Abstract
Aim
Post-traumatic headache (PTH) attributed to mild traumatic brain injury (mTBI) frequently co-occurs with other physical, cognitive, and emotional symptoms. This study assessed associations between PTH improvement and these symptoms in individuals with acute headaches (APTH) and compared symptoms between those who had recently developed persistent PTH (PPTH) to those who had long-standing PPTH.
Methods
This study included 105 individuals with APTH (mean age = 43.4, SD = 15.8; 70 females, 35 males) and 46 individuals with PPTH (mean age = 38.2, SD = 10.9; 16 females, 30 males) with an average duration of 11.3 years from two prospective longitudinal studies. For those who had APTH, PTH improvement was assessed at three months post-enrollment using an electronic headache diary. Participants completed questionnaires evaluating physical (including headache symptoms, photosensitivity, hyperacusis, insomnia, autonomic symptoms, and disability), emotional (including depression, anxiety, pain catastrophizing, and post-traumatic stress disorder screening), and cognitive symptoms (including memory and attention). The APTH cohort completed questionnaires at baseline, follow-up #1 (three to four months from baseline), and follow-up #2 (six to seven months from baseline). Mixed Effects Models were used to analyze the temporal trend of symptoms among the APTH improved and non-improved groups.
Results
Among the 105 individuals with APTH, 60 experienced PTH improvement and 45 did not. At baseline, compared to those who had PTH improvement, those who did not have PTH improvement had higher SCAT total scores and physical subscores, greater levels of hyperacusis, pain catastrophizing, anxiety and depression. At follow-up #1 (three to four months), these individuals continued to show more pronounced physical and emotional symptoms and worse cognitive function and insomnia. Most of these group differences persisted at follow-up #2 (six to seven months). Most symptoms showed progressive improvement over three months for individuals who had PTH improvement but not for those without PTH improvement. Furthermore, individuals with long-standing PPTH showed more severe insomnia, pain catastrophizing, and PTSD compared to those who had recently developed PPTH.
Conclusion
Individuals with APTH who did not experience PTH improvement at three months had more severe physical, cognitive, and emotional symptoms at baseline compared to those who had PTH improvement. While most symptoms normalized in individuals with PTH improvement, symptoms overall persisted at three and at six months for those with PTH persistence. Individuals who had long-standing PPTH had more severe insomnia and more negative cognitive/emotional responses to pain compared to those who had recently developed PPTH.
This is a visual representation of the abstract.
Keywords
Introduction
Acute post-traumatic headache (APTH) is a common symptom following mild traumatic brain injury (mTBI). While some individuals experience resolution quickly, others develop persistent PTH (PPTH) lasting months or even years, leading to substantial long-term health burden (1,2). However, PTH is not the only symptom that can significantly affect daily functioning following mTBI. In fact, other symptoms, such as non-headache physical symptoms and cognitive and emotional symptoms frequently co-occur along with APTH (3–5). However, the relationship between these co-occurring post-mTBI symptoms and PTH persistence (or PTH non-improvement) is insufficiently understood.
In this study, we assessed physical, cognitive and emotional symptoms in individuals with APTH following mTBI and tracked PTH outcomes over six months using an electronic headache diary (eDiary). We compared longitudinal symptoms between individuals with APTH improvement and non-improvement across three timepoints, immediately after mTBI (baseline), at three to four months (follow-up #1), and again at six to seven months (follow-up #2), and further compared symptoms of those who have recently developed PPTH to those who had PPTH for over a decade. The goal of this study was to assess whether non-headache physical, emotional, and cognitive symptoms are different over time in individuals who will have PTH improvement compared to individuals who will have persisting PTH (headache non-improvement) at three-months. We further aimed to determine whether individuals who experience PTH improvement at three-months also show more rapid resolution of physical, emotional, and cognitive symptoms over time, compared to those with persisting PTH. Lastly, we assessed whether physical, emotional, and cognitive symptoms are more severe in individuals with long-standing PPTH (on average over a decade) compared to those who have PTH persistence for only three or six months.
Methods
Participant enrollment
This work uses data from two prospective longitudinal studies. The studies were approved by the Mayo Clinic and Phoenix VA Institutional Review Boards and the United States Department of Defense Human Research Protection Office. Individuals with PTH were identified from the Mayo Clinic in Arizona, Minnesota, and Florida, and the Phoenix VA healthcare system. All participants provided written informed consent prior to participation.
Enrollment for the APTH cohort
Enrollment for the APTH cohort occurred between 2019 and 2025. Eligible participants were adults between 18 and 70 years of age. Individuals were excluded for history of moderate or severe TBI and were not excluded for history of migraine prior to mTBI. Individuals were seen at three time-points: baseline (0–59 days post-mTBI), follow-up #1 (three to four months after baseline), and follow-up #2 (six to seven months after baseline visit).
Enrollment for the PPTH cohort
Enrollment for the PPTH cohort occurred between 2014 and 2018. Eligible participants were adults between the ages of 18 and 65. Individuals were excluded for history of moderate and severe TBI and history of migraine prior to injury. Individuals with history of infrequent episodic or tension-type headache were not excluded. The average duration of PTH in the PPTH group was greater than ten years. APTH and PPTH attributed to mTBI were diagnosed according to the most recent version of the International Classification of Headache Disorders available at the time of participant enrollment (ICHD-3 beta or ICHD-3) (6,7).
A subset of individuals with PTH included in this study were included in prior publications (8,9) but this study was the first to assess the longitudinal progression of physical, cognitive and emotional symptoms in individuals with short-term and long-standing PTH.
Questionnaires
All individuals completed a comprehensive list of questionnaires assessing physical, emotional and cognitive symptoms following mTBI. Headache and TBI characteristics were collected using case report forms containing Common Data Elements developed by the National Institute of Neurological Disorders and Stroke including a modified PTH headache symptom questionnaire (10). TBI history was confirmed using the Ohio State University TBI identification method, a standardized questionnaire assessing the lifetime history of TBI for an individual (11).
Physical symptoms were assessed using the following validated questionnaires: Traumatic brain injury-related symptoms: Sports Concussion Assessment Tool – 5th Edition (SCAT-5) (12), including total score, number of symptoms, and physical subscore. Sound sensitivity: Hyperacusis Questionnaire (12), total score. Light sensitivity: Photophobia Assessment Questionnaire (PAQ) (13), photophobia subscore. Headache-related disability: Migraine Disability Assessment Scale (MIDAS) (14), total score. Sleep disturbances: Insomnia Severity Index (ISI) (15), total score. Autonomic symptoms: Autonomic Symptom Questionnaire (COMPASS-31) (16), total score. Post-traumatic stress symptoms: PTSD Checklist (PCL-5) (17).
Emotional symptoms were assessed using the following instruments: Depression: Beck Depression Inventory (BDI) (18), total score. Anxiety: State-Trait Anxiety Inventory (STAI) (19), Form Y-1 (state anxiety) and Form Y-2 (trait anxiety), total scores. Pain catastrophizing: Pain Catastrophizing Scale (PCS) (20,21), including total score and subscores for helplessness, magnification, and rumination. Self-reported emotional symptoms: SCAT-5 emotional subscore.
Cognitive symptoms were assessed using the following measures: Memory recall: Rey Auditory Verbal Learning Test (RAVLT) (22), z-scored immediate and delayed recall scores, Attention: Trail Making Test (TMT, Parts A and B) (4), z-scored performance, and self-reported cognitive symptoms: SCAT-5 cognitive subscore.
Outcome definition
All individuals with APTH completed a daily electronic headache diary (eDiary) over the first three months after enrollment and again in the four weeks prior to their follow-up visit, to determine if individuals had headache improvement or non-improvement at follow-up #1 (three to four months) and follow-up #2 (six to seven months). Headache improvement vs. non-improvement (i.e., persistence) was determined at three months post-enrollment using a method that considered an individual's headache history before and after their most recent mTBI, as described in detail in our prior publications (8). Briefly, for individuals with pre-existing headaches before mTBI, improvement was defined as either a 50% or greater reduction in additional headaches (post-TBI headaches minus pre-TBI headaches), or a reduction of additional headaches to 2.5 or fewer at the time of assessment. The determination of whether the additional headaches had been reduced to 2.5 or fewer was made by comparing the number of monthly headaches reported prior to the mTBI with the number recorded during the first month following study enrollment. As natural month-to-month variation in headache frequency is expected (23), an increase of 2.5 or fewer additional headaches was considered to fall within the range of normal fluctuation for individuals with pre-existing headaches. For individuals without pre-TBI headaches, improvement was defined as a 50% or greater reduction in headaches occurring within the first month after enrollment.
Statistical analysis
Longitudinal analysis of symptoms’ association with PTH outcome
This analysis examined changes in physical, cognitive, and emotional symptoms over time in individuals with APTH, comparing those who experienced PTH improvement to those who had PTH persistence at three months. Symptoms were assessed using questionnaires collected across three time-points: at baseline, follow-up #1, and follow-up #2. We used Mixed Effect Models (MEM) (24) to analyze the trend of symptoms over time for different groups of individuals with APTH while controlling for covariates. Specifically, we modeled each symptom (
In this model,
To maximize data utilization, we fit separate MEM models to evaluate symptom changes in individuals with APTH in the first three months post-enrollment and in the later three months post-enrollment. Specifically, we first applied MEM models to each symptom using data collected at baseline and follow-up #1 to compare individuals with vs. without PTH improvement at these two time points, while also analyzing symptom trends over the first three months. Next, we applied MEM models using data from the follow-up #1 and #2 to evaluate symptom trends from three to six months and to compare groups at six months.
The p-value of the coefficients were used to determine the statistical significance of each interaction term in the model based on a significance threshold of 0.05. To account for multiple comparisons, Holm-Bonferroni correction was applied to model coefficients with significant p-values to control the family-wise error rate.
Symptom comparison between groups with varying duration of PTH
In this analysis, we investigate symptom differences between those who had long-standing PPTH (on average for over a decade) to individuals in the APTH cohort who had not shown improvement at three or six months. Mixed Effect Models cannot be used in this analysis as longitudinal data was not collected for the PPTH cohort. Instead, each symptom was analyzed using a linear regression model modelling the symptom as the dependent variable and group (long-standing PPTH vs. APTH with three or six-month of PTH persistence) as the independent variable, while controlling for the covariates of age, sex, and elapsed time from mTBI injury to enrollment. Holm-Bonferroni correction was not applied in this analysis due to the smaller sample size of the groups being compared (25). Instead, we used a more stringent significance threshold of 0.01.
We compared the demographics and headache characteristics of the PTH groups using two-sided t-tests for continuous variables and chi-square tests for binary variables. All analyses were performed using Python release 3.11.0 (Python Software Foundation, Wilmington, DE, USA). Statistical testing was conducted with the scipy package (26), MEM and regression modelling with the statsmodels package (27).
Results
One hundred and five participants with APTH due to mTBI (mean age = 43.4, SD = 15.8; 70 females, 35 males) were enrolled on average 31 days post-mTBI (SD = 15.1), with six individuals (6%) enrolled within seven days post-mTBI, nine individuals (9%) enrolled between eight to 14 days, 39 individuals (37%) enrolled between 15 to 30 days, and 32 individuals (30%) enrolled between 31 to 45 days, and 19 individuals (18%) enrolled more than 45 days from injury. Onset of APTH occurred within seven days of injury in all individuals. Based on the outcome assessment at three months post-enrollment, 60 participants with APTH had PTH improvement (mean age=44.2, SD = 16.0; 38 females/22 males) and 45 had PTH non-improvement (mean age=42.3, SD = 14.4; 32 females/13 males). Re-assessment at six months post-enrollment showed that 43 participants with APTH had PTH improvement (mean age=43.4, SD = 15.0; 27 females/16 males), 24 had headache non-improvement (mean age=49.2, SD = 13.9; 13 females/11 males), 37 individuals did not provide enough information to determine their six month outcome, and one individual was still participating in the study and did not yet complete their sixth-month headache diary. There were no significant differences in years of education at baseline between individuals with PTH improvement vs. non-improvement. Sixty individuals in the APTH group had a single lifetime mTBI and 45 individuals had at least one prior mTBI. In terms of mechanism of mTBI, 40 individuals had TBI due to falls, 30 due to motor vehicle accidents, five due to sports-related injuries, seven due to direct impact blow to head, 11 due to direct impact head against object, two due to acceleration/deacceleration, and 10 due to other causes. The headache phenotype acutely following mTBI among the APTH group was as follows: 31 individuals had tension-type headache, 42 had probable migraine, and 32 had migraine-like headache. Among those with migraine-like headache 12 individuals reported migraine with aura.
The PPTH cohort included 46 participants with PPTH with an average duration of 11.3 years (mean age = 38.2, SD = 10.9; 16 females/30 males). Among these PPTH participants, 27 individuals had a single lifetime mTBI and 19 individuals had at least one prior mTBI. Twenty two individuals had TBI due to a blast, 11 due to falls, eight due to sports-related injuries, and five due to motor vehicle accidents. The headache phenotype of those with PPTH were: one individual had tension-type headache, nine had probable migraine, and 36 had migraine-like headache. Among the PPTH individuals with migraine-like headaches, 18 reported migraine with aura. Migraine-like headache was more common in the cohort with PPTH as compared to the cohort with APTH. While only 11.4% of individuals with APTH reported migraine with aura following mTBI, 39% of those with PPTH reported migraine with aura.
Seven individuals (7%) with APTH and 29 individuals (63%) with PPTH were enrolled from the Phoenix VA Healthcare System. Of those, four (57%) with APTH and 21 (72%) with PPTH had migraine-like headache. PTSD was more common among participants enrolled from the Phoenix VA Healthcare System, with four individuals (57%) with APTH and 24 (83%) individuals with PPTH. Besides military background, PTSD also differed by mechanism of injury. Among individuals with APTH, 23 (22%) had PTSD with the most common mechanisms of injury being motor vehicle accidents (n = 9) and falls (n = 6). In the PPTH group, 30 (65%) develped PTSD with the most common mechanism of injury being blast-type injuries (n = 20).
Table 1 summarizes the demographics of the groups being compared in the subsequent analysis. The group average of physical, emotional, and cognitive symptoms scores from the 12 questionnaires collected across three visits for individuals in the APTH cohort are summarized in Table 2.
Demographics and baseline headache characteristics of different PTH groups: (i) individuals from the APTH cohort with PTH improvement at three months, (ii) individuals from the APTH cohort with PTH persistence at three months, (iii) individuals from the APTH cohort with PTH persistence at six months, and (iv) individuals with long-standing PPTH.

History of prior TBIs = Traumatic Brain Injuries sustained before the current injury associated with PTH (yes/no); TTH = Tension-type headache; PM = probable migraine-like headache; M = migraine-like headache.
Group comparison between (i) and (ii).
Group comparison between (ii) and (iv).
Group comparison between (iii) and (iv).
Years of Education was not collected for those with PPTH.
Average scores of physical, emotional, and cognitive symptoms from 12 questionnaires collected across three visits (baseline, follow-up #1, follow-up #2) for individuals in the APTH cohort who had PTH improvement (group 1, N = 60) and who had PTH non-improvement (group 2, N = 45) at three months at three months.

During the COVID-19 pandemic, a phone visit was conducted in place of an in-person visit, resulting in 17 individuals with APTH not completing the TMT assessments at their follow-up #1 and 30 individuals not completing it at their follow-up #2. In addition, 41 individuals were not administered the RAVLT, MIDAS, and COMPASS tests. At baseline, one individual with APTH did not complete the ISI questionnaire. At follow-up #1, one individual with APTH did not complete any questionnaires, three individuals did not complete the BDI questionnaire, one individual did not complete the COMPASS-31. 35 individuals from the APTH cohort did not complete their follow-up #2 visit by the time of the analysis. Additionally, nine individuals did not complete a portion of the questionnaires at follow-up #2. The MEM models for variables with missing data were fitted using the available data, excluding the participants with missing values for the corresponding outcome.
Symptom comparison between individuals with APTH with and without PTH improvement across three visits
Group differences in symptom burden (physical, emotional, and cognitive, respectively) between individuals with PTH who experienced PTH improvement and those who did not have PTH improvement at three months across three visits are visualized in Figures 1, 2 and 3. All reported p-values reflect Bonferroni-corrected results.
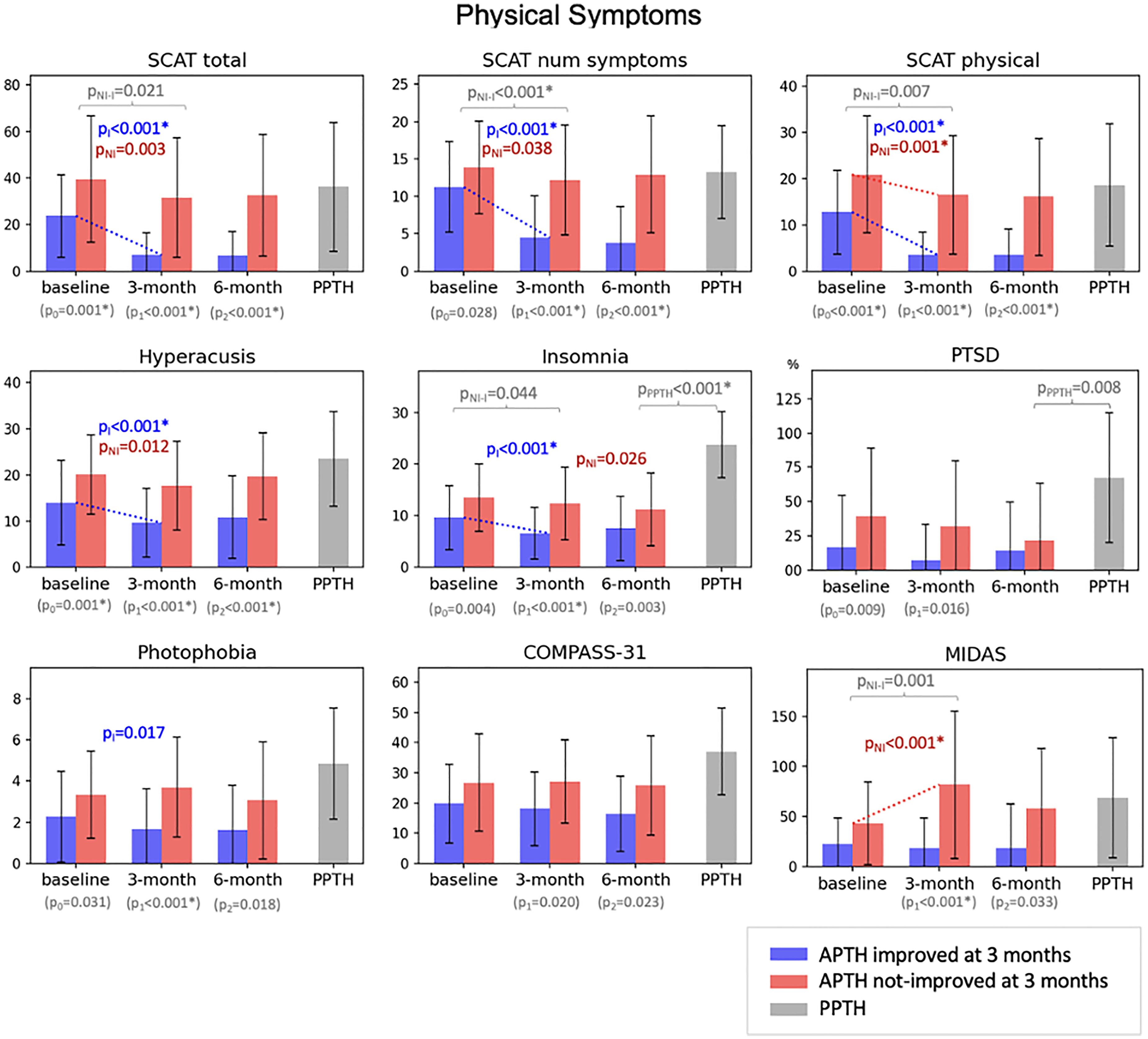
Comparison of physical symptoms between APTH individuals with PTH improvement at three months (blue), those with PTH non-improvement at three months (red), and those with longstanding PPTH (gray). P-values are shown for statistically significant comparisons with * indicating significance after multiple testing correction: p0, p-value of the comparison between red and blue at baseline; p1, p-value of the comparison between red and blue at the follow-up #1; p2, p-value of the comparison between red and blue at the follow-up #2; pNI, p-value of the symptom change from baseline to three months for red; pI, p-value of the symptom change from baseline to three months for blue; pNI-I, p-value of the difference in symptom change from baseline to three months between red and blue; pPPTH, p-value of the comparison between red at the six months and gray. All test results are based on Mixed Effect Models except pPPTH is based on linear regression model.

Comparison of emotional symptoms between APTH individuals with PTH improvement at three months (blue), those with PTH non-improvement at three months (red), and those with longstanding PPTH (gray). P-values are shown for statistically significant comparisons with * indicating significance after multiple testing correction: p0, p-value of the comparison between red and blue at baseline; p1, p-value of the comparison between red and blue at the follow-up #1; p2, p-value of the comparison between red and blue at the follow-up #2; pNI, p-value of the symptom change from baseline to three months for red; pI, p-value of the symptom change from baseline to three months for blue; pNI-I, p-value of the difference in symptom change from baseline to three months between red and blue; pPPTH, p-value of the comparison between red at the six months and gray. All test results are based on Mixed Effect Models except pPPTH is based on linear regression model.

Comparison of cognitive symptoms between APTH individuals with PTH improvement at three months (blue), those with PTH non-improvement at three months (red), and those with longstanding PPTH (gray). P-values are shown for statistically significant comparisons with * indicating significance after multiple testing correction: p0, p-value of the comparison between red and blue at baseline; p1, p-value of the comparison between red and blue at the follow-up #1; p2, p-value of the comparison between red and blue at the follow-up #2; pNI, p-value of the symptom change from baseline to three months for red; pI, p-value of the symptom change from baseline to three months for blue; pNI-I, p-value of the difference in symptom change from baseline to three months between red and blue; pPPTH, p-value of the comparison between red at the six months and gray. All test results are based on Mixed Effect Models except pPPTH is based on linear regression model.
Baseline
At baseline, individuals with APTH who did not have PTH improvement at three months exhibited more severe physical, emotional, and cognitive symptoms compared to those who improved. The non-improvement group had significantly higher SCAT scores (total score, p = 0.001), SCAT physical (p < 0.001) subscores and more severe hyperacusis (p = 0.001). The non-improvement group also had higher levels of pain catastrophizing (PCS total score, p = 0.001) anxiety (TRAIT, p < 0.001), depression (BDI, p < 0.001), as well as self-reported mood symptoms (SCAT emotional subscore, p < 0.001). For clinical interpretation, mean levels of depression symptoms remained below the threshold for clinical significance for those with headache improvement (normal range) and were slightly elevated indicating mild depression for those with headache persistence. In terms of cognitive function as measured by the RAVLT or the TMT, mean scores for those with PTH improvement and non-improvement were in the normal/average range and there were no differences at baseline in those who had PTH improvement compared to those with PTH persistence at three months. Mean scores on the Insomnia symptom index revealed mild/subthreshold insomnia in individuals both with and without headache improvement.
Follow-up visit #1
Individuals who did not have PTH improvement at three months continued to show worse physical, emotional and cognitive symptom scores. Physical symptoms included higher SCAT scores (total score, p < 0.001; number of symptoms, p < 0.001; physical subscore, p < 0.001), greater symptoms of hyperacusis (p < 0.001), insomnia (p < 0.001), photophobia (p < 0.001), and higher headache-related disability (MIDAS score p < 0.001). Emotional symptom scores included higher levels of pain catastrophizing (PCS total, p < 0.001; helplessness, p < 0.001; magnification, p < 0.001; rumination, p < 0.001) greater self-reported emotional symptoms (SCAT emotional subscore, p < 0.001), higher levels of anxiety (TRAIT, p < 0.001; STATE, p < 0.001) and depression (BDI, p < 0.001) although mean BDI scores for individuals with and without headache improvement were in the average/normal range. Those who did not have headache improvement at three-months reported worse cognitive functioning (SCAT cognitive subscore, p < 0.001). For clinical interpretation, mean scores on the Insomnia symptom index revealed no insomnia in individuals with headache improvement and mild/subthreshold insomnia for those with headache non-improvement.
Follow-up visit #2
Most of these symptoms continue to show group differences at six to seven months, with individuals who did not have PTH improvement at three-months continuing to report more overall symptoms including higher SCAT scores (total symptoms, p < 0.001; number of symptoms, p < 0.001; physical symptoms, p < 0.001), more severe symptoms of hyperacusis (p < 0.001), and higher levels of depression (BDI, p < 0.001), anxiety (TRAIT, p < 0.001), and pain catastrophizing (PCS total score, p < 0.001; PCS helplessness, p = 0.001; PCS magnification, p = 0.001; PCS rumination, p = 0.001) compared to those who had PTH improvement at three-months. The report of worse cognitive symptoms persisted for those who did not have PTH improvement (SCAT cognitive subscore, p < 0.001). For clinical interpretation, mean scores on the Insomnia symptom index revealed no insomnia in individuals with headache improvement and mild/subthreshold insomnia for those with headache non-improvement.
Symptom trend for individuals with APTH
For individuals who had PTH improvement at three months, most symptoms showed significant improvement trend at follow-up visit #1, including physical symptoms measured by SCAT (SCAT total score, p < 0.001; SCAT number of symptoms, p < 0.001; SCAT physical, p < 0.001), symptoms of hyperacusis (HQ, p < 0.001) and insomnia (ISI, p < 0.001), emotional symptoms including depression (BDI, p < 0.001), anxiety (TRAIT, p = 0.002; STATE, p < 0.001), pain catastrophizing (PCS, p < 0.001; helplessness, p < 0.001; magnification, p < 0.001; rumination, p < 0.001), self-reported emotional symptoms (SCAT emotional subscore, p < 0.001), as well as cognitive function (TMT, p = 0.003; SCAT cognitive, p < 0.001). However, for individuals whose PTH persisted at three months, no symptoms significantly improved at the follow-up visits #1 or #2 except for a reduction in the SCAT physical subscore (p = 0.001). Moreover, the headache-related disability worsened over time for those without PTH improvement (MIDAS, p < 0.001).
The trend comparison analysis showed that patients who had PTH improvement had a significantly faster improvement on the SCAT number of symptoms in the first three months compared to those who had PTH persistence (p < 0.001). Neither group exhibited any significant trend changes in physical, emotional, and cognitive symptoms from three months to six months post-enrollment.
Symptom comparison between recent vs. long-standing PPTH
Amongst 105 participants from the APTH cohort, 45 individuals had PTH non-improvement at three months, and 24 had PTH non-improvement when re-assessed at six months. Table 3 summarizes the group comparison of physical, emotional, and cognitive symptoms between individuals in the APTH cohort who experienced PTH for three months or six months and individuals in the PPTH cohort who experienced long-standing PTH. Compared to those who had persisting PTH for only three months so far, individuals with long-standing PPTH had more severe symptoms of insomnia (p < 0.001), pain catastrophizing (total score, p < 0.001; helplessness subscore, p < 0.001; rumination subscore, p = 0.002), and reported more PTSD (p = 0.001). Similarly, compared to those in the APTH group who did not have PTH improvement at six months, individuals with decade long PPTH had more severe symptoms of insomnia (p < 0.001) and PTSD symptoms (p = 0.008). Mean scores on the ISI and the BDI revealed severe clinical insomnia and mild depression for those with decade long PPTH.
Group comparison between individuals in the APTH cohort who experienced PTH for three months or six months and individuals in the PPTH cohort who experienced long-standing PTH.

P-values obtained from linear regression models controlling for age, sex, and elapse time from injury to enrollment. Significant one-sided p-values according to the 0.01 threshold are bolden.
Discussion
In individuals with APTH following mTBI, those who did not have PTH improvement at three months had significantly greater symptom burden immediately following mTBI (i.e., at baseline) across physical, emotional, and cognitive domains as compared to those who had PTH improvement. Longitudinal analyses revealed that individuals with PTH improvement showed significant recovery for most physical and emotional symptoms between baseline and three months. In contrast, individuals with non-improving PTH had limited improvement of the same symptoms, with significant longitudinal change observed only for self-reported physical symptoms from baseline to three months whereas headache-related disability (MIDAS) significantly worsened. Additionally, individuals with long-standing PPTH reported more severe insomnia, higher cognitive and emotional pain-related response (as measured by the PCS), and PTSD, compared to those who had persisting PTH for a few months so far. In individuals who have PTH persistence, these heightened responses to pain, including pain-related rumination and helplessness, as assessed using the PCS, may relate to heightened threat perception and reduced coping resources, particularly when recovery is delayed or uncertain. Importantly, we clarify that PCS assessments were conducted after the mTBI, and the resulting scores reflect post-injury cognitive and emotional responses to pain rather than pre-existing psychological traits. In our interpretation, these findings are best understood as evidence that attention to pain and emotional responses to pain may evolve as symptoms persist and interfere with daily life, rather than indicating pre-injury psychological traits.
Our findings are consistent with previous studies demonstrating that PTH following mTBI is associated with a multidimensional range of physical, cognitive, and emotional symptoms, often described collectively as “post-concussion syndrome” (4,5,28–31). Merritt et al. found that greater symptom severity immediately following injury predicted a higher overall symptom load during the first week post-mTBI (32). McConnell et al. reported higher SCAT scores in a pediatric cohort with PTH following mTBI compared to those with mTBI without headache (29). Chan and Woldeamanuel (33) reported that PTH-related comorbidities, including physical symptoms (neck pain) and psychological conditions like PTSD were predictive of PPTH. Consistent with findings by Yilmaz et al., who reported that those with PPTH reported higher levels of anxiety and depression in those with headache persistence following mTBI (34), our results suggest sustained emotional symptoms in individuals with PTH non-recovery months and years following mTBI.
Higher cognitive and/or emotional responses to pain in those who had PTH persistence for months or years may develop as symptoms become chronic and interfere with daily functioning (35). These cognitive-affective processes have long been interpreted as solely negative psychological reactions to pain but are increasingly understood as psychophysical constructs with neurobiological correlates. Functional neuroimaging studies demonstrate that pain rumination and helplessness are associated with increased activity in brain regions implicated in affective regulation and central pain modulation, including the anterior insula, amygdala, and prefrontal cortex (36,37). This supports the notion that these responses are not purely subjective experiences but are underpinned by measurable alterations in brain function.
Notably, individuals with years of PPTH had higher levels of insomnia, and more severe symptoms of helplessness. These findings suggest that certain symptoms can become more entrenched or exacerbated over time, potentially reflecting the cumulative impact of persisting headache on psychophysical function. The persistence and progression of these symptoms highlight the substantial long-term burden of individuals living with PPTH and underscore the importance of early intervention and comprehensive symptom management. Although not the primary focus of the current study, it is noteworthy that 11.4% of individuals in the APTH cohort reported migraine with aura following mTBI, compared to 39% in the PPTH cohort. The higher prevalence of aura among those with PPTH is of particular interest. Prior work by Metti et al. (38) demonstrated that aura alone differentiated PTH from other headache types in a military population, suggesting that aura may associate with PTH persistence.
This study has several limitations. Although headache characteristics and TBI-related symptoms were systematically assessed using National Institutes of Health Common Data Elements (CDEs) for PTH and TBI, recall bias is a concern. In particular, individuals with decade-long PPTH may have had difficulty accurately recalling details on their TBI or their headache patterns over the course of a decade, which could affect the reliability of self-reported data. The SCAT-5 was originally developed as a sideline assessment tool for sports-related concussions and was designed to be interpreted relative to a baseline evaluation. In our study, we utilized the symptom checklist component of the SCAT-5 to assess the severity of post-injury symptoms following mTBI. As no pre-injury symptom data were available for participants, we acknowledge that the SCAT-5 has not been specifically validated for in-office use.
The APTH and PPTH cohorts were recruited separately and differed in several key demographic and clinical characteristics, including sex distribution, headache phenotype, and mechanism of injury. Additionally, the PPTH cohort was recruited as part of a prior study that had the objective to compare individuals with migraine without history of TBI to those with PPTH, and to enable this comparison, PPTH individuals with prior history of migraine were excluded. While this design choice allowed us to isolate PTH mechanisms independent of pre-existing migraine, it does introduce an important limitation to the current study, specifically because history of migraine prior to TBI is very common in those with PTH and believed to contribute to symptom persistence (39). For the APTH cohort, we elected to enroll individuals within two months of injury. While it is correct that this timeframe is somewhat arbitrary given the absence of formal guidelines defining ‘acute’ PTH, it was chosen to optimize recruitment feasibility and overall enrollment success. These differences may have introduced confounding effects and limited the comparability between the groups, although we attempted to account for them by including relevant covariates in our statistical models. Data collection was also impacted by the COVID-19 pandemic, which required some follow-up visits to be conducted via phone rather than in person. As a result, 17 participants were unable to complete the in-person cognitive assessments, and certain questionnaires were not completed by a significant subset of participants, leading to a reduced sample sizes in the corresponding MEM analyses and limiting statistical power for those outcomes. A total of 38 individuals did not have six-month follow-up data, representing a limitation common to longitudinal studies. However, longitudinal analyses of trend between three to six months were restricted to participants with complete data at both the three- and six-month timepoints while controlling individual differences as random effects to help mitigate potential bias from missing data. This analysis did not investigate PTH characteristics (e.g., migraine phenotype, headache frequency, PTH intensity, etc.) and mTBI characteristics (e.g., injury mechanism) that might associate with PTH improvement or non-improvement. The generalizability of the results to different subgroups of individuals with PTH is yet to be investigated in future studies. Additionally, sociodemographic factors such as income, access to care, and whether participants were pursuing Worker's Compensation claims or were involved in litigation were not collected. Years of education data were collected for the APTH cohort but not for the PPTH cohort.
Future work incorporating both clinical and sociodemographic variables will be important to better account for confounders in the assessment of PTH outcomes. Baseline physical and emotional symptoms that differed significantly between groups in this study may provide insights into factors associated with PTH persistence; however, their potential predictive value will need to be examined in future research.
Conclusion
This study demonstrates that individuals with APTH who fail to have PTH improvement within three months post-injury exhibit significantly greater severity of non-headache symptoms, including physical, cognitive, and emotional disturbances, compared to those who improve. These symptom differences persist at both follow-ups #1 and #2. The symptoms improve along with PTH over the first three months in individuals who have PTH improvement. Moreover, individuals with long-standing PPTH have even more pronounced impairments, particularly in sleep and emotional regulation. These findings suggest that early symptom burden may be predictive of prolonged headache and functional impairment, highlighting the need for early, comprehensive assessment and multidisciplinary intervention strategies to mitigate the risk of PPTH.
Clinical implications
Non-headache physical, cognitive, and emotional symptoms experienced by individuals with acute post-traumatic headache (APTH) in the first weeks following mTBI, may help identify individuals at risk for persistent post-traumatic headache (PPTH), supporting the need for early, targeted screening after mTBI.
Persistent symptoms beyond three months post-mTBI highlight the importance of ongoing monitoring and tailored support for individuals with delayed recovery, particularly Interventions that address sleep disturbance and pain-related cognitive/emotional regulation.
Footnotes
Acknowledgements
We are grateful to the study participants for their dedication to this project
Ethical considerations
The studies were approved by the Mayo Clinic and Phoenix VA Institutional Review Boards and the United States Department of Defense Human Research Protection Office.
Consent to participate
All participants provided written informed consent prior to participation.
Author contributions
L.M.: Data curation; Formal analysis; Writing – original draft.
J.L.: Methodology; Formal analysis; Supervision – review & editing.
T.J.S.: Conceptualization; Supervision; – review & editing.
T.W.: Methodology; Supervision; – review & editing.
K.R.: Methodology; Project administration – review & editing.
G.D.: Data Curation; Formal Analysis – review & editing.
D.C.S.: Data curation; Project administration– review & editing.
C.D.C.: Conceptualization; Funding acquisition; Methodology; Supervision; Project administration; Writing – original draft; Writing – review & editing. M.L.: Data curation, Project administration-review & editing. R.C.: Data curation, Project administration-review & editing. A.S.: Methodology, Project administration-review 7 editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Institutes of Health, National Institute of Neurological Disorders and Stroke, Award Number 1R61NS113315–01 and the United States Department of Defense, W81XWH-19-0534 and W81XWH-15-1-0286.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.M., J.L., G.D., D.S., M.L., R.C., T.W., K.R. declare no competing interests. C.D.C. has received research funding from Amgen, National Institutes of Health, U.S. Department of Defense, and the American Heart Association. She serves as a Junior Editor on the Editorial Board of Cephalalgia. A.S. has received consulting fees from AbbVie, Allergan, Amgen, Amneal, Axsome Therapeutics, Eli Lilly, eNeura, Everyday Health, Impel, Lundbeck, MedIQ, Medscape, Miller Medical, Neurolief, Novartis, Pfizer, Salvia, Satsuma, Teva, Theranica, UpToDate, WebMD, and Woodberry Associates. T.S., within the prior 24 months, has received consulting fees from AbbVie, Linpharma, Lundbeck, Salvia BioElectronics, and Scilex, and royalties from UpToDate. He holds stock options in Allevalux and Nocira. He has received research funding from the American Heart Association, Flinn Foundation, Henry Jackson Foundation, National Headache Foundation, National Institutes of Health, Patient Centered Outcomes Research Institute, Pfizer, Spark Neuro, and United States Department of Defense.
Data availability statement
De-identified data may be made available from the corresponding author upon reasonable request and with approval from the Mayo Clinic Institutional Review Board, in accordance with applicable guidelines and regulations.
Supplemental material
Supplemental material for this article is available online.

