Abstract
Background
Persistent post-traumatic headache most commonly has symptoms that overlap those of migraine. In some cases, it can be clinically difficult to differentiate persistent post-traumatic headache with a migraine phenotype from migraine. The objective of this study was to develop a classification model based on questionnaire data and structural neuroimaging data that distinguishes individuals with migraine from those with persistent post-traumatic headache.
Methods
Questionnaires assessing headache characteristics, sensory hypersensitivities, cognitive functioning, and mood, as well as T1-weighted magnetic resonance imaging and diffusion tensor data from 34 patients with migraine and 48 patients with persistent post-traumatic headache attributed to mild traumatic brain injury were included for analysis. The majority of patients with persistent post-traumatic headache had a migraine/probable migraine phenotype (77%). A machine-learning leave-one-out cross-validation algorithm determined the average accuracy for distinguishing individual migraine patients from individual patients with persistent post-traumatic headache.
Results
Based on questionnaire data alone, the average classification accuracy for determining whether an individual person had migraine or persistent post-traumatic headache was 71.9%. Adding imaging data features to the model improved the classification accuracy to 78%, including an average accuracy of 97.1% for identifying individual migraine patients and an average accuracy of 64.6% for identifying individual patients with persistent post-traumatic headache. The most important clinical features that contributed to the classification accuracy included questions related to anxiety and decision making. Cortical brain features and fibertract data from the following regions or tracts most contributed to the classification accuracy: Bilateral superior temporal, inferior parietal and posterior cingulate; right lateral occipital, uncinate, and superior longitudinal fasciculus. A post-hoc analysis showed that compared to incorrectly classified persistent post-traumatic headache patients, those who were correctly classified as having persistent post-traumatic headache had more severe physical, autonomic, anxiety and depression symptoms, were more likely to have post-traumatic stress disorder, and were more likely to have had mild traumatic brain injury attributed to blasts.
Discussion
A classification model that included a combination of questionnaire data and structural imaging parameters classified individual patients as having migraine versus persistent post-traumatic headache with good accuracy. The most important clinical measures that contributed to the classification accuracy included questions on mood. Regional brain structures and fibertracts that play roles in pain processing and pain integration were important brain features that contributed to the classification accuracy. The lower classification accuracy for patients with persistent post-traumatic headache compared to migraine may be related to greater heterogeneity of patients in the persistent post-traumatic headache cohort regarding their traumatic brain injury mechanisms, and physical, emotional, and cognitive symptoms.
Keywords
Introduction
Patients with persistent post-traumatic headache (PPTH) and patients with migraine have similar phenotypes (1–3). Migraine is the most common headache phenotype in patients with PPTH in clinic-based studies (4). In our clinical experience, these similarities between both headache disorders can make it difficult to distinguish a patient with new-onset PPTH of a migraine-like phenotype, based on headache characteristics alone, from a migraine exacerbation triggered by a traumatic brain injury (TBI). The clinical distinction can be challenging when an individual had migraine prior to their head injury and when there is a delayed or uncertain timing of headache onset following TBI. Prior studies have found differences between patients with migraine and patients with PPTH in dynamic and static functional connectivity (5), brain volume and area (6), and fibertract integrity (7), suggesting that both headache disorders, despite sharing similar headache characteristics, have differences in their underlying neuropathology.
This study aimed to determine the utility of classifying individual patients as having migraine versus PPTH based on a battery of clinical questionnaires and brain structural (T1-weighted imaging and diffusion tensor imaging) data. If successful, disease classification based on imaging and questionnaire data would provide important pathophysiological insights into the disease mechanisms underlying migraine and PPTH and help clinicians make objective, non-biased treatment decisions. It was hypothesized that: i) patients could be accurately classified as having migraine or PPTH using machine-learning classification-based algorithms that include a combination of clinical and neuroimaging features; and ii) a classifier containing clinical data and neuroimaging data would be more accurate than a classifier containing clinical data alone.
Methods
Subjects were recruited for participation from patient clinics at the Phoenix VA Health Care System and from the Mayo Clinic in Arizona. Subjects completed written informed consent prior to study participation. Data from subjects included in this dataset have been included in prior publications (5,6,8,9). Prior publications did not interrogate classification accuracies based on clinical data or/and MRI-based features to distinguish patients with migraine from patients with PPTH. The study enrolled male and female subjects ages 18–65 years with a diagnosis of migraine or PPTH attributed to mild TBI (mTBI). International Classification of Headache Disorders, 3rd edition (beta version) (ICHD-3 beta) symptom criteria (10) were used to assign headache phenotypes, since these were the most recent criteria available at the time of patient enrolment. Headache diagnoses were assigned by a headache specialist at Mayo Clinic (TS). Exclusion criteria were as follows: a) migraine group: History of TBI; b) PPTH group: History of moderate or severe TBI, history of migraine or another headache disorder prior to concussion except for infrequent episodic tension-type headaches. Migraine patients and patients with PPTH were not excluded for taking preventive or abortive medication, or for having comorbid depression, anxiety, or post-traumatic stress disorder. Migraine patients were not excluded if they had occasional tension-type headaches (≤ 3 tension-type headaches per month).
Questionnaires
All subjects completed a comprehensive clinical symptoms battery to investigate memory (Ray Auditory Verbal Learning Test: RAVLT, immediate and delayed memory recall (11)), attention (Trails A and B (12)), symptoms of depression (Beck Depression Inventory, BDI (13)), anxiety (State-Trait Anxiety Inventory, Form Y-1 and Form Y-2 (14)), TBI history (Ohio State University TBI identification method, a standardized questionnaire assessing the lifetime history of TBI for an individual (15)), sound sensitivity (Hyperacusis Questionnaire (16)), light sensitivity (Photophobia Assessment Questionnaire, PAQ (17)), the Allodynia Symptom Checklist (ASC-12 (18)), the Migraine Disability Assessment Scale (MIDAS (19)), the pain catastrophizing scale (PCS) (20,21), the Insomnia Severity Index (22), the COMPASS 31-autonomic symptom questionnaire (23), a validated post-traumatic stress disorder checklist (DSM-5) (24), and a structured headache interview developed by headache specialists at Mayo Clinic, which included questions about the number of headache days per month, the number of headache-free days per month, when headaches first began (year/month), the location, quality and intensity of headaches, the presence/absence of aura, the presence/absence of light and/or sound hypersensitivity, questions specific to cognitive and physical symptoms during headache, the date of the most recent headache, current pain level, and family history of migraine. Headache and TBI characteristics were collected using case report forms containing Common Data Elements (CDE) developed by the National Institute of Neurological Disorders and Stroke (NINDS) and included a 22-item symptom evaluation checklist from the Sport Concussion Assessment Tool (SCAT) 5th edition (25).
MRI-based imaging
Migraine patients and patients with PPTH underwent neuroimaging at Mayo Clinic hospital, Phoenix, Arizona. Imaging was conducted on a 3-Tesla Siemens MAGNETOM (Erlangen, Germany) scanner. Imaging sequences were acquired by trained Mayo Clinic Radiology technologists and assisted by research study personnel. Subjects were scanned during a 26-month time period (2016–2018). Imaging sequences were collected using a 20-channel head/neck coil.
MRI sequence collection included the following: 3D T1-weighted sagittal MP-RAGE (TE = 3.03 ms, TR = 2400 ms, flip angle = 8°), 128 slices, slice thickness = 1.25 mm (no gap), voxel dimensions = 1 mm × 1 mm, field of view (FOV) = 256 mm2, matrix size = 256 × 256. Axial T2-weighted imaging: TE = 84 ms, TR = 6800 ms, flip angle = 150°, 38 slices, voxel dimensions = 1 × 1 × 4 mm3, slice thickness = 4 mm, FOV = 256 mm2, matrix size = 256 × 256. Diffusion Tensor Imaging (DTI): TE = 71 ms, TR = 5100 ms, 38 number of slices, slice thickness = 4 mm, voxel dimensions = 1.7 × 1.7 × 4 mm3, FOV = 220 mm2 matrix size = 128 × 128. Thirty diffusion weighted (b = 1000 s/mm2) non-linear directions and one image without diffusion weighting (diffusion-unweighted volume; b = 0 s/mm2). T2-weighted imaging and diffusion tensor imaging were acquired with two averages. An MRI fiducial marker was placed on subjects’ right temple to avoid accidental image-flipping during the post-processing stream.
T1 and T2 images were reviewed by a board certified neuroradiologist. Subjects with presence of gross anatomical abnormalities on imaging (including T2 hyperintensities) were excluded from the final analysis.
MRI data post-processing for T1-weighted data: All imaging data were post-processed using FreeSurfer analysis suite (version 6.0) and the integrated automated global tractography toolbox TRACULA (TRActs Constrained by Underlying Anatomy) (http://surfer.nmr.mgh.harvard.edu/) (26). Post-processing of anatomical T1-weighted imaging was conducted using the automated “recon-all” brain segmentation and parcellation stream and the Desikan-Killiany Atlas, which segments each subject’s T1-weighted brain data into 34 regions in each hemisphere for the estimation of cortical thickness, volume, area, and curvature (27). Data processing steps of this technique include: Removal of non-brain tissue (skull stripping) (28), Talairach transformation (29), segmentation and parcellation of gray and white matter (30), intensity normalization and brain boundary tessellation (30), and surface deformation (31).
MRI data post-processing for DTI data: The TRACULA package within FreeSurfer has been shown to accurately and reliably identify white matter tracts in healthy cohorts and patient populations (26,32). TRACULA reconstructs 18 major white matter pathways and associated diffusion metrics (fractional anisotropy, mean diffusivity, axial diffusivity and radial diffusivity). The fibertracts include: Bilateral corticospinal tract, bilateral uncinate fasciculus, bilateral anterior thalamic radiations, bilateral cingulum angular bundles, bilateral cingulum cingulate gyri, bilateral superior longitudinal fasciculi – parietal, bilateral superior longitudinal fasciculi – temporal, bilateral inferior longitudinal fasciculi, and the forceps major and forceps minor tracts.
All white matter tracts were automatically reconstructed using the T1 anatomical priors of each individual. TRACULA uses probabilistic tractography from FSL for the standard pre-processing of diffusion data, which includes the following pre-processing steps (https://surfer.nmr.mgh.harvard.edu/fswiki/trac-all): Image correction to correct for B0 inhomogeneities, eddy current and head motion correction, brain extraction using ‘bet’ and ‘bedpostx’ to fit the ball-and-stick model for calculating voxel-based diffusion parameters. Bvecs are rotated for eddy current correction. Within-subject registration is performed by affine registration of each subject’s low-b diffusion to the individual’s T1-weighted imaging and then co-registering to a template for between-group comparisons. Diffusion gradient directions were manually checked in ‘fslview’ for correctness before including subject data in the final analysis. Using the structural brain anatomy of each person in combination with a pre-labelled training set, the distribution of fibertracts was estimated using a Markov Chain Monte Carlo algorithm (34). To prevent post-processing irregularities stemming from using multiple workstations (34), all image post-processing was conducted on a single Mac workstation (3.5 Ghz 6-core Xeon E5) installed with OS X Sierra (10.12.6). The accuracy of the brain segmentation output was manually inspected by a neuroimaging scientist (CC) before including subject data for group analyses. This ensures the accuracy of the automated brain reconstruction process and prevents the inclusion of erroneous datasets. All imaging was assessed for excessive motion, and subjects which moved > 2 mm in either direction during scanning were excluded from the final analysis. Furthermore, to ensure that movement in the scanner was not more prominent in patients with PPTH compared to migraine patients, between-groups differences for movement in the scanner were assessed using two-sided t-tests, which showed no significant group differences in movement parameters in any direction (p > 0.5).
Statistical analysis
Proper measures were taken to avoid adding bias to the modelling algorithm: Any group-identifying data or questionnaires that included characteristics obviously known to be more prevalent in patients with migraine or patients with PPTH were not added as features to the classification model. For example, items from the Ohio State TBI Identification Method, which determine TBI history, were not included in the modelling, since eligibility criteria stipulated that all PPTH patients had a history of TBI and all migraine patients had no history of TBI. Furthermore, history of post-traumatic stress disorder (known to be common in military personnel who suffered concussions during active duty) was not included as a feature in the classification model. Lastly, medication history was not included as a feature, as most patients with migraine were taking migraine preventives. Questionnaires and demographic data (including age and sex) were used as features in the machine-learning classification algorithm (see Table 1). Group differences for subject demographics, headache characteristics and anxiety and depression symptoms were assessed via two-sided t-tests, except for sex, which was assessed using a χ2 test (Table 2).
Component measures.

ASC-12: Allodynia Symptom Checklist; BDI: Beck Depression Inventory; COMPASS-31: Autonomic Symptom Questionnaire; MIDAS: Migraine Disability Assessment Scale; OSU TBI-ID: Ohio State University TBI Identification Method; PAQ: Photosensitivity Assessment Questionnaire; PCS: Pain Catastrophizing Scale; PCL-5: Post-Traumatic Stress Disorder Checklist; RAVLT: Ray Auditory Verbal Learning Test; SCAT: Sport Concussion Assessment Tool; STAI: State and Trait Anxiety Inventory; Trails A and B: Trail making version A and B.
Subject demographics, headache characteristics, and anxiety and depression symptoms.

f: female; m: male; BDI: Beck Depression Inventory; SD: standard deviation; STATE: State anxiety inventory; TRAIT: Trait anxiety inventory; Headache frequency: average number of headache days per month; years with headache: years lived with headache; PPTH: persistent post-traumatic headache.
Note: Group differences were assessed via two-sided t-tests except for sex, which was assessed using a chi square test. Italics signifies standard deviation.
Machine-learning classification
Due to the limited sample size and the very large number of variables (both clinical and imaging), a logistic classifier with a sparsity constraint was used on the learned coefficients (35). This was done after the feature set was reduced using principal components analysis (PCA) – the feature dimension was determined by the minimum number of features required to maintain 90% of signal energy. The classification model has one hyperparameter, the strength of the sparsity constraint, which is optimized during training. The sparsity constraint ensures a parsimonious model that only captures the relevant questionnaire and imaging features for the task at hand and is more robust to risks of overfitting data. One of the benefits of sparsity-constrained models is that they jointly perform feature selection and classification. That is, the model only gives non-zero coefficient values to feature variables that improve classification performance and the number of non-zero coefficients is inversely proportional to the strength of the hyperparameter. The model is evaluated using leave-one-out cross-validation (36). Data from all subjects were used to train the model. For each cross-validation iteration, data from all of the subjects except for one were used to train the sparse logistic model (including setting the hyperparameter value), and the resultant model was evaluated on the single out-of-sample subject. This process is repeated until the model has evaluated all data and the average error was computed after all folds were completed.
Since PCA results in a linear combination of features, it becomes difficult to determine which individual features are driving classifier performance. As a result, a secondary analysis was conducted for each type of data (questionnaire, MRI data) to determine the optimal features by data type. We use the information gain criterion to determine the optimal features based on their ability to differentiate between PPTH and migraine (37).
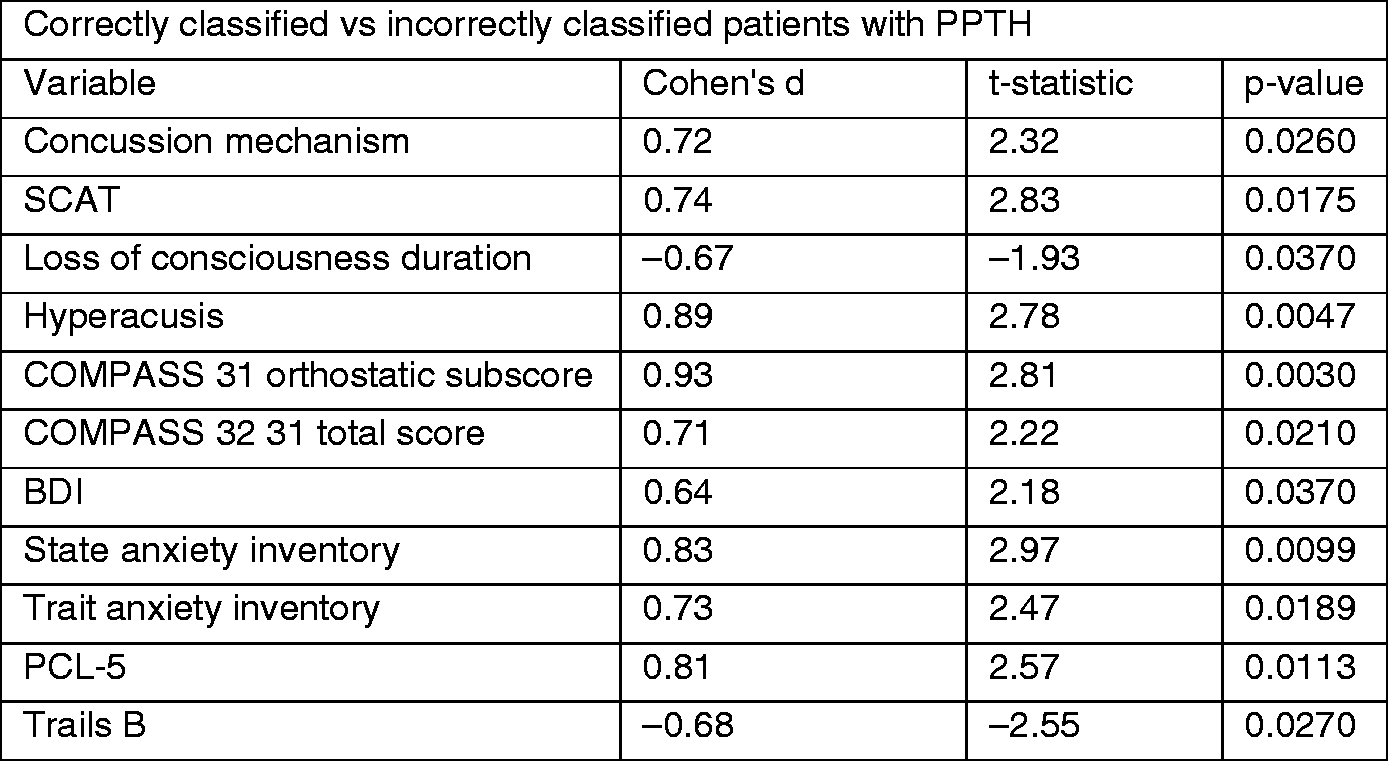
In order to better understand the differences between PPTH patients that were correctly classified compared to patients with PPTH that were incorrectly classified, a series of two-tailed t-tests were conducted on the best physical, cognitive, and psychological measures resulting from the post-hoc analysis, as well as measures that were not included in the classification model since they were characteristics unique to the PPTH cohort. These measures included demographic information and patient history (age, sex, PPTH headache phenotype, headache frequency, headache duration in hours, years lived with post-traumatic headache, family history of migraine), concussion mechanism (sports-related, motor vehicle accident, fall, blast-type injuries), number of concussions, and concussion-related details (loss of consciousness, presence/absence of post-traumatic stress disorder).
Results
This dataset included demographic information, questionnaire data and brain structural data from 34 patients with migraine (mean age = 41.7; SD = 10.9; 12 male, 22 female) and from 48 patients with PPTH (mean age = 38.1; SD = 11.1; 31 male, 17 female). There were no significant differences for age between patients with migraine and patients with PPTH (p = 0.14). There were significant differences in sex ratios between subject groups (migraine: 64.6% female and 35.3% male; PPTH: 64.6% male and 35.4% female; p = 0.009), significant differences in depression raw scores (BDI; migraine: Mean = 9.0, SD = 6.0; PPTH: mean = 17.0, SD = 9.1; p < 0.001), headache frequency (average headache frequency per month; migraine: Mean = 20.4, SD = 6.2; PPTH: Mean = 16.1, SD = 8.4; p = 0.01), and years lived with headache (average years lived with headache; migraine: Mean = 25.0, SD = 14.4; PPTH: Mean = 10.6, SD = 8.2; p < 0.001). There were no group differences in general anxiety (TRAIT Anxiety Inventory; migraine: Mean = 40.0, SD = 10.3; PPTH: Mean = 45.0, SD = 13.7; p = 0.062) or situational anxiety (State Anxiety Inventory; migraine: Mean = 34.8, SD = 8.9; PPTH: Mean = 37.7, SD = 14.3; p = 0.24).
See subject demographics, Table 2. Thirty patients had chronic migraine and four patients had episodic migraine. Eighteen patients had migraine with aura and 16 patients had migraine without aura. For patients with PPTH, 37 patients had a headache phenotype that was migraine-like, nine patients had a headache phenotype that was probable migraine-like, and two patients had a phenotype that was tension-type headache-like. Ten patients with PPTH had sports-related concussions, seven patients had motor vehicle accidents, 10 patients had falls, and 21 patients had blast-related concussions. Ten patients had a single concussion, 19 patients had two concussions, 15 patients had 3–10 concussions, and four patients had over 10 concussions. Out of the four people with a lifetime history of over 10 concussions, two people suffered concussions via repetitive sports-related injuries and two people suffered repeat concussions during military training exercises and active duty blast-related injuries. For those with multiple concussions, patients developed post-traumatic headache following their most recent concussion.
The average classification accuracy for correctly identifying individual patients as having migraine versus PPTH based on questionnaire data alone was 71.9%. The classification accuracy for correctly identifying individual patients as having migraine versus PPTH based on questionnaire data and T1-weighted structural imaging data was also 71.9%. The classification accuracy based on questionnaire data, T1-weighted structural imaging data, and diffusion tensor imaging fibertract data was 78%. Figure 1 shows that patients with migraine were correctly classified as having migraine with an average accuracy of 97.1%, while patients with PPTH were accurately classified as having PPTH with an average classification accuracy of 64.6%. Figure 2 shows the confusion matrix and the average classification accuracy for distinguishing migraine patients from patients with PPTH when using questionnaire data, morphometric data, and fibertract data.

Classification accuracies based on clinical data only, clinical data + regional brain morphometrics, and clinical data + regional brain morphometrics and fibertract data.
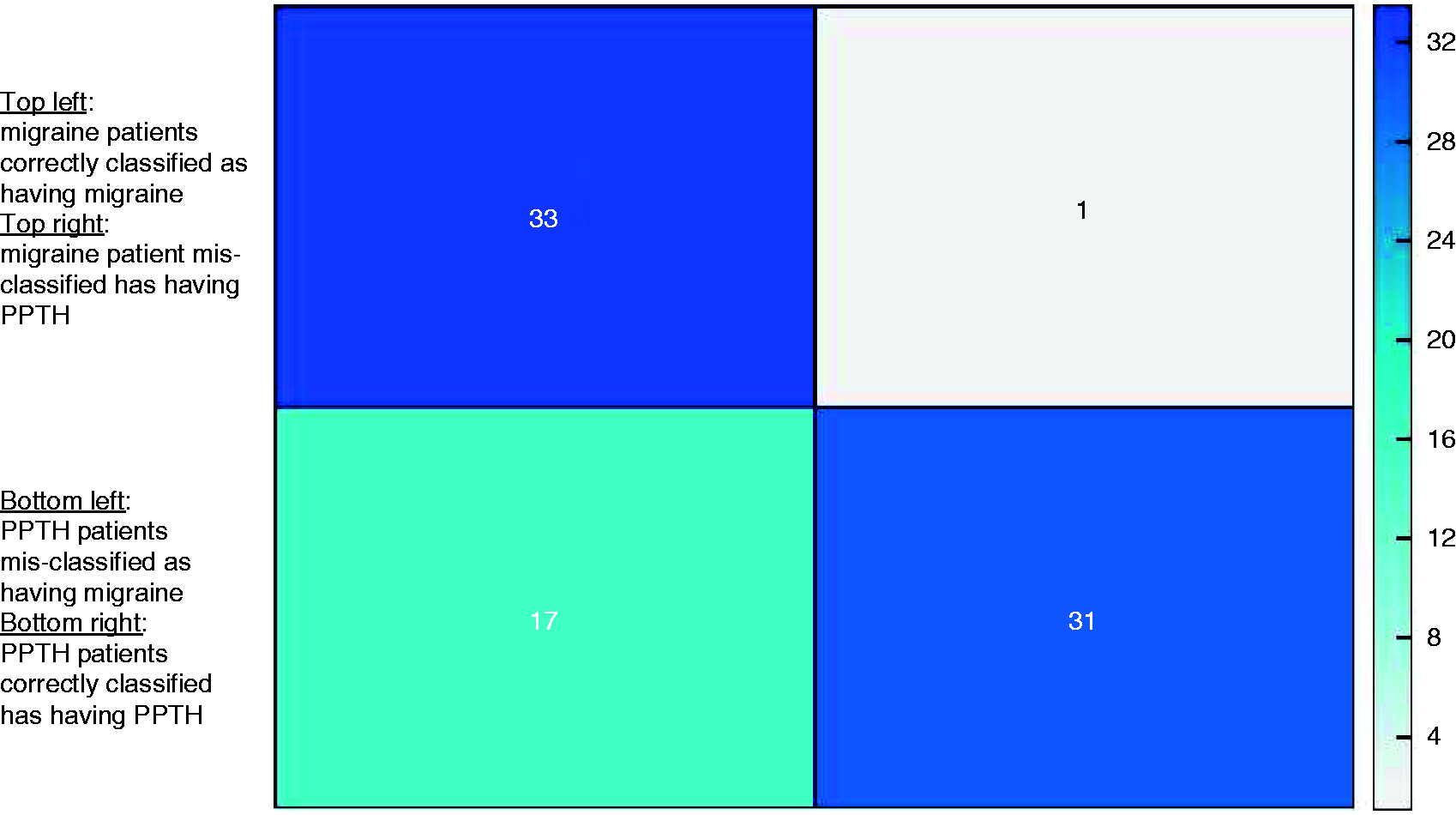
Confusion Matrix indicating model performance when using clinical data, brain morphometric data and fibertract data for classifying individual patients as either having migraine or persistent-post-traumatic headache (PPTH). This model has an average accuracy of 78.0%. Patients with migraine and patients with PPTH were classified with an accuracy of 97.1% (migraine) and 64.6% (PPTH), respectively.
Amongst the physical, psychological, and cognitive component measures that contributed to classification, the two best features identified in the information gain analysis were answers to questions about anxiety on the SCAT and decision making on the BDI. The top features were answers to question #22 on the Symptom Evaluation questionnaire of the SCAT, which asks patients to score themselves on “nervous or anxious” symptoms, based on how they feel at the moment. Patients with PPTH that were correctly classified as PPTH had more severe symptoms of nervousness or anxiety based on ranked responses (0 = none, 1–2 = mild, 3–4 = moderate, 5–6 = severe) compared to PPTH patients that were misclassified as having migraine. The other most important questionnaire feature included answers to question #13 on the BDI about decision making (0 = I make decisions about as well as I ever could; 1 = I put off making decisions more than I used to; 2 = I have greater difficulty in making decisions than before; 3 = I can’t make decisions at all any more). Patients with PPTH who were correctly classified scored higher on this answer compared to patients with PPTH who were misclassified as having migraine (see Table 3).
(a) Top features from clinical component measures and from features of fibertract data that contributed to the classification accuracy of distinguishing patients with persistent post-traumatic headache (PPTH) from patients with migraine.

Brain regions and fibertracts are named according to FreeSurfer convention. lh: left hemisphere; rh: right hemisphere. (b) Means and variance for features of patients with PPTH that were accurately classified as PPTH. (c) Means and variance for features of patients with PPTH that were misclassified as patients with migraine. SCAT: Sport Concussion Assessment Tool.
Note: Diffusion measures are expressed in scientific notation.
The best two structural features by modality when all features were evaluated by modality with the information gain criteria included right precentral thickness and left total mean thickness, right precental area and left white matter surface area, left hemisphere isthmus cingulate volume and estimated total intracranial volume, right hemisphere inferior parietal and lateral orbitalfrontal curvature. However, these brain structure features did not contribute significantly to increase the classification accuracy, as shown by the lack of improvement in overall classification accuracy from the clinical data-only model to the clinical + structural model.
The fibertracts that contributed most to classification when evaluated independently were the right anterior thalamic radiations (mean diffusivity) and the right superior longitudinal fasciculus (radial diffusivity).
Figure 3 shows those measures that were significantly different between PPTH patients that were correctly classified and those that were incorrectly classified using both t-statistics and Cohen’s d for effect size interpretation (38,39). The post-hoc plots in Figures 4 and 5 show that patients who were incorrectly classified as having PPTH had symptoms that were more similar to patients with migraine. In contrast, those patients who were correctly classified as having PPTH had more elevated symptoms of depression (BDI), anxiety (state and trait anxiety), more severe physical symptoms (SCAT, hyperacusis), more severe autonomic symptoms (COMPASS 31 orthostatic sub-score and total COMPASS 31 score), lower cognitive processing speed (Trails B, z-score), and were more likely to have post-traumatic stress disorder. Figure 6 shows the histogram of the concussion mechanism for patients with PPTH. The results indicate that the majority of correctly classified patients suffered blast-type injuries, whereas the majority of incorrectly classified patients with PPTH had mTBI due to sports-related activities or falling.

Measures of depression and anxiety. Differences between correctly classified patients with persistent post-traumatic headache (PPTH) and incorrectly classified patients with PPTH relative to patients with migraine.

Measures of physical and cognitive function. Differences between correctly classified patients with persistent post-traumatic headache (PPTH) and incorrectly classified patients with PPTH relative to patients with migraine.

Concussion characteristics. Differences between correctly classified patients with persistent post-traumatic headache (PPTH) and incorrectly classified patients with PPTH.
Discussion
This study interrogated the utility of classifying individuals with headache as either having migraine or PPTH attributed to mTBI using questionnaire data and brain structural (T-1 weighted and DTI) data. Results indicate that the combined use of questionnaire and brain imaging DTI data provided reasonable accuracy (78%) for distinguishing between migraine patients and patients with PPTH and that the model that combines both data types outperformed the classification model, which was based only on questionnaire data (71.9%).
Out of the three types of questionnaire measures included in the model (physical, psychological, and cognitive) the two most important features that helped to distinguish between PPTH and migraine were related to anxiety and decision-making difficulties. It is surprising that several of the clinical symptoms, including headache characteristics, hypersensitivity symptoms, sleep disturbances, and physical symptoms, were not “best” features to contribute to the classification accuracy, especially as previously published results of a similar but larger PPTH cohort have shown differences in patients with PPTH compared to migraine patients, with patients with PPTH showing more severe symptoms of autonomic dysfunction and sleep disturbances (9,40).This finding highlights how difficult it can be in a clinic setting to use symptoms alone to differentiate PPTH from a migraine exacerbation when someone had migraine prior to their TBI. This finding underscores the likely need for a multidimensional approach using imaging, blood-based biomarkers, neurophysiology, and other modalities that address similarities and differences between PPTH and migraine.
Adding measures of cortical thickness, volume, and area to the clinical features did not significantly contribute to increase the classification accuracy. However, regions that were important features included the posterior and isthmus cingulate, the inferior parietal region, and the banks of the superior temporal sulcus. These regions, which structurally wrap the limbic system, are integral to pain processing and multisensory integration and are areas previously implicated in showing alterations in patients with migraine and PPTH, which might indicate underlying pathophysiology unique to each disorder (6,41–46).
The fibertract data improved accuracy overall, a lot for migraine (from 79.4% to 97.1%) but not for PPTH (from 66.7% to 64.6%). It is therefore possible that questionnaire data plus DTI data would have had equal classification accuracy to questionnaire data plus morphometric data plus DTI data. Although these findings need to be validated in future studies, this may suggest that “trimming” morphometric data from the classifier would not negatively impact classification accuracy and would provide for optimal (and shorter) imaging protocols relevant for classification accuracy, which is essentially the goal in the development of an imaging biomarker.
It is noteworthy that for patients with PPTH adding brain morphometric data to the model slightly lowered the classification accuracy. It is possible that some brain metrics, including regional curvature measures, which are sensitive for identifying neurobiological components, have utility for identifying migraine structural biomarkers (for a recent chronic migraine biomarker review, see Filippi and Messina (47)) but are less useful for identifying structural biomarkers for PPTH. Future studies that develop classification models based on questionnaire data, brain structural and functional data will be needed to investigate whether the addition of functional brain connectivity patterns increases classification success for distinguishing between migraine and PPTH.
The right anterior thalamic radiations and the right superior longitudinal fasciculus were of significant importance for classifying migraine patients from patients with PPTH. There is prior evidence indicating abnormal diffusion characteristics for the anterior thalamic radiations and the superior longitudinal fasciculus in patients with migraine (48,49) as well as in patients with PPTH (7,50). In fact, our previous study results indicated that although migraine and patients with PPTH show alterations of the same tracts relative to healthy controls there were disease-specific differences in tract damage along the length of the fibertracts, which may suggest differences in the neuropathological mechanisms underlying PPTH versus migraine.
Limitations: Although the results of this study show a reasonable classification accuracy based on questionnaire and neuroimaging data, our results will need to be validated using an independent dataset of patients with PPTH and migraine. For this dataset, the classification accuracy was significantly better for migraine patients than for patients with PPTH. Migraine patients had a high classification accuracy of 97.1%, while the classification accuracy based on the combination of questionnaire, brain morphometric, and fibertract data for patients with PPTH was 64.6%. Several factors could account for the lower classification accuracy in patients with PPTH, including the variance associated with the injury mechanism (i.e. concussions related to falls vs. motor vehicle accidents vs. sports-related vs. blast-related), years lived with PPTH, and headache frequency. Indeed, post-hoc analyses indicated differences in measures of depression, anxiety, physical and cognitive symptoms between PPTH patients that were correctly classified versus PPTH patients that were incorrectly classified. Correctly classified PPTH patients had a higher symptom load and were more likely to have post-traumatic stress disorder and mTBI due to blast injuries. These differences within the PPTH cohort as well as differences in enrolment patterns could have contributed to the variance in patients with PPTH. Approximately half of the patients with PPTH were enrolled from Mayo Clinic compared to the other half, who were enrolled from the Phoenix VA. In contrast, the majority of migraine patients were enrolled through Mayo Clinic, thus yielding a more homogeneous cohort sample. There are limitations relevant to the imaging sequences used; we acknowledge that fibertracking and structural measurements based on non-isotropic voxels could have had a negative impact on the accuracy of the diffusion and brain structural results. Several disease factors were not assessed as part of this study, which could have influenced our results. For example, we did not account for the use of preventive medications in the classification model nor asked patients to complete a headache diary, which would have allowed us to determine the phase of the migraine/headache cycle that each patient was in during testing.
Conclusion
A classification model based on questionnaire, brain morphometric, and fibertract data distinguished individual patients with migraine from individual patients with PPTH with an average accuracy of 78%. Questionnaire measures of anxiety and decision making and fibertracts were important contributors to the classification accuracy. Classification accuracy of migraine was higher than that of PPTH. The heterogeneity of the PPTH cohort relative to psychological, physical and cognitive symptom load and the variance within the cohort as to the concussion mechanism may have played a role in the lower classification accuracy for patients with PPTH.
Article highlights
The combination of questionnaire, brain morphometric and fibertract data had good accuracy (78%) for distinguishing individual patients with migraine from individual patients with PPTH. Questions on mood and decision making and DTI measures of the anterior thalamic radiations and superior longitudinal fasciculus were the most important features that contributed to the classification accuracy. A classification model that included questionnaire and brain structural data was more accurate than a model including questionnaire data only for distinguishing individual patients with migraine from individual patients with PPTH.
Footnotes
Acknowledgements
We are grateful to the study participants, imaging technologists and study coordinators who have dedicated their time to this study. We would like to thank Jonalle Sauer for her design expertise.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Office of the Assistant Secretary of Defense for Health Affairs, through the Peer Reviewed Medical Research Program under Award No. W81XWH-15-1-0286 and Award No. W81XWH-19-1-0534. Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense. The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014 is the awarding and administering acquisition office.
