Abstract
Introduction
Anlotinib, a tyrosine kinase inhibitor, has demonstrated significant anti-tumor activity in hepatocellular carcinoma (HCC); nevertheless, the optimal combination therapy is still contested. Consequently, we synthesized the efficacy and safety of anlotinib in combination therapy for advanced HCC via meta-analysis, offering more extensive data for clinical application.
Methods
We systematically searched PubMed, Embase, Web of Science, and Cochrane Library. The primary efficacy indicators included median overall survival (mOS), median progression-free survival (mPFS), overall response rate (ORR), and disease control rate (DCR). The relevant safety indicators were adverse events (AEs). We conducted a meta-analysis utilizing the STATA 14.0 statistical software.
Results
A total of 14 studies involving 863 patients with HCC were included. The combination therapy with anlotinib showed an ORR of 33.8% (95% confidence intervals (CI): 26.8%-40.7%) and a DCR of 77.1% (95% CI: 70.1%-84.2%). The mPFS and mOS were 6.9 months (95% CI: 6.2-7.6) and 17.1 months (95% CI: 15.6-18.6), respectively. Common AEs associated with anlotinib combination therapy included liver function abnormalities 52.9% (95% CI: 37.7%-68.0%), hand-foot skin reaction 37.2% (95% CI: 26.2%-48.2%), and decreased appetite 32.0% (95% CI: 18.7%-45.3%).
Conclusions
Anlotinib-based combination therapy demonstrated promising efficacy with manageable safety in advanced HCC patients.
Introduction
Hepatocellular carcinoma (HCC) is a major global health issue, ranking as the sixth most common cancer and the third leading cause of cancer-related death, with approximately 800 000 annual deaths worldwide.1,2 Advanced HCC remains particularly challenging to manage due to its complex biological characteristics, limited early diagnostic methods, and restricted treatment options. 2 For over a decade, sorafenib served as the only systemic therapy to improve survival in advanced HCC patients. However, its efficacy is modest—extending median overall survival (mOS) by less than 3 months (10.7 months vs 7.9 months in the placebo group, per the SHARP study) and achieving an objective response rate (ORR) of less than 5%. 3 Since 2017, new tyrosine kinase inhibitors (TKIs) such as lenvatinib, regorafenib, and cabozantinib have been approved for advanced HCC,3,4 but none have demonstrated superior efficacy to sorafenib: lenvatinib showed non-inferior mOS (13.6 months), while cabozantinib only extended mOS to 10.2 months in second-line settings.5-7 As Amit G. et al. Noted in the context of HCC treatment paradigms, local therapies like transarterial chemoembolization (TACE) are further limited by their inability to address advanced-stage tumors with microvascular invasion or distant metastasis—such tumors either harbor occult micrometastases beyond TACE’s local action scope or have distant spread that TACE cannot target, making the therapy insufficient to improve outcomes in these patient groups. 8
The core limitations of existing HCC treatments stem from tumor heterogeneity and acquired resistance. HCC frequently harbor diverse genetic aberrations and activate alternative pro-tumor signaling pathways—including platelet-derived growth factor receptor (PDGFR), fibroblast growth factor receptor (FGFR), and stem cell factor receptor (c-Kit)—that bypass the inhibition of single targets (eg, VEGFR targeted by sorafenib). 9 This pathway redundancy leads to treatment failure, highlighting an urgent need for therapies that can cover multiple pathogenic pathways and target both local and systemic tumor progression.
Anlotinib, a novel small-molecule multi-target TKI, is uniquely positioned to address these unmet needs. It effectively inhibits four key pro-tumor kinase families (VEGFR, PDGFR, FGFR, c-Kit), 10 directly counteracting tumor heterogeneity and acquired resistance by blocking alternative escape pathways that single-target agents miss. This broad mechanism enables dual anti-tumor effects: suppressing angiogenesis via inhibition of VEGFR/PDGFR/FGFR, and directly inhibiting tumor cell proliferation via c-Kit and modulation of the extracellular signal-regulated kinase/protein kinase B (ERK/AKT) pathway. 11 The APOLLO study, which evaluated anlotinib as first-line therapy for advanced HCC, confirmed its clinical value—significantly prolonging patients’ mOS and median progression-free survival (mPFS) compared to controls, with hypertension as the most common manageable adverse event. 12
Notably, the generalizability of randomized controlled trials (RCTs) like APOLLO is limited by strict inclusion criteria—often excluding patients with comorbidities, poor performance status, or abnormal laboratory values. To address this evidence gap and validate the real-world efficacy and safety of anlotinib-based combination therapy in advanced HCC, we conducted a systematic meta-analysis. Our findings aim to provide generalizable evidence for clinical practice and expand treatment options for advanced HCC patients.
Methods
Search Strategy
This systematic review and meta-analysis strictly followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines to ensure the integrity of the study and its reporting. The PRISMA standard provides key support throughout the process—from formulating the research plan to presenting the results—via its 27-item checklist and systematic methodology. This approach ensures the review is comprehensive and transparent. 13 The literature management of the article is completed by EndNote.
We searched all literatures with publication dates up to May 20, 2025. The search was conducted in four major electronic databases: PubMed, Embase, Web of Science, and Cochrane Library. We designed our search strategy using key terms based on the (Population, Intervention, Comparison, Outcome) PICO framework: “anlotinib” AND ((neoplasms, Hepatic) OR (neoplasms, Liver) OR (Cancer of Liver) OR (Hepatocellular Cancer) OR (Liver Cancer)). This approach aimed to fully capture studies on the efficacy and safety of anlotinib combination therapy for advanced HCC. We didn’t restrict by language to include international research. We used Boolean operators like “AND” and “OR” to refine searches. Full strategies for the databases are available in eAppendix. Additionally, we checked reference lists of relevant studies to find more papers. This multi-step method helped us comprehensively collect all potential studies.
Eligibility Criteria
Inclusion Criteria
Inclusion Criteria: (1). Study Design: RCTs, cohort studies, or case-control studies evaluating the efficacy and/or safety of anlotinib combination therapy in advanced HCC. (2). Population: Studies involving patients with advanced HCC. (3). Intervention: Focus on anlotinib combination therapy. (4). Outcome: Studies must report at least one of the following: overall survival rate, progression-free survival rate, response rate, or safety profile (including adverse event frequency and types). (5). Language and Publication Status: No restrictions on language or publication status; both published and unpublished studies are included to minimize publication bias.
Exclusion Criteria
(1). Irrelevant Study Design: Editorials, letters, review studies, laboratory, comments and animal studies focusing only on pharmacokinetics or dosage determination were excluded. (2). Ineligible Population: Studies including patients with non-advanced HCC or other cancer types were excluded. (3). No anlotinib Combination Therapy: Studies not specifically evaluating anlotinib combination therapy or using it as an intervention were excluded. (4). Inadequate Data: Studies lacking clear efficacy/safety outcomes or insufficient data for extraction and analysis were excluded. (5). Redundant Data: Studies with duplicate or overlapping data were excluded to avoid double-counting.
Data Extraction and Quality Assessment
We used a systematic data extraction process to ensure accuracy and comprehensiveness. Two independent reviewers screened literature and extracted data, each evaluating studies to obtain relevant information. They then cross-checked findings to ensure consistency. If any disagreement persists without consensus, a third reviewer will be appointed to make an impartial decision. Data extracted from each study included author names, publication years, and case numbers. Clinical and safety outcomes were evaluated based on the overall response rate (ORR), disease control rate (DCR), median overall survival (mOS), median progression-free survival (mPFS), incidence of any adverse events (AEs), and incidence of grade 3 or higher AEs.
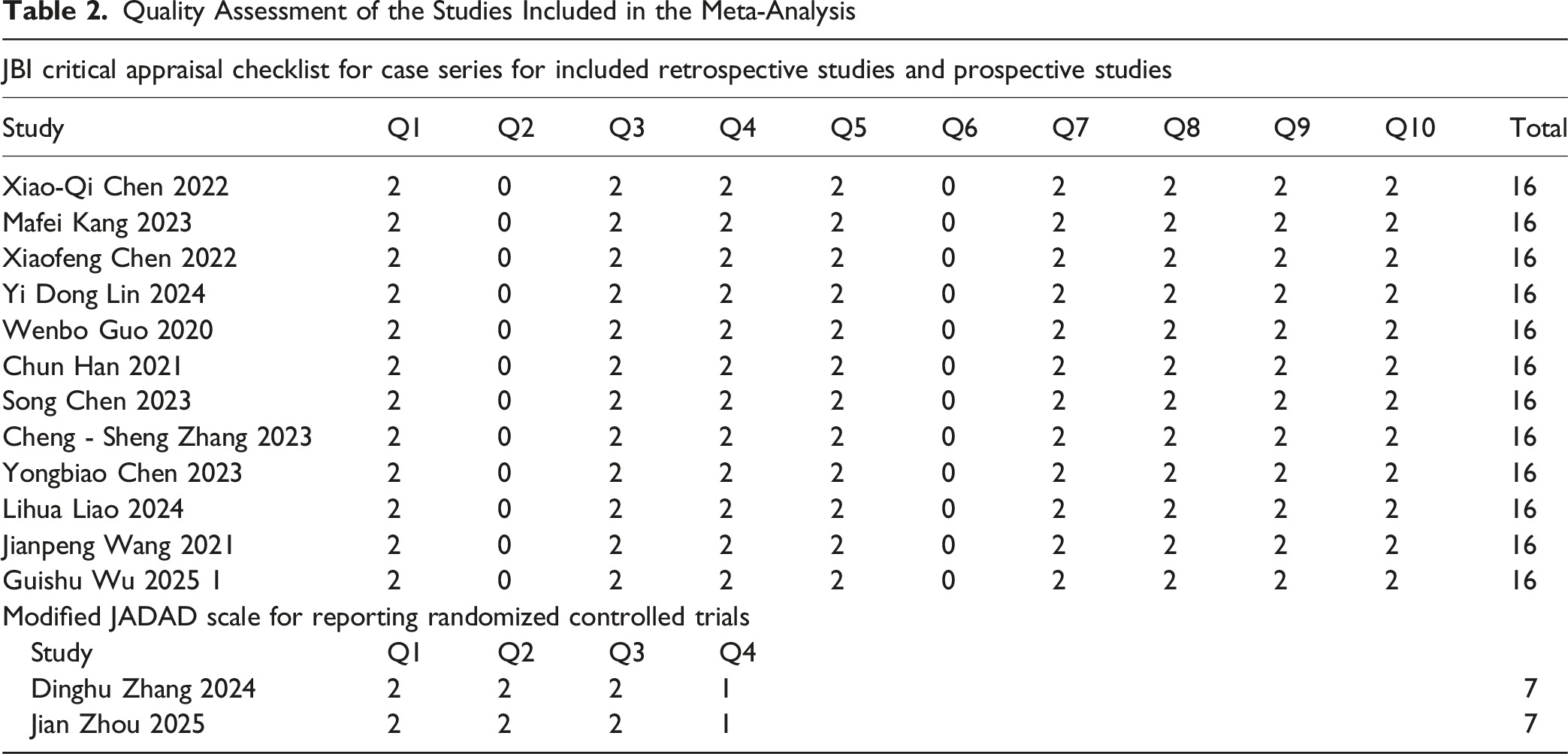
Two independent reviewers assessed study quality using the Joanna Briggs Institute Critical Appraisal Checklist for Patient Series and the JADAD scale.
The Joanna Briggs Institute (JBI) Critical Assessment Checklist for Patient Series including Retrospective Studies and Prospective Studies
Number Q1-Q10 indicates the following query: Q1, have the criteria for inclusion in the case series been clearly defined? Q2, Have all participants in the case series been evaluated in a consistent and reliable manner? Q3, Have reliable methods been used to identify the status of all participants in the case series? Q4, Does the case series continuously include participants? Q5, Have the participants been included in the case series? Q6, Is the demographic report of the research participants clear? Q7, Has the clinical information of the participants been clearly reported? Q8, Is the case result or subsequent discovery clearly recorded? Q9, Is the demographic information of the display location/clinic clearly recorded? Q10, Is statistical analysis conducted properly?
JADAD Scale for Reporting Randomized Controlled Trials
Numbers Q1-Q4 in heading signified: Q1: Was the study described as randomized? Q2: Was the method of randomization appropriate (eg, computer-generated random numbers)? Q3: Was the study described as double-blind? Q4: Was there a description of withdrawals and dropouts?
Statistical Analysis
This meta-analysis used multiple statistical methods to ensure result reliability. Cochran’s Q-test and the I2 statistic were used to assess inter-study heterogeneity. If I2 < 50% and P ≥ .10, a fixed-effects model was applied; otherwise, a random-effects model was used. For binary outcomes, the summary effect size was calculated proportionally and presented with 95% confidence intervals (CIs). Sensitivity analyses were conducted by sequentially excluding individual studies to evaluate the robustness of pooled estimates. Publication bias was detected using Begg and Egger linear regression tests (P < .05 indicated potential bias). AEs rates were evaluated via summary ratio analysis, for which appropriate models were selected based on heterogeneity levels. All analyses were performed using Stata 14.0, with two-sided P < .05 defined as statistically significant and 95% CIs used to assess estimation precision.
Ethics Approval and Consent to Participate
This meta-analysis complied with the principles of the Helsinki Declaration. In the included studies, all participants provided informed consent, and the research was approved by the respective institutional ethics committees. As this study involved a meta-analysis of published data, additional informed consent was not required.
Results
Study Selection
During the initial stages of this systematic review and meta-analysis, an extensive search identified 366 potentially relevant studies. After removing duplicate entries, the titles and abstracts of the remaining studies were screened based on predefined inclusion and exclusion criteria—considering study design, participant characteristics, outcome measures, and methodological quality. This process shortlisted 43 studies for full-text review. Following independent evaluation by multiple reviewers, 29 studies were excluded: 3 review articles, 8 case reports, and 18 studies with insufficient data. Ultimately, 14 studies met all inclusion criteria and were included in the final meta-analysis (Figure 1).12,14-26 The Flow Diagram of the Meta-Analysis
Baseline Characteristics of Included Studies
Baseline Characteristics of 14 Studies in Network Meta-Analysis

ECOG-PS, eastern cooperative oncology group performance status; CTCAE, common terminology criteria for adverse events; RCTs, randomized controlled trials; TACE, transarterial chemoembolization; SBRT, stereotactic body radiotherapy; PD-1, programmed cell death protein 1.
Quality Assessment
Quality Assessment of the Studies Included in the Meta-Analysis
Tumor Response
We conducted a meta-analysis of 14 studies to assess the ORR in patients receiving anlotinib combination therapy. The analysis showed an overall ORR of 33.8% (95% CI: 26.8%-40.7%, I2 = 78.7%, P = .000) (Figure 2A). For the DCR, 14 studies were included, yielding an overall DCR of 77.1% (95% CI: 70.1%-84.2%, I2 = 85.5%, P = .000) (Figure 2B). Forest Plot Depicting Objective Response Rate (ORR) and Disease Control Rate (DCR) of Anlotinib Combination Therapy in Advanced Hepatocellular Carcinoma (HCC). (A) ORR; (B) DCR
Survival
The aggregated median survival rate was estimated because most studies gave survival estimates as median values and confidence ranges. A total of 6 and 4 investigations, respectively, presented mPFS and mOS. Nevertheless, certain studies failed to provide confidence ranges for mPFS and mOS, resulting in their exclusion from the meta-analysis. The aggregated mPFS and mOS were 6.9 months (95% CI: 6.2-7.6; I2 = 0.0%, P = .843) and 17.1 months (95% CI: 15.6-18.6; I2 = 0.0%, P = .510), respectively (Figure 3A–B). Forest Plot of the Pooled (A) median Progression-free Survival (mPFS) and (B) Median Overall Survival (mOS) According to the Treatment Regimen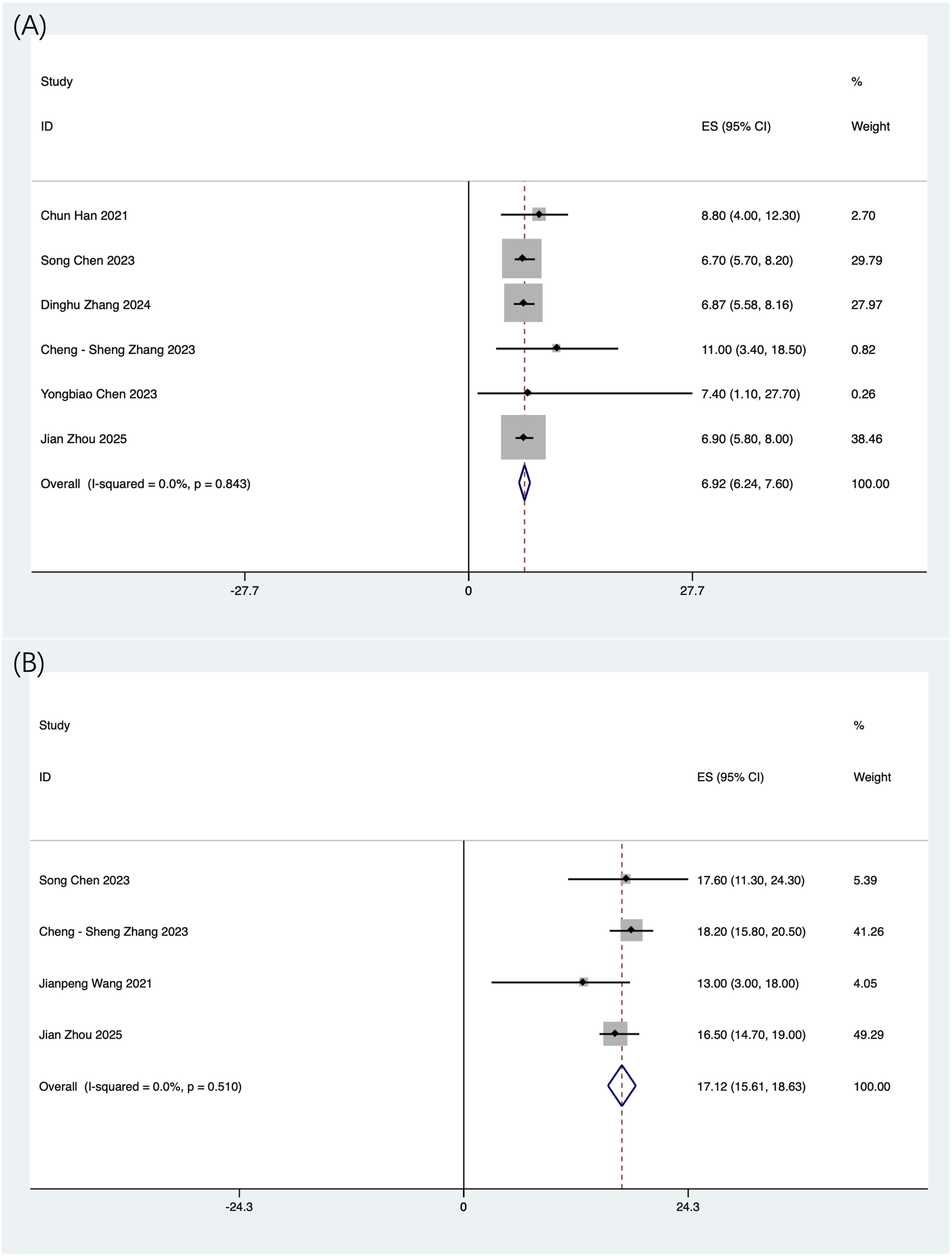
For survival data, 6 studies reported mPFS and 4 studies reported mOS. However, some studies lacked confidence intervals for mPFS and mOS, so they were excluded from the meta-analysis. The pooled mPFS was 6.9 months (95% CI: 6.2-7.6; I2 = 0.0%, P = .843), and the pooled mOS was 17.1 months (95% CI: 15.6-18.6; I2 = 0.0%, P = .510) (Figure 3A–B).
Subgroup Analysis
We hypothesized that heterogeneity in ORR and DCR might originate from differences in therapy types—such as combinations with immune checkpoint inhibitors (ICIs), radiation therapy (RT), or TACE—or differences in study design (retrospective vs prospective). To evaluate these factors, meta-regression was conducted. The results indicated that neither therapy type (ORR: P = .242; DCR: P = .211) nor study design (ORR: P = .209; DCR: P = .670) significantly contributed to the heterogeneity, implying that no single factor was solely responsible. Instead, the heterogeneity likely arose from the interaction of multiple factors. Subgroup analyses were therefore performed to visually examine relevant groupings for patterns and trends in ORR, DCR, mPFS, and mOS outcomes. These analyses provided important contextual interpretation of the heterogeneity and yielded clinically useful hypotheses for future validation studies.
The ORR for combination therapy was 27.1% (95% CI: 20.7%-33.4%, I2 = 46.7%, P = .081) (Figure 4A). For combination RT, the ORR was 41.1% (95% CI: 12.7%-69.5%, I2 = 92.6%, P = .000) (Figure 4A), while combination TACE yielded an ORR of 36.9% (95% CI: 29.9%-43.9%, I2 = 12.1%, P = .000) (Figure 4A). By study design, retrospective studies had an overall ORR of 38.1% (95% CI: 28.6%-47.6%, I2 = 71.3%, P = .001) (Figure 4B), and prospective studies showed an ORR of 28.4% (95% CI: 20.4%-36.3%, I2 = 67.3%, P = .005) (Figure 4B). Forest Plot of the Pooled Objective Response Rate (ORR) and Disease Control Rate (DCR). (A) ORR of Subgroups With Different Combination Therapy Methods; (B) ORR of Subgroups Using Different Research Methods; (C) DCR of Subgroups Treated With Different Combination Therapies; (D) DCR of Subgroups Using Different Research Methods
The DCR for combination therapy was 74.2% (95% CI: 64.0%-84.4%, I2 = 83.2%, P = .000) (Figure 4C). For combination RT, the DCR was 67.0% (95% CI: 54.4%-79.6%, I2 = 49.4%, P = .139) (Figure 4C). The pooled DCR for the TACE subgroup was 87.5% (95% CI: 79.4%-95.5%, I2 = 73.1%, P = .000) (Figure 4C). By study design, retrospective studies showed a DCR of 75.0% (95% CI: 63.9%-86.1%, I2 = 84.3%, P = .000) (Figure 4D), and prospective studies had a DCR of 79.0% (95% CI: 68.6%-89.4%, I2 = 88.7%, P = .000) (Figure 4D).
For patients receiving combination ICIs, the mPFS was 7.1 months (95% CI: 6.0-8.2, I2 = 0.0%, P = .407), and the mOS was 17.3 months (95% CI: 15.7-18.7, I2 = 8.6%, P = .269). Patients treated with combination RT had an mPFS of 7.4 months (95% CI: 1.1-27.7). Those receiving combined TACE had an mPFS of 6.8 months (95% CI: 5.9-7.7, I2 = 0.0%, P = .853) and an mOS of 15.6 months (95% CI: 10.7-20.5, I2 = 0.0%, P = .364) (Figure 5A–B). Forest Plot of the Pooled Median Progression-Free Survival (mPFS) and Median Overall Survival (mOS). (A) mPFS of Subgroups Treated With Different Combination Therapies; (B) mOS of Subgroups Treated With Different Combination Therapies; (C) mPFS of Subgroups Treated With Different Research Methods; (D) mOS of Subgroups Treated With Different Research Methods
In prospective studies, the mPFS was 7.0 months (95% CI: 6.2-7.8, I2 = 0.0%, P = .759), and the mOS was 17.3 months (95% CI: 15.6-18.9, I2 = 8.6%, P = .296) (Figure 5C–D). In retrospective studies, the mPFS was 6.7 months (95% CI: 5.7-8.2), and the mOS was 15.6 months (95% CI: 10.7-20.5, I2 = 0.0%, P = .364) (Figure 5C–D).
Toxicities
Adverse Events of the Studies Included in the Meta-Analysis

AEs, adverse events.

Forest Plot of the Pooled Adverse Events (AEs). (A) All Grades of AEs; (B) AEs of ≥ Grade 3
The three most common AEs across all grades were abnormal liver function (52.9%, 95% CI: 37.7%-68.0%, I2 = 93.2%, P = .000), hand-foot skin reaction (37.2%, 95% CI: 26.2%-48.2%, I2 = 82.8%, P = .000), and decreased appetite (32.0%, 95% CI: 18.7%-45.3%, I2 = 91.0%, P = .000). These results highlight the need for regular liver function monitoring and assessment of patients’ quality of life during treatment.
The incidence of grade 3 or higher AEs were within an acceptable range, indicating manageable safety for this combination therapy. The most common grade 3 or higher AEs were hypertension (9.2%, 95% CI: 3.1%-15.3%, I2 = 87.2%, P < .001), thrombocytopenia (6.8%, 95% CI: 1.6%-12.1%, I2 = 84.4%, P < .001), and abnormal liver function (6.7%, 95% CI: 2.1%-11.4%, I2 = 75.1%, P < .001). Although these severe AEs occurred, they were infrequent and can be managed with appropriate clinical supervision.
Sensitivity Analysis
All included studies underwent sensitivity analysis to evaluate the stability of pooled results. The analysis showed no significant changes in outcomes when individual trials were excluded one by one (Figure 7). Sensitivity Analysis Based on (A) Objective Response Rate (ORR); (B) Disease Control Rate (DCR); (C) Median Progression-free Survival (mPFS); (D) Median Overall Survival (mOS); (E) Adverse Events (AEs); (F) AEs Level 3 or Above
Publication Bias
To ensure the robustness of the meta-analysis findings, potential publication bias was determined using the Begg’s and Egger’s tests. The P-values for Begg’s and Egger’s tests for different parameters were as follows: ORR, 0.133 and 0.024, respectively; DCR, 0.002 and 0.083, respectively; mPFS, 0.260 and 0.078, respectively; mOS, 0.734 and 0.536, respectively. Begg’s test indicated the potential for slight publication bias in the DCR outcomes (P = .002), which may be due to small-study effects or other heterogeneity; however, the trim and fill method estimated that zero studies needed to be imputed, suggesting that the asymmetry was not substantial enough to significantly alter the pooled effect size. These results together imply that while some bias may be present, its overall impact on the meta-analysis conclusions is likely limited.
For the ORR, after correcting publication bias using the trim-and-fill method, three studies were imputed after five iterations, resulting in 18 bias-free studies. The pooled effect was 1.33 (95% CI, 1.26-1.41) and, which is similar to the previous results. Those indicates unstable outcomes for ORR and DCR in some meta-analyses, with the potential for new data to alter the conclusions. Contributing factors may include differences in response evaluation criteria (eg, response evaluation criteria in solid tumors (RECIST) vs international response evaluation criteria in solid tumors (iRECIST)), variations in treatment protocols (eg, dosing schedules or combination therapies), and heterogeneity in patient populations across studies. Additionally, small sample sizes or short follow-up periods in some trials may limit the ability to fully assess durable responses or delayed progression, further contributing to variability in ORR and DCR estimates.
Discussion
This meta-analysis of 863 advanced HCC patients shows anlotinib-based combination therapy has favorable efficacy (ORR 33.8%, DCR 77.1%, mPFS 6.9 months, mOS 17.1 months) and manageable safety. Despite substantial advancements in the treatment paradigm of HCC following the introduction of ICIs, the second-line or subsequent management of HCC continues to pose challenges, particularly for patients exhibiting disease progression after first-line therapy. The SHARP study identified sorafenib as the first-line standard treatment, demonstrating a mOS of 10.7 months in the sorafenib cohort compared to 7.9 months in the placebo cohort 3 ; the REFLECT trial indicated that the efficacy of lenvatinib was comparable to that of sorafenib, with a mOS of 13.6 months. 27 The TACTICS trial established that TACE in conjunction with sorafenib extended mPFS (25.2 months vs 13.5 months). 28 The CELESTIAL study showed that second-line therapy with cabozantinib can extend mOS (10.2 months vs 8.0 months). 29 The above research expands treatment options for HCC, though the clinical efficacy of these medications does not surpass sorafenib. Thus, improving efficacy without significantly increasing side effects has become a key priority. The limited efficacy of existing HCC therapies often results from tumor heterogeneity and acquired resistance, particularly through the activation of alternative signaling pathways beyond VEGFR. Anlotinib addresses this limitation by simultaneously inhibiting multiple kinases (VEGFR, PDGFR, FGFR, c-Kit), offering broader anti-angiogenic activity and potentially overcoming escape mechanisms that compromise single-target agents. 10 This multi-target strategy provides a strong rationale for its use in overcoming resistance in advanced HCC. Anlotinib, an oral antineoplastic drug, has shown significant therapeutic effects against multiple solid tumors—including HCC, non-small cell lung cancer, soft tissue sarcoma, esophageal cancer, gastrointestinal malignancies, and renal cancer. Based on these promising results, anlotinib has been approved in China for treating advanced liver cancer patients.
This study evaluated the efficacy and safety of anlotinib combination therapy in advanced HCC patients. A meta-analysis of 14 studies showed an overall ORR of 33.8% and DCR of 77.1% for anlotinib, which are higher than the rates for second-line anlotinib monotherapy (ORR: 8.3%; DCR: 66.7%) in the ALTER-0802 trial. However, mOS and mPFS for anlotinib combination therapy were 17.1 months and 6.9 months, respectively—shorter than the 18.0-month mOS observed with second-line anlotinib monotherapy in ALTER-0802. 30 The ALTER-0802 RCT used strict inclusion criteria, screening patients with favorable baseline characteristics (eg, good performance status, normal organ function, low tumor burden), so the monotherapy group’s mOS (18.0 months) likely reflects a prognostically selected population. In contrast, combination therapy meta-analyses may include broader patient populations—including advanced-stage, drug-resistant, or poor-baseline cases (such as those with more prior treatment lines, poorer physical status assessed by ECOG-PS, or higher tumor burden)-diluting overall survival data. Subgroup analysis revealed that prospective studies yielded superior results compared to retrospective analyses, further supporting the influence of rigorous patient selection on treatment efficacy. Prospective studies typically enroll individuals with better disease control and physical tolerance, leading to improved outcomes. These findings highlight the need for additional real-world studies to assess anlotinib combination therapy in broader patient populations, examine the impact of baseline characteristics, monitor treatment patterns, and validate its clinical benefits beyond controlled trial settings.
Ongoing Trials of Anlotinib Combination Therapy in Advanced Hepatocellular Carcinoma
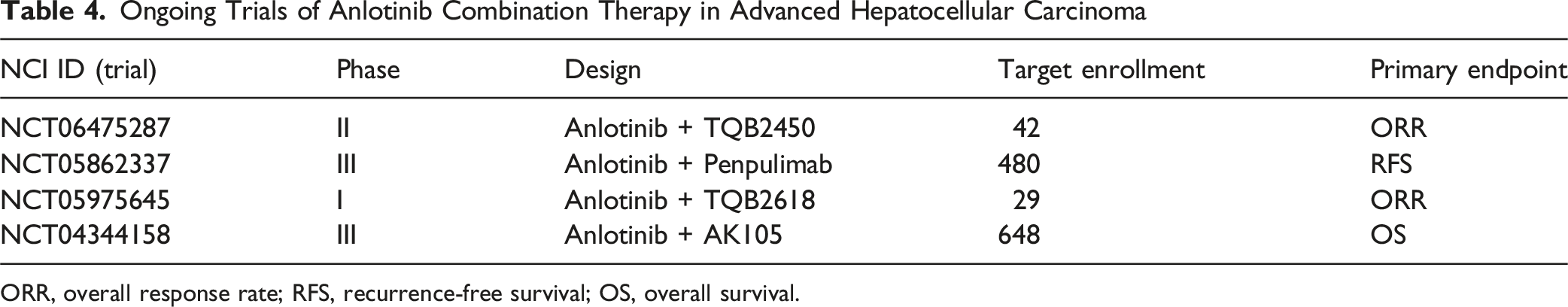
ORR, overall response rate; RFS, recurrence-free survival; OS, overall survival.
Notably, for regimens with higher initial ORR (eg, RT-anlotinib and TACE-anlotinib), patients who exhibit better tumor response are more likely to complete the planned treatment courses with sufficient frequency and longer duration. This forms a positive cycle: favorable efficacy supports sustained treatment, while prolonged and standardized treatment further consolidates therapeutic effects. In contrast, the immunotherapy-anlotinib regimen, despite its relatively lower initial ORR, may require prolonged treatment duration to fully exert the synergistic effect of immune activation and immunosuppression reversal, thereby achieving survival extension. This observation also reflects the correlation between effective tumor response, treatment adherence (including treatment times and duration), and final outcomes.
AEs for Subgroups and ALTER-0802

AEs, adverse events.
Anlotinib, a multitarget TKI, suppresses tumor angiogenesis and proliferation by blocking vascular endothelial growth factor receptors (VEGFR1-3), 34 epidermal growth factor receptor (EGFR), fibroblast growth factor receptors (FGFR1-4), platelet-derived growth factor receptors (PDGFR α/β), C-Kit, and Ret. Experimental evidence demonstrates its dual mechanisms: enhancing antitumor effects through apoptosis induction and autophagy inhibition in lung cancer, 35 and reinforcing antiangiogenic activity via the JAK2/STAT3/VEGFA pathway. 36 Additionally, anlotinib inhibits proliferation, migration, and invasion in small cell lung cancer by targeting c-Met and ERK1/2 signaling, 36 and suppresses liver cancer cell growth through ERK/AKT pathway modulation. 37 In colorectal cancer, it attenuates multidrug resistance by blocking PI3K/AKT signaling, highlighting its potential for combination therapies. Clinical trials and meta-analyses consistently confirm its therapeutic efficacy and safety profile.34,38
This study has limitations. First, heterogeneity was observed in efficacy endpoints including ORR and DCR. We therefore performed subgroup analyses and meta-regression to explore potential sources. The results suggest that heterogeneity likely arises from multiple factors rather than a single source. For example, subgroup differences were found between types of combination therapy (eg, with immune checkpoint inhibitors, radiotherapy, or TACE) and study designs (prospective vs retrospective). These indicate that variation in treatment protocols—such as use of different anti-PD-1 agents (eg, Sintilimab, Penpulimab, Toripalimab) or radiation techniques (eg, stereotactic body radiotherapy (SBRT), intensity-modulated radiotherapy (IMRT))—contribute significantly to heterogeneity. Additionally, differences in patient baseline features, including previous lines of therapy, Child-Pugh class (A or B), and BCLC stage (B or C), may affect outcomes. However, inconsistent reporting and definitions of these variables across studies introduce potential measurement and reporting bias. Other baseline characteristics such as age, sex, and ECOG performance status also varied across studies and were not fully adjusted for, possibly leading to confounding bias. Methodological limitations exist as well: most included studies were retrospective, and some prospective trials did not adequately describe randomization or blinding. Sample sizes were generally small, which may affect the reliability and generalizability of the findings. Future large-scale, prospective, randomized controlled trials that rigorously control and report key patient characteristics are needed to confirm the efficacy and safety of anlotinib combination therapy.
This study shows that anlotinib combination therapy can significantly improve ORR and DCR, and prolong OS and PFS in previously treated HCC patients. The incidence of AEs is within an acceptable range. However, due to the significant heterogeneity among the included studies and methodological limitations in some studies, these findings need to be carefully evaluated when applied in clinical practice. More high-quality randomized controlled trials are required to validate these results.
Conclusions
This meta-analysis confirms the significant clinical efficacy of anlotinib-based combination therapy for advanced HCC, demonstrating strong synergistic antitumor effects through improved ORR and DCR, as well as prolonged mOS and mPFS. These findings position anlotinib combination therapy as a promising strategy in the advanced HCC treatment landscape. While acknowledging the limitations of this study, the novel insights contribute meaningfully to evolving treatment paradigms. The results offer evidence-based support for the development of multidisciplinary, precision medicine-guided comprehensive therapy frameworks, providing critical guidance for both clinical practice and future research. These findings not only support the integration of anlotinib-based combination regimens into current treatment algorithms but also underscore the imperative for future prospective studies to validate its efficacy across diverse patient populations and to explore predictive biomarkers for personalized therapeutic approaches.
Supplemental Material
Supplemental Material - The Efficacy and Safety of Anlotinib Combination Therapy for Advanced Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis
Supplemental Material for The Efficacy and Safety of Anlotinib Combination Therapy for Advanced Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis by Ting Chen, Ran Cui, and Xinlin Yu in Cancer Control
Footnotes
Author Contributions
(I) Design of the study: X Yu and R Cui. (II) Study screening, data extraction, or data analysis: X Yu and T Chen. (III) Manuscript drafting: X Yu. (IV) Figure or table editing: R Cui. (V) Revision of the manuscript: T Chen. (VI) Final approval to the manuscript: all authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data are included in the manuscript.
Trail Registration
PROSPERO registration: CRD42024590395.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
