Abstract
Aim
To describe headache phenotypes and treatment patterns in adults with moderate to severe post-traumatic headache and a high burden of other post-concussion symptoms two to four months after mild traumatic brain injury.
Methods
A cross-sectional study was conducted using patient-reported questionnaire data from 218 individuals, aged 18–60 years, with moderate to severe post-traumatic headache, a headache duration between two and four months, and a high burden of other post-concussion symptoms. The study was based on data from a cohort study with a nested randomized controlled trial called Get Going After Concussion 2.0. Individuals were recruited from emergency departments and general practitioners in Central Denmark Region (1.4 million inhabitants). Phenotyping was performed using an algorithm linking the questionnaire data to diagnostic criteria of primary headache disorders.
Results
Among the 218 individuals, 62% had acute- and 38% had persistent post-traumatic headache. A migraine-like or probable migraine-like phenotype was identified in 88% of the individuals with no significant differences in phenotype distribution between the acute- and persistent headache subgroups (p = 0.414) or between sexes (p = 0.994). Headache frequency of ≥15 days per month was reported by 78% and average headache intensity was moderate or severe in 94% of all individuals. A pressing and/or tightening headache quality was the most frequent (85%). No significant differences were found between the acute- and persistent headache subgroups regarding pharmacological treatment use or efficacy (p = 0.776). Non-opioid analgesics had been used by 88% of all individuals and 81% were still using them; 52% reported lack of efficacy. Triptans had been or were currently used by 4% of all individuals. Preventive medication, used by 7% of the study population, was reported to have lack of efficacy but all continued treatment. Probable medication overuse headache was found in 27% of the individuals. Overall, 33% had tried two or more non-pharmacological treatments, and 6% had tried between four and six.
Conclusion
A migraine-like phenotype was the most common presentation in adults with moderate to severe post-traumatic headache and a high symptom burden of other post-concussion symptoms two to four months after trauma. Overall, limited effect of both pharmacological and non-pharmacological treatment was reported. Preventive medication and triptans were used by very few individuals, despite being recommended in current treatment guidelines. The study population exhibited a high health care-seeking behavior, and a large proportion had probable medication overuse headache. Consequently, further insights into disease-specific treatment approaches of post-traumatic headache is needed.
This is a visual representation of the abstract.
Keywords
Introduction
Post-traumatic headache (PTH) is one of the most frequent and disabling consequences of mild traumatic brain injury (mTBI) (1). The estimated lifetime prevalence of this secondary headache disorder is around 5% in the general population (2,3). Although highly prevalent and linked to significant disability, sick leave and health-care costs, PTH remains an understudied and underrecognized disorder (4). According to The International Classification of Headache Disorders, third edition (ICHD-3) (5), PTH is defined as a headache caused by a traumatic injury to the head and/or neck or caused by craniotomy, with onset within seven days post-trauma. PTH is further classified as acute PTH (aPTH) if the duration is less than three months and persistent PTH (pPTH) if the duration exceeds three months. Notably, the ICHD-3 definition of PTH relies solely on the temporal association with head trauma, without consideration of clinical characteristics. (5).
Regarding phenotypes of PTH, a migraine-like phenotype has been reported as the most common in pPTH (6,7). A migraine-like phenotype has also been reported as the most prevalent from one week to three months following mTBI (8), but knowledge about aPTH phenotypes remains limited. Moreover, most previous studies have been restricted to specific populations, such as sports athletes and military personnel in which a migraine-like phenotype of pPTH has been reported as the most common clinical presentation (9,10). Consequently, phenotyping studies in the early phase, defined as the period from two to four months post-trauma, which includes both aPTH and pPTH, in the broad adult population are lacking. PTH frequently co-occurs with other post-concussion symptoms such as impaired concentration and sleep disturbances (11,12). Up to 30% of mTBI patients develop persistent post-concussion symptoms (PPCS), defined as symptoms lasting more than one month (13,14). A high mTBI symptom burden is a recognized risk factor of PPCS (15,16). Additionally, moderate to severe headache intensity has been found highly prevalent in pPTH (7), and headache severity in aPTH-patients may be an important prognostic predictor of pPTH (15–17). Nevertheless, phenotyping studies of PTH populations with moderate to severe headache and a high burden of other post-concussion symptoms remains limited.
The pathophysiological mechanisms underlying PTH and its persistence are not fully understood, though proposed hypotheses include disruption of descending pain modulatory systems and structural neuroplastic changes (1,18). Consequently, early management of PTH may be crucial to reduce the risk of developing pPTH. PTH is treated as the primary headache disorder it resembles the most (19,20). However, high-quality studies investigating the efficacy of the current expert-opinion based guidelines are lacking (10,18,19,21). Regarding the use of non-pharmacological treatments, most previous studies have been focusing on treatment efficacy in relation to the broad range of mTBI symptoms rather than PTH-specific outcomes (22).
Elucidating the use and efficacy of pharmacological- and non-pharmacological treatments in a PTH-population consisting of both individuals with aPTH and pPTH in the early phase may provide valuable insights into the management of PTH.
The aim of this study is therefore to describe PTH phenotypes and treatment patterns in adults with moderate to severe PTH and a high burden of other post-concussion symptoms two to four months after mTBI based on patient-reported questionnaire data.
Methods
Study design
This study was a cross-sectional study based on data from a cohort study with a nested randomized controlled trial called Get Going After Concussion 2.0 (GAIN 2.0) (23). The present study was conducted in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for cross-sectional studies. The studies are listed in the record of processing activities for research projects in Central Denmark Region (reg.no. 1-16-02-657-20 and 1-16-02-69- 21, respectively). The trial has been approved by The Central Denmark Region Committees on Health Research Ethics (approval number: 1-10-72-330-20). All participants gave written informed consent.
Setting
The cohort study recruited individuals from the 1.4 million inhabitants in Central Denmark Region, Denmark from May 2021–December 2022 (Figure 1). Individuals were recruited two to four months post-trauma via two sources: 1. Individuals with PPCS could be referred from general practice. 2.Individuals diagnosed with mTBI at one of the five emergency departments in Central Denmark Region were identified through an administrative hospital register.
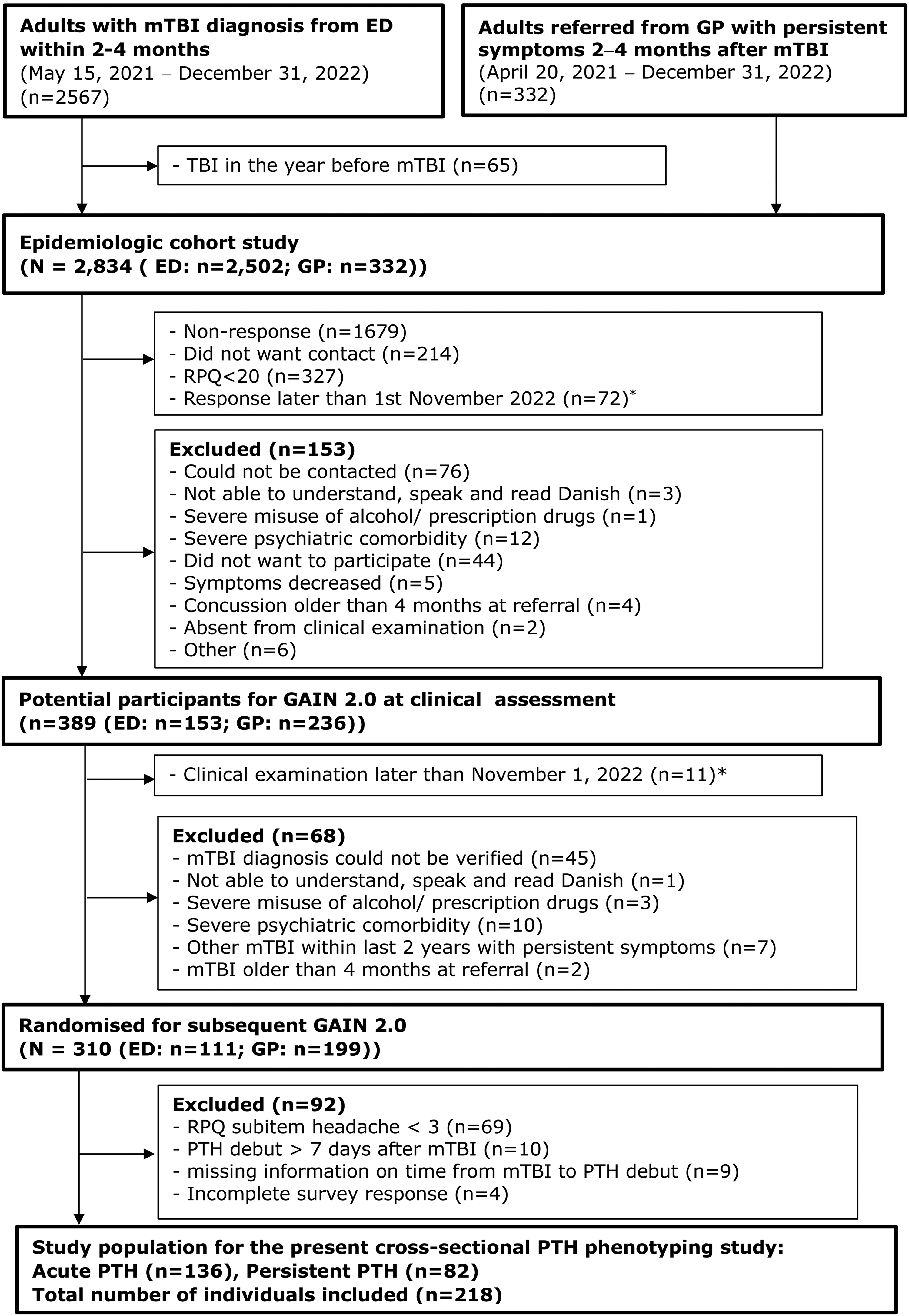
Flowchart of the study population for the present cross-sectional PTH phenotyping study. Abbreviations: GP: General practice, ED: Emergency Department, TBI: Traumatic brain injury, RPQ: Rivermead post-concussion symptoms questionnaire, mTBI: Mild traumatic Brain injury, PTH: Post-traumatic headache, GAIN 2.0: Randomised controlled trial called Get Going After Concussion 2.0. * November 1, 2022: cut-off date for inclusion in GAIN 2.0.
Participants
Potential participants received the Rivermead Post-Concussion Symptoms Questionnaire (RPQ) measuring the severity of 16 post-concussion symptoms [range 0–64, higher scores indicate worse symptoms] (24) via email to assess symptom severity (23). For the present study, a high symptom-burden of other post-concussion symptoms was defined as a total RPQ score ≥ 20, and further a moderate to severe PTH was defined as a RPQ subitem headache score > 2. Consequently, individuals with a total RPQ score ≥ 20 and a RPQ subitem headache score > 2 [1 item, range 0–4, higher scores indicate worse symptoms] received a supplementary headache questionnaire focusing on headache characteristics and headache treatment patterns as well as The Headache Impact Test Questionnaire (HIT-6) (see Variables section) (25). Individuals with a total RPQ score ≥ 20 irrespective of their score on RPQ subitem headache were invited to a clinical assessment for potential inclusion in the GAIN 2.0 project (23,26). The inclusion criteria for the present study were: (1) An mTBI diagnosis based on the recommendations by the WHO Task Force (27), but with the amendment, that there must have been a direct contact between the head and an object, (2) age 18 to 60 years at the time of the trauma, (3) a total RPQ score ≥ 20 within 1 week before enrolment in the GAIN 2.0 study and, (4) identified from registers of the emergency departments or referred by general practitioners (GPs) within two to four months after mTBI, (5) RPQ subitem headache score > 2 since this was a criteria for receiving the supplementary headache questionnaire and (6) headache onset within 7 days following mTBI in accordance with the diagnostic criteria of PTH (28). All six inclusion criteria should be met to be included. Exclusion criteria were (1) objective neurological findings and/or acute trauma CT scan indicating neurological disease or brain damage linked to the mTBi; (2) previous mTBI within the last 2 years with ongoing post-concussion symptoms at the time of the present mTBi; (3) severe misuse of alcohol, prescription drugs and/or illegal drugs; (4) severe psychiatric co-morbidity defined as contact to psychiatric department or private psychiatrist within the last two years before mTBi; (5) severe neurological disease (e.g, multiple sclerosis) that impedes participation in the program; and (6) inability to start the intervention within the maximum time for eligibility (4 months) plus the maximum possible waiting time from assessment to start of intervention (3 months), i.e, 7 months post-trauma. The two to four months cut-off was chosen to ensure a maximum symptom duration of seven months, as the GAIN 2.0 study hypothesized intervention efficacy within this timeframe. Furthermore, individuals with PPCS lasting more than two years from a previous mTBI was excluded to ensure a maximum symptom duration of seven months. The two years cut-off was selected because most mTBI cases are expected to remit within this period (14).
Variables
Data were collected and managed using Research Electronic Data Capture (REDCap) which is a secure, web-based software platform designed to support data capture for research studies hosted at Aarhus University, Denmark (29). All data for the present study including data on demographic characteristics and trauma-related characteristics were collected two to four months post mTBI with the exception of additional patient-reported data on loss of consciousness in relation to mTBI and comorbid disorders pre-trauma which was collected two to seven months post mTBI as part of the clinical assessment in GAIN 2.0. Apart from the patient-reported data collected as part of the clinical assessment, all data in the present study are patient-reported questionnaire data.
Headache data
A headache questionnaire based on the ICHD-3 criteria for primary headache disorders has previously been developed for a PTH phenotyping study in adolescents and young adults (30). A modified version of this questionnaire was developed for the present study. The questionnaire consisted of 56 questions with further specifying subitems covering the following: Trauma mechanism that lead to PTH (traffic accident, fall, direct blow to the head (not violence), violence/assault, other), headache history (pre- and post-mTBI), headache duration, headache frequency (days within the last month), headache localization (binary variables of unilateral- and bilateral headache localization was made based on a five-point Likert scale (1 = never, 2 = seldom, 3 = sometimes, 4 = very often, 5 = always) where “no” was defined as 1 and “yes” was defined as ≥2. Furthermore, questions regarding headache quality (pressing, throbbing, tightening, pulsating, stabbing), headache mean intensity within the last 4 weeks measured on a numeric rating scale (NRS) ranging from 0–10 divided into mild (1–3), moderate (4–7) and severe (8–10) pain (31), worsening factors (physical activity, housework, sudden movements, head movements), accompanying symptoms (phonophobia, photophobia, dizziness, nausea, insomnia, tinnitus, vomiting, continuous symptoms was defined as being constantly present, independent of headache), aura symptoms, autonomic symptoms and other trigger factors (unspecified stress, bright lights, insomnia, neck pain, alcohol).
Additionally, questions covering previous and current use of pharmacological treatment (paracetamol, acetylsalicylic acid, other nonsteroidal anti-inflammatory drugs (NSAIDs), codeine, tramadol, morphine, triptans, anticonvulsants, antidepressants, beta blockers, and calcium channel blockers), and non-pharmacological treatment strategies (physiotherapy, chiropractor, acupuncture, psychologist, hypnosis, and craniosacral therapy) to alleviate the headache frequency was included in the questionnaire. Medication usage data was based on the questions; “Have you previously used or are you currently using one or more of the following types of medicine for treating your headache after concussion?” and “How long did you use/have you been using the following treatment?”. Efficacy of pharmacological- and non-pharmacological treatments was based on the question; “Which effect, measured as a reduction in the number of headache days, have you experienced in relation to the following pharmacological/non-pharmacological treatment” based on the following scale: 0% = no effect, 25% = little effect, 50% = good effect, 75% = very good effect and 100% = headache free. Lack of efficacy was defined as “no effect” (0%) or “little effect” (25%). Effective treatment was defined as “good effect” (50%), “very good effect” (75%) or “headache free” (100%). The full-length questionnaire is available upon request for research purposes.
Data on the headache impact test (HIT-6) (25) was also collected. HIT-6 is used for assessing the headache impact on social functioning, role functioning, vitality, cognitive functioning and psychological distress. We used the recommended item response weights (never=6, rarely=8, sometimes=10, very often=11 and always=13). The final HIT-6 score is obtained from simple summation of the six items and ranges between 36 and 78 (higher score indicating higher impact on functioning) (25).
Phenotyping of PTH based on the ICHD-3 criteria for primary headache disorders
An algorithm linking the headache questionnaire data variables to the ICDH-3 criteria of primary headache disorders was developed by CN, ABN and MK supported by JF, SFK and HS (Figure 2). The algorithm was manually checked by CN. Based on this algorithm, the individuals were classified into one of the 13 pre-defined phenotypes in accordance with ICHD-3 (5). The algorithm follows the diagnostic hierarchy of the ICHD-3. Consequently, fulfilment of the diagnostic criteria for migraine-like, TTH-like or TACs-like phenotypes trumps fulfilment of the criteria for probable migraine-like-, probable TTH-like-, and probable TACs-like phenotypes.

Overview of the PTH phenotyping algorithm based on ICHD-3 criteria for primary headache disorders, with specified modifications. The algorithm follows the ICHD-3 hierarchy. No individuals were classified into phenotypes marked with an asterisk.
Some modifications of the ICDH-3 was made in the phenotyping algorithm to take differences between PTH and primary headache disorders. Given PTH's typically high monthly headache frequency, (6,32) distinguishing attack frequency and duration via patient-reported questionnaires was not feasible. Consequently, the A criteria of 1.1. migraine without aura and 1.2 migraine with aura was not included in the migraine-phenotypes of PTH. Since PTH often exceeds 72 h(6), the upper time limit in the B-criteria for 1.2 migraine with aura was omitted in the migraine with aura-like phenotype. Regarding the tension type headache (TTH)-like phenotype, the A-criteria of 2.1 infrequent tension type headache and 2.2 frequent tension type headache was excluded to reduce the risk of recall bias when assessing headache frequency over the past three months. TACs-like phenotype was modified accordingly.
Additionally, probable medication overuse headache (pMOH) was in accordance with ICDH-3 recommendations (5) assessed by the question; “How many days have you been using paracetamol, ibuprofen etc. within the last month”. High risk of pMOH was defined as a use of paracetamol, ibuprofen or acetylsalicylic acid ≥ 15 days/month within the last month, a use of codeine ≥10 days/month or a use of triptans > 9 days/month within the last month. Low risk of pMOH was defined as a medication use below these limits within the last month.
Statistical analysis
Continuous data variables were not normally distributed and are therefore presented as median and interquartile range (IQR). Categorical data are presented as frequency counts and/or percentages. Group comparisons were performed using Fisher's exact test when expected cell counts were below five; otherwise, the chi-square test was applied. In cases with multiple comparisons, p-values were adjusted using Bonferroni correction.
All statistical analysis were performed using StataCorp. 2023. Stata 18. Statistical Software. StataCorp LLC. The Stata code used for the phenotyping algorithm is available upon request for research purposes.
Results
Demographics, trauma-related characteristics and pre-trauma comorbidity
Eligibility for the present cross-sectional study was assessed among the 310 individuals randomized for the subsequent GAIN 2.0 study (Figure 1). 69 participants had a baseline RPQ subitem headache < 3 and were therefore not invited to fill out the baseline headache questionnaire. Headache onset more than 7 days from mTBI was reported by 10 individuals, who therefore did not fulfill the diagnostic criteria of PTH. The study population for the present study consisted of 218 individuals with PTH (Figure 1).
The median age of the individuals was 36 years and 74% were females (Table 1). In the study population 62% had aPTH and 38% had pPTH. Within the last four weeks, 54% had been on full-time sick leave, and 70% of the study population had been referred by GPs. The median time from mTBI to questionnaire completion was 82 days. The most frequent injury mechanism was a direct blow to the head not caused by violence (46%). Median RPQ and HIT-6 scores were 39 and 64, respectively. Pre-mTBI, 38% of the individuals had a psychiatric diagnosis, 26% a functional disorder, and 46% a somatic diagnosis (Table 1). A pre-trauma diagnosis of migraine was reported by 10% of the study population, and 22% reported a pre-trauma diagnosis of TTH.
Baseline characteristics of the study population (n = 218).

Values are median (IQR) or n (%). Percentages may not total 100% due to rounding. Abbreviations: PTH: Post-traumatic headache, IQR: interquartile range, RPQ: Rivermead Post-Concussion Questionnaire, HIT-6: Headache Impact Test.
Individuals with missing data on at least one subitem in the HIT-6 were not included (n = 3).
Based on the question “Have a medical doctor at any timepoint in your life prior to mTBI given you a diagnosis of..” (yes/no):
Includes attention deficit hyperactivity disorder (ADHD), anxiety, autism, bipolar disorder, depression, obsessive-compulsive disorder (OCD), psychosis or schizophrenia, eating disorder, personality disorder.
Includes whiplash, irritable bowel syndrome, chronic fatigue syndrome, fibromyalgia.
Includes cancer, diabetes, hypertension, heart disease, stroke, acute myocardial infarction, neurological disorder, asthma, psoriasis, osteoarthritis, rheumatoid arthritis, chronic obstructive pulmonary disease), osteoporosis.
Headache characteristics and other clinical characteristics
Within the past month, 78% of all individuals reported having had ≥ 15 headache days (Table 2). Average headache intensity was reported as moderate or severe by 94%. Headaches were bilateral in 89%. However, 71% reported that the headache could also have a unilateral localization. A pressing and/or tightening headache quality was the most common (85%). Continuous nausea, photophobia, and phonophobia were reported by 10%, 31%, and 36% of individuals, respectively. The most frequent headache triggers were screen use (70%) and stress (69%). Post-mTBI, 63% reported persistent or recurrent neck pain and 11% reported persistent of recurrent low back pain with a duration above three months.
Headache characteristics and other clinical characteristics of the study population (n = 218).
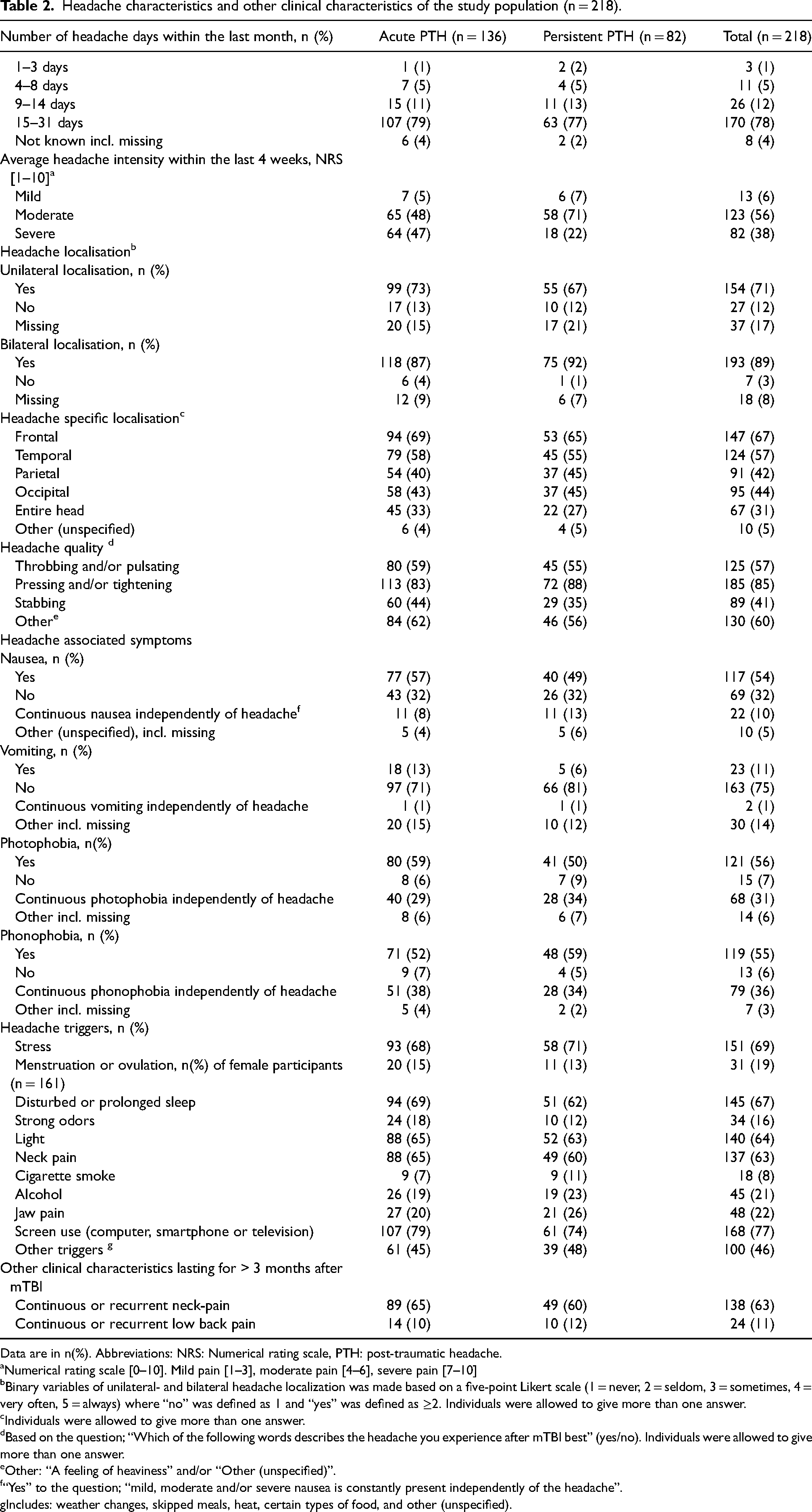
Data are in n(%). Abbreviations: NRS: Numerical rating scale, PTH: post-traumatic headache.
Numerical rating scale [0–10]. Mild pain [1–3], moderate pain [4–6], severe pain [7–10]
Binary variables of unilateral- and bilateral headache localization was made based on a five-point Likert scale (1 = never, 2 = seldom, 3 = sometimes, 4 = very often, 5 = always) where “no” was defined as 1 and “yes” was defined as ≥2. Individuals were allowed to give more than one answer.
Individuals were allowed to give more than one answer.
Based on the question; “Which of the following words describes the headache you experience after mTBI best” (yes/no). Individuals were allowed to give more than one answer.
Other: “A feeling of heaviness” and/or “Other (unspecified)”.
“Yes” to the question; “mild, moderate and/or severe nausea is constantly present independently of the headache”.
Includes: weather changes, skipped meals, heat, certain types of food, and other (unspecified).
PTH phenotypes
Of the 218 individuals, 88% were classified as having a migraine-like or probable migraine-like PTH phenotype (Figure 2; Table 3). No significant difference in phenotype distribution was found between the aPTH- and pPTH subgroups (p = 0.414) (Table 3). Consequently, results are presented for the overall study population. A migraine without aura-like phenotype was the most common (38%). A TTH-like phenotype was found in 6% of the study population and 3% had a probable TTH-like phenotype. Only one individual was classified as having both a migraine-like and TTH-like phenotype. A cluster-headache like phenotype was found in 2% and no individuals fulfilled the criteria for the other defined TAC's-like phenotypes (Figure 2). A migraine-like phenotype of PTH was found in 62% of the individuals reporting a pre-mTBI diagnosis of migraine (Table 1). No significant difference in phenotype distribution was found between males and females (p = 0.994) (Supplementary table 1).
Phenotypes of post-traumatic headache in the study population (n = 218).

Data are in n(%).
Abbreviations: PTH: post-traumatic headache, TTH-like: tension-type headache like, TACs-like: Trigeminal autonomic cephalalgias-like.
Pharmacological treatment patterns and treatment efficacy
No significant differences in treatment efficacy of acute- and preventive treatment between the aPTH and pPTH subgroups (Table 4) and between males and females (Supplementary table 1) were found. Similarly, no significant difference in treatment efficacy was found when stratifying participants into non-migraine-like and migraine-like phenotype of PTH (Supplementary table 3). Consequently, results are presented for the overall study population. A history of acute medication use was reported by 88% and 81% reported a current use (Table 4). Triptans had been used or were currently used by 4% of the individuals and 75% of the users reported it to be an effective treatment. A pre-mTBI diagnosis of primary migraine was reported by 75% of the triptan-users. A current or previous use of non-opioid analgesics was reported by 88% with 52% reporting this treatment to have a lack of efficacy. A current or previous use of opioids were reported by 9%, with 40% reporting lack of efficacy. Preventive medication was used by 7%, all of whom continued treatment despite high rates of reported inefficacy: 100% for beta-blockers, 83% for anticonvulsants, and 56% for antidepressants. In the study population, 27% were categorized with pMOH.
The use of pharmacological treatment and its efficacy in the study population stratified by acute- and persistent PTH.
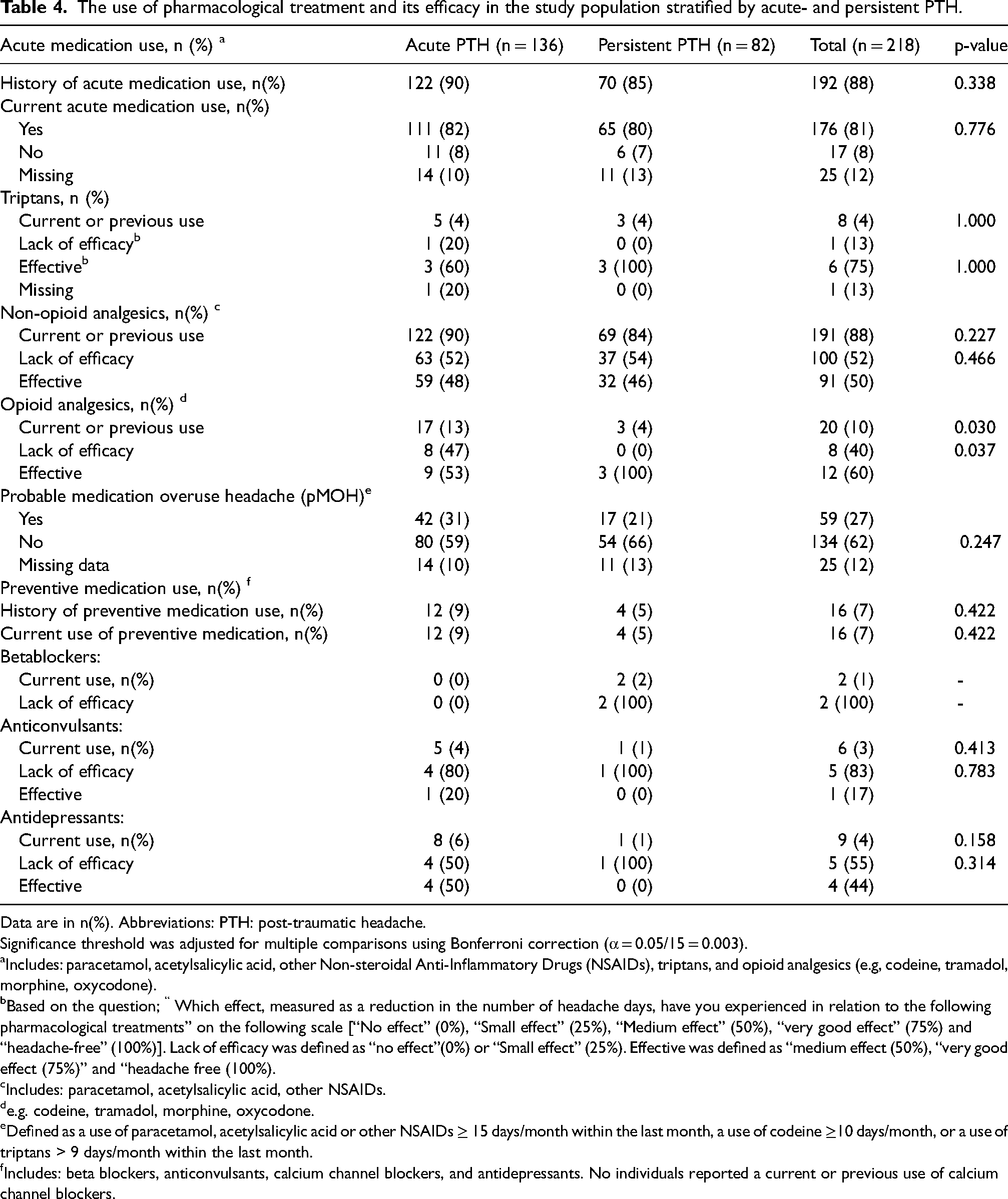
Data are in n(%). Abbreviations: PTH: post-traumatic headache.
Significance threshold was adjusted for multiple comparisons using Bonferroni correction (α = 0.05/15 = 0.003).
Includes: paracetamol, acetylsalicylic acid, other Non-steroidal Anti-Inflammatory Drugs (NSAIDs), triptans, and opioid analgesics (e.g, codeine, tramadol, morphine, oxycodone).
Based on the question; “ Which effect, measured as a reduction in the number of headache days, have you experienced in relation to the following pharmacological treatments” on the following scale [“No effect” (0%), “Small effect” (25%), “Medium effect” (50%), “very good effect” (75%) and “headache-free” (100%)]. Lack of efficacy was defined as “no effect”(0%) or “Small effect” (25%). Effective was defined as “medium effect (50%), “very good effect (75%)” and “headache free (100%).
Includes: paracetamol, acetylsalicylic acid, other NSAIDs.
e.g. codeine, tramadol, morphine, oxycodone.
Defined as a use of paracetamol, acetylsalicylic acid or other NSAIDs ≥ 15 days/month within the last month, a use of codeine ≥10 days/month, or a use of triptans > 9 days/month within the last month.
Includes: beta blockers, anticonvulsants, calcium channel blockers, and antidepressants. No individuals reported a current or previous use of calcium channel blockers.
Non-pharmacological treatment patterns and treatment efficacy
With 51% having tried physiotherapy, this was the most frequently tried non-pharmacological treatment in the study population (Figure 3). Lack of efficacy was reported by 67%. The second most frequently tried non-pharmacological treatment was craniosacral therapy (24%), for which 58% reported lack of efficacy. There were no significant difference in treatment efficacy between the aPTH and pPTH subgroups (Supplementary table 2). Two or more non-pharmacological treatments had been tried by 33% of the study population and 6% reported to have tried ≥4 and ≤ 6 different non-pharmacological treatments (Table 5).

The use and efficacy of non-pharmacological treatment in the study population (n = 218). Numbers in the bars are n(%) of the individuals with a previous or current use. Numbers to the right of each bar are n(%) of the total study population. Efficacy was based on the question; “Which effect, measured as a reduction in the number of headache days, have you experienced in relation to the following non-pharmacological treatments” on the following scale [“No effect” (0%), “Small effect” (25%), “Medium effect” (50%), “very good effect” (75%) and “headache-free” (100%)]. Lack of efficacy was defined as “no effect”(0%) or “Small effect” (25%). Effective was defined as “medium effect (50%), “very good effect (75%)” and “headache free (100%).
Number of non-pharmacological treatments tried in the study population (n = 218).

Data are in n(%). Abbreviations: PTH: post-traumatic headache.
Non-pharmacological treatments includes: physiotherapy, chiropractor, acupuncture, psychologist, hypnosis and craniosacral therapy.
Discussion
In the present study, the majority had a migraine-like phenotype of PTH two to four months post-trauma. The most prevalent headache characteristics were a headache frequency of ≥15 days/month, a moderate headache intensity, and a pressing and/or tightening headache quality. Acute non-opioid medication was used by the majority of individuals whereas few used preventive medication and triptans. The majority had tried at least one non-pharmacological treatment, while one-third had tried two or more. Overall, limited effect of both pharmacological- and non-pharmacological treatment was reported.
Headache phenotypes and other clinical characteristics
In the present study, a migraine-like phenotype of PTH was the most common in both the aPTH- and the pPTH subgroup which is in line with most previous findings irrespective of study population and disease duration (8,10). However, some studies have found a TTH-like phenotype and a combination of migraine- and TTH-like phenotype to be the most prevalent in pPTH (33,34). Additionally, other studies found a migraine-like phenotype followed by a TTH-like phenotype to be the most prevalent in a pPTH (6,11). The few previously conducted studies on PTH phenotyping in the early phase have, similar to the present study, identified a migraine-like phenotype as the most prevalent (6,30). Consequently, the present study supports previous evidence that a migraine-like phenotype of PTH is highly prevalent two to four months post-trauma. The low prevalence of cluster-headache-like phenotype is also in alignment with previous findings (35). Interestingly, the present study found a much lower prevalence of a TTH-like phenotype and a combined migraine- and TTH-like phenotype compared to previous studies (6,36). These differences may be due to methodological differences between semi-structured interview-based phenotyping and questionnaire-based phenotyping. In the present study, the modifications of the ICHD-3 criteria in the phenotyping algorithm may have led to an overestimation of the migraine-like phenotypes of PTH. Additionally, the low prevalence of a TTH-like phenotype in the present study might partly be explained by the large proportion reporting continuous nausea and photophobia. Furthermore, the differences could be caused by the large inhomogeneity between studies with regard to differences in disease duration and differences within PTH subpopulations. In the present study, the majority reported a headache frequency > 15 days per month and a moderate to severe headache intensity which is in alignment with previous studies (6,8). Regarding headache quality, 85% reported a pressing and/or tightening headache quality and 57% of individuals reported a throbbing and/or pulsating headache quality in line with previous findings (6). Interestingly, 31% experienced continuous photophobia and 36% experienced phonophobia independent of headache as part of their post-concussion symptoms. A high prevalence of these continuous symptoms have also previously been described (6). This highlights the importance of differentiating between continuous symptoms and aura symptoms. However, this could be challenging when phenotyping PTH and may partly explain the differences in the prevalence of migraine-like and TTH-like phenotypes between previous studies. With 11% of all individuals reporting post-mTBI low back pain and 63% reporting post-mTBI neck-pain, this was highly prevalent in the present study although to a less degree than reported in the more chronic PTH population (6).
Pharmacological treatment patterns and efficacy
The current treatment guidelines of PTH is based on expert consensus due to the lack of controlled trials on PTH (10,19). Consequently, a primary headache phenotype-guided treatment approach of PTH is recommended without differentiating between aPTH and pPTH (10,37,38). However, the efficacy of migraine-specific preventive medications and acute medication has previously been found insufficient (6,20,39,40). Consistent with these findings, preventive medication was found to lack efficacy in the present study; however, these findings was based on only 7% of the study population using preventive medication. Similarly, in the present study, triptans were used by only 4% of individuals even though a migraine-like phenotype of PTH was highly prevalent. Notably, 75% of the triptan users reported a pre-mTBI diagnosis of migraine, suggesting that triptans are rarely prescribed solely for PTH management. Consequently, these results could indicate a discrepancy between the current treatment guidelines of PTH and clinical practice. These findings are in line with a migraine study highlighting that the therapeutic guideline on acute migraine treatment in migraine patients is rarely followed (41). Consequently, further studies investigating the efficacy of triptans in PTH-are are needed since low efficacy of triptans has been reported in the pPTH population (10). In the present study, 27% of the study population had pMOH. High prevalences of medication overuse have previously been found in pPTH populations (30,34,42). Furthermore, the prevalence of co-existing medication overuse headache (MOH) has been found to be as high as 42% in pPTH populations (34,43,44). However, it should be emphasized that not all individuals with pMOH will necessarily develop MOH. Nevertheless, these findings highlight the need for clinicians to recognize the use of low-efficacy pharmacological treatments and potential side effects, including MOH in the PTH-population, as these may complicate PTH management.
Non-pharmacological treatment patterns and efficacy
In the present study, 33% of all individuals had tried two or more non-pharmacological treatments. This high health care seeking behavior in PTH patients has to the authors knowledge not previously been described. Overall, the non-pharmacological treatments were reported to have low efficacy. In line with these findings, a large review on non-pharmacological treatment of PPCS found only weak recommendations for graded physical exercise, manual treatment of neck- and back pain, psychological treatment and interdisciplinary coordinated rehabilitative treatment (22).
Strengths and limitations
This is to the authors knowledge the first PTH phenotyping study two to four months after mTBI, on a large sample of adults with moderate to severe PTH and a high symptom burden of other post-concussion symptoms recruited through both emergency wards and GP's. However, it was a limitation that the that individuals experiencing severe headache but had a total RPQ score below 20 were not eligible for inclusion. Similarly, individuals reporting headache as a mild problem were not included, since the headache questionnaire was not sent to these individuals. Thus, the study population can be considered a representative sample only of individuals with moderate to severe PTH and a high symptom burden of other post-concussion symptoms. Individuals with mild mTBI symptoms including mild PTH may differ in phenotype or treatment patterns compared to the present study population. Additionally, inclusion of some individuals with PPCS >2 years from a previous mTBI cannot be ruled out, potentially biasing results toward reduced treatment efficacy.
Further, the patient reported headache questionnaire used in this study has not been validated which could have led to some degree of misclassification and diagnostic imprecision in phenotyping. However, to the authors knowledge, no validated phenotyping questionnaires are currently available. Moreover, the modifications of the ICHD-3 criteria in the phenotyping algorithm may also have reduced the diagnostic accuracy (Figure 2). However, the low number of missing data represents an important strength in the present study since this has reduced the risk of selection bias. Patient-reported data on pre-mTBI headache days/month, pre-mTBI primary headache diagnosis, medication use etc. should be interpreted with caution due to the risk of imprecise information and potential recall bias. Although questions targeted headache-specific treatments for PTH, medication use may have been overestimated due to comorbid pain conditions and psychiatric disorders. Interactions between multiple pharmacological and nonpharmacological treatments were not considered in assessing treatment efficacy, and potential self-assessment bias may have influenced results, likely favoring a lack of efficacy.
It was not possible to differentiate between a current or prior use of the acute medication subgroups, limiting comparability with other studies investigating pharmacological efficacy. Similarly, efficacy could not be distinguished between treatment for pre-existing migraine and PTH in individuals with prior migraine who may have continued triptan use after PTH onset. The pre-defined algorithm has strengthened the study as it has secured a systematic phenotyping of the individuals. However, the questionnaire and algorithm could be further improved. A logical next step is a questionnaire validation study before applying it to other Danish PTH populations. For future studies, a combination of prospective headache diaries, supplementary questionnaires, and semi-structured diagnostic interviews would be ideal. Hopefully, e-diaries based on the ICDH-3 criteria can aid both researchers and clinicians in the near future (45).
Conclusion
A migraine-like phenotype was the most prevalent in adults with moderate to severe PTH and a high symptom-burden of other post-concussion symptoms two to four months after mTBI. Phenotype distribution did not differ between aPTH and pPTH or by sex. Overall, the majority reported a limited effect of both non-opioid analgesics and preventive treatments. Preventive treatment and triptans were only used by a minority highlighting a discrepancy with current phenotype-based treatment guidelines of PTH. Further studies are needed to clarify the efficacy of triptans and preventive treatment in the early phase of PTH. The study population demonstrated a high healthcare-seeking behavior, and a considerable proportion had pMOH. These findings underscore the need to monitor both efficacy and side effects of pharmacological treatments in PTH patients. Further research should focus on disease-specific treatment strategies and their effectiveness in the early phase of PTH.
Article highlights
A migraine-like phenotype was most common among adults with moderate to severe PTH and a high post-concussion symptom burden 2–4 months post-trauma. Only a minority used triptans or preventive medication, indicating a discrepancy between treatment guidelines and clinical practice.
Limited effect of pharmacological- and non-pharmacological treatment was reported.
Future studies should investigate disease-specific treatments and their efficacy in early-phase PTH.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261423477 - Supplemental material for Post-traumatic headache two to four months after mild traumatic brain injury: A description of phenotypes and treatment patterns
Supplemental material, sj-docx-1-cep-10.1177_03331024261423477 for Post-traumatic headache two to four months after mild traumatic brain injury: A description of phenotypes and treatment patterns by Charlotte Nygaard, Henrik Winther Schytz, Lene Odgaard, Martin Byskov Kinnerup, Amalie Bilgaard Nicolaisen, Line Amalie Hellemose, Peter Preben Eggertsen, Toril Skandsen, Mille Møller Thastum, Henriette Holm Stabel, Carsten Bøcker Pedersen, Simple Futarmal Kothari and Jørgen Feldbæk Nielsen in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024261423477 - Supplemental material for Post-traumatic headache two to four months after mild traumatic brain injury: A description of phenotypes and treatment patterns
Supplemental material, sj-docx-2-cep-10.1177_03331024261423477 for Post-traumatic headache two to four months after mild traumatic brain injury: A description of phenotypes and treatment patterns by Charlotte Nygaard, Henrik Winther Schytz, Lene Odgaard, Martin Byskov Kinnerup, Amalie Bilgaard Nicolaisen, Line Amalie Hellemose, Peter Preben Eggertsen, Toril Skandsen, Mille Møller Thastum, Henriette Holm Stabel, Carsten Bøcker Pedersen, Simple Futarmal Kothari and Jørgen Feldbæk Nielsen in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024261423477 - Supplemental material for Post-traumatic headache two to four months after mild traumatic brain injury: A description of phenotypes and treatment patterns
Supplemental material, sj-docx-3-cep-10.1177_03331024261423477 for Post-traumatic headache two to four months after mild traumatic brain injury: A description of phenotypes and treatment patterns by Charlotte Nygaard, Henrik Winther Schytz, Lene Odgaard, Martin Byskov Kinnerup, Amalie Bilgaard Nicolaisen, Line Amalie Hellemose, Peter Preben Eggertsen, Toril Skandsen, Mille Møller Thastum, Henriette Holm Stabel, Carsten Bøcker Pedersen, Simple Futarmal Kothari and Jørgen Feldbæk Nielsen in Cephalalgia
Footnotes
Acknowledgments
None.
Author contributions
Study concept and design: CN, HWS, SFK, JFN.
Acquisition of data, data analysis and interpretation: CN, HWS, AM, LO, MBK, CBP, SFK, JFN.
All authors contributed to the drafting and critical revision of the manuscript for important intellectual content and has approved the final version.
Consent to participate
All participants gave written informed consent.
Consent for publishing
The authors agree to publish with Cephalalgia if the manuscript is accepted.
Data availability statement
The full-length questionnaire is available upon request for research purposes. The Stata code used for the phenoyping algorithm is available upon request for research purposes.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
The trial has been approved by The Central Denmark Region Committees on Health Research Ethics (approval number: 1-10-72-330-20).
Funding
The study was funded by The Health Research Fund of Central Denmark Region. The funder was not involved in the design of the study; collection, analysis, or interpretation of data; and in the writing of the article. The corresponding author (CN) and the last authors (SFK, JFN) had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Health Research Fund of Central Denmark Region, (grant number A4133).
Open practices
Not applicable
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
