Abstract
Aim
To compare cortical microstructural features in people with persistent post-traumatic headache (PPTH), migraine and healthy controls (HCs) using quantitative magnetic resonance imaging (qMRI).
Methods
This cross-sectional study included 103 participants with PPTH, 296 with migraine and 155 HCs. All underwent 3.0 Tesla qMRI using standardized sequences to generate quantitative T2 (qT2), quantitative T1 (qT1) and apparent diffusion coefficient (ADC) maps. Intracortical voxel values were projected onto the cortical surface for surface-based analysis. Group comparisons were adjusted for age and sex, with correction for multiple comparisons. Associations with clinical variables were explored in regions showing significant group differences.
Results
Compared with HCs, participants with PPTH showed elevated qT2 values in the left lateral occipital cortex (pcluster = 0.0002). Compared with migraine, those with PPTH exhibited lower ADC values in the left superior frontal cortex (pcluster = 0.0020). Relative to migraine with aura, additional reductions in qT1 and ADC values were found in the right middle and inferior temporal gyri (all pcluster < 0.01), and, relative to migraine without aura, lower ADC values were detected in the right middle temporal gyrus (pcluster = 0.0002).
Conclusions
Cortical microstructural alterations in PPTH suggest underlying neurobiological processes distinct from both migraine and healthy adults. The findings support a pathophysiological model involving chronic low-grade neuroinflammation.
Keywords
Introduction
Persistent post-traumatic headache (PPTH) is an increasingly recognized sequela of mild traumatic brain injury (mTBI), often persisting for months or even years after the initial trauma (1). Despite its widespread prevalence and disabling nature (1,2), the biological mechanisms underlying PPTH remain inadequately characterized (1).
Although the clinical features of PPTH frequently overlap with those of primary headache disorders, particularly migraine (3–5), accumulating neuroimaging data suggest that PPTH might arise from partially distinct neurobiological processes (6–9). These differences raise important questions about its pathogenesis and underscore the need for studies that can disentangle shared and unique mechanisms.
One hypothesized contributor to PPTH, as well as a potential differentiator from migraine, is neuroinflammation (1). Animal models have consistently demonstrated prolonged activation of microglia in regions associated with sensory processing (10–12). These pro-inflammatory responses are accompanied by extracellular edema and the release of cytokines, suggesting sustained disruption of cortical function. In parallel, positron emission tomography (PET) data have revealed prolonged microglial activation extending months or years following moderate-to-severe TBI (13,14). However, neuroinflammatory profiles specific to PPTH have not yet been characterized.
Quantitative magnetic resonance imaging (qMRI) provides a non-invasive, in vivo technique for assessing tissue-level changes associated with neuroinflammation (15). By simultaneously assessing multiple imaging parameters, qMRI can detect microstructural signatures of edema, cellularity and iron content (16,17). Despite its potential, multimodal qMRI has not been used to assess cortical microstructural alterations in PPTH. Addressing this gap is essential for clarifying the neurobiological substrates of PPTH and for distinguishing it from migraine.
Here, we report the first multimodal qMRI investigation of adults diagnosed with PPTH, comparing them with persons with migraine and healthy controls (HCs). Furthermore, we investigate associations between qMRI outcome measures and clinical features, aiming to establish biologically meaningful relationships that could inform our disease understanding.
Methods
The study protocol received approval from the ethics committee of the Capital Region of Denmark (Identifier: H-20033264) and all participants provided their written informed consent prior to any study procedures. Data on adults with migraine and HCs were obtained from the MRI Core of the prospective Registry for Migraine (REFORM) study (18,19). A previous analysis from this sample reported cortical microstructural alterations in migraine (including subtypes) compared to age- and sex-matched HCs (20). The current investigation extends those findings by examining cortical microstructure in participants with PPTH, relative to both those with migraine and HCs from the REFORM study. All procedures adhered to the principles of the Declaration of Helsinki and its subsequent revisions.
Design and participants
This cross-sectional study compared adults with PPTH to those with migraine and healthy controls. Enrollment occurred between November 2020 and October 2023. Participants with PPTH and migraine were primarily identified through referrals from neurologists at a national referral hospital. HCs were recruited via public advertisements on a national research volunteer website (https://forsoegsperson.dk). Detailed inclusion and exclusion criteria for each group are outlined in the supplementary material (Tables S1–S3).
PPTH group
Participants met the diagnostic criteria for persistent headache attributed to mild traumatic injury to the head, as defined by the International Classification of Headache Disorders, 3rd edition (ICHD-3) (21). Eligibility required a minimum PPTH duration of one year and an average of at least four monthly headache days during the three months prior to enrollment. Exclusion criteria included a history of more than one mild TBI, any moderate-to-severe TBI, prior whiplash injury, or comorbid neurological or severe systemic disorders. Individuals with a history of primary headache disorder were excluded, except those with infrequent episodic tension-type headache.
Migraine group
Eligible individuals had a diagnosis of migraine without aura, migraine with aura or chronic migraine according to ICHD-3 criteria (21). Participants also required a migraine history of at least one year and a minimum of four migraine days per month during the three months preceding enrollment. Individuals with comorbid neurological or serious systemic disease were excluded.
HC group
Eligible HCs were adults with no personal history of primary or secondary headache disorders, aside from infrequent episodic tension-type headache. Individuals were excluded if they had first-degree relatives with a diagnosed primary headache disorder, clinically significant medical conditions or regular medication use.
Procedures
All participants and HCs completed standardized physical and neurological examinations. Site investigators conducted a semi-structured interview with participants experiencing PPTH or migraine. This interview documented headache characteristics, current and past medication use, and relevant medical history. Participants also provided information on the timing of their most recent headache or migraine, presence of aura symptoms, and, when relevant, menstrual cycle status.
Participants and HCs were instructed to abstain from specific substances before MRI scanning. Use of analgesics, acute migraine treatments, anti-inflammatory medications, sedatives or antihistamines was prohibited for at least 48 hours before the scan. Caffeine or caffeinated products were not permitted within 12 hours prior to imaging.
MRI procedures
Imaging was performed on a 3.0 Tesla MAGNETOM Prisma scanner (Siemens, Munich, Germany) using a 32-channel head coil. Foam pads were placed at the temples to minimize head motion, and participants were instructed to remain still.
The imaging protocol included magnetization-prepared two rapid acquisition gradient echo (MP2RAGE), generalized autocalibrating partially parallel acquisition and model-based accelerated relaxometry by iterative non-linear inversion (GRAPPATINI), and diffusion tensor imaging (DTI). It also included magnetization-prepared rapid acquisition gradient echo (MPRAGE) and fluid-attenuated inversion recovery (FLAIR). These sequences enabled high-resolution structural and microstructural brain assessments. Sequence parameters are provided in the supplementary material (Table S4). Total scan duration was about 55 min.
Preprocessing
FreeSurfer 7.2.0 (https://surfer.nmr.mgh.harvard.edu/pub/dist/freesurfer/7.2.0) and MRtrix3 (https://www.mrtrix.org), running on macOS Big Sur 11.6 (Apple Inc., Cupertino, CA, USA), performed all preprocessing and subsequent analyses (22). Pial and white-matter surfaces were reconstructed from MPRAGE and FLAIR images to facilitate registration of the quantitative data with the cortical ribbon. Segmentation errors underwent manual correction by blinded assessors. GRAPPATINI generated quantitative T2 (qT2) maps (23); DTI provided apparent diffusion coefficient (ADC) maps; MP2RAGE produced quantitative T1 (qT1) maps (24).
Using FreeSurfer, a customized volume-to-surface algorithm next registered quantitative values at every 25th percentile depth of the cortical ribbon, to the cortical surface of each individual, and then to a common template (fsaverage), as previously detailed (20). This yields quantitative cortical values that unlike qualitative measure are comparable between subjects, as described elsewhere (16,17). Surface maps were smoothed with a 10-mm full-width-at-half-maximum kernel, except for qT1 data, which required a 5-mm kernel.
Outcome measures
The analysis focused on three quantitative MRI indices: qT2, qT1 and ADC. Cortical qT2 reflects tissue water content and increases in the presence of edema or elevated cellularity. Cortical qT1 also correlates with tissue water in addition to loss of structural tissue integrity. Both qT2 and qT1 are inversely associated with tissue iron content. The ADC quantifies water molecule diffusion, increasing with extracellular water and decreasing with intracellular water. Elevated intracellular water might result from cellular swelling or increased cellularity (16,17).
Outcomes
The outcome measures (qT2, qT1 and ADC) were compared between participants with PPTH and six groups: (I) healthy controls and (II) all participants with migraine. Comparisons were also performed to investigate differences in relation to specific subgroups of migraine, who were classified by frequency (episodic or chronic), aura status (with or without aura) and headache status at the time of scan (ictal, non-migraine headache, headache-free). These categories were not mutually exclusive. If a participant experienced both migraine with and without aura, they were included in the group with migraine with aura for analysis. These additional comparisons were performed between participants with PPTH and (III) those with migraine with aura; (IV) those with migraine without aura; (V) those with chronic migraine; and (VI) those with episodic migraine. These analyses had the goal of investigating the potential effects of migraine frequency and aura upon the results.
Post-hoc analyses
Finally, post-hoc analyses were conducted to compare (VII) participants with PPTH using vs. not using preventive medication and (VIII) participants with PPTH vs. ictal participants with migraine.
Statistical analysis
For sociodemographic and clinical data, normally distributed variables are reported as the mean ± SD. Non-normally distributed variables are reported as medians with interquartile ranges. The Shapiro–Wilk test was used to assess normality. Depending on distribution, continuous variables were compared using either Student's t-test or the Wilcoxon rank sum test. Categorical variables were compared using chi-squared. The age distributions between groups were compared using the two-sample Kolmogorov–Smirnov test. All analyses of sociodemographic and clinical data were performed in R, version 4.2.0 (R Foundation, Vienna, Austria).
General linear models (GLMs) in FreeSurfer were used to examine whole-cortex differences for all study comparisons. Each model included age and sex as covariates and was run separately for each hemisphere. The comparisons with migraine or migraine subtypes were additionally adjusted for monthly headache days and preventive medication use as covariates (with these variables not being present in the HC group). For the comparison with ictal participants with migraine only monthly headache days were adjusted for because insufficient group sizes resulted in rank deficiency when preventive use was incorporated. Correction for multiple comparisons was applied across the entire cortex. The cortex was parcellated using the Desikan–Killiany atlas. Multiple comparison correction relied on a cluster-determining threshold of p < 0.05 and a clusterwise threshold of p < 0.05, implemented via Monte Carlo simulation. This method aligns with the approach used in previous qMRI studies (20,25,26). Additionally, these results were corrected using Bonferroni correction for the number of outcomes investigated before significance was accepted.
Associations between altered qMRI measures and clinical variables, including monthly headache days, monthly migraine-like days and disease duration, were then evaluated. Analyses focused on clusters with significant differences between participants with PPTH and HCs and/or those with migraine. Small volume corrections were applied using Monte Carlo simulations with 10,000 iterations. These models incorporated different-onset, different-slope GLMs with a cluster-determining threshold of p < 0.05 and a clusterwise threshold of p < 0.05.
Results
Participants
In total, 105 participants with PPTH, 306 participants with migraine and 160 HCs were enrolled between November 2020 and October 2023. After excluding individuals with incidental findings, the final sample comprised 103 participants with PPTH, 296 participants with migraine and 155 HCs.
Among participants with PPTH, the mean ± SD age was 41.0 ± 11.6 years, and 76 (73.8%) were female. Preventive migraine medication was used by 39 (37.9%) participants and 13 (12.6%) met criteria for medication-overuse headache. Participants with migraine had a mean ± SD age of 41.6 ± 12.4 years and 261 (88.2%) were female. In the HC group, the mean ± SD age was 41.1 ± 11.7 years, with 133 (85.8%) being female. Sociodemographic and clinical characteristics for all groups are summarized in Table 1 and the supplementary material (Table S5) and PPTH phenotypes are shown in Table 2.
Demographic and clinical characteristics of study populations.

*Including riboflavin and magnesium, approved for preventive treatment in Denmark.
**Medication-overuse headache fulfilling International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria (ICHD-3, code 8.2).
***Headache fulfilling ICHD-3 criteria for migraine without aura (ICHD-3, code 1.1).
****Headache not fulfilling ICHD-3 criteria for migraine without aura (ICHD-3, code 1.1).
NA = not applicable; PPTH = persistent post-traumatic headache.
Clinical presentation of participants with persistent post-traumatic headache.

PPTH = persistent post-traumatic headache.
Participants with PPTH were matched for age and age distribution to both participants with migraine (age: p = 0.343; distribution: p = 0.417) and HCs (age: p = 0.671; distribution: p = 0.934). However, sex distribution differed significantly between participants with PPTH and those with migraine (p < 0.001) as well as HCs (p = 0.016). Moreover, preventive medication was significantly less used amongst participants with PPTH (39 of 103; 37.9%) than those with migraine (167 of 296; 56.4%, p = 0.001).
PPTH vs. HCs
Participants with PPTH showed significantly higher qT2 values in the left lateral occipital cortex, compared to HCs (pcluster = 0.0002; Figure 1 and Table 3). Within this region, qT2 values did not correlate with monthly migraine days, monthly headache days or disease duration (all p > 0.05). No group differences were observed in cortical qT1 or ADC values.

Posterior view of both hemispheres and medial view of the left hemisphere. Participants with persistent post-traumatic headache displayed higher quantitative T2 values (red) in the left lateral occipital cortex compared to healthy controls (pcluster = 0.0002). Cluster-determining threshold of p < 0.05 and clusterwise threshold of p < 0.05. Cluster size of 1981.9 mm2, Montreal Neurological Institute coordinates X: −28.4, Y: −92.4, Z: −4.9.
Regions showing differences in quantitative measures.

Results significant after correction based on Monte Carlo simulation and Bonferroni correction marked in bold.
ADC = apparent diffusion coefficient; MCS = Monte Carlo simulation; MNI = Montreal Neurological Institute; PTH = post-traumatic headache; qT1 = quantitative T1; qT2 = quantitative T2.
*Significant at p ≤ 0.05 after clusterwise correction (Monte Carlo simulation) and Bonferroni correction.
PPTH vs. migraine
Compared to participants with migraine, those with PPTH exhibited significantly lower ADC values in the left superior frontal cortex (pcluster = 0.0020) (Figure 2 and Table 3). However, within the PPTH group the ADC values in this cluster were not associated with monthly migraine-like days, monthly headache days or disease duration (all p > 0.05).
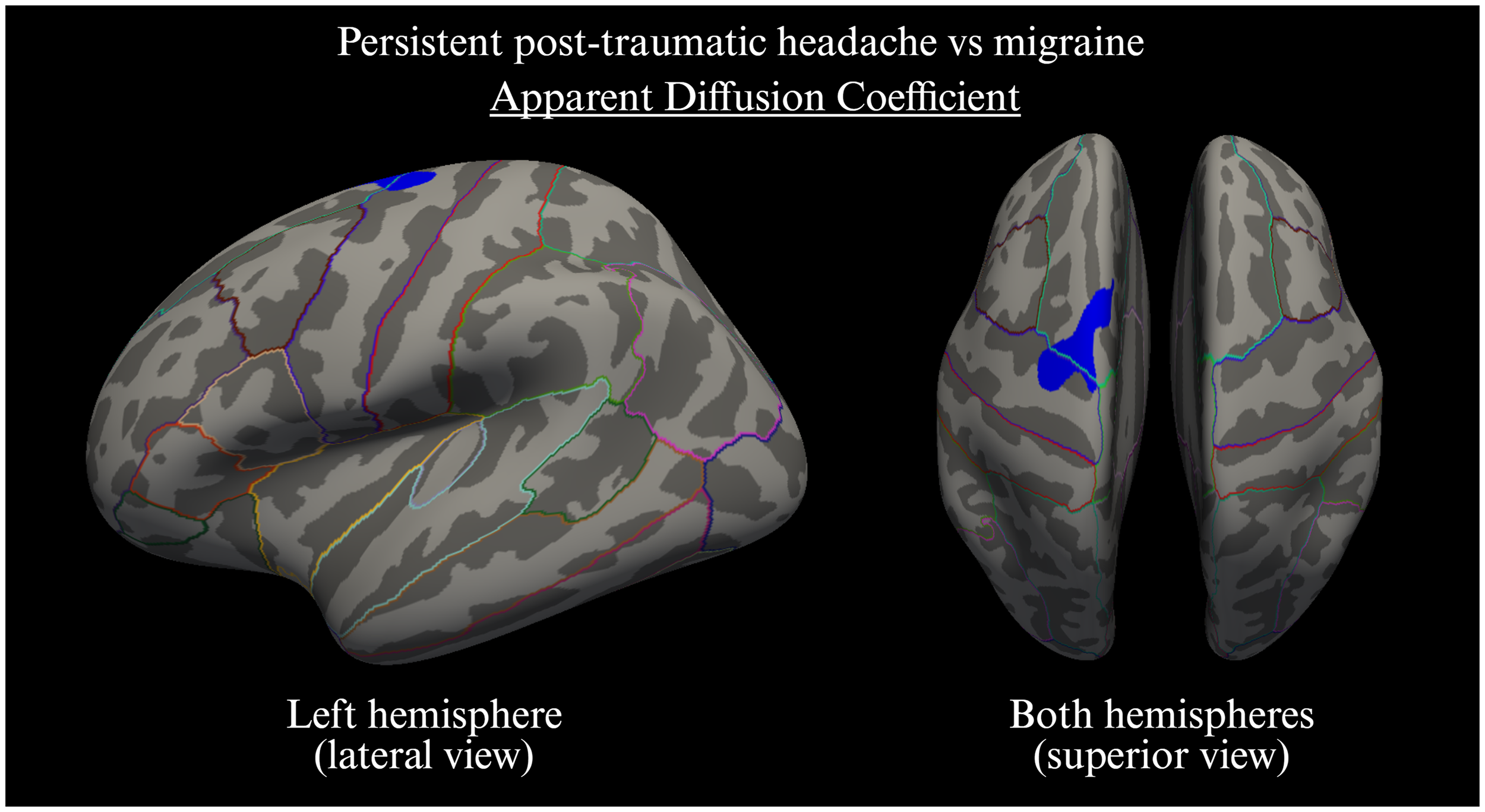
Inferior view of both hemispheres as well as medial and lateral views of the left hemisphere. Participants with persistent post-traumatic headache displayed lower apparent diffusion coefficient values (blue) in the left superior frontal cortex compared to individuals with migraine (pcluster = 0.0020). Cluster-determining threshold of p <0.05 and clusterwise threshold of p <0.05. Cluster size of 594.5 mm2, Montreal Neurological Institute coordinates X: −15.9, Y: −10.2, Z: 63.1.
PPTH vs. migraine subgroups
Participants with PPTH had significantly lower ADC values in the right middle temporal gyrus, compared to those with migraine without aura (pcluster = 0.0002) (Figure 3, upper right, and Table 4). When compared with migraine with aura, participants with PPTH exhibited lower ADC within the right inferior temporal gyrus (pcluster = 0.0052) (Figure 3, upper right, and Table 4), in addition to reduced qT1 values in two clusters within the right middle temporal gyrus (pcluster = 0.0002 and pcluster = 0.0002) (Figure 3, lower left, and Table 4).

Lateral views of the right hemisphere. Upper left: Participants with persistent post-traumatic headache (PPTH) displayed lower apparent diffusion coefficient (ADC) values within the right inferior temporal gyrus compared to participants with migraine with aura (pcluster = 0.0052). Upper right: Participants with PPTH exhibited lower ADC within the right middle temporal gyrus compared to participants with migraine without aura (two clusters, both significant at pcluster = 0.0002). Lower left: Participants with PPTH had lower quantitative T1 values within the right inferior temporal gyrus compared to participants with migraine with aura (pcluster = 0.0002). For area extent and specific coordinate, see Table 4.
Regions showing differences in quantitative measures compared with migraine subtypes.

Results significant after correction based on Monte Carlo simulation and Bonferroni correction marked in bold.
ADC = apparent diffusion coefficient; MCS = Monte Carlo simulation; MNI = Montreal Neurological Institute; PTH = post-traumatic headache; qT1 = quantitative T1; qT2 = quantitative T2.
*Significant at p ≤ 0.05 after clusterwise correction (Monte Carlo simulation) and Bonferroni correction.
No significant differences in qT2, qT1 or ADC values were observed between participants with PPTH and other migraine subgroups. Likewise, there were no significant differences between participants with PPTH using or not using preventive medication.
Discussion
This cross-sectional qMRI study identified cortical microstructural alterations in people with PPTH. Compared with HCs, participants with PPTH exhibited higher qT2 values in the left lateral occipital cortex, suggesting increased water content or cellularity (16,17). Compared with migraine, they had lower ADC values in the left superior frontal cortex, a region involved in cognitive function (27). Additional reductions in qT1 and ADC values were observed in the temporal gyri compared to specific migraine subgroups. These findings indicate that PPTH attributed to mTBI involves a neurobiological profile that differs in part from both migraine and healthy adults. The absence of associations with headache frequency or disease duration might indicate that these imaging features reflect trait-like differences rather than fluctuating disease activity.
Pathophysiological implications
Higher qT2 values in the occipital cortex among participants with PPTH likely reflect increased water content, consistent with extracellular edema or microglial proliferation (16,17). Several lines of research suggest that edema in PPTH could be linked to neuroinflammation following TBI. Less likely causes include relative ischemia, which initially presents with reduced ADC, with increases in chronic stages as qT1 increases, whereas iron deposition linked to neurodegeneration can reduce qT2.
Neuroinflammatory processes are increasingly recognized as contributors to PPTH after mTBI (10). Preclinical studies have shown sustained activation of pro-inflammatory microglia in sensory cortices post-mTBI (10–12). These changes are associated with cytokine release, extracellular edema and neuronal dysfunction (10–12,28). In humans, PET imaging has demonstrated persistent elevation of translocator protein (TSPO) expression in cortical regions, months to years after moderate-to-severe TBI (13,14). Specifically, individuals with repetitive or moderate-to-severe TBIs showed upregulation of TSPO, a marker of microglial activity, in several brain regions including the occipital cortex. Notably, this activity persisted even for months to years following injury. Although these findings are limited by small sample sizes and comparatively greater injury severity, they support a role for chronic microglial activation in PPTH. Preclinical models also suggest that microglial inflammation is instrumental in neuronal changes following TBI (29,30), indicating that neuroinflammation might constitute a potential early treatment target.
Existing evidence and added value
The present study extends former evidence by identifying region-specific cortical microstructural alterations in people with PPTH that align with features of low-grade neuroinflammation. By directly comparing participants with PPTH, migraine and HCs, these qMRI findings provide the first evidence of divergent cortical microstructure between PPTH and migraine.
Occipital inflammation could thus contribute to persistent and unremitting photophobia or visual fatigue, symptoms frequently reported by people with PPTH even outside headache episodes (29–31). Our recent qMRI study has also identified more pronounced occipital qT2 value increases in migraine with aura (20), potentially explaining the lack of difference between migraine and PPTH in this region. Nonetheless, similar though more discrete occipital findings in PPTH might support a broader role for cortical neuroinflammation in photosensitivity. Although the visual cortex is classically involved in migraine with aura, it might have other roles in headache pathophysiology, exemplified for instance by findings of visual cortex hyperexcitability even in migraine without aura (32–34). However, the precise role of microstructure within the visual cortex in PPTH remains to be determined and might be a goal for future studies to investigate.
It merits mentioning that a prior study by Nikolova found decreased T2* in 20 individuals with acute PTH compared to healthy controls, which was interpreted as increased tissue iron content (35). Although superficially at odds with the findings of the present study, several key differences should be noted. First, T2* is sensitive to field inhomogeneities and tissue iron content, whereas the qT2 used in the present study more clearly reflects tissue water. Second, Nikolova (35) investigated acute PTH, whereas we investigated PPTH. Third, Nikolova (35) used a whole-brain analytical approach whereas we relied on a dedicated pipeline to investigate the cortical ribbon.
Our participants with PPTH also exhibited lower ADC values in the superior frontal cortex compared to migraine. This finding suggests extracellular diffusion restriction due to increased intracellular water, which can reflect either cellular swelling or increased cellularity (16,17). Cellular swelling might result from disrupted ion homeostasis and membrane permeability following mTBI. In these scenarios, intracellular water is siphoned from the extracellular space so that the net cortical water as well as qT2 and qT1 often stays the same (36). Cellular swelling is consistent with imaging and histopathological findings in the subacute and chronic stages of TBI (37–39). Persistent changes might results from disruption of ion channels such as NKKC1, AQP4 and SUR1, which contribute to cellular swelling after TBI (40). Such altered fluid homeostasis could theoretically reflect chronic astrocyte adaptations linked to reactive astrocytosis (41,42). Alternatively, persistent microglial or astrocytic proliferation could represent a chronic pro-inflammatory state (10–12). Although a possibility, increased cellularity would often be associated with increases in qT2 or qT1, which we did not observe in the frontal cortex.
Finally, persistent changes in neural activity might promote ADC decreases. Recent findings demonstrate reproducible task-related decreases in ADC during activation in healthy volunteers (43), and prior studies have shown altered functional connectivity with frontal regions in PTH (8). Because the superior frontal cortex supports working memory and executive control (27), microstructural alterations might contribute to cognitive symptoms such as concentration difficulties often reported in people with PPTH (2). Although the ADC value difference was not observed relative to HCs, the larger migraine sample could have increased power to detect subtle effects. Future studies are therefore needed to confirm this finding in larger cohorts.
Further differences were noted when comparing individuals with PPTH to the separate subgroups of migraine with and without aura. Specifically, individuals with PPTH displayed reduced ADC within the right middle temporal gyrus compared to migraine without aura. Similar findings were made within the inferior temporal gyrus when comparing with migraine with aura, in addition to a reduction in qT1. The middle and inferior temporal gyri are involved in more complex visual-auditory processing, including semantic memory, visual object recognition and social cognition (44,45). These myriad functions could theoretically relate to diffuse cognitive burden and concentration difficulties in individuals with PPTH (46). However, given that differences were absent when comparing PPTH with the whole group of participants with migraine, additional neuroimaging data will be necessary to validate these findings and clarify their relevance to disease mechanisms.
Together, our results suggest that PPTH is not simply a post-traumatic analogue of migraine but shares certain overlapping cortical features at the same time as possessing others that are unique. The distribution of alterations across sensory and executive cortices aligns with a model of low-grade, chronic neuroinflammation sustaining persistent symptoms following mTBI.
Strengths and limitations
This study is the first to apply multimodal qMRI to individuals with PPTH to directly compare cortical microstructure across PPTH, migraine and HCs. All imaging was acquired using a standardized, high-resolution protocol on a single 3.0 Tesla MRI scanner, reducing inter-subject variability. T2 mapping was performed using anisotropic voxels, the standard for this sequence, which improves in-plane resolution at the same time as maintaining a scan time that minimizes motion artifacts (47). Accelerated T2 mapping techniques might further enhance spatial resolution in future studies (47). The combined use of qT2, qT1, and ADC values enabled multimodal assessment of cortical microstructure, providing indirect markers of neuroinflammation and tissue integrity. Surface-based analysis enhanced anatomical precision, and all statistical models were adjusted for age and sex to account for demographic confounding.
Several limitations must be acknowledged. First, the cross-sectional design precludes inference about causal relationships or temporal evolution. Longitudinal data are needed to clarify whether the observed alterations precede or follow symptom persistence. Second, sex imbalance across groups could have introduced bias, although this was accounted for by adjusting all models for sex differences. Third, qMRI measures differences in tissue microstructures, which can arise from more than one contributing factor (16,17). Fourth, participants with PPTH were not matched to comparison groups by injury mechanism or site of trauma. Lastly, the PPTH cohort exhibited heterogeneity in medication use and trauma characteristics, and most (91.3%) were experiencing a headache, although the character differed. Although we might speculate that this increases biological variability, it enhances the generalizability of findings to the broader clinical population with PPTH after mTBI.
Conclusions
Our multimodal qMRI findings demonstrate distinct cortical microstructural alterations in people with PPTH. Higher qT2 values in the occipital cortex, compared to HCs, might reflect edema or increased cellularity. Lower ADC values in the superior frontal cortex, relative to migraine, could indicate cellular swelling and potentially relate to cognitive symptoms. These alterations suggest a pathophysiological basis involving chronic low-grade neuroinflammation. Taken together, our results support a model, in which PPTH represents a biologically distinct disorder, partially diverging from migraine in both cortical profile and underlying mechanisms.
Article highlights
Persistent post-traumatic headache was associated with higher qT2 in the occipital cortex, potentially related to edema or greater cellularity.
Lower ADC in the frontal cortex differentiated post-traumatic headache from migraine and might reflect cellular perturbations and swelling after brain injury.
These findings might indicate low-grade neuroinflammation and cellular swelling in persistent post-traumatic headache, which might contribute to visual and cognitive symptoms.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251409317 - Supplemental material for Cortical microstructure in persistent post-traumatic headache, migraine and healthy controls: A quantitative magnetic resonance imaging study
Supplemental material, sj-docx-1-cep-10.1177_03331024251409317 for Cortical microstructure in persistent post-traumatic headache, migraine and healthy controls: A quantitative magnetic resonance imaging study by Rune Häckert Christensen, Haidar Muhsen Al-Khazali, Messoud Ashina and Håkan Ashina in Cephalalgia
Footnotes
Author contributions
Study concept and design: MA and HA. Data acquisition and analysis: RHC and HMA. Drafting of the manuscript: RHC, HMA and HA. Critical revision of the manuscript: RHC, HMA, MA and HA. Funding acquisition: MA and HA.
Data availability
The corresponding author will provide necessary data and materials of the study upon reasonable request from a qualified investigator.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HMA has received personal fees from Lundbeck, Pfizer, and Teva, outside of the submitted work. RHC has received personal fees from Teva, Lundbeck and Pfizer, outside of the submitted work, and serves as Section Editor for the Journal of Pain Research. MA has received personal fees from AbbVie, Amgen, AstraZeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva, outside of the submitted work. MA is also a Primary Investigator for ongoing clinical trials sponsored by AbbVie, Lundbeck and Pfizer. MA also serves as an Associate Editor of Brain and The Journal of Headache and Pain. HA has received personal fees from AbbVie, Lundbeck, Pfizer and Teva, outside of the submitted work. HA also serves as an Editorial Board Member of The Journal of Headache and Pain.
Ethical considerations
The study protocol received approval from the ethics committee of the Capital Region of Denmark (Identifier: H-20033264). All participants provided their written informed consent prior to any study procedures.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was received support by research grants from the Lundbeck Foundation (R403-2022-1352 to HA and R310-2018-3711 to MA).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
