Abstract
Background
Several studies show that the long non-coding RNA HOX transcript antisense RNA (HOTAIR) was upregulated in human cancer, which was associated with several clinical features and may have the potential to be prognostic markers. However, the significance of HOTAIR in hepatocellular carcinoma remains unclear. We performed a meta-analysis and bioanalysis to further investigate the association between HOTAIR and hepatocellular carcinoma.
Methods
Eligible literature was systematically retrieved from PubMed, Embase, and Web of Science databases. The pooled hazard ratios with 95% confidence intervals were used to evaluate to the effect. Raw data on HOTAIR expression were obtained from The Cancer Genome Atlas data portals. All bioinformatics analyses were performed using R software (version 4.3.1).
Results
We identified eight studies in this meta-analysis with a total of 399 patients. High-level HOTAIR expression was found to be significantly related to advanced tumor node metastasis stage, distant metastasis, poor tumor differentiation, and patients with hepatitis. Correspondingly, HOTAIR was also associated with poor overall survival and relapse-free survival. Subsequently, in bioanalysis, HOTAIR expression was higher in hepatocellular carcinoma as well as poor overall survival. High HOTAIR expression was strongly correlated with tumor node metastasis stage. Kyoto Encyclopedia of Genes and Genomes analysis revealed that the differentially expressed genes related to HOTAIR may be involved in the cancer-associated signaling pathway.
Conclusion
HOTAIR may be a potential biomarker for HCC prediction and is expected to become a new choice for clinical HCC prediction..
Introduction
Liver cancer, as a common malignancy, is the second cause of cancer-related death worldwide, 1 which poses a serious threat to human health. Depending on the type of pathology, hepatocellular carcinoma (HCC), which is aggressive in nature, accounts for 90% of primary liver cancers. 2 There are approximately 626,000 new cases diagnosed per year. Even more concerning is that 70%–80% of patients are at an advanced-stage at diagnosis.3,4 Hepatitis, alcohol, aflatoxin, and aristolochic acid are the most important causes of HCC. 5 Although the effective techniques for HCC diagnosis and treatment were application, the prognosis of HCC patients still remains grim. 6 Therefore, urgent identification of more sensitive and specific biomarkers for early detection of HCC is essential.
In recent decades, long non-coding RNAs (lncRNAs) have become an important regulatory factor in biology fields. 7 The lncRNA is a non-protein coding RNA with 200 nucleotides in length. 8 Numerous studies have demonstrated that lncRNA plays a significant role in biological processes including cell differentiation, development, gene expression, and cell cycle control. 9 Emerging evidence has suggested that the abnormal versions of cancer-related lncRNAs including HOX transcript antisense RNA (HOTAIR), HOTTIP, GAS5, BANCR, and SNHG3 are associated with prognosis in HCC.10–14 Among them, HOTAIR is one of the most well-studied lncRNAs. HOTAIR was initially discovered through microarray analysis in 2007 by Rinn et al. 15 HOTAIR is a 2.2 kb long and consists of 6 exons lncRNA, which is derived from an antisense strand of HOXC gene cluster on chromosome 12q 13.13. Numerous studies have suggested that HOTAIR expression may play a negative prognostic role in human cancers, including breast cancer, cervical cancer, colorectal cancer, endometrial cancer.16–19 Pioneering studies have identified that HOTAIR is notably upregulated in HCC tissue and may be applied as a biomarker for HCC screening and prognostic prediction. 20 So far, due to discrete outcome and little sample size, none of these studies have completely interpreted the role of HOTAIR expression in HCC. Thus, we conducted this meta-analysis to validate the expression of HOTAIR and its relationship with HCC. Most importantly, we further verified the results of the meta-analysis through bioinformatics analysis.
Materials and methods
Meta-analysis
Data sources and search strategy
To obtain articles relevant to this meta-analysis, we searched through electronic databases including PubMed, Embase, Web of Science, and Clinical Key. The terms for the search were: “HOTAIR,” “HOX transcription antisense RNA,” “liver cancer,” “hepatocellular carcinoma,” and any combination of these search terms with the deadline of March 30, 2023.
Inclusion and exclusion criteria
Inclusion criteria were as follows: (a) Any patient diagnosed with HCC by cytology or pathology, regardless of TNM stage; (b) the association of HOTAIR expression levels with clinicopathological features or survival outcomes of liver cancer were evaluated; (c) the studies were restricted to human experiments, and the type of sample had to be tissues; (d) sufficient data were provided to estimate the hazard ratio (HR) with their 95% confidence intervals (CIs) of the relationship between HOTAIR and clinicopathological parameters. Studies were excluded for the following reasons: (a) reviews, expert opinions, case reports, editorials, and commentaries; (b) the raw data presented were inadequate or were animal model studies.
Date extraction and quality assessment
The systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Evaluation and Meta-Analysis (PRISMA) guidelines. To ensure the reliability of the extracted data, initial data were screened and extracted independently by two authors through cross-checking. When disagreements arose, we resolved them by group discussion. The collection of each study included the following information: first author, year, country, type of cancer, tumor node metastasis (TNM) stage, sample, case number, follow-up (month), detection method, and outcomes. We also collected clinicopathological features data including age, gender, TNM stage, tumor number, tumor size, lymph node metastasis, distant metastasis, tumor differentiation, cirrhosis, hepatitis, alpha-fetoprotein (AFP) level and portal vein tumor thrombosis (PVTT). When survival data were not reported in some studies, Engauge Digitizer v10.8 software was used to extract data and calculate corresponding survival data, which was required according to the relevant method. 21
We assessed the quality of each included study by the Newcastle–Ottawa (NOS). 22 Studies with NOS scores ranging from 0 to 9 and NOS scores ≥6 were considered to be of high quality.
Statistical methods
This meta-analysis was conducted by software Stata 14.0 and a P value <0.05 was considered statistically significant. The I² statistic was used to estimate heterogeneity among the included studies. Also, an I² value > 50% was considered as existing heterogeneity between studies; in that case, a random-effects model was applied. In contrast, the fixed-effects model was used. 23 The effects magnitude used odds ratio (OR) as effect indicators, and the OR was combined with individual HRs and their related 95% Cis.
Bioinformatics analysis
Gene expression analysis of HOTAIR
The TIMER database (http://timer.comp-genomics.org/timer/, accessed on August 8, 2023) is an analysis network, which provides the HOTAIR expression in various tumor tissues through the “Gene_DE” module. 24 The HOTAIR expression data were downloaded from The Cancer Genome Atlas (TCGA) database (https://www.cancer.gov/about-nci/organization/ccg/research/structural-genomics/tcga). The 292 samples of liver HCC (LIHC) were divided into high-HOTAIR expression and low-HOTAIR expression groups based on the median expression value. Differentially expressed genes (DEGs) between the two groups were analyzed using the “limma” (V3.56.2) R package. with log-fold change absolute P value >1.5 and P value < 0.05 as the threshold parameters. The volcano plots of the DEGs were visualized using the “ggplot2” (V3.4.3) R package.
The prognostic analysis and clinicopathologic features of HOTAIR in LIHC
The clinical information of 292 cases were downloaded from the TCGA database. The patients were separated into high- and low-HOTAIR groups according to the median-HOTAIR expression value with 50% cut-off high and 50% cut-off low. Kaplan–Meier survival analysis was conducted to explore the correlation between HOTAIR and survival prognosis using the “survminer” and “survival” packages. The HOTAIR gene expression levels in LIHC patients with different clinicopathological features are shown by box plots, which were analyzed by the “ggpubr” package. The value of P < 0.05 was considered to be a statistically significant difference (postscript: P ≥ 0.05; *P < 0.05; **P < 0.01; ***P < 0.001).
The KEGG analysis
We performed KEGG pathway enrichment analysis of HOTAIR genes by using the cluster profile (V4.8.3) R package and visualized the results.
Results
Meta-analysis
Eligible studies and characteristics
As shown in Figure, a total of 221 published studies were revealed from four databases with relevant terms. After a layer-by-layer screened according to the inclusion and exclusion criteria. As a result, 8 published studies were included into our meta-analysis.20,25–31 The basic characteristics of included studies are shown in Table 1. In summary, a total of 399 subjects from 8 studies were enrolled. The level of HOTAIR expression was measured with quantitative real-time polymerase chain reaction in all of the included studies. In all 8 studies, patients were divided into high- and low-HOTAIR expression groups.
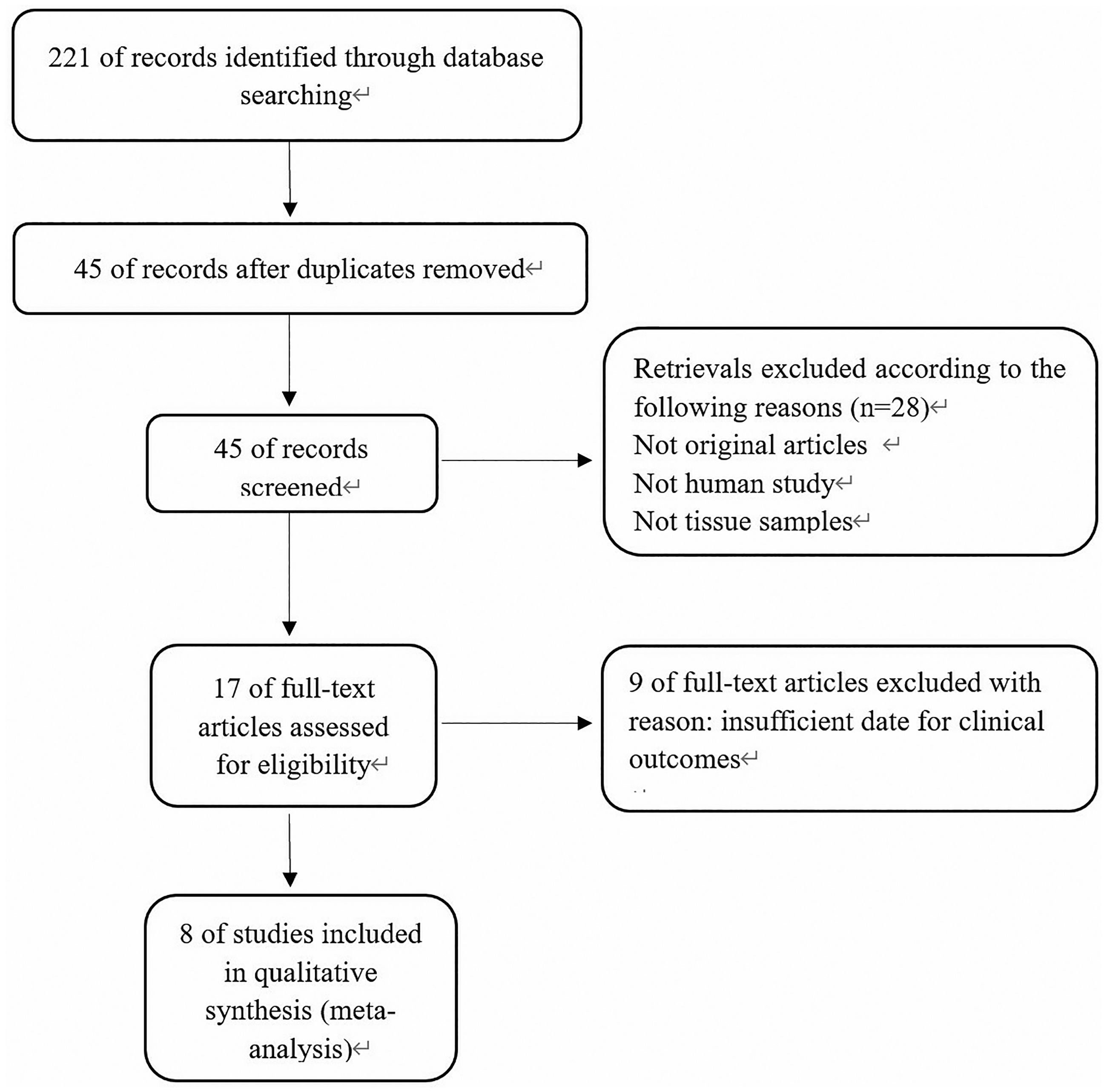
Flow diagram of the selection process.
Baseline characteristics of the included studies.

A: age; AFP: alpha-fetoprotein; CCA: cholangiocarcinoma; D: tumor differentiation; G: gender; H: hepatitis; HCC: hepatocellular carcinoma; L: lymph node metastasis; M: distant metastasis; N: tumor number; NA: not applicable; NM: not mention; NOS: Newcastle–Ottawa score; OS: overall survival; P: PVTT: portal vein tumor thrombosis; qRT-PCR: quantitative reverse transcription polymerase chain reaction; Ref. reference; RFS: recurrence-free survival; S: clinical stage; T: tumor size.
The association between increased HOTAIR expression and clinicopathological parameters
There were 8 studies assessing the association between HOTAIR and clinicopathological characteristics of liver cancer (Table 2). Through statistical analysis, we found that upregulated HOTAIR was closely related to advanced TNM stage (OR) 1.45; 95% CI 1.02, 2.05; P = 0.038), distant metastasis (OR = 2.51; 95% CI 1.02, 6.14; P = 0.044), poor tumor differentiation (OR = 1.59; 95% CI 1.21, 2.10; P = 0.001) and with hepatitis (OR = 1.42; 95% CI 1.21, 11.79; P = 0.03). However, there was no noteworthy correlation found between HOTAIR expression level and age, gender, tumor number, tumor size, lymph node metastasis, AFP level, and PVTT.
Risk estimates of the association between increased HOTAIR expression and clinicopathologic parameters.
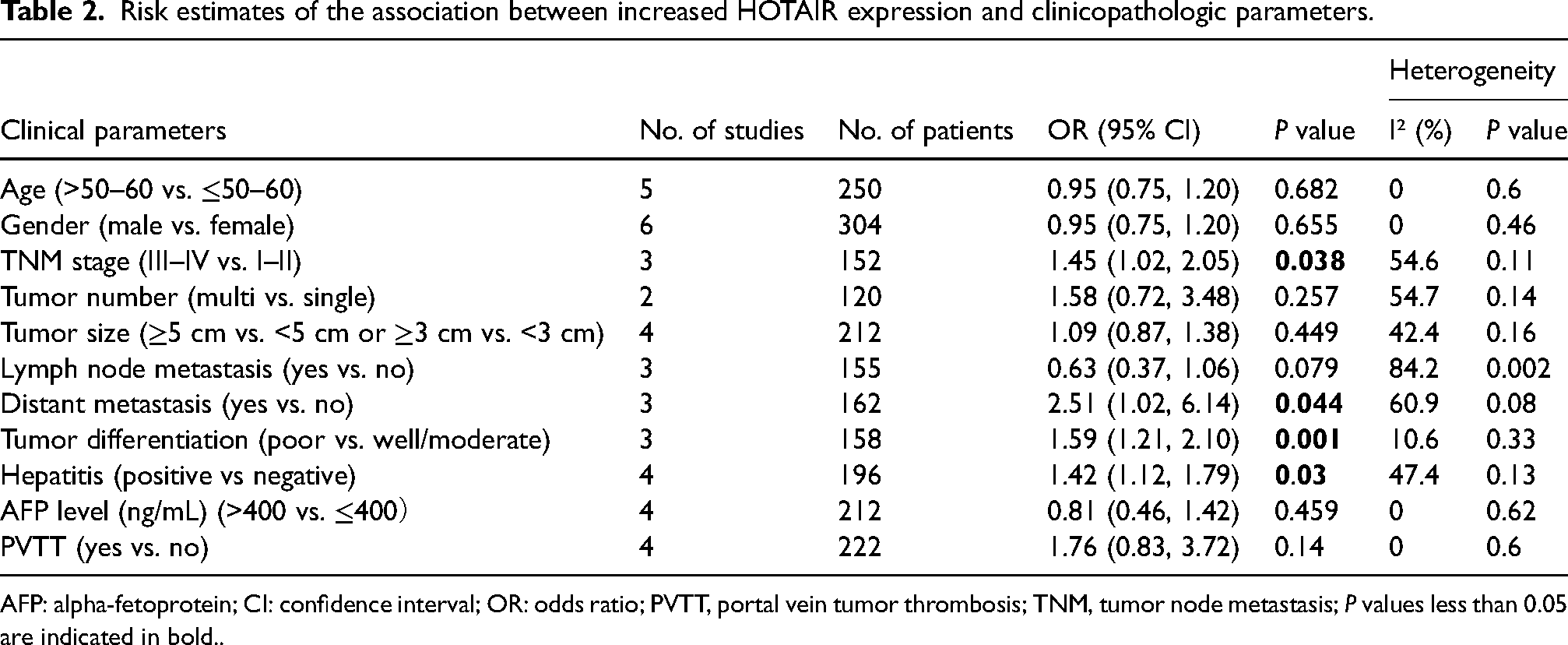
AFP: alpha-fetoprotein; CI: confidence interval; OR: odds ratio; PVTT, portal vein tumor thrombosis; TNM, tumor node metastasis; P values less than 0.05 are indicated in bold..
Correlation between HOTAIR and HCC survival
Among the 8 eligible articles, the correlation between overall survival (OS) and HOTAIR expression was reported in 3 articles. None of the patients underwent radiotherapy and chemotherapy treatment before operative treatment. The results show that high-level HOTAIR was strongly related to poor OS of HCC with a pool of HR = 1.69; 95% CI 1.14, 2.51; P = 0.009. The heterogeneity test showed homogeneity of I² = 50.1%; P = 0.135 (Supplementary Figure 1).
In addition, we also detect that there was a significant association between HOTAIR expression and recurrence-free survival (RFS). The RFS were extracted from 4 studies. Negative correlation was observed between high level HOTAIR and RFS (HR = 1.89; 95% CI 1.37, 2.59; P = 0.000) with low heterogeneity (I² = 31.5%; P = 0.223) (Supplementary Figure 2).
Bioinformatics analysis
HOTAIR gene expression analysis
To better understand the expression of HOTAIR in various cancer types, we employed the TIMER 2.0 website to explore the expression level of HOTAIR between tumor tissues and corresponding normal samples (Figure 2). The results show that HOTAIR expression was higher in LIHC (P < 0.01). Furthermore, HOTAIR expression was significantly higher in the liver cancer tissues compared to the adjacent peritumoral liver tissues in the TCGA datasets (P < 0.01, Supplementary Figure 3).
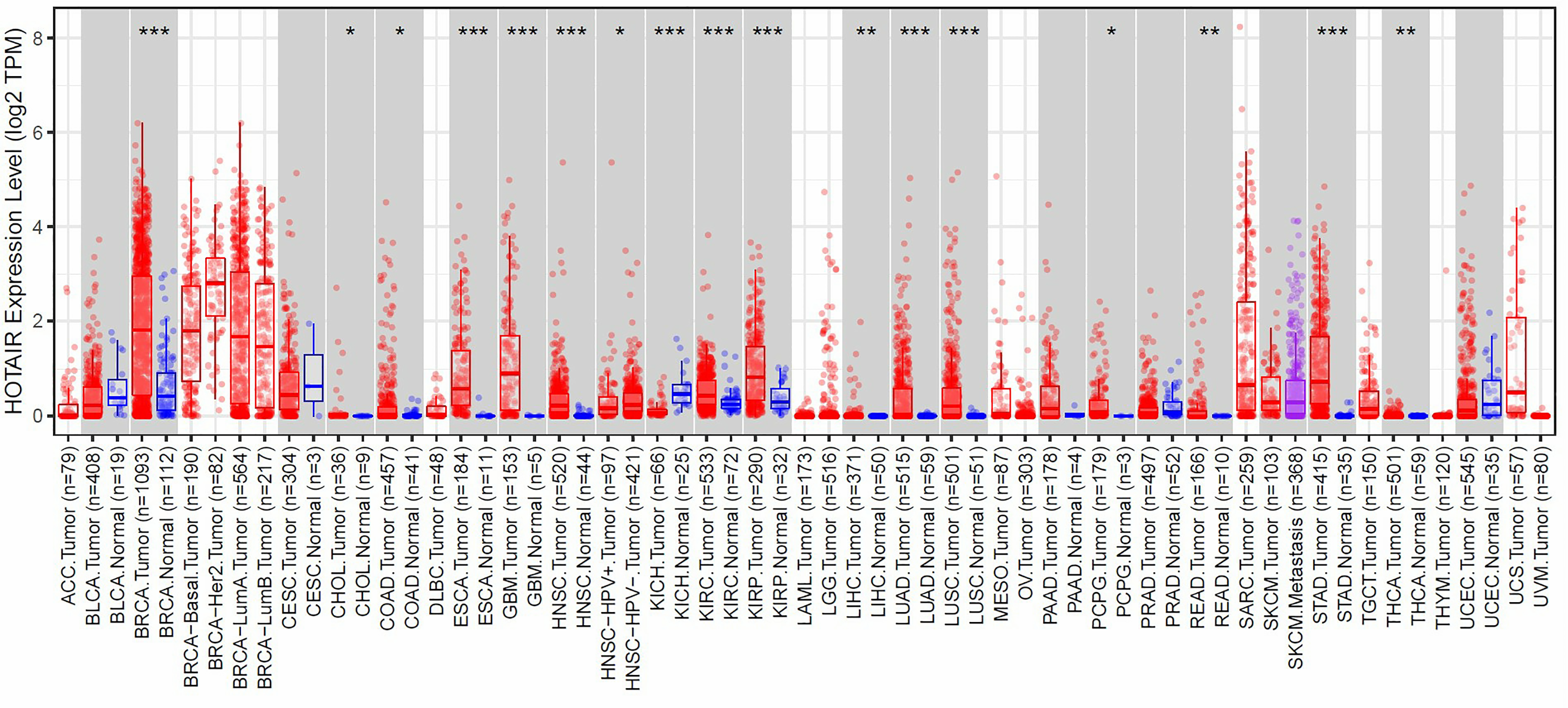
HOTAIR expression in pan-cancer from TCGA database were analyzed by TIMER2.0. (*P < 0.05, **P < 0.01, ***P < 0.001).
Prognostic value of HOTAIR in liver cancer
To further elucidate the impact of HOTAIR expression on the prognosis of LIHC, we downloaded TCGA RNA-seq and clinical data. While Kaplan–Meier survival curves were applied to assess the relation between HOTAIR expression and OS, the result was consistent with previous studies (P = 0.025, Supplementary Figure 4).
To verify the differences of OS between cases with high- and low-HOTAIR expression in the same TNM stage, the patients were further divided into two groups: stage I–II and stage III–IV; the OS of high- and low-HOTAIR groups were respectively evaluated. The results show that high-HOTAIR was related to poor OS of LIHC with stage III–IV (P = 0.02, Supplementary Figure 5(a)). However, there was no noteworthy correlation found between HOTAIR expression level and OS in LIHC with stage I–II (P = 0.292, Supplementary Figure 5(b)).
The correlation between HOTAIR expression and clinicopathological parameters
To further validate the clinical value of HOTAIR in liver cancer, we explored the association between HOTAIR expression and the clinicopathological parameters of liver cancer based on the TCGA dataset (Supplementary Figure 6). HOTAIR expression has significant correlations with histologic grade (G1 vs. G4), TNM (M0 vs. M1) status and tumor stage (stage I vs. stage IV). Additionally, we investigated the age factor, and there was no significant difference between the HOTAIR expression and LIHC.
Functional enrichment analysis of HOTAIR in LIHC
We then performed a KEGG signaling pathway analysis of HOTAIR in the LIHC patients. We discovered that HOTAIR is significantly enriched in neuroactive ligand–receptor interaction, calcium signaling pathway and PI3 K/AKT signaling axis in cancer signaling pathway (Figure 3) (P < 0.01).
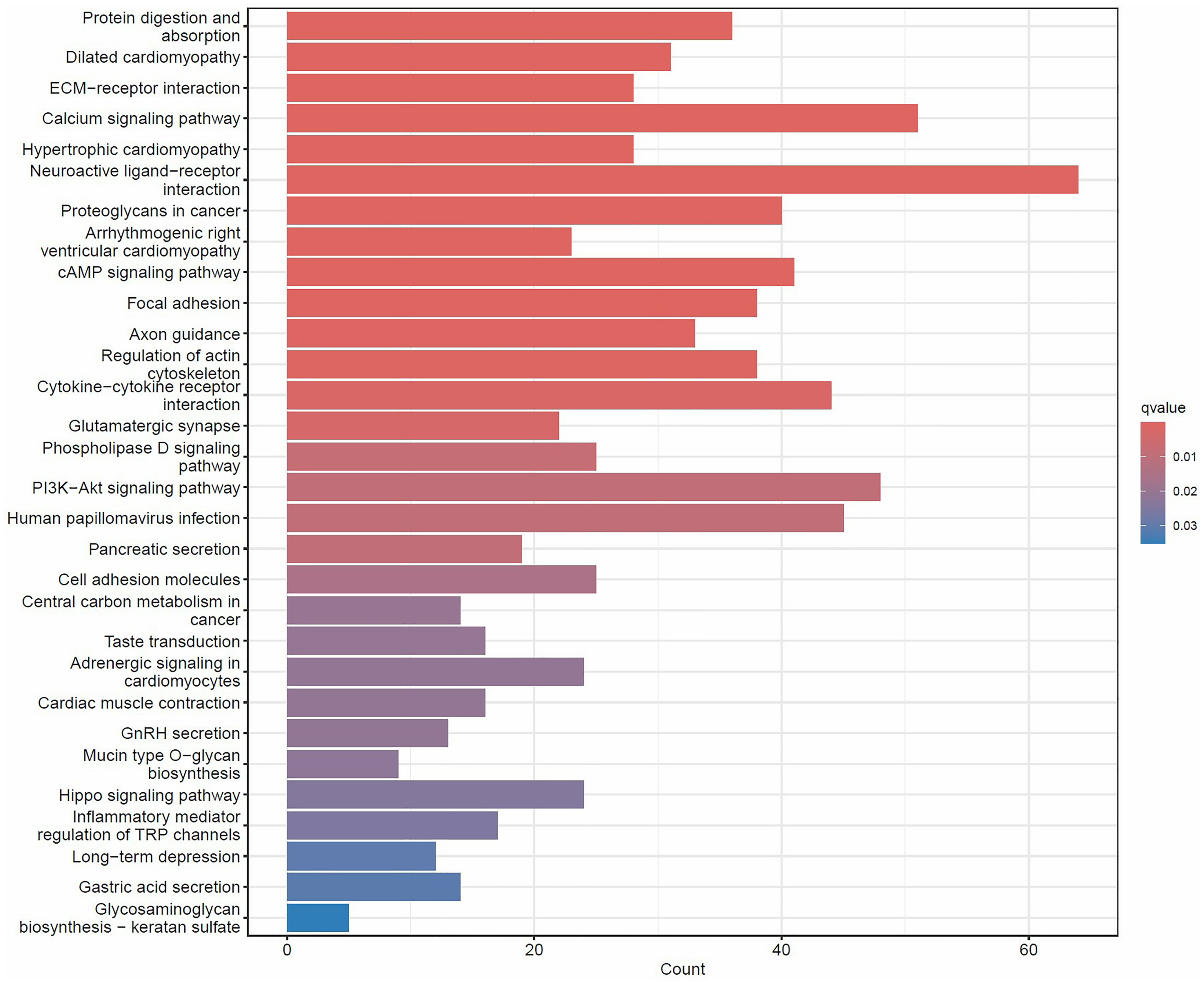
KEGG pathway analysis for hub genes.
Discussion
It has been demonstrated that overexpressed HOTAIR exhibits pro-oncogenic activity and is associated with several characteristics of cancer that inhibit apoptosis, cellular differentiation, tumor growth, invasion, metastasis, and even radiosensitivity.32–34 Numerous studies revealed that the genetic variants of HOTAIR may affect regulatory factors and cause the overexpression of HOTAIR. Moreover, it could cause the overexpression of HOTAIR, which acts on cancer prognosis. Wu et al. 35 have shown that HOTAIR is important in the OS of oral carcinoma through the regulation of the PRC2. X.S. Ge et al. 36 have shown that HOTAIR could promote tumor aggressiveness through the inhibition of WIF-1 through the regulation of the Wnt signaling pathway in esophageal squamous cell carcinoma. Recently a study confirmed that HOTAIR could influence radiosensitivity of pancreatic cancer through regulating autophagy. 37 Furthermore, Wang et al. 38 have demonstrated that HOTAIR might serve as a diagnostic biomarker for cancer recurrence of esophageal squamous cell carcinoma patients who undergo tumor resection. Thus, it is meaningful and necessary to perform a comprehensive meta-analysis to evaluate the clinic significance with HCC.
Previous meta-analyses have investigated the association between HOTAIR expression and clinicopathological parameters. However, most studies focus on malignancies of the digestive tract,39,40 but the diversity of cancer types may reduce its specificity in results. In this meta-analysis, a total of 8 studies including 399 patients were included. The combined results showed that a higher expression of HOTAIR was correlated with an advanced TNM stage, distant metastasis, poor tumor differentiation, and hepatitis infection. We found that high-expression HOTAIR was associated with a shorter OS in patients with liver cancer, as well as poorer RFS. We also noticed that HOTAIR was upregulated in LIHC by analyzing relational databases. Our work confirmed the expression of HOTAIR in LIHC and normal tissue samples; the expression of HOTAIR was higher in liver cancer tissues compared to normal tissues. Subsequently, a high-HOTAIR expression observed poor OS of liver cancer patients through bioinformatics analysis. Moreover, the expression level of HOTAIR correlated with the clinicopathological TNM status, histologic grade, and tumor stage, which is consistent with the results of the our meta-analysis.
Through KEGG analysis, we found that HOTAIR was involved in the enrichment of multiple tumor-related signaling pathways. Just as related literature shows that the neuroactive ligand−receptor interaction pathway may influence the immune status of the tumor. 41 The PI3 K/AKT signaling pathway plays a pivotal role in regulating various pathophysiological processes such as cell proliferation, migration, and drug resistance. 42 It has been reported that HOTAIR activates the Akt phosphate by down-regulating miR-34a (a downstream target of P53) in HCC, and the Akt kinase signaling pathway can induce drug resistance in HCC through PI3 kinase or Akt itself, suggesting that the HOTAIR/PI3 K/AKT signaling pathway may be a potential therapeutic target in HCC. 43 In addition, abnormal changes in intracellular ion homeostasis in HCC cells—especially intracellular Ca2+ as a second messenger, whose ion channels can simultaneously respond to oxidative stress and regulate intracellular ion homeostasis—Ca2+ ion channels are considered to be an important factor in hepatocellular injury, cells continue to multiply and HCC drug resistance. However, the mechanism of its interaction with HOTAIR needs to be further explored. 44
It is well known that serum AFP is widely used for screening suspicious HCC lesions, and has been proven to have the capability of prefiguring the prognosis. However, studies have shown that AFP has low diagnostic sensitivity, particularly in the diagnosis of early-stage HCC, and that its level is raised in some other conditions, such as fibrotic chronic liver diseases. 45 A recent study by El-Shendidi et al. 46 shows that serum HOTAIR was clearly over-expressed in patients with HCC compared to healthy groups, and that the increase of serum HOTAIR was connected with the advancement of HCC stage. The results suggest that the detection of circulating HOTAIR can be used or combined with serum AFP, which is helpful for the diagnosis and staging of HCC. Hence, HOTAIR might be a promising biomarker for HCC diagnosis.
Based on our current study, the clinical significance of HOTAIR in patients with HCC can be summarized as follows. First, HOTAIR could be considered in combination with other biomarkers to enhance the accuracy of predicting prognoses, including OS and RFS. Second, HOTAIR could be applied to indicate the clinicopathological characteristics of the tumor, such as TNM stage and degree of cell differentiation. Finally, HOTAIR more than participates in the regulation of HCC pathways; based on the activities of the HOTAIR targeted block, it may be an important way to resolve tumor resistance.
However, the study does have some limitations. First, the sample size included in the meta-analysis and bioinformatics is relatively small. Therefore, further larger scale studies are needed to confirm the results of this study. Second, most patients included in the meta-analysis were Chinese; however, studies in other races are rare, which might cause publication bias. Third, since the raw values of HR and 95% CIs are not given, some of these can only be acquired by calculation from the data or extraction from the Kaplan–Meier curve, which may cause some inaccuracies. Fourth, there is no consensus regarding a cut-off definition of high- and low-HOTAIR expression, since it varies in different studies. Finally, as negative results do not tend to be published, the role of HOTAIR in cancer may be exaggerated, which might lead to a potential publication bias, and experimental research remains to be further confirmed.
Given the limitations of our study, we look forward to more large-scale, better-designed studies and related experiments to explore the mechanisms of HOTAIR in preventing the occurrence and development of liver cancer.
Conclusion
To sum up, our study provides a theoretical foundation and a new direction for the clinical diagnosis and treatment of HCC, and indicates that HOTAIR may well be a prospective biomarker for assessing clinical tumor stage and prognosis.
Supplemental Material
sj-jpg-1-jbm-10.1177_03936155241252458 - Supplemental material for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis
Supplemental material, sj-jpg-1-jbm-10.1177_03936155241252458 for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis by Ping Wen, Xiyu Qi and Ruzhen Zheng in The International Journal of Biological Markers
Supplemental Material
sj-jpg-2-jbm-10.1177_03936155241252458 - Supplemental material for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis
Supplemental material, sj-jpg-2-jbm-10.1177_03936155241252458 for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis by Ping Wen, Xiyu Qi and Ruzhen Zheng in The International Journal of Biological Markers
Supplemental Material
sj-jpg-3-jbm-10.1177_03936155241252458 - Supplemental material for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis
Supplemental material, sj-jpg-3-jbm-10.1177_03936155241252458 for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis by Ping Wen, Xiyu Qi and Ruzhen Zheng in The International Journal of Biological Markers
Supplemental Material
sj-jpg-4-jbm-10.1177_03936155241252458 - Supplemental material for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis
Supplemental material, sj-jpg-4-jbm-10.1177_03936155241252458 for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis by Ping Wen, Xiyu Qi and Ruzhen Zheng in The International Journal of Biological Markers
Supplemental Material
sj-jpg-5-jbm-10.1177_03936155241252458 - Supplemental material for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis
Supplemental material, sj-jpg-5-jbm-10.1177_03936155241252458 for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis by Ping Wen, Xiyu Qi and Ruzhen Zheng in The International Journal of Biological Markers
Supplemental Material
sj-jpg-6-jbm-10.1177_03936155241252458 - Supplemental material for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis
Supplemental material, sj-jpg-6-jbm-10.1177_03936155241252458 for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis by Ping Wen, Xiyu Qi and Ruzhen Zheng in The International Journal of Biological Markers
Supplemental Material
sj-doc-7-jbm-10.1177_03936155241252458 - Supplemental material for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis
Supplemental material, sj-doc-7-jbm-10.1177_03936155241252458 for Value of the HOTAIR expression assay in predicting therapy target in hepatocellular carcinoma: A meta-analysis and bioinformatics analysis by Ping Wen, Xiyu Qi and Ruzhen Zheng in The International Journal of Biological Markers
Footnotes
Author contributions
PW drafted the manuscript, contributed to article designs and the data analysis. XQ participated in the coordination of the study. RZ conceived the study and participated in its design. All authors made substantial contributions to the acquisition of data and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical consideration
The data is collected from public database sources and does not violate any ethical guidelines.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
