Abstract
Background
FOXO3a (previously termed FKHRL1), plays an evolutionarily conserved role in the control of biological process, including DNA damage, apoptosis, and cell cycle regulation. However, the role of FOXO3a in tumors remains controversial. This meta-analysis was conducted to evaluate the prognostic value of FOXO3a expression in patients with solid tumors.
Methods
A systematic literature search of the PubMed, Web of Science, Embase, and Cochrane Library databases was performed. Eligible publications on FOXO3a and cancer prognosis were collected and screened according to the eligibility criteria. The combined odds ratios (ORs) or hazard ratios (HRs) with corresponding 95% confidence intervals (CIs) were used to assess the prognostic value of FOXO3a. Stata 12.0 software was used for statistical analysis.
Results
A total of 4058 patients from 21 articles on a variety of solid tumors were included. Meta-analysis showed that the increased FOXO3a expression level was associated with longer overall survival (HR = 0.62; 95% CI: 0.46–0.85). The pooled ORs indicated high expression level of FOXO3a in tumors was significantly associated with lymph node metastasis (OR = 0.46; 95% CI: 0.30–0.71), TNM stage (OR = 0.37; 95% CI: 0.25–0.54), tumor differentiation (OR = 0.46; 95% CI: 0.26–0.80), distant metastasis (OR = 0.44; 95% CI: 0.32–0.61), and age (OR = 1.28; 95% CI: 1.08–1.51). However, we did not observe a significant correlation between the high expression of FOXO3a and sex or tumor size.
Conclusions
The high expression level of FOXO3a was associated with better clinical outcomes in solid tumors. FOXO3a may therefore serve as a potential prognostic biomarker and a promising molecular target.
Introduction
Cancer has become the most common disease and a serious threat to human life. Approximately 28.4 million new cancer cases, with a 47% increase from 19.3 million in 2020, are estimated to occur in 2040. 1 Estimated by the World Health Organization (WHO) in 2019, cancer ranked first or second as the leading cause of human death before age of 70 in 112 of 183 countries, partly indicating the declining mortality rates of stroke and coronary heart diseases. 2 Although new treatments and early screening have made great developments, the prognosis of cancer has not been improved significantly. Moreover, the incidence of cancer in developed countries has increased gradually in recent years. 3 Therefore, there is an urgency to identify novel prognostic biomarkers and molecular targets for human solid tumors.
FOXO3a, also named as FOXO3 or forkhead in rhabdomyosarcoma-like 1 (FKHRL1), is a member of forkhead box class O (FOXO) transcription factor family and originally identified in human placental cosmid. The FOXO3a gene is located on chromosome 6 (6q21).4,5 FOXO3a is a key transcription factor that mediates a variety of physiological and pathological processes, including apoptosis, 6 proliferation, 7 cell cycle progression, 8 survival, 9 and DNA damage. 10 In addition, FOXO3a is closely related to human longevity. 11 The multiple functions of FOXO3a showed that deregulation in FOXO3a expression and/or activity can lead to various diseases, in particular with cancer. 12
The prognostic results of FOXO3a in tumors are still conflicting. A growing number of studies have shown that FOXO3a, as a key mediator of tumorigenesis, can be used as a potential biomarker for the diagnosis and treatment of multiple malignant tumors. Many studies also reported that the deregulation of FOXO3a is associated with a series of malignancies, indicating its association with cancer prognosis. Some studies showed that the level of FOXO3a expression acted as a prognostic biomarker in multiple cancers. 13 Overexpression of FOXO3a is associated with poor prognosis in triple-negative breast cancer, 14 hepatocellular carcinoma, 15 glioblastoma, 16 and upper tract urothelial carcinoma. 17 IT is also correlated with unfavorable clinicopathological features such as advanced tumor node metastasis (TNM) stages, histological grades, and vessel invasion. In addition, tumors with highly expressed FOXO3a appear to be more prone to metastasis. However, the role of FOXO3a in tumors is controversial; several studies have shown that low expression of FOXO3a is associated with poor prognosis in glioma 18 and ovarian cancer 19 patients. Therefore, we conducted this meta-analysis to systematically assess the potential prognostic value of FOXO3a in solid tumors.
Methods
Literature search
This meta-analysis was performed based on the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement. 20 A comprehensive literature search was conducted in the Web of Science, PubMed, Embase, and Cochrane library databases, with a deadline of November 2021 to identify eligible studies. The keywords for the search included (“Forkhead box O3a” OR “FOXO3a” OR “Forkhead in rhabdomyosarcoma-like 1” OR “FKHRL1” OR “Forkhead box O3” OR “FOXO3” OR “AF6q21 protein”) AND (“cancer” OR “carcinoma” OR “neoplasm” OR “tumor”) AND (“prognosis” OR “survival”). Additional relevant articles were retrieved by manually examining the reference lists of the included articles.
Study selection
The inclusion criteria for this meta-analysis were: (a) the study investigated the role of FOXO3a in the development of human solid tumors; (b) associations of FOXO3a expression with prognosis were depicted; (c) the expression level of FOXO3a in human solid tumor tissues was determined by immunohistochemistry (IHC); (d) the patients were separated into high and low FOXO3a expression groups; (e) the study reported available data for calculating odds ratio (OR) with the corresponding 95% confidence interval (CI); and (f) only the most recent study or the study with most comprehensive data was included if the authors had published multiple articles with overlapping sample data from the same institutions. The exclusion criteria were as follows: (a) duplicate publications; (b) cell-lines, animal experiments, or studies lacking valuable/sufficient data; (c) case reports, reviews, letters, comments, expert opinions, or conference abstracts; and (d) only the serum levels of FOXO3a were determined.
Date extraction and quality assessment
The name of first author; publication year; country; cancer type; total number of patients; outcome measures; the criteria for high FOXO3a expression; detection method; clinical features such as tumor size, TNM stage, lymph node metastasis, distant metastasis, age and sex; follow-up period; hazard ratio (HR) and corresponding 95% CI were collected from each study. If a study provided the results of both univariate and multivariate analysis, then only the latter was applied directly because it improved the accuracy of interpreting confounding factors. Any study that reported Kaplan–Meier curves only but did not provide multivariate data was excluded. Disagreements were resolved by consensus from all authors. Two investigators (LZ and CW) performed quality evaluation independently for all eligible studies, and any discrepancies were resolved through discussion until consensus was achieved. The Newcastle–Ottawa Quality Assessment Scale (NOS) tool, 21 which scores from 0 to 9, was used to evaluate the quality of the studies. Studies with an NOS score ≥ 6 were designated as high quality, and studies with low NOS scores (scores 1–5) were deemed as low quality.
Statistical analysis
Stata SE12.0 (Stata Corp., College Station, TX, USA) was used to perform the meta-analysis. We evaluated the heterogeneity across the included studies using the Chi-squared-based Q test and I2 statistic. 22 A P-value of less than 0.05 for the Q test and an I2 value of more than 50% were considered to indicate significant heterogeneity. For studies with no obvious heterogeneity (Ph > 0.05, I2 < 50%), the fixed-effects model was adopted, and the random-effects model was applied for others (Ph ≤ 0.05, I2 ≥ 50%). Begg’s and Egger’s tests 23 were used to assess potential publication bias. The stability of the results was assessed using sensitivity analysis. Differences with P-values of less than 0.05 were considered statistically significant.
Results
Study characteristics
The flowchart for study selection in this meta-analysis is presented in Figure 1. The literature retrieval process obtained 21 eligible articles,14,15,17,19,24–40 encompassing 4058 cancer patients, published from 2008 to 2021. The mean of sample size was 193, ranging from 60 to 934 patients. These studies were conducted in Asia (n = 20) and Europe (n = 1). Studies involving 13 different solid tumor types were evaluated, including 3 on gastric cancer, 5 on breast cancer, 2 on ovarian carcinoma, 2 on hepatocellular carcinoma, and 1 each on nasopharyngeal carcinoma, salivary gland adenoid cystic carcinoma, esophageal cancer, bladder cancer, glioma, pancreatic ductal adenocarcinoma, lung adenocarcinoma, extrahepatic cholangiocarcinoma, and upper tract urothelial carcinoma. All cancerous specimens were well preserved and the studies relied on pathology findings to make a diagnosis. The quality of the studies included in this meta-analysis varied from 5 to 9, with a mean value of 6.5. The basic characteristics of the eligible studies are summarized in Table S1.
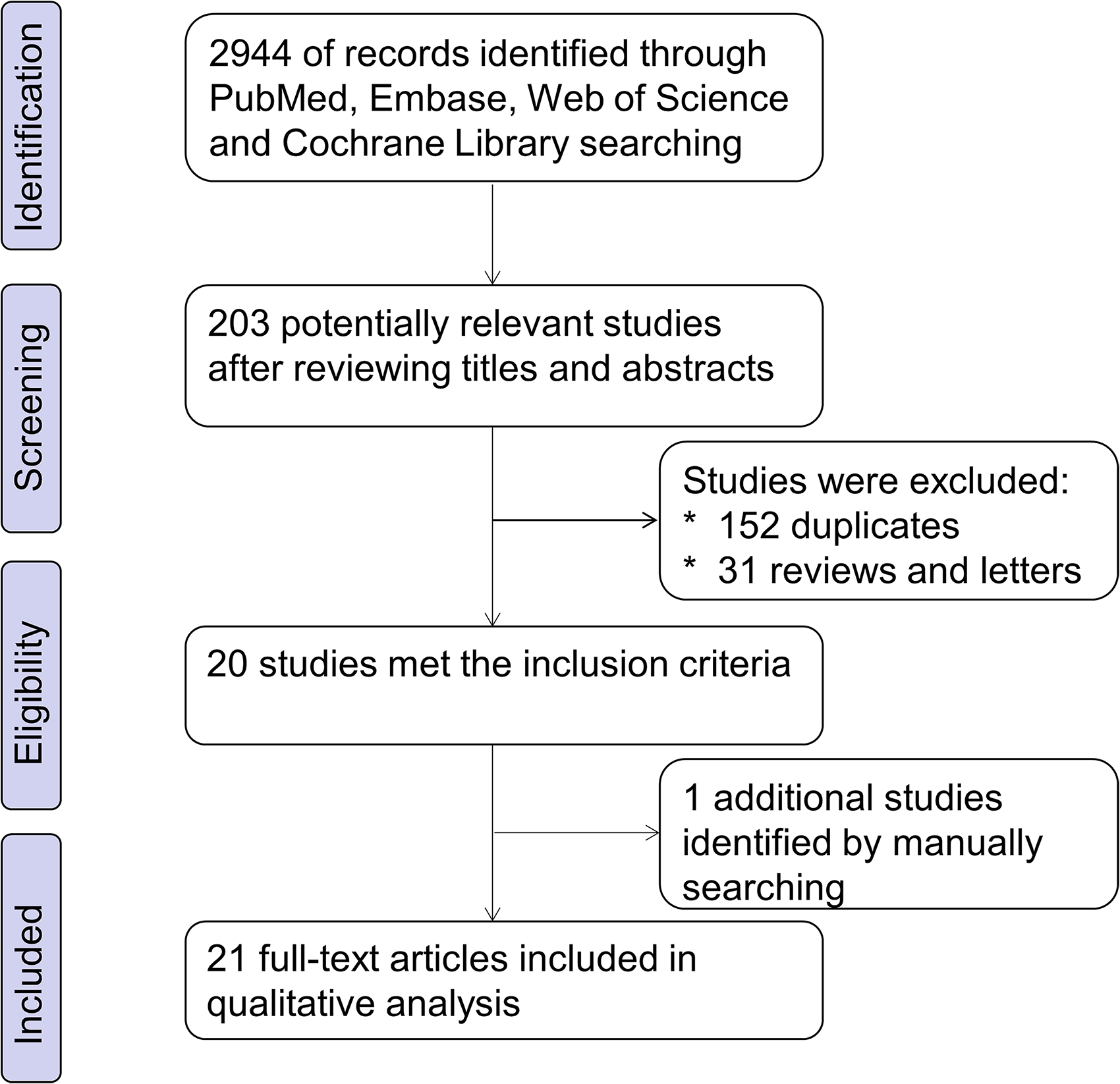
Flowchart representation of the steps of the literature search and selection in this meta-analysis.
Prognostic role of FOXO3a expression
The relationship between overall survival (OS) and FOXO3a expression was reported in 17 articles. We adopted the random-effects model to estimate the pooled HR and corresponding 95% CI. There was obvious heterogeneity across studies (I2 = 79.9%, Ph < 0.001). The HR for the high expression group versus the low expression group was 0.62 (95% CI: 0.46–0.85) (Figure 2(a)). After stratification by study type, a significant association was observed between enhanced expression of FOXO3a and poor OS with HR = 0.61 in retrospective studies (95% CI: 0.42–0.89) and HR = 0.67 in prospective studies (95% CI: 0.40–1.12) (Figure 2(b)). After stratification by prognosis, the HRs for the high versus the low FOXO3a expression group were 0.44 (95% CI: 0.32–0.61) in good prognostic group, 1.36 (95% CI: 1.01–1.82) in no impact group and 2.14 (95% CI: 1.17–3.94) in harm prognostic group (Figure 2(c)). Moreover, a significant association was found between higher expression of the FOXO3a and better OS in surgery (S) (HR = 0.67, 95% CI: 0.48–0.92), compared with the surgery plus chemotherapy (C), plus endocrine therapy (E) (S + C + E) (HR = 0.39; 95% CI: 0.20–0.76), and surgery plus chemotherapy plus radiotherapy (S + C + R) (HR = 0.36; 95% CI: 0.16–0.78) (Figure 2(d)). A significantly longer OS was observed in patients with high FOXO3a expression versus those with low FOXO3a expression. Thus, we concluded that high expression of FOXO3a was associated with better OS.

Forest plots of HR for the relationship between high FOXO3a expression and OS. (a) The relationship between FOXO3a expression and OS in 21 articles included in this study. (b) The relationship between FOXO3a expression and OS in retrospective studies and prospective studies. (c) The relationship between FOXO3a expression and OS according to good prognosis, no impact, harmful prognosis studies. (d) The relationship between FOXO3a expression and OS in surgery, surgery plus chemotherapy plus endocrine therapy (S + C + E) and surgery plus chemotherapy plus radiotherapy (S + C + R).
Correlations between FOXO3a expression with clinical and pathological features
The pooled results (Table S2) indicated that increased FOXO3a was significantly associated with lymph node metastasis (OR = 0.46; 95% CI: 0.30–0.71) (Figure 3(a)), TNM stage (OR = 0.37; 95% CI: 0.25–0.54) (Figure 3(b)), tumor differentiation (OR = 0.46; 95% CI: 0.26–0.80) (Figure 3(c)), distant metastasis (OR = 0.44; 95% CI: 0.32–0.61) (Figure 3(d)), and age (OR = 1.28; 95% CI: 1.08–1.51) (data not shown). However, no significant correlation was observed between increased FOXO3a expression and tumor size and sex (data not shown). We failed to detect a relationship between overexpression of FOXO3a and other clinical and pathological parameters due to insufficient data.

Forest plots of HR for the relationship between high FOXO3a expression and clinicopathological parameters. (a) The relationship between FOXO3a expression and lymph node metastasis. (b) The relationship between FOXO3a expression and TNM stage. (c) The relationship between FOXO3a expression and tumor differentiation. (d) The relationship between FOXO3a expression and distant metastasis.
Sensitivity analysis
Sensitivity analysis for the meta-analysis of the association between the FOXO3a expression level and OS was performed by sequentially removing individual studies from the analysis. The purpose of this process was to assess the impact of each deleted data set on the overall HRs. The final result was not significantly affected by the exclusion of any study individual, demonstrating the robustness of the results (Figure 4).
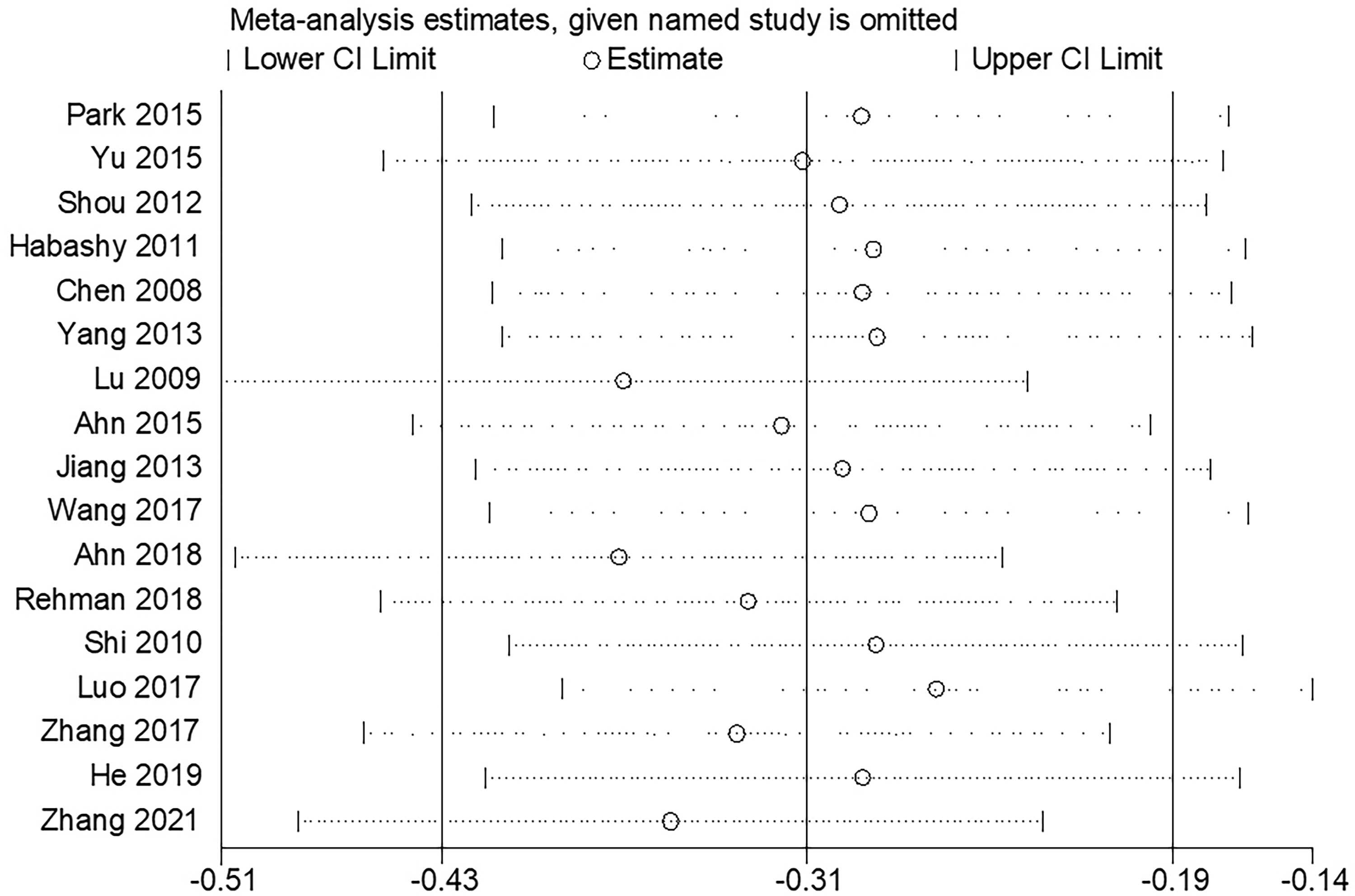
Sensitivity analysis of studies regarding OS.
Publication bias
To corroborate the meta-analysis of the correlation between FOXO3a expression level and OS, publication bias was tested using Begg’s and Egger’s tests. The results indicated that there was no publication bias among the included studies (Figure 5).
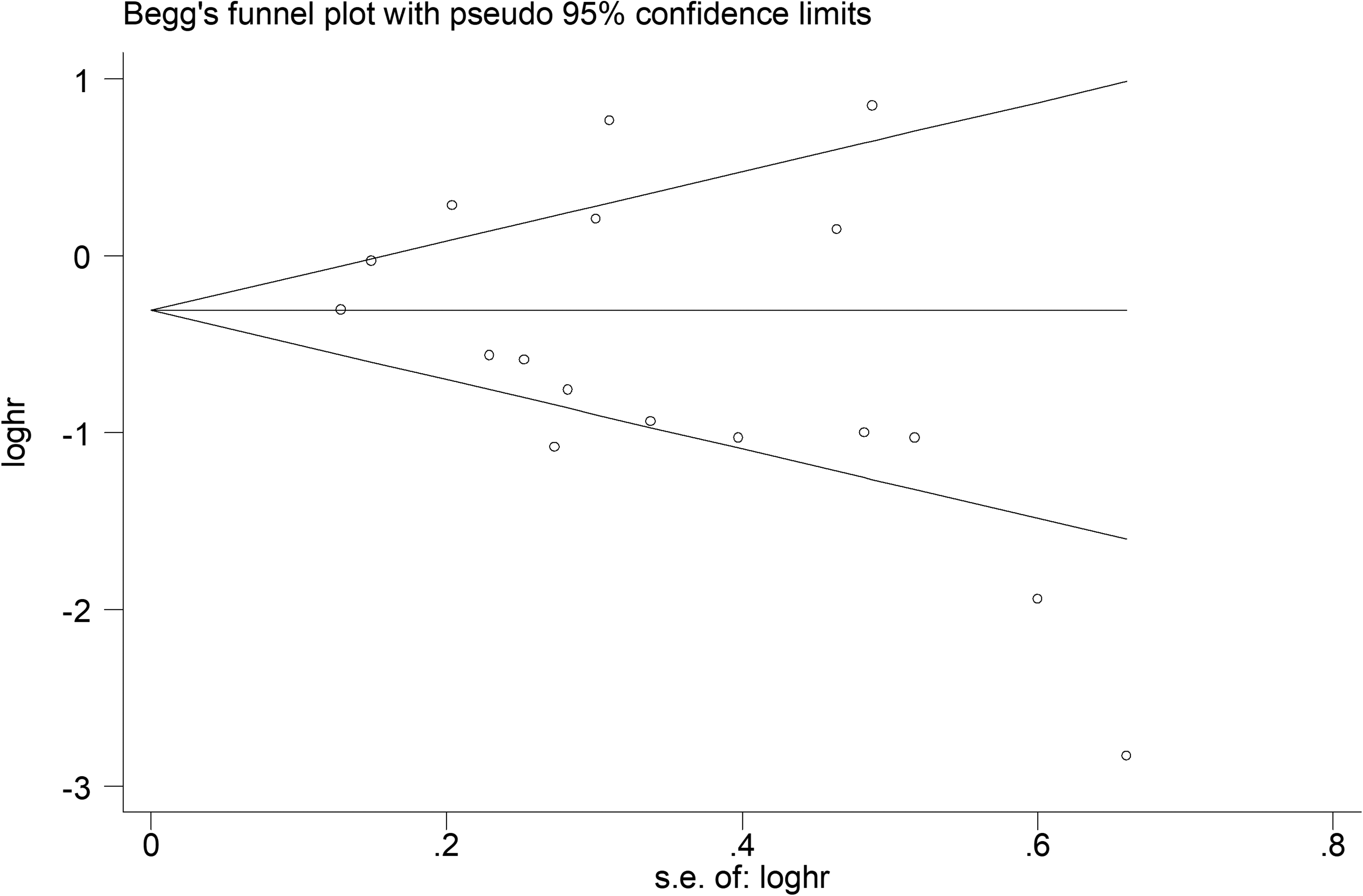
Results of the analysis of publication bias for OS.
Discussion
FOXO3a is a crucial regulator in various physiological and pathological processes by directly inducing or mediating the expression of genes related to cell proliferation, growth, and survival. 41 The disorder of FOXO3a contributes to the development and progress of many diseases including cancer. Post-translational modifications of FOXO3a can regulate its transcriptional activity and nuclear location, enabling it as a potential target for novel strategy in cancer treatment.42,43 Some studies reported that high expression of FOXO3a was associated with improved OS in patients with solid tumor. However, other studies indicated that the high expression of FOXO3a was related to poor OS in comparable patients. 13 Although the inconsistencies among these findings were not explained in detail, the differences may be, in part, due to the fact that the tested samples were from individual tumor tissues or that FOXO3a functions differently in divergent tumor origins. Cytoplasmic (inactive FOXO3a) and nuclear (active FOXO3a) expression of the transcription factor among different types or subtypes of the same type of cancer, which seems to have a protective role in estrogen receptor positive, luminal A breast cancer, but associates with poor prognosis in triple negative breast cancer.14,26,28,44,45 Regulation of FOXO3a expression and localization plays a significant role in homeostasis, involving posttranslational modifications like phosphorylation, acetylation/deacetylation as well as DNA binding efficiency and downstream gene activations. 46 In addition, small differences in the design of experiments in the studies may also have affected the observations. It is worth noting that an insufficient number of tumor samples in a study can result in limited statistical efficacy and reduce the apparent prognostic value of FOXO3a. In this meta-analysis, we assessed the relationship between survival and FOXO3a expression in patients with solid tumors. The result showed that high expression of FOXO3a was associated with longer OS of patients with solid tumor, suggesting that FOXO3a may be a potential prognostic biomarker and molecular target for solid tumors.
The present meta-analysis had some limitations. First, there was significant heterogeneity of FOXO3a and OS, but the source cannot be identified by subgroup analysis. FOXO3a might act differently in different tumor types/subtypes depending on the cellular context as well as the transcriptional machinery involved. Second, due to the relatively small sample size in total, we could not analyze the relationship between FOXO3a expression and the other clinical and pathological parameters, except lymph node metastasis, TNM stage, tumor differentiation, distant metastasis, age, tumor size, and sex. Third, the number of included studies is small and the statistical strength of the meta-analysis is therefore limited. Although the results indicated that there was no publication bias, the limited number of studies that could be included may still cause the possibility of secondary selection bias. In addition, the methods, platforms, and judgment criteria for IHC testing were inconsistent. Various sample sources and different types of diseases may also lead to deviations in the results of this meta-analysis. Moreover, most of the studies included in the meta-analysis were conducted in Asia, which may limit the applicability of our conclusions. Finally, the prognostic value of the combination of FOXO3a and other tumor biomarkers has not been evaluated. Therefore, further large multicenter studies with higher quality and uniform criteria, as well as specific location (cytoplasmic or nuclear expression) in divergent tumor types/subtypes for determining the prognostic role of FOXO3a expression, are necessary for validating our findings.
Conclusions
In this study, a meta-analysis was used to evaluate the prognostic role of FOXO3a in solid tumors. Our results suggest that FOXO3a may be a useful prognostic biomarker and molecular target. However, further studies are still required to determine the potential impact of FOXO3a in different solid tumors.
Footnotes
Availability of data and materials
The datasets used and/or analyzed in this study are available from the corresponding author on reasonable request.
Authors’ contributions
LZ, CW, and PPJ participated in the collection and analysis of data. XHT, YFJ, YCX, and LZ performed the statistical analyses. LZ, CW, and XHD conceived the study and designed the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Funding
The research received grants from the Natural Science Foundation of Jiangxi Province (grant number 20202BABL206076) and the Doctoral Research Fund of Ganzhou People’s Hospital (grant number BSQD2020008).
