Abstract
Introduction
Although migraine and persistent post-traumatic headache often share phenotypic characteristics, few studies have interrogated the pathophysiological differences underlying these headache types. While there is now some indication of differences in brain structure between migraine and persistent post-traumatic headache, differences in brain function have not been adequately investigated. The objective of this study was to compare static and dynamic functional connectivity patterns in migraine versus persistent post-traumatic headache using resting-state magnetic resonance imaging.
Methods
This case-control study interrogated the static functional connectivity and dynamic functional connectivity patterns of 59 a priori selected regions of interest involved in pain processing. Pairwise connectivity (region of interest to region of interest) differences between migraine (n = 33) and persistent post-traumatic headache (n = 44) were determined and compared to healthy controls (n = 36) with ANOVA and subsequent t-tests. Pearson partial correlations were used to explore the relationship between headache burden (headache frequency; years lived with headache) and functional connectivity and between pain intensity at the time of imaging and functional connectivity for migraine and persistent post-traumatic headache groups, separately.
Results
Significant differences in static functional connectivity between migraine and persistent post-traumatic headache were found for 17 region pairs that included the following regions of interest: Primary somatosensory, secondary somatosensory, posterior insula, hypothalamus, anterior cingulate, middle cingulate, temporal pole, supramarginal gyrus, superior parietal, middle occipital, lingual gyrus, pulvinar, precuneus, cuneus, somatomotor, ventromedial prefrontal cortex, and dorsolateral prefrontal cortex. Significant differences in dynamic functional connectivity between migraine and persistent post-traumatic headache were found for 10 region pairs that included the following regions of interest: Secondary somatosensory, hypothalamus, middle cingulate, temporal pole, supramarginal gyrus, superior parietal, lingual gyrus, somatomotor, precentral, posterior cingulate, middle frontal, fusiform gyrus, parieto-occiptal, and amygdala. Although there was overlap among the regions demonstrating static functional connectivity differences and those showing dynamic functional connectivity differences between persistent post-traumatic headache and migraine, there was no overlap in the region pair functional connections. After controlling for sex and age, there were significant correlations between years lived with headache with static functional connectivity of the right dorsolateral prefrontal cortex with the right ventromedial prefrontal cortex in the migraine group and with static functional connectivity of right primary somatosensory with left supramarginal gyrus in the persistent post-traumatic headache group. There were significant correlations between headache frequency with static functional connectivity of left secondary somatosensory with right cuneus in the migraine group and with static functional connectivity of left middle cingulate with right pulvinar and right posterior insula with left hypothalamus in the persistent post-traumatic headache group. Dynamic functional connectivity was significantly correlated with headache frequency, after controlling for sex and age, in the persistent post-traumatic headache group for one region pair (right middle cingulate with right supramarginal gyrus). Dynamic functional connectivity was correlated with pain intensity at the time of imaging for the migraine cohort for one region pair (right posterior cingulate with right amygdala).
Conclusions
Resting-state functional imaging revealed static functional connectivity and dynamic functional connectivity differences between migraine and persistent post-traumatic headache for regions involved in pain processing. These differences in functional connectivity might be indicative of distinctive pathophysiology associated with migraine versus persistent post-traumatic headache.
Keywords
Introduction
Post-traumatic headache (PTH) is the most common symptom following a mild traumatic brain injury (mTBI) (1). PTH is defined according to its onset within seven days of the TBI (2). When PTH continues for 3 months or longer, it is classified as persistent PTH (PPTH) (2). Other than the time interval between TBI and the onset of PTH, there are no clinical characteristics that assist with classifying PPTH. Often, the symptoms of PPTH resemble those of migraine, yet only a few studies have investigated brain structural differences underlying PPTH and migraine (3,4), and functional differences between migraine and PPTH are unknown.
To date, most functional connectivity studies in patients with migraine or PPTH have explored static functional connectivity (sFC), where a single measurement of connectivity is generated across the entire time-series of functional data. Static functional connectivity is often explored by calculating the Pearson correlation between two region-of-interest (ROI) signals, yielding a single (time-invariant) correlation value per ROI pair. More recent efforts have explored connectivity that changes from moment to moment over time, termed dynamic functional connectivity (dFC). Dynamic functional connectivity is defined as time-varying connectivity, and it is typically evaluated with sliding window correlations.
Static and dynamic functional connectivity analyses provide datasets that are complementary to one another when evaluating brain functional connectivity. Intermittent connectivity and variations in connectivity strength over time may be obscured by measures of sFC, but observable with measures of dFC. For example, two regions could have a strong periodic connection that is obscured when averaged over the entire time-series.
For this paper, we measured sFC and dFC in PPTH attributed to mild traumatic brain injury (mTBI), migraine, and healthy controls. The goal of this study was to compare sFC and dFC between migraine and PPTH. The sFC and dFC of ROI pairs that differed between migraine and PPTH were compared to healthy controls for interpretation purposes. For ROI pairs that showed differences in connectivity between migraine versus PPTH and between migraine or PPTH versus healthy controls, correlations between connectivity and headache burden were investigated.
Methods
Research participants
Research participants were enrolled from Mayo Clinic Arizona, and from the Phoenix VA Health Care System using convenience sampling. Healthy controls were enrolled from the Phoenix area using advertisements and word of mouth. Participants were men and women aged 18 to 65 years. Individuals with PPTH were excluded if they had a history of moderate or severe TBI, history of migraine, or any other headache type prior to injury with the exception of infrequent episodic tension-type headaches (3 or fewer days per month) being allowed. Those with migraine and healthy controls were excluded if they had a personal history of TBI. Furthermore, healthy controls were excluded if they had any history of migraine or if they had tension-type headaches on 4 or more days per month. A headache specialist confirmed all headache diagnoses using International Classification of Headache Disorders 3rd edition, beta version (ICHD-3 beta) diagnostic criteria (2).
This study was approved by the Mayo Clinic Institutional Review Board, the Phoenix VA Health Care System Institutional Review Board, and the U.S. Department of Defense Human Research Protection Office. All research participants provided written informed consent, prior to study participation. Data from some of the research participants included in this study have been included in other publications (4–6).
Due to the high frequency of headaches experienced by research participants in this study, participants underwent brain imaging regardless of their pain level at the time of imaging. Current pain intensity was collected at the time of imaging. Patients were asked to report their current pain level on a scale of 0 to 10 where 0 is no pain and 10 is the most severe pain imaginable.
Questionnaires
All research participants were screened for TBI using the Ohio State TBI Identification method (7). Participants with migraine and those with PPTH provided detailed information about their headaches. All participants completed the Beck Depression Inventory and the State-Trait Anxiety Inventory (8–10).
Imaging data acquisition
All participants were imaged on a 3-Tesla Siemens (Erlangen, Germany) MAGNETOM Skyra scanner located at Mayo Clinic Arizona. Structural sequences included: a) high-resolution 3D T1-weighted sagittal magnetisation prepared rapid gradient echo (MP-RAGE) series with TE (echo time) = 3.03 ms, TR (repetition time) = 2400 ms, 1 × 1 × 1.3 mm3 voxels, and 256 × 256 mm field-of-view (FOV); b) T2-weighted images in axial plane with TE = 84 ms, TR = 6800 ms, 1 × 1 × 4 mm3 voxels, and 256 × 256 mm FOV; and c) blood oxygenation level dependent (BOLD) resting-state sequence with TE = 27 ms, TR = 2500 ms, and 4 × 4 × 4 mm3 voxels, and 256 × 256 mm FOV. T2 images were used in conjunction with T1 images to rule out gross anatomical abnormalities.
Resting-state collection and data preprocessing
Ten minutes of BOLD resting-state imaging data were collected for each participant. Prior to the scan, participants were instructed to keep their eyes closed but to remain awake, to relax, and to try to clear their minds. All imaging was preprocessed using SPM 8 (Wellcome Department of Cognitive Neurology, Institute of Neurology, London, UK) and the SPM toolbox DPARSF (11) and interfaced with MATLAB version 11.0 (MathWorks, Natick, MA, USA). All data were processed on a single Macintosh computer (OS X Lion 10.7. software) to avoid postprocessing inconsistencies that may arise when using more than one workstation (12).
Resting-state data were processed according to standard SPM methodology (13), which included the following: Data were slice-time corrected, corrected for motion, and realigned to the first volume; skull and non-brain tissue were removed, and data were spatially smoothed to 6 mm, full width and at half maximum (FWHM); the resting-state images of each participant were first aligned to their own T1-weighted scan and then transformed to the standardised Montreal Neurological Institute (MNI) 305 template to enable signal averaging across all participants. To capture low-frequency components, data were bandpass filtered to between 0.01 to 0.1 Hz (14). As dFC is prone to noise artifacts due to motion, cardiac, and respiratory rhythms (15), signals of no interest (including white matter signal, cerebrospinal fluid signal, and global mean signal) were regressed from the data. Variance due to head motion was regressed using a framewise displacement model (16). In addition, prior to including participants in the final analysis, all scans were checked for motion using the DPARSF head motion output file, allowing for data from participants that exceeded the predetermined motion limit ( > 2 mm movement in scanner) to be excluded.
X, y, z-coordinates are labeled in Montreal Neurological Institute (MNI) space. An 8 mm sphere was drawn around each ROI to explore functional connectivity patterns.

DLPFC: dorsolateral prefrontal cortex; Inf: inferior; Lat: lateral; ROI: region of interest; Sup: superior; VMPFC: ventromedial prefrontal cortex.
Static functional connectivity analysis
Eight millimetre spheres were drawn around each of the 59 ROIs, then time courses over each seed region were extracted, and Spearman correlation matrices were computed. For each group, the sFC among the ROIs was explored, and only those ROI pairs that had an absolute mean static correlation above 0.1 for at least one of the participant groups (i.e. PPTH, migraine, controls) were included for further analysis. This thresholding technique ensured that those ROIs that were not meaningfully connected in any group were excluded from further analysis.
Analyses of variance with subsequent t-tests, controlling for sex and age, were used to compare the sFC differences between migraine and PPTH, as well as the difference in sFC between PPTH and healthy controls and between migraine and healthy controls. The Benjamini-Hochberg procedure was performed to correct for multiple comparisons (28). For the ROI pairs that had significant sFC differences between migraine and PTH and between migraine or PPTH with healthy controls, age- and sex-corrected correlations (using Pearson's r) were explored between headache characteristics (years with headache and headache frequency) and sFC and between pain intensity at the time of imaging and sFC.
Dynamic functional connectivity analysis
A sliding window correlation analysis (29) (Figure 1) was performed for each ROI pair, and the standard deviation of the resulting time course values were calculated. Ten minutes of resting-state data was collected, and window lengths of 60 seconds were selected to increase confidence of capturing time-varying fluctuations that can be missed using shorter window lengths (30). These dFC values were compared between groups (migraine vs. PPTH; migraine vs. healthy controls; PPTH vs. healthy controls) using ANOVA and subsequent t-tests and controlling for sex and age. The Benjamini-Hochberg procedure was used to correct for multiple comparisons. For those ROI pairs with significant dFC differences between migraine and PPTH and between migraine or PPTH with healthy controls, correlations (using Pearson's r) corrected for sex and age were explored between dFC and headache characteristics (years with headache and headache frequency) and between pain intensity at the time of imaging and dFC.
Illustration of how the dynamic functional connectivity measure is derived for a single patient and a single region-of-interest (ROI) pair. The figure only shows the first 100 seconds of BOLD data collection. The first window encompasses the first 24 measurements. The correlation between the two ROI time courses over the first 24 measurements (r = −0.23) is considered the correlation within the first window. The window then slides forward 1 measurement to encompass measurements 2 through 25 and a second sliding window correlation (r = −0.28) is generated. This process is repeated over the entire time course (not shown). The standard deviation of the time course is the measure of dynamic functional connectivity for a single patient for the given ROI pair. In this example, the standard deviation of the sliding window correlations for this first 100 seconds is 0.08. B.H.: Benjamini-Hochberg correction.
Statistical analyses of demographic and headache characteristic data
Analyses of variance with subsequent two-tailed t-tests, Kruskal-Wallis with subsequent Mann Whitney U tests, or Fisher's exact tests were used as appropriate for comparing demographic data among participant cohorts.
Results
Research participant characteristics
Subject demographics.
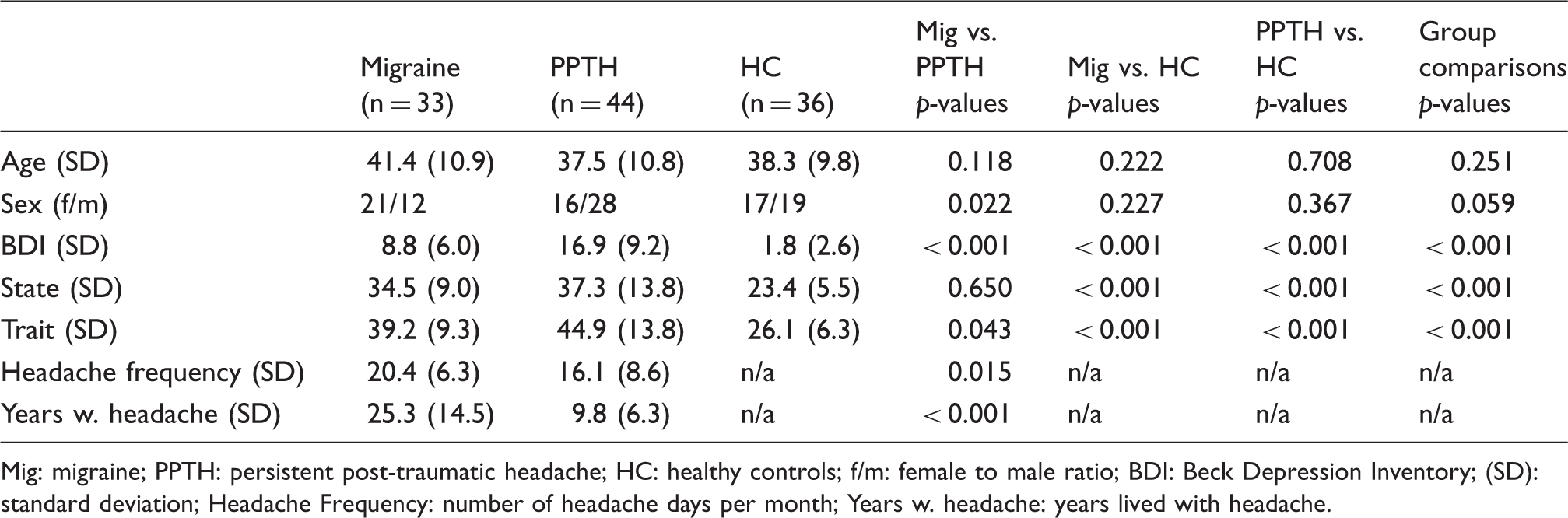
Mig: migraine; PPTH: persistent post-traumatic headache; HC: healthy controls; f/m: female to male ratio; BDI: Beck Depression Inventory; (SD): standard deviation; Headache Frequency: number of headache days per month; Years w. headache: years lived with headache.
Static functional connectivity (sFC)
The sFC amongst the 59 ROIs in migraine and PPTH is illustrated as a heatmap in Figure 2. Of the 1711 total ROI pairs, 1008 ROI pairs showed sFC above the preset 0.1 threshold in one or more of the cohorts. Analyses of variance, analysing differences between any of the three participant cohorts, indicated main effect sFC differences for 54 ROI pairs after controlling for sex and age. Of the 54 ROI pairs with significant ANOVA test results, 23 pairs had a significant (p < 0.05) difference in sFC between migraine and PPTH, as evaluated by t-tests. The network plot in Figure 3 shows the 23 ROI pairs where the sFC differed significantly between migraine and PPTH. Seventeen pairwise comparisons between migraine and PPTH remained significant after Benjamini-Hochberg FDR correction (see Table 3 and Figures 3 and 4). These ROI pairs included primary somatosensory, secondary somatosensory, posterior insula, hypothalamus, anterior cingulate, middle cingulate, temporal pole, supramarginal gyrus, superior parietal, middle occipital, lingual gyrus, pulvinar, precuneus, cuneus, somatomotor, ventromedial prefrontal cortex (VMPFC), and dorsolateral prefrontal cortex (DLPFC) regions.
Heatmap of static functional connectivity (sFC) between 59 regions-of-interest in migraine and persistent post-traumatic headache. Compared to cooler colours, warmer colours represent stronger static functional connectivity between regions. Region-to-region connections with significant (unadjusted) static functional connectivity differences between migraineurs and patients with persistent post-traumatic headache (PPTH). The numbered circles represent the regions of interest. The size of the circle represents the number of connections a specific node has (larger circles = more connections). The number within the circle corresponds to the name of the region, as indicated in Table 1. The placement of the circles on the brain template is meant for illustration purposes only and does not precisely reflect the location of the region on the brain. B.H.: Benjamini-Hochberg correction. Boxplots showing static functional connectivity (sFC) differences for 17 region pairs that significantly differed (following correction for multiple comparisons) between migraineurs and persistent post-traumatic headache (PPTH). A healthy control (HC) cohort was included to interpret the differences between migraine and PPTH. Circles indicate means; black bars indicate medians. Region pairs with significant differences in static functional connectivity between migraine and PPTH groups. indicates significance after Benjamini-Hochberg false discovery rate correction. Mig: Migraine; PPTH: persistent post-traumatic headache; DLPFC: dorsolateral prefrontal cortex; TPJ: temporal parietal junction; VMPFC: ventromedial prefrontal cortex; R: right; L: left; Sup: superior; Lat: lateral; FC: functional connectivity.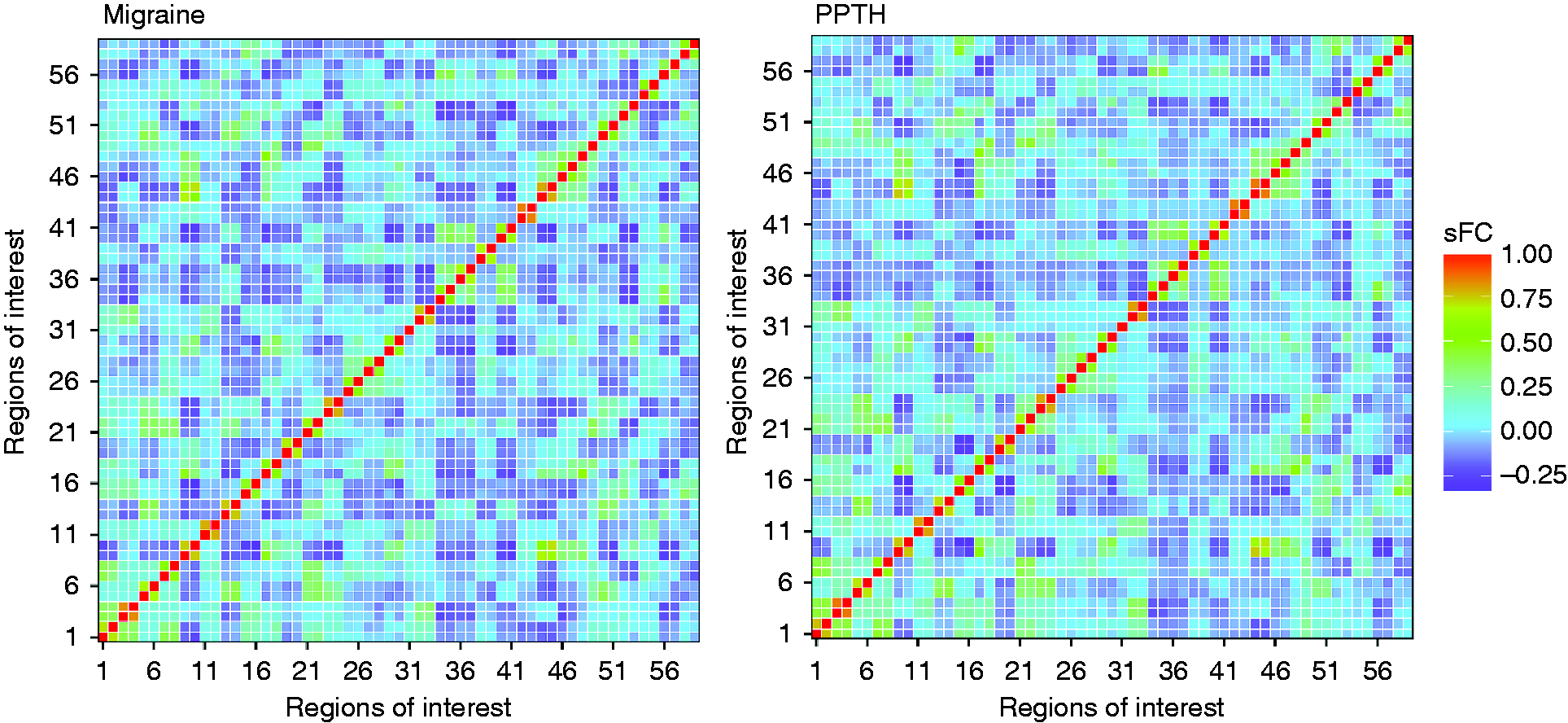

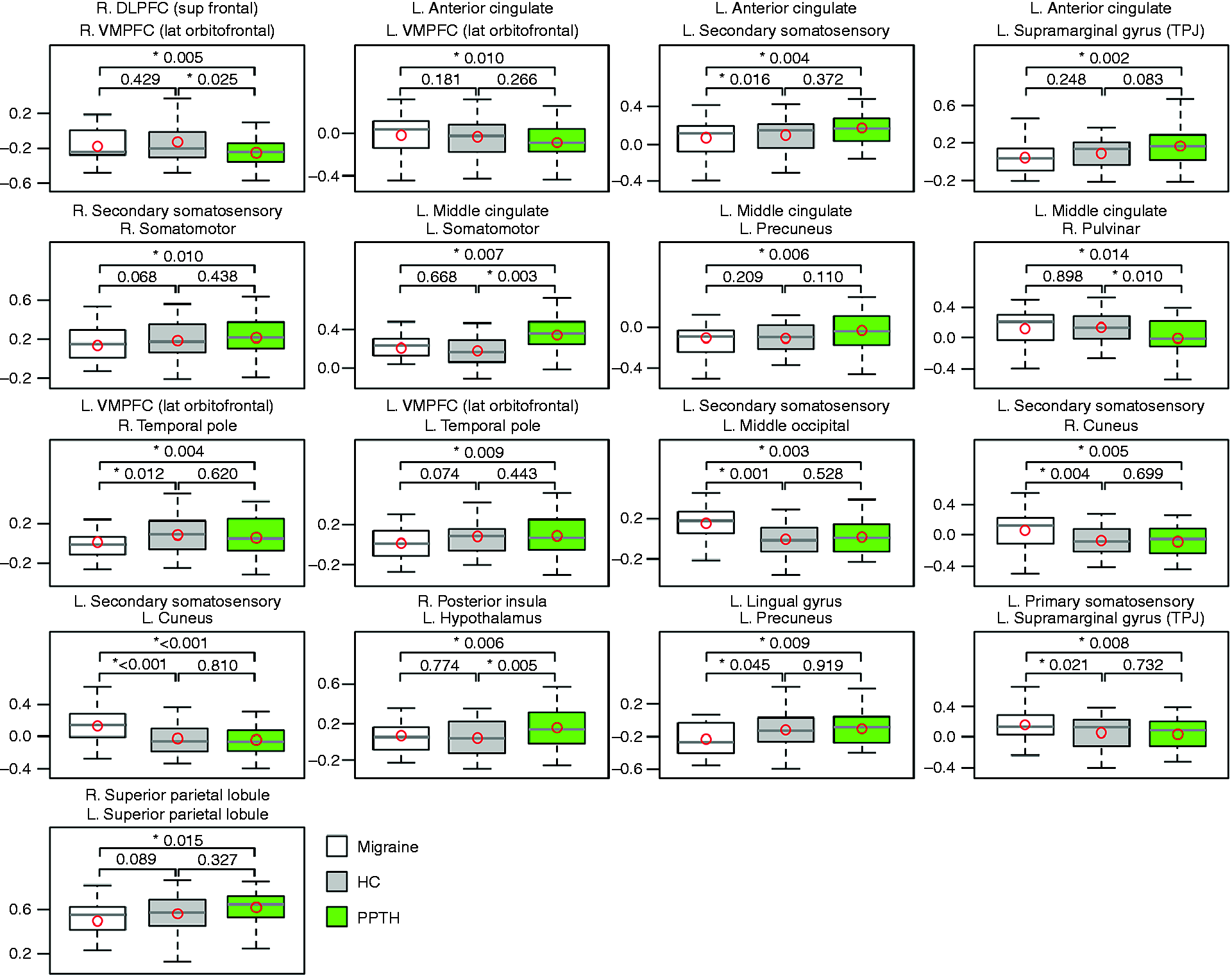

As shown in Figure 4, amongst these 17 ROI pairs with significant sFC differences between migraine and PPTH, four ROI pairs (right DLPFC with right VMPFC; left middle cingulate with left somatomotor and right pulvinar; right posterior insula with left hypothalamus) also showed significant sFC differences between healthy controls and PPTH. An additional seven ROI pairs (right primary somatosensory with left supramarginal gyrus; left VMPFC with right temporal pole; left secondary somatosensory with left middle occipital, right cuneus, left cuneus, and left anterior cingulate; left lingual gyrus with left precuneus) showed significant sFC differences between healthy controls and migraine. The other pairwise differences were not significant.
For each of the ROI pairs with sFC differences between migraine and PPTH and between migraine and healthy controls or PPTH and healthy controls, correlations between sFC and headache burden (i.e. years with headache and headache frequency), controlling for sex and age, and between sFC and pain intensity at the time of imaging, were calculated. In the migraine group, the correlation between years with headache and sFC, after controlling for sex and age, was significant for right DLPFC with right VMPFC (r=−0.37, p = 0.033); in the PPTH group, the correlation was significant for right primary somatosensory with left supramarginal gyrus (r=0.37, p = 0.014). In the migraine group, headache frequency was significantly correlated with sFC of left secondary somatosensory with right cuneus (r=0.35, p = 0.044), while in the PPTH group headache frequency was significantly correlated with sFC of left middle cingulate with right pulvinar (r=0.32, p = 0.034) and right posterior insula with left hypothalamus (r=−0.32, p = 0.035). These correlations were not significant after correction for multiple testing. There were no significant correlations between pain intensity at the time of imaging and static functional connectivity.
Dynamic functional connectivity (dFC)
The dFC amongst the 59 ROIs in migraine and PPTH is illustrated in a heatmap in Figure 5. Analyses of variance showed 88 ROI pairs with significantly different group mean dFC values (p < 0.05) amongst the three participant cohorts. Of those, PPTH and migraine had different dFC for 29 ROI pairs (p < 0.05). Significant group differences remained for 10 of these ROI pairs following Benjamini-Hochberg correction (see Table 4 and Figures 6 and 7). These 10 ROI pairs included the secondary somatosensory, hypothalamus, middle cingulate, temporal pole, supramarginal gyrus, superior parietal, lingual gyrus, somatomotor, precentral, posterior cingulate, middle frontal, fusiform gyrus, parieto-occiptal, and amygdala regions.
Heatmap of dynamic functional connectivity (dFC) between 59 regions-of-interest in migraine and persistent post-traumatic headache. Compared to cooler colours, warmer colours represent greater dynamic functional connectivity (i.e. more variability in functional connectivity). Region-to-region connections with significant (unadjusted) dynamic functional connectivity (dFC) differences between migraineurs and patients with persistent post-traumatic headache (PPTH). The numbered circles represent the regions of interest. The size of the circle represents the number of connections a specific node has (larger circles = more connections). The number within the circle corresponds to the name of the region, as indicated in Table 1. The placement of the circles on the brain template is meant for illustration purposes only and does not precisely reflect the location of the region on the brain. Boxplots showing dynamic functional connectivity (dFC) differences for 10 region pairs that significantly differed (following correction for multiple comparisons) between migraineurs and persistent post-traumatic headache (PPTH). A healthy control (HC) cohort was included to interpret the differences between migraine and PPTH. Circles indicate means; black bars indicates medians. Region pairs with significant difference in dynamic functional connectivity between migraine and PPTH groups. indicates significance after Benjamini-Hochberg false discovery rate correction. Mig: migraine; PPTH: persistent post-traumatic headache; DLPFC: dorsolateral prefrontal cortex; VMPFC: ventromedial prefrontal cortex; R: right; L: left; Lat: lateral; Inf: inferior; Sup: superior; FC: functional connectivity.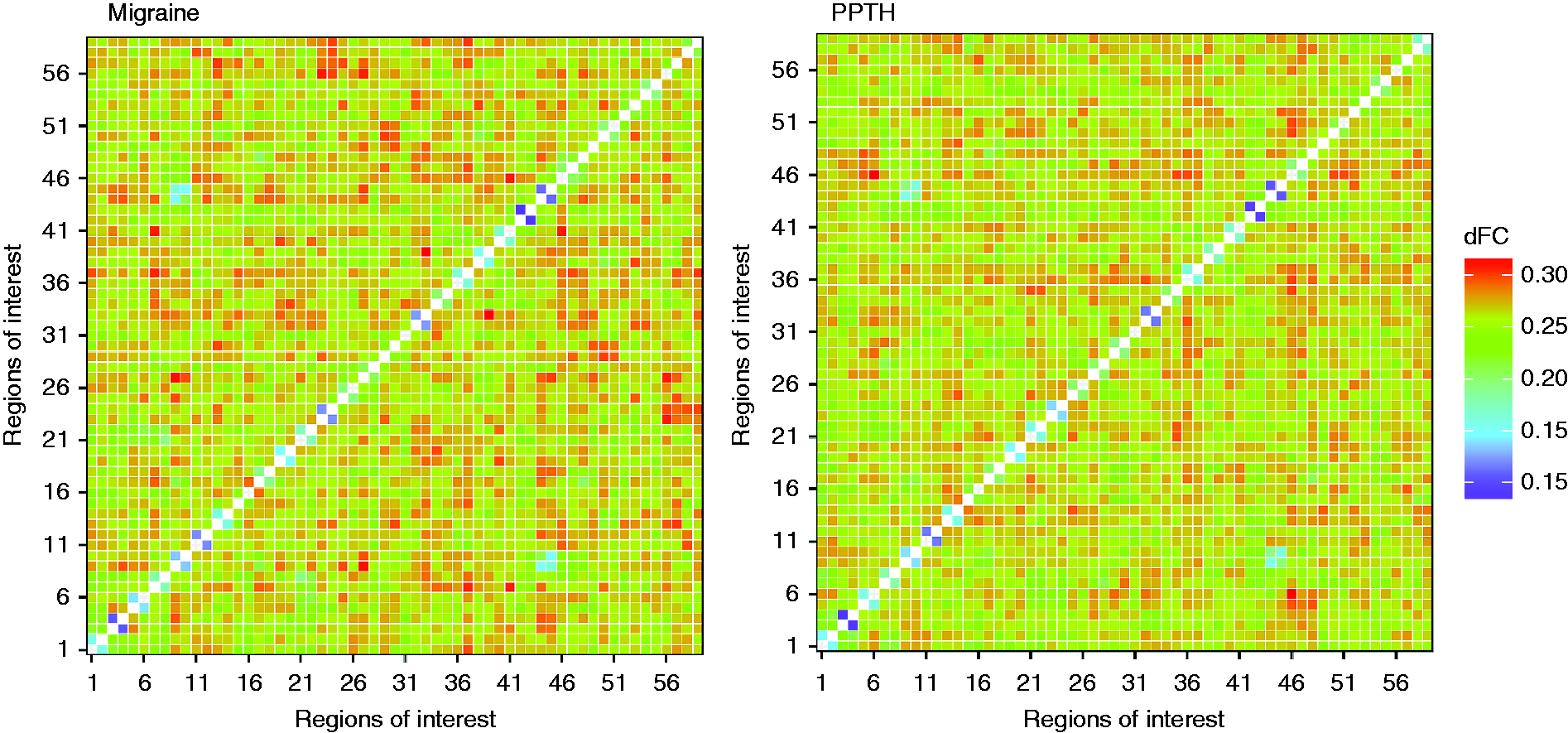

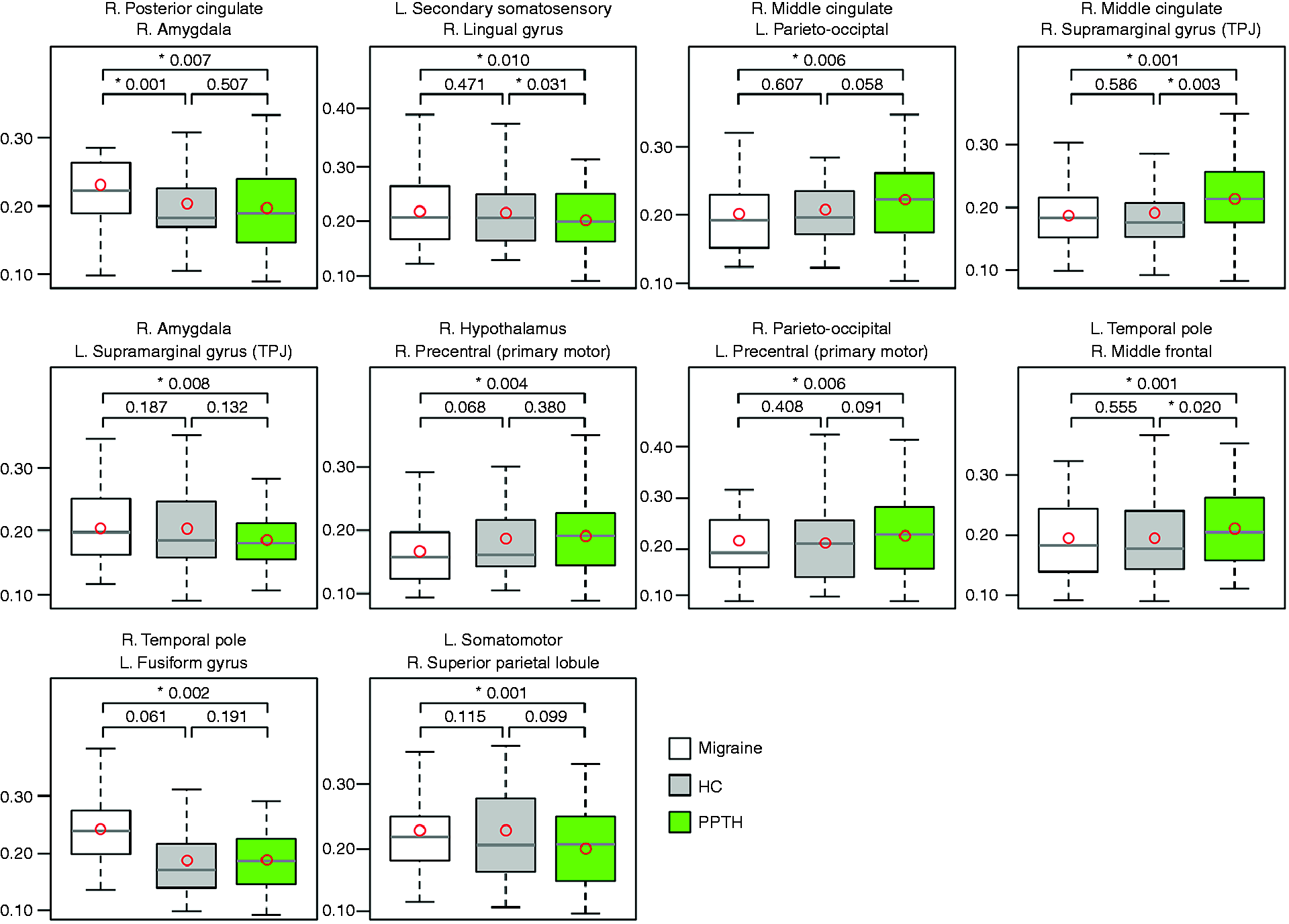

As shown in Figure 7, one of these 10 ROI pairs that differed between migraine and PPTH showed a significant difference between healthy controls and migraine but not between PPTH and healthy controls; three showed a difference between healthy controls and PPTH but not migraine and healthy controls; and the remaining three ROI pairs did not show a significant difference between healthy controls and PPTH or between healthy controls and migraine.
For each of the four ROI pairs with dFC differences between PPTH and migraine where there was also a difference between migraine or PPTH and healthy controls, correlations between years with headache and headache frequency with dFC and between dFC with pain intensity at the time of imaging were explored. In the PPTH group, there was a significant correlation between headache frequency with dFC of the right middle cingulate with right supramarginal gyrus (r = 0.32, p = 0.035). In the migraine group, there was a significant correlation between pain intensity at the time of testing with dFC of the right posterior cingulate with right amygdala (r = −0.51, p = 0.002). This correlation remained significant after correction for multiple testing.
Discussion
This study evaluated static and dynamic resting-state brain functional connectivity patterns for 59 ROIs in individuals with PPTH relative to those with migraine. For interpretation purposes, a healthy control cohort was included to be able to interpret the connectivity aberrations found in migraine and PPTH. Significant group differences between migraine and PPTH were found for both sFC and dFC amongst regions that participate in processing pain. Although there was overlap in individual regions, those ROI pairs that showed sFC differences between migraine and PPTH were dissimilar from the ROI pairs that showed dFC differences between the groups. These findings concur with other studies that have compared sFC and dFC patterns in patient groups relative to healthy controls in that they further indicate that sFC and dFC are complementary techniques that reflect different brain dynamics. Thus, both analytical techniques can be useful for exploring pathophysiological mechanisms in PPTH and migraine (31).
There have been many publications based on resting state sFC analyses of migraine, but few that have investigated dFC (32). Since sFC analyses average over an entire time-series, they do not capture moment-to-moment changes in connectivity strength between brain regions. Dynamic functional connectivity analyses calculate the connectivity across overlapping time segments throughout the time-series and then determine the variance of the correlations (i.e. variance of the connectivity strength). Region pairs or networks that have low variance are those that maintain stable connectivity strength (whether that connectivity strength is weak or strong), while those with high variance have substantial fluctuations in connectivity strength over time. It has been suggested that homologous brain regions, those with bidirectional anatomic connections, and those within known resting state networks show the least amount of connectivity variance over time, while regions with unidirectional or no anatomic connections, those that are part of different functional networks, and higher order regions that require a high degree of flexibility have greater connectivity variance (33,34).
As would be expected with sFC analyes, we found varying magnitude and directionality of ROI-to-ROI differences in sFC when comparing PPTH, migraine, and healthy controls. For some of these region pairs, sFC was “stronger” (i.e. further from zero) in PPTH versus migraine, while for others sFC was weaker (i.e. closer to zero). Data from healthy controls were used to determine whether the sFC in the PPTH or migraine group was closer to “normal”. A notable finding in this study was that several of the region pairs that differed in sFC between migraine and PPTH and also between migraine and healthy controls included connectivity between sensory-discriminative pain processing regions with visual processing regions. For example, the migraine group had stronger sFC between secondary somatosensory cortex with middle occipital and cuneus regions. These findings could be reflective of the multifaceted relationships between visual processing and pain processing that are perhaps inherent to the migraine brain, such as visual stimuli triggering and worsening migraine headache pain.
Although the interpretation of functional connectivity strength can certainly be debated, altered connectivity in migraine could be indicative of underlying differences in the migraine brain that predispose patients to development of migraine and individual migraine attacks. Alternatively, recurrent concomitant activation of brain regions with repetitive migraine attacks could lead to strengthening of the functional connections between pain processing regions over time. Several of the regions that were found to have sFC that differed in migraine versus PPTH in this study have previously been identified as having sFC that differs in migraine versus healthy controls (17,35–39). For PPTH, a secondary headache, altered functional connectivity could be a result of the underlying mTBI, such as weakening of connectivity due to the injury itself or a strengthening of connectivity to compensate for functional connectivity losses amongst other brain regions. Alternatively, like in migraine, altered connectivity in PPTH could be a result of having recurrent concomitant brain activations with each headache episode.
Relative to PPTH, migraine was associated with more variability in functional connectivity (higher standard deviation of windowed correlations) for half of the ten ROI pairs that significantly differed between the cohorts. For all of these ROI pairs, there was also more variability in functional connectivity for migraine relative to healthy controls (significantly so, for right posterior cingulate with right amygdala). For the other half of ROI pairs, there was more variability in functional connectivity in PPTH compared to migraine and in PPTH compared to HC (significantly so for right middle cingulate with right suparmarginal gyrus, and left temporal pole with right middle frontal).It is possible that greater dFC reflects an enhanced degree of cognitive flexibility, perhaps a compensatory mechanism to frequent pain. Correlation between headache frequency and dFC of posterior cingulate with amygdala in the PPTH group suggests impact of pain frequency on dFC of this region pair. Alternatively, the magnitude of dFC could be an inherent property of the migraine brain or a result of mTBI in the PPTH group that is not impacted by recurrent migraine attacks or post-traumatic headaches. It is also possible that greater dFC reflects a mind-wandering state or less ability to achieve a “resting state” during scanning. Since fMRI was performed with participants in the “resting state” (i.e. absence of task or stimulus), one might expect the functional connectivity between two regions to remain stable over time, whereas changes in brain function during BOLD signal collection would result in the connectivity strength between regions being dynamic.
There are a paucity of studies investigating functional connectivity in PPTH and comparing brain function or structure in migraine versus PPTH. Thus, we took a broad approach to our investigation, including multiple brain regions that have been shown to participate in pain processing and/or that have been demonstrated to be involved in migraine. Results from studies such as this one should allow for future analyses to be focused on brain regions that might be specifically involved in PPTH versus migraine. The relative lack of previously published data comparing brain structure and function in PPTH versus migraine makes it difficult to put the results from this current study in context with existing findings. Our prior publication on brain structure showed migraine versus PPTH differences in regions of the right lateral orbitofrontal, left middle frontal caudal, left precuneus, right supramarginal gyrus, and left superior frontal cortices (6). Realising that some of the participants included in that analysis are also included in the analysis reported here, there is overlap in the findings of differences in brain structure with those of differences in functional connectivity of frontal regions, precuneus, and supramarginal gyrus when comparing PPTH to migraine. Further work is necessary to investigate why these brain areas might be differentially involved in migraine versus PPTH.
Whereas it is likely that the differences in functional connectivity identified between PPTH and migraine at least relate to the headache disorders themselves, there are many potential confounders that we did not control for, including but not limited to, anxiety, depression, post-traumatic stress disorder, medication use, and other medical conditions/symptoms that are commonly present amongst individuals with PPTH and those with migraine. In this study, it is unlikely that the entire difference in functional connectivity between PPTH and migraine can be directly attributed to the headache disorders. It is more likely that the headaches contribute to the identified differences and that other characteristics also contribute to the differences.
By restricting our analysis to a priori selected pain-processing ROIs as opposed to analysing all brain voxels, it is possible that we might have missed relevant areas of connectivity aberrations between migraine and PPTH. However, we chose to pursue a hypothesis-driven ROI approach. Although we found differences in sFC and dFC patterns in migraine relative to PPTH for a number of regions involved in pain processing, whether sFC and dFC analyses could provide a connectivity marker specific to migraine or whether it is useful for differentiating migraine from PPTH will need to be explored in future studies using multivariate predictive modeling. This study is not able to dissect functional connectivity findings that might be due to PPTH versus those that might be due to the underlying mTBI. To do so, future analyses should include a cohort of individuals who do not have PTH but do have persistent symptoms attributed to mTBI. Most participants were currently experiencing pain at the time of brain imaging. Pain intensity and the proportion of individuals with pain at the time of testing, was similar for the PPTH and migraine cohorts although there was a small difference in pain intensity between the subject groups. In the migraine group, pain at time of testing did correlate with dFC for one ROI pair. We did not collect the information that would be required to determine the proportion of participants who had an active migraine (or migraine-like headache) at the time of imaging. The participants in this study had migraine or PPTH for many years and had high frequency headaches. Future studies should investigate whether the findings from this study are generalisable to less severely affected populations of people with migraine and PPTH. Headache preventive medications can likely impact functional connectivity findings. Approximately 50% of individuals in the PPTH and migraine cohorts were currently using preventive medications. Larger sample sizes are required to evaluate potential impacts of migraine preventive medication use on functional connectivity. Finally, since most of the research participants either had bilateral headaches or unilateral headaches that switched sides from attack to attack, we were not able to match across participants for the side of headache pain (e.g. flip images).
Conclusions
Static and dynamic functional connectivity differed between migraine and PPTH for a number of pain-processing and visual-processing regions. Although there was some overlap in the dFC and sFC differences between migraine and PPTH, sFC and dFC analyses provided results that were complementary, suggesting that both methods of analyses might be useful when studying migraine and PPTH. Additional research is necessary to validate the current findings and to determine whether sFC and dFC analyses can be used for accurately differentiating PPTH from migraine.
Footnotes
Key findings
Static and dynamic functional connectivity of pain-processing and visual-processing regions differs between migraine and persistent post-traumatic headache.
Static and dynamic functional connectivity analyses might both be useful for interrogating brain function in migraine and PPTH and perhaps for differentiating PPTH from migraine.
Acknowledgements
The authors would like to thank all study participants and research personnel for their time and dedication to this project.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Office of the Assistant Secretary of Defense for Health Affairs, through the Peer Reviewed Medical Research Program under Award No. W81XWH-15-1-0286. Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense. The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014 is the awarding and administering acquisition office.
