Abstract
Objective
Peripheral systemic inflammatory, nutritional, and coagulation biomarkers have prognostic and predictive value in various malignancies. We evaluated the prognostic and predictive roles of systemic inflammatory, nutritional, and coagulation biomarkers in the circulating blood of patients with advanced cervical cancer.
Methods
A retrospective study of 795 patients with cervical cancer who received concurrent chemoradiation therapy was performed. Overall survival was evaluated by the Kaplan–Meier estimator. Univariate and multivariate Cox regression models were used to determine prognostic factors associated with overall survival.
Results
The median follow-up time was 76 months. In the univariate analysis, overall survival showed positive prognostic value in patients with a platelet-to-lymphocyte ratio (PLR) <164.29 (P = 0.010), and a plasma fibrinogen (FIB) level <4 g/L(P = 0.012). In the multivariate analysis, the PLR (P = 0.036), and FIB level (P = 0.047) maintained their significance for overall survival. Therefore, the PLR and FIB levels are independent prognostic factors in patients with advanced cervical cancer.
Conclusions
Systemic inflammatory and coagulation biomarkers could help to understand survival differences in the clinical treatment of advanced cervical cancer. The PLR and FIB levels are independent prognostic factors of poor survival in patients with advanced cervical cancer.
Introduction
Cervical cancer is the fourth most common female malignant tumor in the world. 1 According to recent worldwide research in 2018, approximately 570,000 women are diagnosed with cervical cancer, which causes 311,000 deaths worldwide. 2 The established standard of care for advanced cervical cancer is concurrent chemoradiation. 3 At present, the International Federation of Gynecology and Obstetrics (FIGO) staging system has been proven to be able to predict overall survival (OS) in cervical cancer patients. Advanced cervical cancer has a worse prognosis than stage IA–IIA cervical cancer. Because of the poor prognosis of advanced cervical cancer, the identification of prognostic biomarkers remains a challenge.
Systemic inflammation, nutrition, and coagulation have emerged as potential prognostic factors for patient survival because they play roles in tumor promotion and progression.4,5 In recent years, several inflammation-based scores, nutritional statuses, and coagulation indices have been confirmed as significant predictors in cervical cancer and other tumors.5–9 Several prognostic indices have been proposed, such as the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), body mass index (BMI), and serum albumin (ALB) level. All these biomarkers can be acquired easily by peripheral blood biochemical tests and have great advantages for clinical applications.
However, few studies have focused on the correlation between these indices and survival in patients with advanced cervical cancer. Thus, we aimed to evaluate the prognosis of patients with advanced cervical cancer using inflammatory, nutritional, and coagulation indices.
Methods
Patients
We conducted a retrospective analysis of patients who were pathologically diagnosed with cervical cancer at Shandong Cancer Hospital between October 2009 and December 2014. We retrospectively collected clinical characteristics, hematological examination results, and survival data. We excluded (a) patients who underwent hysterectomy or cervical resection surgery; (b) who had other tumors, hematological diseases or severe liver or renal dysfunction; or (c) who did not undergo peripheral blood tests at diagnosis.
One point to be emphasized is that the tumor stage of all patients was redefined according to the FIGO 2018 criteria for cervical cancer. We evaluated the lymph node status though imaging methods such as computed tomography (CT) scans and positron emission tomography-CT scans. Among these patients, 6 were previously diagnosed with stage IB disease, 12 with stage IIA disease, and 85 with stage IIB disease but were restaged with stage III disease. A total of 795 patients with stage IIB–IVA disease were included in this study.
This study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants. This retrospective study was approved by the Ethics Committee of Shandong Cancer Hospital.
Study design and data collection
A total of 795 patients who had received concurrent chemoradiation therapy were included in this study. The clinical and pathological data collected included age, BMI, tumor size, histotype, grade, FIGO stage, and hematological parameters including the neutrophil count, monocyte count, lymphocyte count, platelet count, plasma fibrinogen (FIB) level, lactate dehydrogenase (LDH) level and ALB level. In this study, the NLR, PLR, MLR, systemic inflammation response index (SIRI), and aggregate index of systemic inflammation (AISI) were calculated as follows: NLR = neutrophil count/lymphocyte count; PLR = platelet count/lymphocyte count; MLR = monocyte count/lymphocyte count; SIRI = neutrophil count ∗ monocyte count/lymphocyte count; and AISI = neutrophil count ∗ monocyte count ∗ platelet count/lymphocyte count. We obtained all the raw values at the same time in each patient.
Because of the lack of validated cut-off values, NLR, MLR, PLR, SIRI, and AISI values were used median value as the cutoff values for OS. The laboratory reference values of PLT, LDH, ALB, and FIB were used as the cut-off values for OS. Patients included in this study were followed-up every 6 months for the first 2 years then annually until death, and all patient follow-ups were ceased in May 2020. It was difficult to accurately acquire the time of recurrence and metastasis. Thus, we ultimately decided to use the OS rate to determine patient prognosis. OS was defined as the time from the initial diagnosis to last contact or death.
Statistical analysis
Statistical analyses were performed using Statistical Product and Service Solutions 22.0 software (IBM Corp., Armonk, NY, USA) and GraphPad prism 8.0 (Windows, GraphPad Software, San Diego, CA, USA; www.graphpad.com). Estimates of OS were calculated using the Kaplan–Meier method. Significant prognostic variables in the univariate and multivariate analyses were entered into a Cox regression model. The variables used in the multivariate analysis were obtained from the univariate analysis. P < 0.05 was considered statistically significant in all analyses.
Results
Patient characteristics
The clinical characteristics of the 795 patients are listed in Table 1. The median age of the patients at diagnosis was 51 years (range: 21–69 years). The median follow- up time was 76 months (range: 2–127 months).
Characteristics of the study population (N = 795).
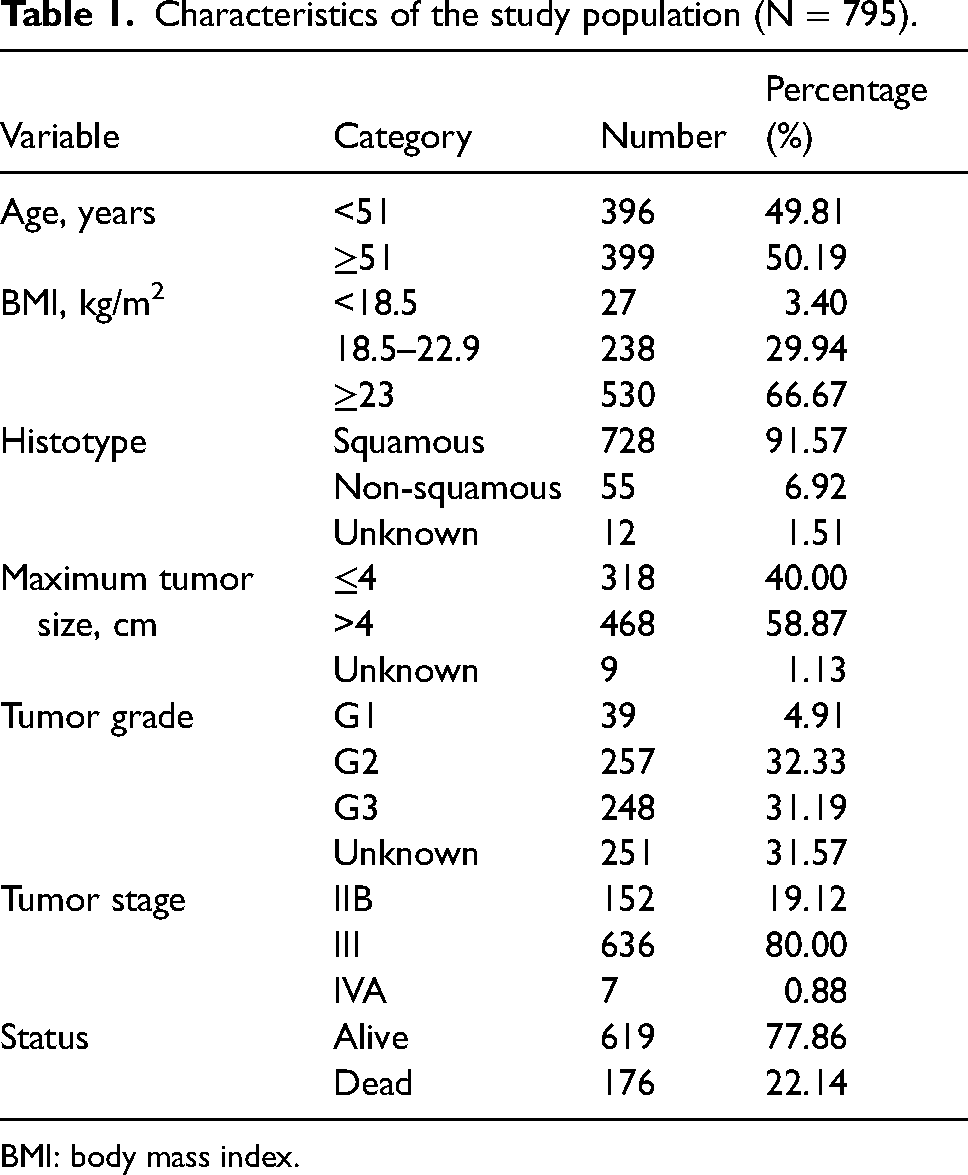
BMI: body mass index.
Univariate and multivariate analyses for OS
In the univariate risk analysis model, a PLR <164.29 (log-rank test P = 0.010), and an FIB level <4 g/L (P = 0.012) showed significantly positive prognostic value for OS (Figures 1 and 2 and Table 2). Other clinically meaningful parameters such as the histological types (P < 0.001), and early tumor stage (III vs. IIB P = 0.001; IV vs. IIB P = 0.010) were also significant prognostic factors.
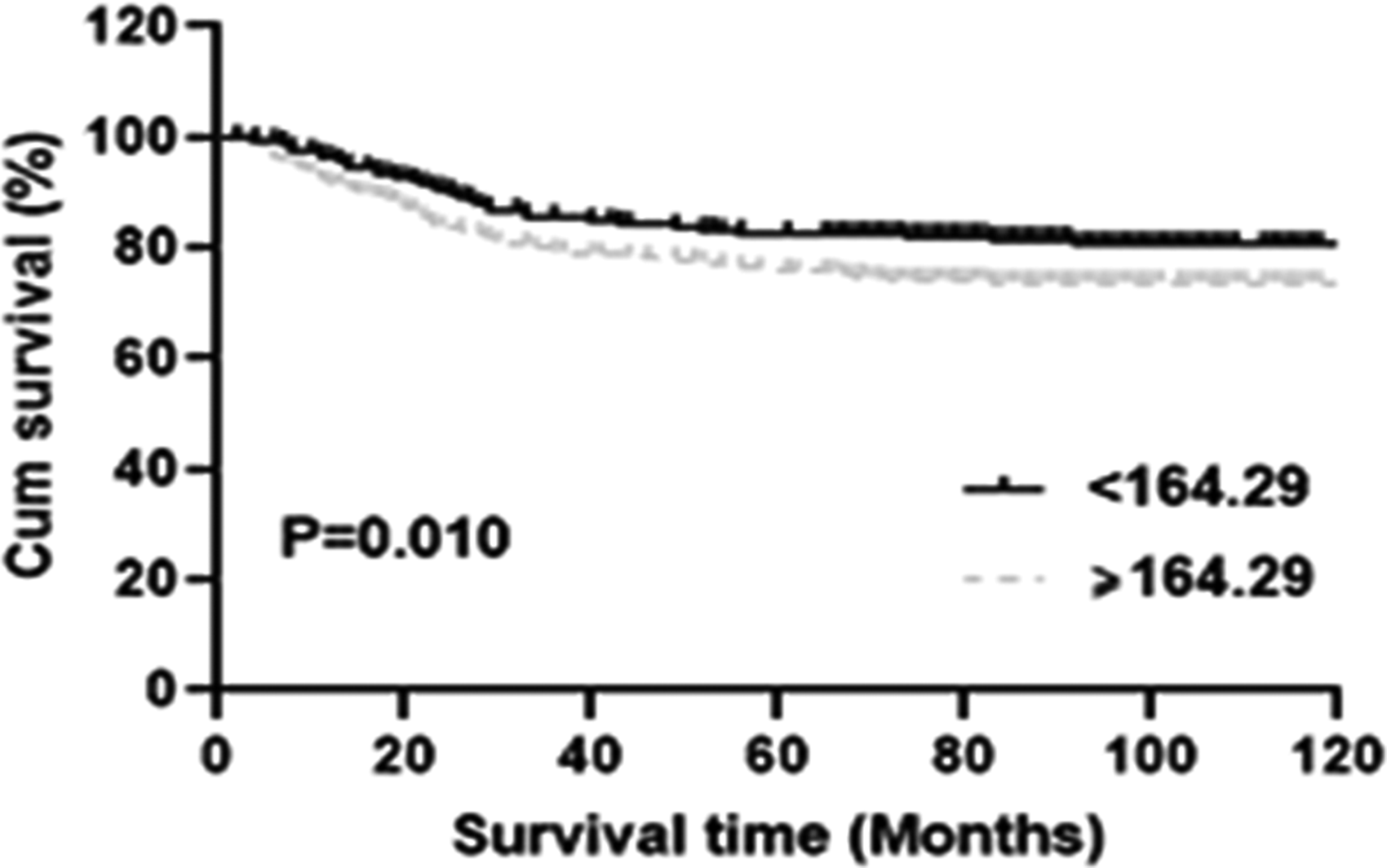
Kaplan–Meier survival curve for overall survival (OS) according to platelet-to-lymphocyte ratio (PLR) values.
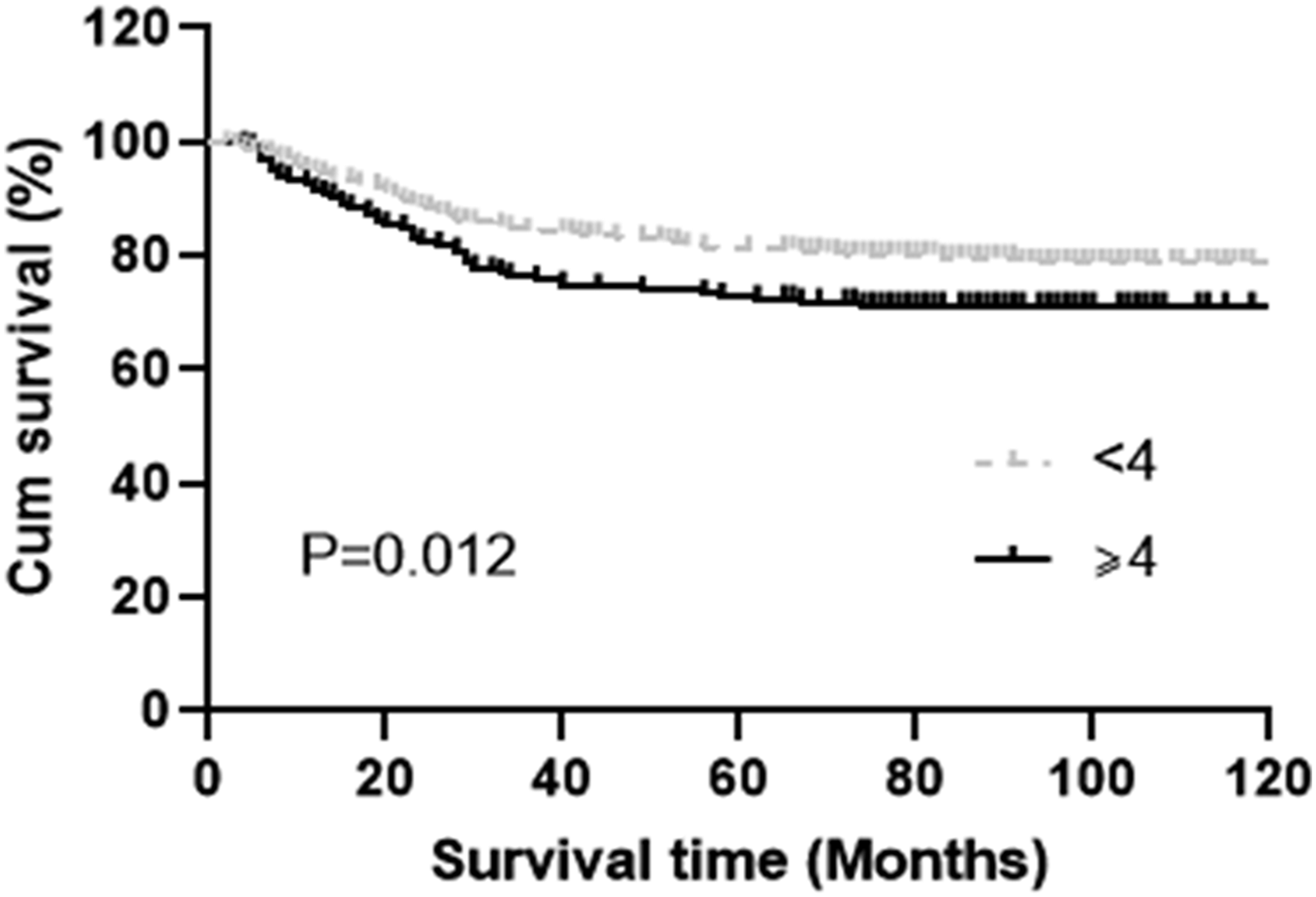
Kaplan–Meier survival curve for overall survival (OS) according to fibrinogen (FIB).
Univariate Cox regression analysis in advanced cervical cancer patients.
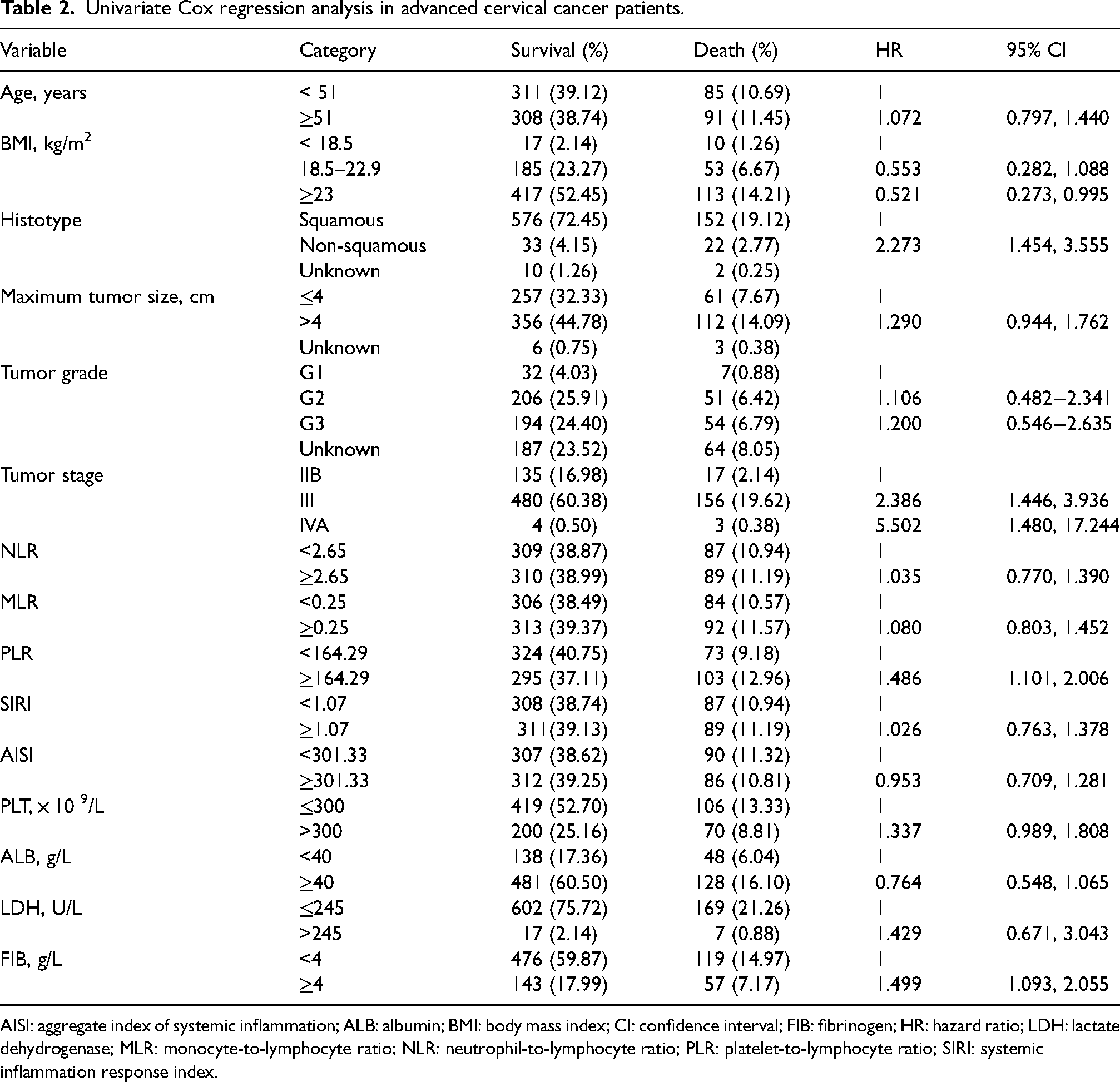
AISI: aggregate index of systemic inflammation; ALB: albumin; BMI: body mass index; CI: confidence interval; FIB: fibrinogen; HR: hazard ratio; LDH: lactate dehydrogenase; MLR: monocyte-to-lymphocyte ratio; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; SIRI: systemic inflammation response index.
In the multivariate analysis, the tumor histological types, stage, PLR, and FIB levels maintained their significance for OS. Therefore, the tumor histological type, stage, PLR, and FIB level were associated with prognosis and should be used as independent prognostic factors according to our series (Table 3).
Multivariate Cox regression analysis in advanced cervical cancer patients.
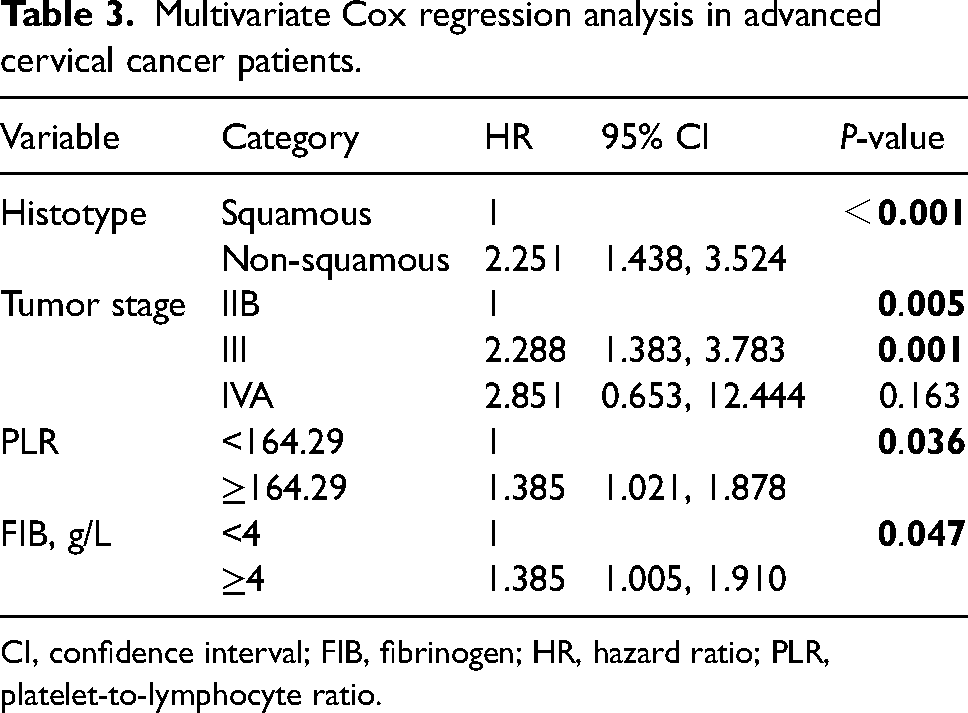
CI, confidence interval; FIB, fibrinogen; HR, hazard ratio; PLR, platelet-to-lymphocyte ratio.
Discussion
Although the incidence of cervical cancer has tended to decrease, it also continues to constitute a major public health problem. 2 Exploring the prognostic factors of cervical cancer, and improving clinical treatment measures are of utmost importance. In recent years, many genetic, laboratory, clinical, and pathological studies have examined the prognostic factors of cervical cancer.10–12 In recent studies, a large number of clinical indicators such as race, age, histological type, grade, and tumor volume seemed to be associated with prognosis. 13 In our study, age, tumor size, and grade were not correlated with prognosis. Moreover, the histological type and tumor stage were identified as independent prognostic factors of poor survival. We believe these results may be related to the characteristics of advanced tumors. Tumor differentiation grade and tumor volume may be better prognostic factors in women with early stage cervical cancer than in those with advanced-stage cervical cancer.
The current study also demonstrated a rare association of the PLR, NLR, MLR, AISI, and SIRI and PLT, FIB, LDH, and ALB levels with OS in patients with advanced cervical cancer. This study also showed that elevated PLR and FIB levels were useful indicators of a poor prognosis.
A significant amount of evidence shows that inflammatory markers have critical value in evaluating the prognosis of most of the common cancers. Inflammation often contributes to the development and progression of cancer. 14 Inflammatory cells are an important component of the tumor microenvironment and participate in the neoplastic process, fostering proliferation, survival, and migration. In addition, tumor cells have co-opted certain signaling molecules of the immune system, such as selectins, chemokines, and their receptors, which stimulate tumor growth and progression.15,16
Platelet counts influence the metastatic potential of tumor cells. Platelets prevent tumor cells from contacting the host's immune system, disturb natural killer cell recognition, and facilitate extravasation of tumor cells. 17 Some studies have reported that increased platelets have a significant effect on patient survival.18–20 Lymphocytes are a critical part of the immune response, and there is no doubt that a low number of lymphocytes can lead to tumor progression.
To explore simple and effective prognostic indicators for assessing the prognosis of cervical cancer patients and guiding clinical treatment, our study examined and analyzed many indicators based on inflammation. The results showed that a high PLR was significantly associated with poor OS in patients with advanced cervical cancer who were treated with concurrent chemoradiation. Li et al. 21 summarized 33 cohort studies including 8215 patients with advanced cancers—including biliary tract cancer, pancreatic cancer, nasopharyngeal carcinoma, and gastric cancer—and concluded that patients with a high pretreatment PLR had poor OS and progression-free survival (PFS). Regarding cervical cancer, Ma et al. 22 researched a total of 12 studies that included 3886 patients and concluded that the pretreatment PLR could serve as a predictive biomarker of a poor prognosis for cervical cancer patients, although most of them were in the early stage. Our result, which was derived mainly from patients with advanced cervical cancer, was the same as that described above and indicates that the PLR is significantly associated with poor survival in cervical cancer patients.
Malnutrition is common in patients with malignant tumors. Scientists have suspected for decades that nutrition has an important influence on the risk of developing cancer. 23 Malnutrition, a subacute or chronic state in cancer patients, may impair immune function and increase susceptibility to infection and complications of treatment, thus leading to an increased mortality rate of cancer patients. 24 Some studies were conducted to evaluate the association between nutrition and cancer. 25 Serum albumin levels and BMI are common nutritional indicators. Some reports showed that a reduction of in BMI and hypoalbuminemia were related to the poor prognosis of malignancy,26–28 while other reports showed that albumin levels and BMI were not independent prognostic indicators for OS.29,30 Our results are congruent with the latter results. Therefore, the relationship between cancer and nutrition is puzzling and needs more study for verification.
There are emerging links between malignancy and the hemostatic system. 31 Fibrinogen plays a critical role in achieving and maintaining hemostasis and is fundamental for effective clot formation. 32 Fibrinogen is also a classical positive acute-phase reactant protein. 33 Tumor cells and tumor-derived inflammatory cells may subclinically activate the coagulation cascade, 34 and there are relationships between hemostatic factors and advanced tumor stage, large tumor size, and tumor biology. 35 Fibrinogen promotes the cellular responses of adhesion, proliferation, and migration during angiogenesis and tumor cell growth by participating in the formation of the extracellular matrix and supporting the binding of growth factors.36–39 Elevated plasma platelet levels are a common phenomenon in a variety of malignancies.37,40
In our present study, we retrospectively analyzed 795 patients with advanced cervical cancer whose pretreatment plasma fibrinogen levels were measured at diagnosis. We found that plasma fibrinogen levels are significantly correlated with a poor prognosis. Polterauer et al. 41 reported that plasma fibrinogen levels were an independent parameter in 313 cervical cancer patients, including 141 patients with advanced cervical cancer, which was in accordance with our study. Zhao et al. 42 concluded that hyperfibrinogenemia may be a valuable parameter for predicting the recurrence of early stage cervical cancer. We verified this finding in a larger number of patients with advanced cervical cancer.
There were some limitations to this study. First, our study lacked data on PFS, which can reflect the status related to cancer. Second, it was a single-center, retrospective study, and its generalization is limited. Third, the detailed concurrent chemoradiation regimen has some heterogeneity and can lead to different clinical results. Third, because of the lack of a unified standard, the cut-off values of biomarkers for prognosis vary in different reports with unknown underlying mechanisms. Further studies are needed to validate the prognostic impact of markers in patients with advanced cervical cancer.
Conclusions
The inflammatory response and coagulating reaction can predict OS in cervical cancer patients. In particular, the PLR and FIB levels evaluated before treatment play a significant prognostic role in advanced cervical cancer. The PLR and FIB levels are inexpensive and readily available biomarkers that could provide additional prognostic information and thus have promising prospects for application. Larger prospective studies with adequate sample sizes are needed to validate this observation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosure
The authors report no conflicts of interest in this work.
Ethics approval and consent to participate
This retrospective study was approved by the Ethics Committee of Shandong Cancer Hospital.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Technology Research and Developmental Program of China (Program Nos. 2022YFC2704400 and 2022YFC2704401).
