Abstract
Objective
The term “post-traumatic migraine” (PTM) has been used to describe post-traumatic headaches (PTHs) that have associated migraine features, but studies of this relationship are lacking. The objective of the present study was to determine whether PTH correlates strongly with migraine symptoms among youth with concussion.
Methods
Twenty-three symptoms were analyzed from a retrospective cohort of 1953 pediatric patients with concussion. A principal component analysis (PCA) with oblique Promax rotation was conducted to explore underlying symptom relationships in the full cohort and in subcohorts stratified by the presence (n = 414) or absence (n = 1526) of premorbid headache.
Results
The mean patient age was 14.1 years; 63% were male. Headache was the most common postconcussion symptom, acknowledged by 69.4% of patients. When considering the full cohort, the PCA demonstrated clustering of headache with photophobia, phonophobia, nausea, dizziness, and neck pain. Similar clustering was present among patients without premorbid headaches. Repeating the analysis in the patients with preconcussion headaches led to elimination of neck pain from the cluster.
Conclusions
PTH correlates strongly with other migraine symptoms among youth with concussion, regardless of premorbid headaches. This clustering of migraine symptoms supports the existence of PTM as a distinct clinical entity in some patients.
Introduction
Concussions result in a complex constellation of somatic, cognitive, and emotional symptoms, with headache being the most common symptom reported. The term “post-traumatic migraine” (PTM) has been used over the past several decades to describe postconcussion headaches with associated migrainous features (including nausea, vomiting, photophobia, and phonophobia) that develop during injury recovery (1–5) or, in rarer cases, within minutes of head impact (6,7). The International Classification of Headache Disorders, third edition (ICHD-3) classifies headache attributed to trauma, or post-traumatic headache (PTH), by the nature and severity of the traumatic brain injury (TBI), the temporal relationship between TBI and headache onset, the headache duration following injury, and the presence or absence of a whiplash injury (8). The classification system does not address specific headache features or recognize PTM as a distinct clinical entity. All references to PTM in this manuscript should be considered theoretical. In an ICHD-3 context it currently represents PTH with a migraine-like phenotype.
The majority of patients who report headaches following concussion have episodes that resemble either migraine or tension-type headaches (5,9–12). The common clinical features shared between PTH and migraine in some patients suggest that common biological processes may be involved in generating both headache types. Previous research has shown that PTH characteristics may serve as predictors of concussion outcomes (13–17). Patients with the clinical constellation of PTM tend to have greater overall postconcussion symptom burdens and protracted concussion recoveries when compared to patients with PTH without migraine features and patients without PTH (14–17).
Several gaps exist, however, in the research tying together PTH and PTM. For example, what if certain concussions simply lead to more numerous postconcussion symptoms? The grouping of PTH with one or more associated features of migraine (nausea, vomiting, photophobia, or phonophobia) would occur commonly by chance alone and would occur more often with the injuries that cause higher overall symptom burdens. This chance occurrence might appear to represent PTM; yet, the apparent migraines following concussion would be no more likely than PTH associated with non-migraine postconcussion symptoms such as sadness, fatigue, or difficulty concentrating. Research designs that recruit individual patients based on their migraine-like presentations after concussion cannot answer whether PTM represents this chance occurrence or a distinct clinical entity. To resolve the complex interrelationships among postconcussion symptoms, all symptoms from a large number of concussion patients must be analyzed in aggregate.
Principal component analysis (PCA) is a technique that identifies clusters of variables or principal components, such that variables within a given cluster correlate strongly with other variables in the same cluster and correlate weakly with variables in different clusters. PCA can be used to study the interrelationships among postconcussion symptoms, even though all symptoms exhibit some degree of correlation. Both PCA and factor analysis techniques have been used in the development of postconcussion symptom scales. These studies sought to maximize parsimony among scale items and optimize scale validity. To accomplish these aims investigators used symptom scoring from non-concussed individuals to derive factor structure (18,19), focused on the factors related to cognitive domains only (20), included TBIs of any severity (21), limited recruitment to patients with acute concussion (<7 days) (22), extended recruitment to patients with distant injuries (21,23), or discarded symptoms because they did not fit the factor solution (18,19,22,23). A study has not been conducted that explores the specific relationships among migraine symptoms following concussion in youth.
The objective of the current study was to determine whether PTH correlates strongly with migraine symptoms among youth with concussion. Using PCA to demonstrate the inter-relationships among all postconcussion symptoms, we hypothesized that PTM symptoms (headache, nausea, vomiting, photophobia, and phonophobia) would cluster as a principal component when considering the full cohort and when stratifying patients by the presence or absence of premorbid headaches. We further hypothesized that headaches would be more likely to occur when migraine symptoms were present and that the probability of headache would not correlate merely with postconcussion symptom frequencies or mean symptom scores. The finding of a strong correlation among migraine symptoms would support the existence of PTM as a distinct clinical entity in some patients with concussion. If PTM exists, these patients might benefit from migraine-specific therapies.
Materials and methods
Standard protocol approvals
The study was approved by the institutional review board at Nationwide Children’s Hospital.
Participants
We conducted a retrospective cohort study of pediatric patients with concussion presenting to a pediatric sports medicine clinic at a large children’s hospital in Columbus, Ohio, between June 2012 and September 2014. The cohort comprised 1953 patients, aged 10–19 years, with the initial clinical evaluation occurring within 30 days of the concussive injury. The mean patient age was 14.1 years (median 14 years, SD 2.09); 63.4% were male. The mean interval between concussion injury and the initial clinical evaluation was 10.1 days (median nine days, range one to 30 days, SD 6.32). The concussion diagnosis was determined clinically for each patient, based on the injury mechanism(s), history, and examination. Diagnoses generally followed the operational definition of concussion set forth in the Consensus Statement on Concussion in Sport (24). Patients were excluded from the study if they did not have a TBI, they had clinical features consistent with moderate or severe TBI, or their concussion occurred > 30 days before their initial clinical evaluation. When patients had more than one concussion within the 28-month study period, only the symptom scores from the first documented injury were analyzed.
Measures
Postconcussion symptom frequencies, symptom scores, and headache probabilities for each symptom.

Statistical analysis
Descriptive statistics were calculated. Conditional headache probabilities were calculated for each postconcussion symptom. Relationships between symptom frequencies, mean symptom scores, and the conditional headache probabilities were assessed using Pearson product-moment correlation coefficients.
A PCA was conducted to explore the interrelationships among the 23 postconcussion symptoms present on the day of clinical evaluation. All R-matrix correlations were <0.8. The Kaiser-Meyer-Olkin (KMO) measure verified the sampling adequacy, KMO = 0.95. Bartlett’s test of sphericity was significant, p < 0.001. In contrast to previous studies of postconcussion scale validation, the aim of this study was to produce clinically meaningful symptom categories and to fit all symptoms of interest into the model, so a conservative approach to data reduction was used. The initial extraction reduced the 23 postconcussion symptoms to six principal components based on the scree plot and the number of eigenvalues > 0.7 (Jolliffe’s criterion) (28). Had the number of eigenvalues > 1 been used as the primary criterion, five components would have been retained with excessive clustering of symptoms in the first component. The six components explained 68.3% of the variance. We assumed that all postconcussion symptoms would be correlated to some degree and that the reduced components also would be correlated, so an oblique Promax rotation (kappa = 4) with Kaiser normalization was used. Identical six-component analyses were repeated to explore symptom structure among those patients with premorbid headaches (n = 414; KMO = 0.93; Bartlett’s test of sphericity, p < 0.001) and without pre-morbid headaches (n = 1526; KMO = 0.95; Bartlett’s test of sphericity, p < 0.001). Structure matrices and pattern matrices for each corresponding PCA had identical symptoms clustered to each principal component. All statistical analyses were performed using SPSS version 21 (SPSS Inc, Chicago, IL). The significance threshold was set at 5%.
Results
In a cohort of 1953 pediatric patients with concussion, headache was the most common postconcussion symptom, reported by 69.4% of the patients. Loss of consciousness occurred in 16.3%, and retrograde and/or anterograde amnesia occurred in 31.8%. Table 1 lists the postconcussion symptom frequencies, the mean symptom scores, and the conditional probabilities of headache for each of the 23 symptoms. As expected, the symptom frequencies and the mean symptom scores correlate (r = 0.98; p < 0.001). The conditional probabilities of headache across postconcussion symptoms were each higher (range 0.86–0.96) than the headache probability in the full cohort (0.69), reflecting the complex nature of concussion as the cause of a constellation of symptoms that tend to resolve together. However, headache probabilities varied in relation to the type of postconcussion symptom assessed. When patients reported migraine symptoms (vomiting, nausea, photophobia, or phonophobia), the probability was high that headache was also present (Table 1). Lower headache probabilities were seen with nervousness, memory difficulties, and concentration difficulties. When all other non-headache symptoms were denied (n = 289), the probability of headache was only 0.09. There was no evidence that headache occurrence correlated with overall symptom burden; correlations were not present between headache probability and symptom frequencies (r = 0.04; p = 0.86) or headache probability and mean symptom scores (r = 0.05; p = 0.84).
Principal component analysis of the full cohort.

Kaiser-Meyer-Olkin (KMO) 0.949; Bartlett’s test of sphericity, p < 0.001.
Principal component analysis of patients with premorbid headaches.
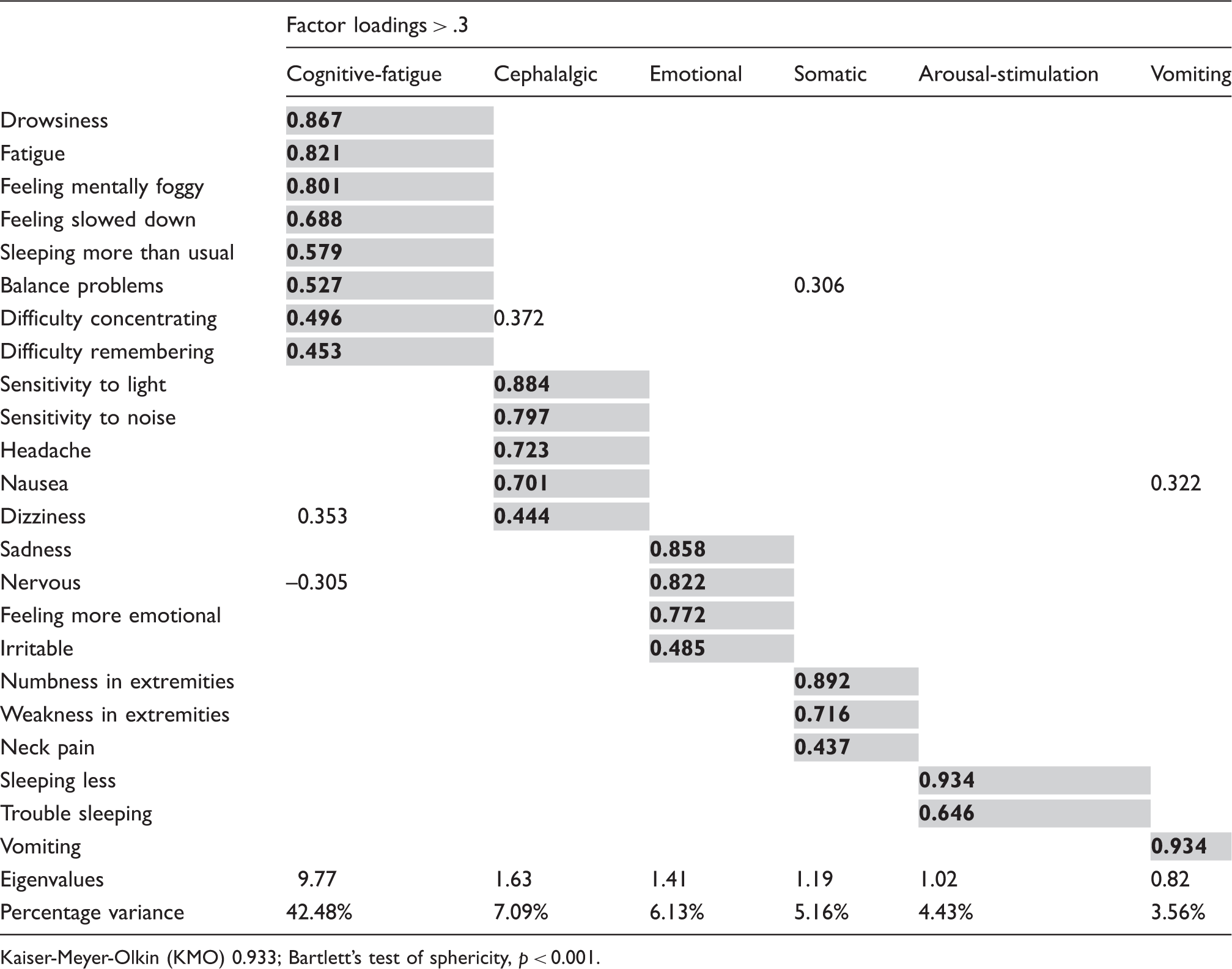
Kaiser-Meyer-Olkin (KMO) 0.933; Bartlett’s test of sphericity, p < 0.001.
Discussion
In a large cohort of youth with postconcussion symptoms of 30 days or less, PCA established underlying symptom structure that links PTHs with migraine symptoms. Analysis of the full cohort demonstrated a strong correlation among headache, nausea, photophobia, phonophobia, dizziness, and neck pain that distinguished these symptoms from other postconcussion symptoms with weaker correlations. The cephalalgic symptom cluster remained regardless of the presence or absence of premorbid headaches. Analysis of the patients with premorbid headaches led to removal of neck pain from the cephalalgic symptom cluster, a refinement that more closely resembles the IHS diagnostic criteria for primary migraine (8). The strong correlation among migraine symptoms following concussion supports the existence of PTM as an etiologic entity in some patients.
If the interrelationships among postconcussion symptoms were related only to the intensity of the concussion injury, the probability of headache would increase as a function of increasing symptom burden (symptom frequency and mean symptom scores), which did not occur. The present study demonstrates that conditional headache probabilities varied according to the type of postconcussion symptom, more so with migraine symptoms and less so with cognitive and emotional symptoms. Additionally, the persistence of the cephalalgic symptom cluster in patients without premorbid headaches argues against PTM representing only an exacerbation of preexisting migraines.
How concussions cause recurrent headaches is largely unknown. Concussion and primary migraine may share some common pathophysiologic pathways (3), which include involvement of the endogenous opioid system (29,30), release of excitatory amino acids (31,32), changes in cerebral blood flow (33), and alterations of magnesium-dependent mechanisms (34). Prior research also supports a relationship between PTH and migraine symptoms, although the study aims differed from the present study. Kontos et al. conducted factor analyses of the 22-item Post-Concussion Symptom Scale (PCSS) to evaluate symptom structure among concussed and non-concussed athletes aged 13–22 years (22). The concussion assessments were limited to acute postconcussion symptoms (≤7 days). Headache, photophobia, and phonophobia clustered with symptoms of cognitive impairment and fatigue in their four-factor solution (22). Nausea was removed from the model because it cross-loaded on two factors. Vomiting and “feeling more emotional” constituted a separate factor. Similarly, Joyce et al. used factor analysis to demonstrate a three-factor solution for a 19-item version of the PCSS (23). All patients had concussion. Age ranged from 6–22 years, and the interval between concussion and symptom reporting ranged from one to 547 days (mean 22.8 days). Headache, nausea, photophobia, and phonophobia loaded with “trouble falling asleep” and “sleeping more than usual” (23). Vomiting did not load onto a factor. Several differences exist between these studies and the present study. By using an interval of 30 days or less from injury to initial assessment, the constellation of early and late postconcussion symptoms in the cohort could be assessed while minimizing the numbers of patients who might misattribute their primary migraine symptoms to a distant concussion injury. Our conservative approach to data reduction also led to all symptoms loading to a principal component with clinically logical symptom clusters.
In our study, however, not all symptoms clustered as we had predicted. The absence of vomiting from the cephalalgic component is a deviation from our hypothesis that all migraine symptoms would correlate strongly, yet vomiting and headache were tightly linked. While only 3.4% of patients reported vomiting, when vomiting was present the conditional probability of headache was higher than with any other postconcussion symptom. Neck pain and dizziness grouped with the other cephalalgic symptoms, even though they are not principal features of migraine. Not all symptoms that cluster with the cephalalgic component necessarily constitute PTM. Both neck pain and dizziness are common with concussion and with PTH. The complaint of neck pain by some of our patients may represent a whiplash-type injury, which distinguishes PTH subtypes (8). Neck pain is commonly associated with primary headaches, and some adolescents acknowledge neck pain as a feature of primary migraine (35,36). Neck pain disability is related to the frequency of migraine attacks (37). Vertigo has long been recognized as an associated symptom of migraine with aura and migraine without aura (38).
We acknowledge several study limitations. We did not evaluate migraine-like symptoms among individual patients following concussion. Instead, we demonstrated that a strong correlation exists among migraine symptoms in the full cohort, regardless of the presence or absence of premorbid headaches. It is possible that the symptoms of interest clustered in the current study, but the individual patients did not have a PTM phenotype, which would mean that the relationship among migraine symptoms does not represent PTM. While possible, this alternate interpretation does not match prior research or our own clinical experience. Neuroimaging was not performed on all patients, so structural brain injury was not ruled out. There have been several neuroimaging studies in mild TBI patients demonstrating macroscopic injuries with magnetic resonance imaging (MRI) (39,40). The presence of brain injury could oppose the apparent biologic and symptomatic similarities between a theoretical PTM phenotype and primary migraines. We included patients with an interval of up to 30 days between their concussion and their clinic evaluations, so we cannot prove from our data that headaches began within seven days of concussion (an ICHD-3 diagnostic criterion for PTH) in all patients (8). The 30-day interval allowed analysis of postconcussion symptoms beyond the acute recovery period of seven days, while excluding patients with distant injuries. It is possible that symptom structure differs during the acute recovery period compared to the subacute period, and this potential difference was not studied. We did not characterize premorbid headaches. Patients with premorbid migraine might differ from patients with premorbid tension-type headaches in terms of postconcussion symptom structure. While we did not account for the mechanisms of injury in this study, the majority of patients had sports-related concussions. It is possible that sports-related concussions differ from other concussion injuries in terms of symptom structure. Finally, our symptom scale has not been validated. We used symptoms that represent an amalgamation of other scales and assessments, all of which include the symptoms of interest: headache, nausea, vomiting, photophobia, and phonophobia.
Conclusion
PTH correlates strongly with other migraine symptoms among youth with concussion. The correlation persists regardless of the presence or absence of premorbid headaches. The clustering of migraine symptoms supports the existence of the theoretical construct of PTM as a distinct clinical entity in some patients, which is worthy of further exploration. These patients might benefit from migraine-specific treatments.
Clinical implications
The present study demonstrates that a strong correlation exists among migraine symptoms in youth with concussion. The migraine symptom cluster persists regardless of the presence or absence of premorbid headaches. The strong correlation among migraine symptoms supports post-traumatic migraine as a distinct clinical entity in some patients following concussion.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
Acknowledgments
We would like to thank the physicians involved in the Division of Sports Medicine at Nationwide Children’s Hospital for allowing us access to their patient data. We would also like to thank Dr Kathi Kemper for her helpful recommendations regarding manuscript preparation.
We declare that all authors made significant contributions toward the study design and/or the acquisition and interpretation of the data. All authors were involved in drafting and/or revising the manuscript. All authors approved the final manuscript version.
