Abstract
Objectives
Often, persistent post-traumatic headache and migraine are phenotypically similar. However, the similarities and differences in the neuropathological underpinnings of persistent post-traumatic headache and migraine require further understanding. We used diffusion tensor imaging (DTI) and a novel method for detecting subtle changes in fibertract integrity by measuring node-by-node parameters along each tract to compare fibertract profiles between those with migraine and those with persistent post-traumatic headache, and compared both cohorts to a group of controls.
Methods
Eighteen fibertracts were reconstructed for 131 subjects, including 49 patients with persistent post-traumatic headache attributed to mild traumatic brain injury, 41 with migraine, and 41 controls. Node-by-node diffusion parameters of mean diffusivity and radial diffusivity were calculated along each tract. Mean diffusivity and radial diffusivity measurements were averaged along quartiles of each tract for statistical interpretation and group comparison. Using a post-hoc analysis, correlations between tract quartile measurements and headache frequency were calculated.
Results
There were significant differences between migraine and persistent post-traumatic headache cohorts for quartile measurements of mean diffusivity or radial diffusivity in the bilateral anterior thalamic radiations, cingulum (angular bundles and cingulate gyri), inferior longitudinal fasciculi, and uncinate fasciculi, the left corticospinal tract, and the right superior longitudinal fasciculi-parietal portion. For migraine patients, there was a significant positive correlation between headache frequency and forceps major mean diffusivity, whereas for persistent post-traumatic headache there was a positive correlation between headache frequency and cingulum angular bundle mean diffusivity and radial diffusivity.
Conclusions
Quartile measurements of radial diffusivity and mean diffusivity indicate unique differences in fibertract profiles between those with migraine vs. persistent post-traumatic headache. Although for both migraine and persistent post-traumatic headache there was a positive relationship between fibertract alterations and headache frequency, there were disease-specific differences between headache frequency and fibertract injury patterns. These findings might suggest potential differences in the neuropathological mechanisms underlying migraine and persistent post-traumatic headache.
Keywords
Introduction
Migraine and persistent post-traumatic headache (PPTH) have similar headache characteristics. In fact, migraine is a common phenotype observed in individuals with post-traumatic headache (PTH) (1–7). Despite these phenotypic similarities, the neuropathologic similarities and differences between migraine and PPTH are yet to be elucidated. Although a number of studies have demonstrated alterations in white matter integrity in patients with migraine (8–15) or in patients with PPTH (16–18), the similarities and differences between migraine and PPTH are insufficiently understood since few studies have directly compared them.
The objective of this study was to estimate white matter integrity using diffusion tensor imaging (DTI) in 18 major brain fibertracts in patients with PPTH attributed to mild traumatic brain injury (mTBI) compared to patients with migraine. A control cohort was included as a comparison group for referencing aberrations of fibertract profiles. Rather than measuring average diffusion characteristics for each tract, we used a novel technique that allows for incremental (node-by-node) measurements of mean diffusivity (MD) and radial diffusivity (RD) along each tract for determining subtle changes in fibertract profiles that might go undetected when diffusion averages are calculated over entire tracts. RD and MD are measures of water diffusion in the brain and provide indirect measures of tract health. RD measures the direction of water movement perpendicular to the main axis of the axon whereas MD, which is directionally nonspecific, measures the average directional diffusion of water within brain tissue. Although the pathomechanism underlying MD and RD is not clearly understood, RD is believed to be reflective of compromised tract integrity purportedly related to demyelination or abnormalities in axonal density (19,20) whereas aberrations in MD values could reflect abnormalities within the intra- and extracellular space or degeneration of the neuropil (21). The main goal of this study was to determine changes in fibertract profiles that are unique to either migraine or to PPTH. Furthermore, in order to better understand whether changes in fibertract profiles relate to disease characteristics, the relationships between headache frequency and alterations in fibertract profiles were explored.
Methods
This study enrolled 138 subjects, all of whom gave written consent prior to being included for study participation. This study was approved by the local institutional review boards at Mayo Clinic and the Phoenix VA Health Care System and the US Department of Defense Human Research Protection Office. Migraineurs and patients with PPTH were enrolled from the Phoenix VA Healthcare system and Mayo Clinic Arizona. The diagnosis of migraine or PPTH was made by a board-certified headache specialist using the diagnostic criteria set forth by the International Classification of Headache Disorders 3 (ICHD-3) beta criteria for “migraine with or without aura” or “persistent headache attributed to mild traumatic injury to the head”, respectively (22). Controls were recruited from within the community or through local advertisement. Subjects were between the ages of 18–65 years. Exclusion criteria for PPTH included history of moderate or severe TBI and history of migraine prior to mTBI. Exclusion criteria for subjects with migraine and controls included history of TBI. Controls were also excluded if they had a history of migraine or if they had tension-type headaches on four or more days per month. Exclusion criteria for all subjects were the following: Moderate to severe head trauma, pregnancy, safety contraindication to MR imaging (i.e. severe claustrophobia, metal in the body, pacemakers, stents), or inability to lie still for longer than 45 minutes. Subjects were not excluded from study participation if they were taking abortive or preventive medications for headache.
Subject demographics.
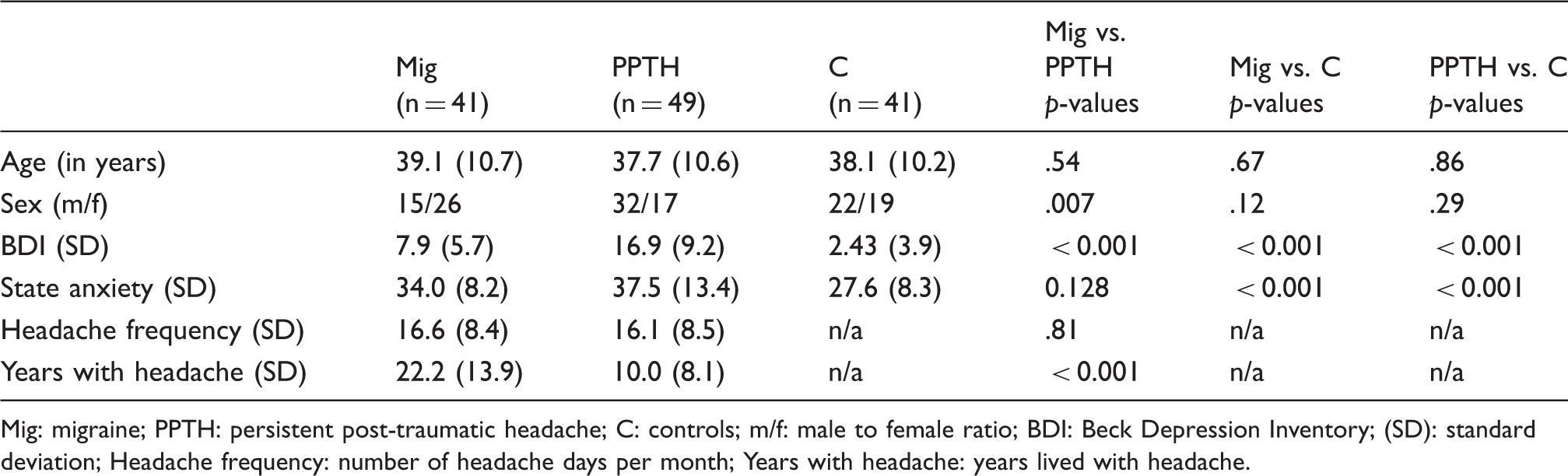
Mig: migraine; PPTH: persistent post-traumatic headache; C: controls; m/f: male to female ratio; BDI: Beck Depression Inventory; (SD): standard deviation; Headache frequency: number of headache days per month; Years with headache: years lived with headache.
Imaging acquisition and postprocessing
All study participants underwent neuroimaging at Mayo Clinic Arizona on a 3-Tesla Siemens (Siemens MAGNETOM Skyra, Erlangen, Germany) scanner using a 20-channel head/neck coil. The magnetic resonance imaging (MRI) protocol included a T1-weighted magnetization prepared rapid image acquisition gradient-echo sequence (MPRAGE): TR = 2400 ms; TE = 3.03 ms; flip angle = 8 degrees, 1 × 1 × 1.3 mm3 voxels. T2-weighted imaging parameters were the following: TR = 6800 ms, TE = 84 ms, flip angle = 150 degrees, 1 × 1 × 4 mm3 voxels. DTI imaging was acquired using a dual-spin-echo, echo-planar imaging (EPI) sequence. Diffusion gradients were applied at 30 non-linear directions (b = 1000 s/mm2). DTI imaging parameters were the following: TR = 5100 ms, TE = 73 ms, flip-angle = 90 degrees, slice thickness = 4 mm; 1.7 × 1.7 × 4 mm3 voxels. One image (b = 0) was acquired without diffusion weighting.
Subject data were excluded if structural abnormalities were detected on routine T1 or T2-weighted imaging. According to commonly accepted imaging standards, DTI data was excluded from the final analysis if subject motion during scanning exceeded more than 2 degrees rotational and/or 2 mm translational movement (26,27).
T1 data and DTI data were post-processed using the open-source software FreeSurfer version 5.3 (28). All imaging was post-processed on a single Macintosh workstation running OS X Lion 10.7.5 software to avoid the potential of post-processing irregularities arising from the use of multiple workstations (29). Scans were post-processed according to the following workflow: T1 and T2 imaging data were checked for brain abnormalities. If none were detected, subjects' imaging scans were included for further post-processing using FreeSurfer software and the integrated tractography toolbox TRACULA (TRActs Constrained by UnderLying Anatomy) (30). As TRACULA uses T1-weighted high-resolution images as anatomical priors for each individual, T1-weighted scans were reconstructed according to the standard “recon-all” stream, which includes skull stripping, transformation into Talairach space, brain segmentation and parcellation, brain intensity normalization, boundary tessellation and surface deformation (31–34). A Markov Chain Monte Carlo algorithm was used to merge each subject's own structural scan to that of a pre-labeled training set for optimizing the accuracy of placement of fibertracts (27,30). To prevent inclusion of compromised data sets, tracts were manually checked by a trained technician for motion artifacts and post-processing errors prior to including subject data for group analyses. Previous studies, including our own, have used this method for identifying tract damage in several patient populations including autism, schizophrenia, temporal lobe epilepsy, and migraine (8,30,31–37) and have demonstrated relationships between tract damage and clinical disease characteristics. The additional advantage of this technique is that it allows for the calculation of whole tract diffusion metrics as well as node-by-node measurements of diffusion metrics along each tract for detecting subtle changes in tract integrity. All fibertracts were reconstructed according to the standard post-processing routine set forth within the TRACULA pipeline, which enables the automatic reconstruction of 18 major fibertract bundles including the bilateral anterior thalamic radiations, bilateral cingulum angular bundles, bilateral cingulum cingulate gyri, bilateral cortico-spinal tracts, bilateral superior longitudinal fasciculi -parietal, bilateral superior longitudinal fasciculi-temporal, bilateral inferior longitudinal fasciculi, bilateral uncinate fasciculi, as well as the forceps major tract and the forceps minor tract.
Fibertract quartile estimates: Node-by-node fibertract metrics of RD and MD were calculated along each white matter tract. Tracts were divided into quartile lengths, and RD and MD values were calculated for each quartile (1st, 2nd, 3rd and 4th quartile means). Quartile measurements for the anterior thalamic radiations were calculated starting from the frontal cortex (MQ1) to the thalamus (MQ4). Mean quartile measurements for the cingulum angular bundle and the cingulum cingulate gyrus were taken from the posterior end (MQ1) to the anterior portion of the tracts (MQ4), quartile measurement for the cortico-spinal tract were taken from the brainstem (MQ1) to the motor cortex (MQ4), the inferior longitudinal fasciculus was measured from the temporal portion (MQ1) to the occipital portion (MQ4), the superior longitudinal fasciculus-temporal was measured from the frontal (MQ1) to the temporal cortex (MQ4), the superior longitudinal fasciculus- parietal was measured from the frontal (MQ1) to the parietal cortex (MQ4), and the uncinate fasciculus was measured from the temporal (MQ1) to the orbitofrontal cortex (MQ4). Both the forceps major and minor were measured from the left (MQ1) to right (MQ4) hemisphere.
The “mean jitter” was also measured for each tract. Jitter is a measure of the absolute difference in a quantity (e.g. RD, MD) between sequential samples of that quantity. For example, for a particular white matter tract, there are N samples of RD along the physical length of the tract. To calculate the jitter along the tract, the absolute value of the difference of RD for all pairs of neighboring samples is taken. Thus, there are N−1 jitter values for an N-sample white matter tract. The mean jitter is the mean of all jitter values along the tract.
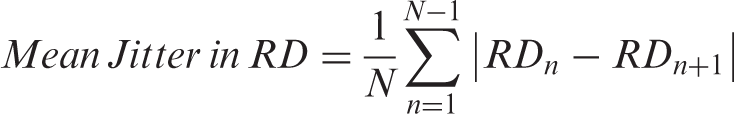
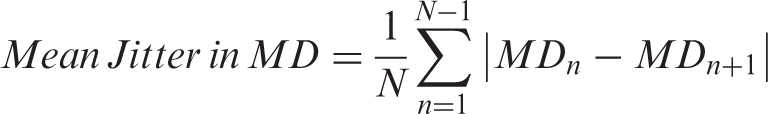
The mean jitter value measures how related a sample of a quantity is to its neighbors. A high mean jitter value indicates a set of quantities that are less related to their neighbors. A low mean jitter value indicates samples of a quantity that are more related to their neighbors, and therefore change more slowly along the length of a tract. The mean jitter is a measurement of the difference in RD or MD values between sequential node samples along the tract and is reflective of the variance within a fibertract.
To capture differences between patient groups while controlling for clinical covariates, a one-way analysis of covariance (ANCOVA) was performed on the DTI features, controlling for differences in sex and years lived with headache (excluded in comparisons with controls). The statistical significance threshold was set to p < 0.05. To mitigate the increased false discovery rate caused by testing multiple hypotheses between patient groups and within each fibertract, a Benjamini-Hochberg p-value adjustment (38) was used on the p-values from ANCOVA testing.
Correlation analysis
To understand the relationship between DTI metrics and headache frequency (defined as the number of headache days per month), diffusion metrics that showed significant differences between migraineurs and controls and between PPTH and controls were correlated with headache frequency. Pearson correlations were calculated separately for migraineurs and PPTH.
Results
Subjects: 138 subjects were initially enrolled. Data collected from seven subjects were excluded for the following reasons: T1-weighted findings of global atrophy (control), hyperintensities on T2-weighted imaging (one patient with migraine and one with PPTH), movement in the scanner exceeding the preset motion threshold (two patients with PPTH) and post-processing errors (one control, one patient with PPTH). Due to the exclusion of seven subjects, this study included a total of 131 subjects: 49 (32 males, 17 females) individuals with PPTH attributed to mTBI, 41 (15 males, 26 females) with migraine (30 with chronic migraine and 11 with episodic migraine), and 41 (22 males, 19 females) controls. Twenty five migraineurs had migraine with aura and 16 had migraine without aura. Ten PPTH patients had one mTBI, 20 had two mTBIs, five had three mTBIs, one had four mTBIs, three had five mTBIs and 10 had six or more mTBIs. Forty-six PPTH patients had no pre-trauma tension-type headaches. One PPTH patient had one tension-type headache per month, one PPTH patient had two tension-type headaches per month and one patient had five tension-type headaches per month. Ninety-six percent of patients with PPTH had a phenotype consistent with migraine (n = 37) or probable migraine (n = 10) and two patients had a headache phenotype consistent with tension-type headache. At the time of scanning, 36.6% of those with migraine were pain free while 63.4% had at least mild pain. Among those with PPTH, 12.3% were pain free and 87.7% had at least mild pain. On average, those with migraine reported a pain level of 2.0 (where 0 = no pain; 10 = worst imaginable pain) while those with PPTH reported a pain level of 3.5.
Subject demographics and group differences are shown in Table 1.
There were no significant age differences between migraineurs and PPTH (p = .54) between migraineurs and controls (p = .67) or between PPTH and controls (p = .86). Between migraineurs and PPTH, there were significant differences in male to female group ratios (p = .007). There were no differences in sex distribution between migraineurs and controls (p = .12) or between PPTH and controls (p = .29). For all group comparisons (migraineurs vs. PPTH, migraineurs vs. controls and PPTH vs. controls) there were significant differences on depression raw scores. Although raw scores for the BDI were significantly different between migraineurs and controls, the average scores of both groups were in the normal, non-depressed range (<10 on the BDI). The average score of 16 for PPTH falls within the range of mild mood disturbances. There were no significant differences between PPTH and migraineurs on days with headache per month (p = .81) but there were significant differences between PPTH and migraineurs on the number of years lived with headache (p < 0.001). For migraineurs relative to PPTH, there were no significant differences on situational anxiety raw scores (p = .13) but there were significant differences between migraineurs and controls (p < 0.001), and between PPTH and controls (p < 0.001).
Fibertract quartile measurements
Table 2 and Figure 1 show the significant tract mean quartile (MQ) differences between migraineurs and those with PPTH, between PPTH and controls, and between migraineurs and controls.
Incremental measurements of mean diffusivity (MD) and radial diffusivity (RD) for the anterior thalamic radiations, corticospinal tracts, inferior longitudinal fasciculi, cingulum angular bundles. 1b: uncinate fasciculi, cingulum cingulate gyri, superior longitudinal fasciculi – parietal portion and the forceps major and forceps minor. C: controls; Mig: migraine; PPTH: persistent post-traumatic headache. On the MRI images, arrows reflect the direction of quartile measurements for each tract. Quartile measurements for the superior longitudinal fasciculus – temporal portion are not shown on the figure, as there were no significant group differences. Differences in tract mean jitter value (MJV) and mean quartile (MQ1, MQ2, MQ3, MQ4) radial diffusivity (RD) and mean diffusivity (MD) measurements between migraineurs (Mig) versus patients with persistent post-traumatic headache (PPTH), between PPTH and healthy controls (C) and between Mig and C. Only those tracts and quartiles for which there was at least one difference between subject groups are included in the table. Tract names and tract orientations are shown in italics. Stars indicate the subject group for which there was a significant positive correlation (p < 0.05) between headache frequency and quartile measurements of MD or RD. p-values and tract names that remain significant after Benjamini-Hochberg correction for multiple comparisons are in bold. L: Left; R: Right; STD: standard deviation. Note: Scientific notation was used to express mean RD and MD values (i.e. −04 indicates 10−4). Quartile measurements for the superior longitudinal fasciculus – temporal portion are not shown, as there were no significant group differences.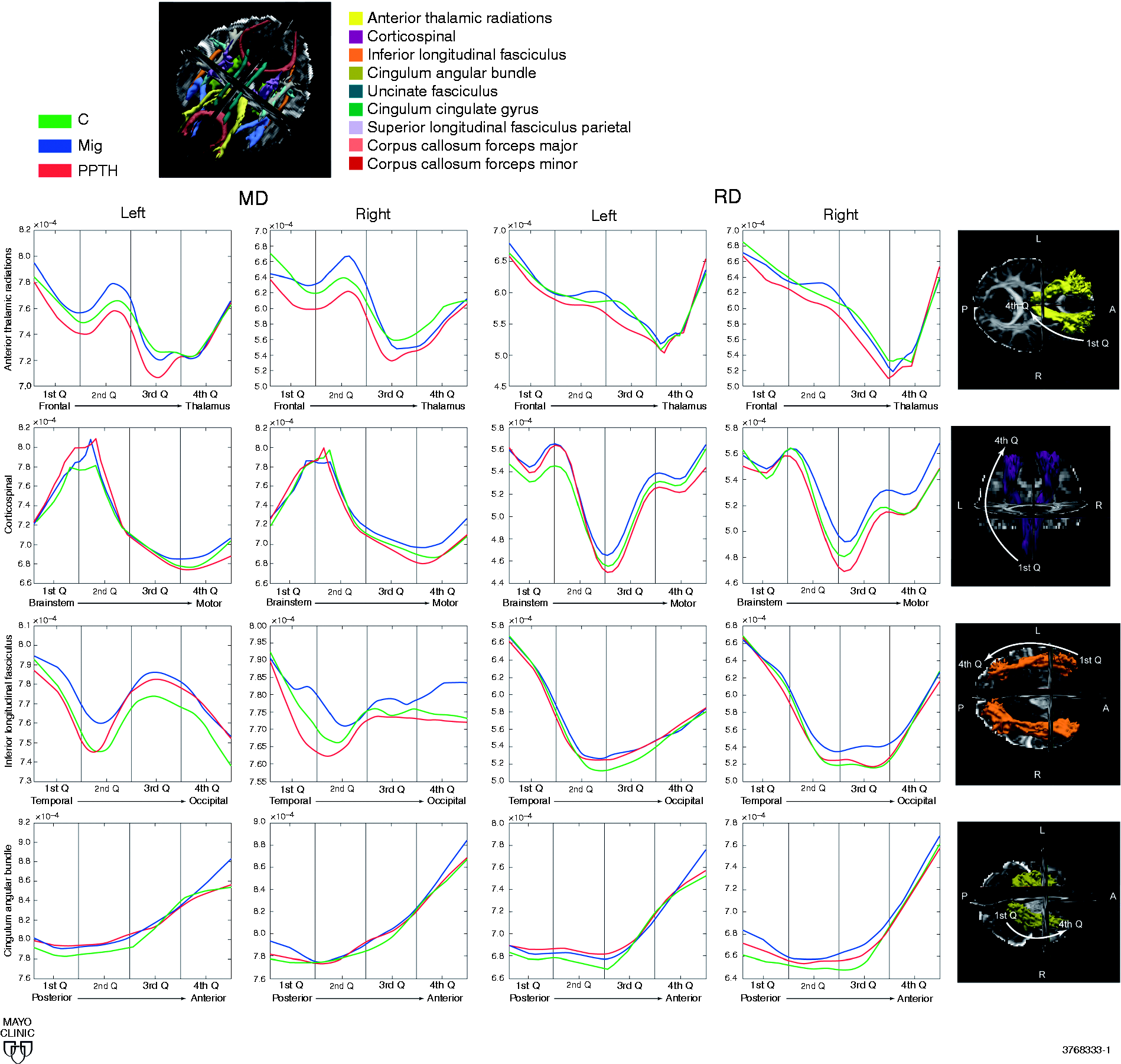
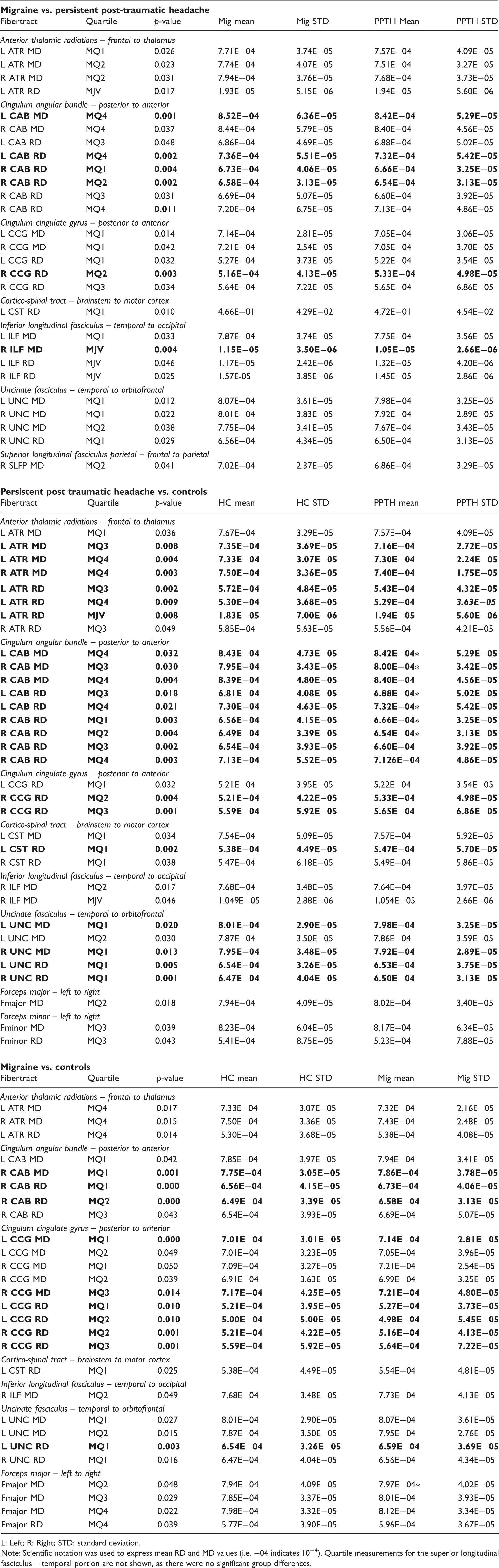
Migraine versus persistent post-traumatic headache
There were significant MQ measurement differences between PPTH and migraine for the following tracts: Left and right anterior thalamic radiation, MD; left anterior thalamic radiation, RD mean jitter; left and right cingulum angular bundle, MD and RD; left and right cingulum cingulate gyrus, MD and RD; left cortico-spinal tract, RD; left inferior longitudinal fasciculus, MD; right inferior longitudinal fasciculus, MD mean jitter, and left and right inferior longitudinal fasciculus, RD mean jitter; left and right uncinate fasciculus, MD; right uncinate fasciculus, RD; right superior longitudinal fasciculus-parietal portion, MD. There were no significant mean quartile tract differences for the fasciculus major, fasciculus minor or the bilateral superior longitudinal fasciculus-temporal portion.
Migraine versus controls
There were significant MQ differences between migraineurs and controls for the following tracts: Left and right anterior thalamic radiation, MD; left anterior thalamic radiation, RD; left and right cingulum angular bundle, MD; right cingulum angular bundle, RD; bilateral cingulum cingulate gyrus, MD and RD; left cortico-spinal tract, RD; right inferior longitudinal fasciculus, MD; left uncinate fasciculus, MD; left and right uncinate fasciculus, RD; forceps major, MD and RD. There were no significant mean quartile differences between migraineurs and controls for the bilateral superior longitudinal fasciculi – temporal portions or the forceps minor tract.
Persistent post-traumatic headache versus controls
There were significant group differences between PPTH and controls for the following tracts: Left and right anterior thalamic radiation, MD; left and right anterior thalamic radiation, RD; left anterior thalamic radiation, RD and mean jitter; left and right cingulum angular bundle, MD and RD; left and right cingulum cingulate gyrus, RD; left cortico-spinal tract, MD; left and right cortico-spinal tract, RD; right inferior longitudinal fasciculus, mean jitter and MD; left and right uncinate fasciculus, MD and RD; forceps major and forceps minor, MD and RD. There were no significant differences between PPTH and controls for the superior longitudinal fasciculus-parietal portion or the superior longitudinal fasciculus – temporal portion.
Post-hoc correlation tests with headache frequency
For PPTH, there were positive relationships between headache frequency and right cingulum angular bundle MD (MQ3: r = 0.3481, p = 0.0142); left cingulum angular bundle MD (MQ4: r = 0.3459, p = 0.0160); right cingulum angular bundle RD (MQ1: r = 0.3438, p = 0.0156; MQ2: r = 0.3251, p = 0.0227); and left cingulum angular bundle RD (MQ3: r = 0.3276, p = 0.0230; MQ4: r = 0.3097, p = 0.0322). These correlations indicate that PPTH patients with more frequent headache had higher MD and RD in the right and left cingulate angular bundle quartiles.
For migraineurs, there was a single positive correlation between headache frequency and forceps major MD (MQ2: r = 0.3291, p = 0.0381).
Discussion
In the present study, we interrogated white matter tract integrity differences between migraineurs and those with PPTH by measuring RD and MD along the length of 18 fibertracts. A normal control cohort was included for referencing the direction of fibertract alterations relative to both patient groups. Our main findings showed significant tract measurement differences between migraineurs and PPTH in the bilateral anterior thalamic radiations, bilateral cingulum (angular bundle and cingulate gyrus), bilateral inferior longitudinal fasciculi, bilateral uncinate fasciculi, the left cortico-spinal tract, and the right superior longitudinal fasciculus – parietal portion. Relative to controls, migraineurs and PPTH had weaker MD in the anterior thalamic radiations and stronger RD in the left corticospinal tract, and stronger MD in the forceps major.
Incremental measurements of tract metrics at different points along each tract allowed the visualization and comparison of tract integrity differences along the length of the tracts amongst three subject cohorts. (Figure 1). As expected, all groups showed similar patterns in diffusion characteristics measured along the length of the tract, with generally stronger MD and lower RD in the middle of the tracts relative to the tract endings. However, there were specific differences in quartile measurements between migraineurs and PPTH, with migraineurs having stronger MD relative to PPTH in a number of tract quartiles. Migraineurs had stronger MD for quartile measurements in the bilateral anterior thalamic radiations and stronger MD in the bilateral cingulum (angular bundles and cingulate gyri), bilateral inferior longitudinal fasciculi and the bilateral uncinate fasciculi. There were no differences in tract quartile measurement where PPTH had stronger MD relative to migraineurs. Additionally, migraineurs also had stronger MD relative to controls for quartile measurements in the bilateral cingulum (angular bundles and cingulate gyri), left uncinate fasciculus, right inferior longitudinal fasciculus and the forceps major. Differences in RD were mixed, with migraineurs having higher RD for most tract quartiles in the cingulum angular bundle relative to PPTH. However, there were also several tract quartiles for which PPTH had higher RD relative to migraineurs (anterior thalamic radiations, cingulum cingulate gyrus, corticospinal tract, and the inferior longitudinal fasciculus). Several DTI studies have shown higher MD and RD in migraineurs relative to controls in previous studies (9,10,39) including our own study of a different subject cohort (8). Although the results of our previous study measured average MD over entire tract lengths (and not for quartile measurements), our previous results and current results showed consistent findings of higher MD in the right inferior longitudinal fasciculus and higher RD in the left corticospinal tract for migraineurs relative to controls.
Most differences between migraineurs and PPTH involved the cingulum and the anterior thalamic radiations. The cingulum resembles a ring-like band that overlies the corpus callosum and is divided into two portions: i) The cingulum cingulate gyrus, which extends from the isthmus of the corpus callosum to the orbitofrontal cortex where the fiber bundle runs anterior and inferior along the genu of the corpus callosum (40), and ii) the cingulum angular bundle, which connects the parahippocampal cortex to the isthmus of the cingulate. In sagittal plane, the angular bundle extends from the temporal pole posteriorly to the parietal lobe and then curves to hug the isthmus of the corpus callosum. Both tracts are implicated in executive function, memory and emotion processing (41,42). Involvement of the cingulum in mTBI is well-documented, in fact several DTI studies have reported structural narrowing or altered cingulum integrity in patients with cognitive impairments following concussion (44–46).
The anterior thalamic radiations connect the thalamus and limbic structures with the frontal cortex. Several studies, including our own, have found abnormal MD and RD within the anterior thalamic radiations in patients with migraine (8,11), in patients during the chronic stage of concussion (48), and aberrations of MD in patients with post-traumatic symptoms relative to those without post-traumatic symptoms (49).
Although the precise neuromechanism underlying diffusion measures remains debated, there is some indication that changes in MD, which describes the average diffusion along the three major diffusion directions, might be related to alterations in the intra- and extracellular space and a thinning of the neuropil layer (21). Instead, higher RD, which describes the diffusion direction perpendicular to the main axis of the tract, might be related to damage of the myelin sheath, axonal damage and gliosis (19,44,50).
Although there were no differences for tract mean jitter between migraineurs and controls, there were mean jitter differences between patients with PPTH and controls and between PPTH and migraine (that is the comparison for which there were the most differences for MJ). With PPTH having higher mean jitter MD in the right inferior longitudinal fasciculus and higher mean jitter RD in the left anterior thalamic radiations, indicating higher fluctuations for consecutive measurements along the tract. Although the neuropathological consequences underlying mean jitter are unknown, larger fluctuations in RD or MD might be interpreted as higher tract inconsistencies (i.e. more dispersed tract damage) within both tracts in patients with PPTH relative to controls. There were also significant differences in mean jitter between migraineurs and patients with PPTH. Migraineurs had higher mean jitter MD and RD in the right inferior longitudinal fasciculus relative to patients with PPTH. However, patients with PPTH had higher mean jitter RD in the left inferior longitudinal fasciculus and higher mean jitter RD in the left anterior thalamic radiations relative to migraineurs.
For migraineurs, there was a positive relationship between headache frequency and forceps major MD, whereas for PPTH there was a positive association between headache frequency and bilateral cingulum angular bundle MD and RD. The difference in correlation patterns between quartile fibertract measurements and headache frequency could suggest differences between migraineurs and PPTH relative to the neurosubstrates that mediate headache patterns.
Study limitations: In order to best match PPTH to the migraine cohort, this study included episodic and chronic migraineurs. Although this could be considered a study limitation, having a wider range in headache frequency in our migraine cohort allowed us to better balance headache frequency in patients with PPTH to our migraine cohort and to determine relationships between fibertract integrity and headache frequency. There were significant differences between migraineurs and PPTH relative to mood, with the cohort of PPTH having mild mood disturbances relative to migraineurs (who were in the normal range). As previous studies have shown a strong comorbidity between PPTH and mood dysfunction (47,51), indicating that mood alterations are either interlinked or are inherent to the disease mechanism underlying PPTH, we decided not to exclude patients with PPTH based on mood alterations in order to recruit a study cohort that is more reflective of the general population with PPTH. Furthermore, at the design stage, we decided not to control our statistical model for potential imbalances in mood alteration, as mood dysfunctions are part of the clinical profile of PPTH. Our study results could have been influenced by several factors; this study did not specifically assess medication-overuse in patients with PPTH or migraine and it is therefore possible that some patients were overusing medication. As several studies have reported structural brain changes associated with medication overuse (52,53) we cannot rule out that medication overuse could have influenced our study findings. Similarly, as we did not assess psychiatric history other than depression or anxiety, history of psychiatric disorder may have influenced our findings. Future studies should investigate the potential relationship between the number of brain injuries experienced by an individual and white matter tract integrity.
Conclusion
Node-by-node measurements along fibertracts are helpful for revealing subtle changes in tract profiles between migraine and PPTH. Although the specific microstructural abnormalities that underlie changes in MD or RD remain uncertain, the unique patterns of MD differences in the anterior thalamic radiations and the cingulum (angular bundle and cingulate gyrus) – with migraineurs having higher MD relative to PPTH and relative to controls – might suggest distinct differences in disease mechanisms for PPTH versus migraine. Future studies using machine-learning algorithms are needed to identify whether these differences have potential for developing disease markers that can distinguish individual migraineurs from individual patients with PPTH.
Key findings
Measurements of MD and RD indicate unique differences in fibertract profiles between migraineurs and those with PPTH. Disease-specific associations between headache frequency and fibertract injury patterns might suggest potential differences in the neuropathological mechanisms underlying migraine and PPTH.
Footnotes
Acknowledgements
The authors would like to thank all study participants, research coordinators and imaging technologists for their time and dedication to this project. We would like to thank Jonalle Sauer for her graphic design expertise.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (NIH) grant NIH K23NS070891 and by the Office of the Assistant Secretary of Defense for Health Affairs, through the Peer Reviewed Medical Research Program under Award No. W81XWH-15-1-0286. Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense. The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014 is the awarding and administering acquisition office.
