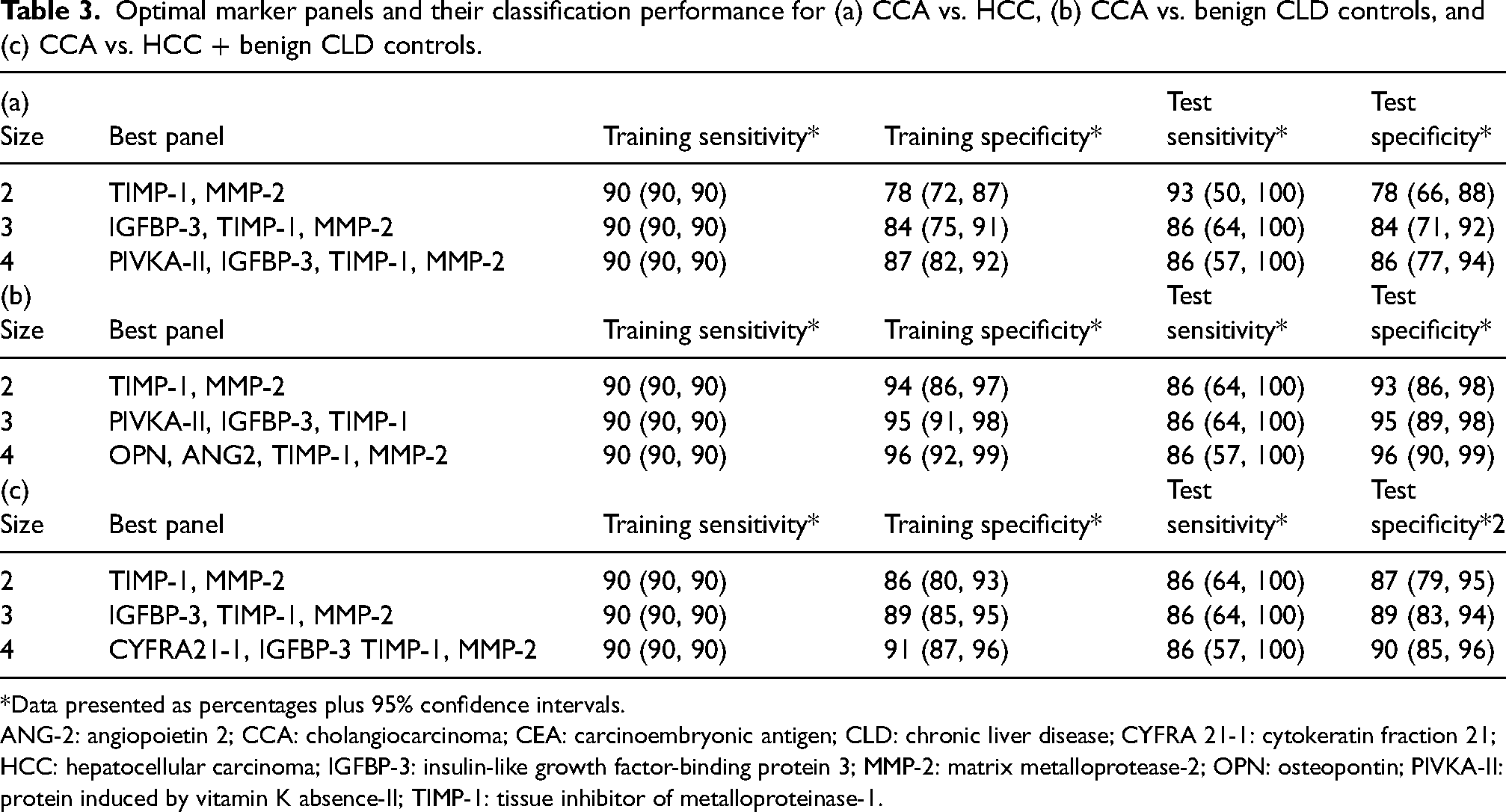
*Data presented as percentages plus 95% confidence intervals.
ANG-2: angiopoietin 2; CCA: cholangiocarcinoma; CEA: carcinoembryonic antigen; CLD: chronic liver disease; CYFRA 21-1: cytokeratin fraction 21; HCC: hepatocellular carcinoma; IGFBP-3: insulin-like growth factor-binding protein 3; MMP-2: matrix metalloprotease-2; OPN: osteopontin; PIVKA-II: protein induced by vitamin K absence-II; TIMP-1: tissue inhibitor of metalloproteinase-1.
For distinguishing between CCA and benign CLD, increases in specificity were observed with increasing panel size until the maximum size of four biomarkers was reached (Table 3(b), Suppl. Figure 3(b)). MMP-2 and TIMP-1 was the best performing two-biomarker panel with an AUC of 97.9% for distinguishing CCA from benign CLD (Figure 2(b)). The best performing four-biomarker panel (OPN, ANG2, TIMP-1, and MMP-2) had an AUC of 98.1% with no loss of sensitivity compared with the two-biomarker panel; therefore, this four-biomarker panel was optimal in this context (Table 3(b)).
For distinguishing between CCA and HCC + benign CLD, MMP-2 combined with TIMP-1 was the best performing two-biomarker panel (AUC of 95.6%) (Figure 2(c)). A four-biomarker panel (CYFRA21-1, IGFBP-3, TIMP-1, and MMP-2) had a slightly higher AUC compared to the two-biomarker panel (Table 3(c)).
Analysis based on samples from Europe only
Due to the high variation in the distribution of ethnicities between the subgroups, a sensitivity analysis was performed using samples from Europe only (Suppl. Figure 4). Both univariate and multivariate results for TIMP-1 and MMP-2 were similar to the overall analysis set. MMP-2 and TIMP-1 as a two-biomarker panel had AUCs of 95.1%, 87.3%, and 92.8% for CCA vs. benign CLD, CCA vs. HCC, and CCA vs. HCC + benign CLD, respectively.
Discussion
Obtaining an early and accurate diagnosis of CCA remains a major challenge given the issues associated with biopsy access due to the anatomic location of these tumors and the relatively poor diagnostic performance of existing biomarkers. 29
For our studies, we selected a group of serum biomarker candidates for diagnosis and differential diagnosis of CCA. The selection was based on guideline recommendations for a given indication (e.g., CA 19-9 and CEA for CCA, and AFP and PIVKA-II for HCC), or on the involvement of the selected protein in the processes of carcinogenesis, immune response and cell migration, angiogenesis, or extracellular matrix remodeling.
In this analysis, we identified AFP and MMP-2 as the best individual biomarkers for distinguishing CCA from HCC, with lower levels indicative of CCA. Sustained elevation of AFP in serum is a known risk factor for HCC, and in patients with CLD undergoing regular screening for HCC, AFP levels are often evaluated alongside liver ultrasounds.14,30 However, AFP is not considered to be sensitive or specific enough to act as a diagnostic biomarker for HCC in isolation: up to 40% of patients diagnosed with HCC have normal AFP levels 31 and elevated AFP levels can also be caused by other malignancies, cirrhosis, hepatitis, and pregnancy.32,33 Although the univariate analysis data here are in agreement with reports in the wider literature,34,35 AFP alone is unlikely to support definitive differential diagnosis of CCA from HCC. Characteristically, in our study, AFP performance was significantly lower in early cases of HCC than for late stages HCC.
For separating CCA and benign CLD, and for separating CCA and the combined HCC + benign CLD cohort, TIMP-1 was the best individual biomarker, with higher levels indicative of CCA. MMP-2, AFP, and TIMP-1 considerably outperformed CA19-9 and CEA in distinguishing CCA from HCC + benign CLD.
Through multivariate analyses, the highest AUCs were achieved with a four-biomarker panel for all analyses; however, compared with a two-biomarker panel, some sensitivity was lost when distinguishing between CCA and HCC or between CCA and HCC + benign CLD. The optimal panel size was therefore considered to be two biomarkers when distinguishing CCA from HCC or CCA from HCC + benign CLD and four biomarkers when distinguishing CCA from benign CLD. For all comparisons, the optimal two-biomarker panel was MMP-2 and TIMP-1, with AUCs ≥90% for all analyses. In an analysis that included all three cohorts, good separation was achieved between CCA and HCC (AUC 91.8%), but even better separation was achieved between CCA and benign CLD (AUC 97.9%).
There were considerable geographic differences between the CCA, HCC, and benign CLD cohorts, with nearly all CCA samples obtained from Europe and a high proportion of HCC and CLD samples obtained from Asia. To explore the potential impact of these differences, a sensitivity analysis was performed using only European samples. The performance results for this sensitivity cohort were similar to the overall analysis set indicating that geographical location had limited impact on the results. While it is known that age and sex can improve the diagnostic performance of biomarker panels for differentiating HCC from controls, as shown in the larger cohort of this trial, 17 we found that these factors did not contribute to distinguishing between HCC and CCA (data not shown) and were therefore not included in the biomarker models.
There are ongoing attempts to identify and validate protein biomarkers for the diagnosis, prediction, and prognosis of CCA; however, such studies are inconclusive and results vary depending on the chosen sample type and methodology. Ince et al. 36 demonstrated that serum MMP-9 and biliary TIMP-1 (based on AUC) were predictive of CCA, but concluded that these biomarkers were ineffective in the differential diagnosis of CCA and benign conditions due to high sensitivity, low specificity, and medium accuracy. In contrast, our study demonstrates high sensitivity and specificity for TIMP-1 both individually and in combination with MMP-2. Recent high-throughput proteomics discovery approaches by Duangkumpha et al. 37 and Lapitz et al. 38 revealed combinations of novel biomarker candidates for the differential diagnosis of CCA from HCC and PSC. However, there is little overlap in the proposed biomarker combinations; potentially as a result of the pre-analytic preparation of samples (whole serum vs. depleted serum vs. serum extracellular vesicles). Due to the lack of consistency observed, further studies are needed to validate biomarker models for the diagnosis of CCA.
The primary function of TIMPs is to inhibit extracellular matrix degradation mediated by MMPs, which may explain these observations. While this would suggest high levels are protective against tumor growth and metastasis, TIMP-1 likely interacts with other proteins and regulates biological processes such as cell growth, apoptosis, and differentiation, independently of its metalloproteinase inhibitory activity. 26 Such a mode of action supports the contribution of TIMP-1 to tumor invasion and metastasis reported for gastrointestinal cancers, with increased expression levels associated with poor survival outcomes (i.e., overall survival and progression-free survival) in gastric and colon cancers. 39 Although TIMP-1 and MMP-2 are both involved in extracellular matrix turnover and remodeling, and TIMP-1 can directly bind and inhibit activity of MMP-2, 26 both biomarkers differ in specificity and sensitivity towards CCA diagnosis. This effect can be caused by the complex interaction network between different MMPs, TIMPs, and further molecules functioning within the tumor microenvironment. In this context, MMP-2 can cross-activate MMP-1, MMP-7, MMP-12 to 17, MMP-24, and MMP-25. 40 TIMP-1 inhibits all types of MMPs, binding particularly strongly to MMP-9 and pro-MMP-9, 36 with the exceptions of MMP-14, MMP-16, MMP-18, and MMP-19. 41 Independently of TIMP-1 and MMP-2, MMP-14 was recently identified as the characteristic gene of intrahepatic CCA development and immune cell infiltration, 42 with MMP-7 and MMP-9 involved in perineural invasion, 43 further confirming the role of metalloproteases in the regulation of the tumor microenvironment. Therefore, it is not surprising that deregulation of one of those molecules can lead to the multiple effects finally reflected by the diagnostic performance of the marker. In addition, Terada et al. 44 previously demonstrated that MMP and TIMP may be neo or overexpressed by intrahepatic bile duct cells after malignant transformation but are not expressed by hepatocytes. Results presented by Nakatsukasa et al. 45 also demonstrated stronger expression of TIMP-1 in CCA compared with HCC tissue, and Chae et al. 46 reported increased activity and expression of MMP-2 in CCA vs. normal livers. Based on these findings and the results of our study, we hypothesize that the increase of TIMP-1 expression and its direct binding and regulation of MMP-2 leads to the extracellular matrix remodeling specific for CCA development, invasion, and metastasis. According to The Human Protein Atlas and earlier reports, expression of MMP-2 is specific for cholangiocytes, but not for hepatocytes, and thus can act as differentiator between HCC and CCA pathobiology.47,48 However, there is a clear need for a greater understanding of the role of TIMPs and MMPs in extracellular remodeling in the context of CCA pathophysiology.
This study has several limitations. First, as with all case-control studies, this study is subject to inherent selection bias. Missing follow-up data for patients with CLD also prevent the analysis of TIMP-1 and MMP-2 as prognostic biomarkers; longitudinal studies could be used to assess this in future research. Second, the relatively small sample size limited our ability to evaluate the performance of these biomarker panels in patients with intra vs. extrahepatic CCA tumors, and due to the limited information on the HCC and CCA tumor sizes, the correlation between biomarker values and tumor size could not be analyzed. Third, even though the highest incidence of CCA typically occurs in Asian countries, CCA samples were obtained almost exclusively from Western countries, which may warrant additional studies to enable comparisons between Western and Asian populations. This may at least partially reflect the exclusion of samples from patients with confirmed parasitic infections. However, none of the criteria used to diagnose and monitor both HCC and CCA (i.e., characteristics on multi-phase imaging, AFP, CA19-9, and tissue biopsy) vary in international guidelines based on ethnicity or underlying etiology of patients. 14 Independent validation of these results is now recommended in clinically relevant populations and prospective studies. Despite these limitations, the study provides clear evidence that serum biomarker panels demonstrate good diagnostic performance with the potential to improve the diagnostic process for CCA, offering lower costs and considerably shorter turnaround times than liquid biopsy-based assays. This approach may be particularly useful in regions where imaging or tissue-based methodologies are limited by availability and cost.
These findings suggest that MMP-2 and TIMP-1 are promising biomarkers to support in the differential diagnosis of CCA. Incorporating these serum-based assays into the diagnostic algorithm could provide additional diagnostic information in a non-invasive, rapid manner, and could supplement existing diagnostic methods. To assess this approach in a real-world setting and to further confirm the observed results, large longitudinal prospective studies—including an increased number of specificity controls for CCA (e.g., samples from patients with PSC) and broad patient populations—are warranted.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155241235185 - Supplemental material for A new biomarker panel for differential diagnosis of cholangiocarcinoma: Results from an exploratory analysis
Supplemental material, sj-docx-1-jbm-10.1177_03936155241235185 for A new biomarker panel for differential diagnosis of cholangiocarcinoma: Results from an exploratory analysis by Bruno Köhler, Marta Bes, Henry Lik-Yuen Chan, Juan Ignacio Esteban, Teerha Piratvisuth, Wattana Sukeepaisarnjaroen, Tawesak Tanwandee, Satawat Thongsawat, Anika Mang, David Morgenstern, Magdalena Swiatek-de Lange and Farshid Dayyani in The International Journal of Biological Markers
Footnotes
Acknowledgments
COBAS is a trademark of Roche. All other product names and trademarks are the property of their respective owners. Editorial support was provided by Steph Carter and Jade Drummond of inScience Communications, Springer Healthcare Ltd, UK, and was funded by Roche Diagnostics International Ltd (Rotkreuz, Switzerland).
Author contributions
Study concept/design: FD, DM, MSdL; data acquisition: MSdL; data analysis and interpretation: AM; review and final approval of manuscript: FD, DM. MSdL, ST, WS, TP, JIE, HL-YC, MB, BK.
Data availability statement
The study was conducted in accordance with applicable regulations. Requests concerning the data supporting the findings of this study can be directed to rotkreuz.datasharingrequests@roche.com for consideration.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BK reports a non-related research grant with AbbVie, support for a clinical trial from Gilead Sciences, speaker's bureau participation for Servier, Takeda, and Pierre Fabre. HL-YC reports consultancy fees from Aligos Therapeutics Arbutus Biopharma, Gilead Sciences, GlaxoSmithKline, Janssen Pharmaceutica, Hepion, Roche, Vir Biotechnology, Vaccitech, and Viron Therapeutics and speaker's bureau participation for Gilead Sciences, Mylan, and Roche Diagnostics. TP reports speaker's bureau participation for Bristol-Myers Squibb, Gilead Science, Bayer, Abbott, and Eisai, plus MSD and research grant/contracts from Gilead Science, Roche Diagnostics, Janssen, FibroGen, and Vir Biotechnology. AM, and MSdL are employees of Roche Diagnostics and hold stock/stock options in F. Hoffman-La Roche Ltd. DM was an employee of Roche Diagnostics and held stock/stock options in F. Hoffman-La Roche Ltd at the time of analysis. MSdL has a patent for a new binding agent and assay for PIVKA-II pending, and a pending patent application on the TIMP-1/MMP-2 panel. FD is a consultant for Genentech/Roche, Array BioPharma, Exelixis, Eisai, QED Therapeutics, and Signatera. JIE, MB, ST, TT, and WS have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This analysis was funded by Roche Diagnostics GmbH (Germany). Editorial support was provided by Steph Carter and Jade Drummond of inScience Communications, Springer Healthcare Ltd, UK, and was funded by Roche Diagnostics International Ltd (Rotkreuz, Switzerland).
Statement of ethics
This study was conducted in accordance with applicable regulations. All patients in the study were pseudonymized (samples from the prospective case control study
17
) or anonymized (samples provided by Biopartners GmbH (Woodland Hills, CA, USA). Ethical approval was obtained for samples from the prospective case control study
17
; for additional samples provided by Biopartners GmbH, samples were completely anonymized and, thus, ethical approval was not necessary. As the study data spanned multiple sites globally, we cannot refer data collection to a single ethics committee. Qualified researchers may request access from the following ethical committees:
Comité Ético de Investigación Clínica del Hospital Universitari Vall d’Hebron (Barcelona, Spain); protocol NT-MIP-HCC-01; email: ceic@vhir.org Siriraj Institutional Review Board (Bangkok, Thailand); protocol NT-MIP-HCC-01 Research Ethics Committee 2, Faculty of Medicine, Chiang Mai University (Chiang Mai, Thailand); protocol NT-MIP-HCC-01 Joint Chinese University of Hong Kong New Territories East Cluster Clinical Research Ethics Committee (Hong Kong, China); protocol NT-MIP-HCC-01; email: crec@cuhk.edu.hk Ethik-Kommission Medizinische Fakultsät Heidelberg (Heidelberg, Germany); protocol NT-MIP-HCC-01; email: ethikkommission-I@med.uni-heidelberg.de Research Ethics Committee, Faculty of Medicine, Prince Songkla University (Hat Yai, Thailand); protocol NT-MIP-HCC-01 Office of the Khon Kaen University Ethics Committee in Human Research (Khon Kaen, Thailand) protocol NT-MIP-HCC-01
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
