Abstract
Background
Migraine headache and complex regional pain syndrome share mechanisms, such as neuroinflammation, central sensitization and loss of inhibitory pain controls, that could provoke or exacerbate symptoms in both disorders. In the present study, it was hypothesized that headaches would worsen after the onset of complex regional pain syndrome and that limb pain would be more severe in patients with co-morbid headaches than in patients who remained headache-free. Notably, complex regional pain syndrome is associated with ipsilateral cranial symptoms such as photophobia and forehead hyperalgesia. Whether shared mechanisms might drive these symptoms was also explored.
Methods
Eighty-eight patients with complex regional pain syndrome were asked about their previous and current headache experience. The spatial distribution of pain was quantified from pain drawings, and hyperalgesia to mechanical and thermal stimulation was assessed in the limbs and forehead. In addition, the visual discomfort threshold was measured separately for each eye.
Results
Sixty-six percent of patients reported that headaches (primarily migraine) had developed or worsened after the onset of complex regional pain syndrome and 22 percent now had daily or near-daily headaches. Limb pain and hyperalgesia were greater in such cases than in those with stable headaches or who remained headache-free. Photophobia and forehead hyperalgesia were greater ipsilateral than contralateral to symptoms of complex regional pain syndrome in patients with stable or worsening headaches but were symmetrical in headache-free patients. In addition, photophobia was symmetrical in patients with recurrent tension-type headaches. Patients with worsening headaches were younger at the onset of complex regional pain syndrome than patients with stable headaches or who were headache-free, in line with greater vulnerability to migraine in younger than older adults. In a subgroup of patients, the pain of complex regional pain syndrome extended from the upper limb to the ipsilateral dorsal cervical region, a documented source of pain in migraine. However, headaches ipsilateral to complex regional pain syndrome also recurred in patients with lower limb pain, indicating involvement of other pain mechanisms.
Conclusions
Together, the findings indicate that headaches with features of migraine develop after the onset of complex regional pain syndrome. In turn, this is associated with ipsilateral cranial symptoms and heightened limb pain. We suggest that shared pathophysiology increases susceptibility to ipsilateral cranial symptoms and exacerbates pain in both disorders, potentially in a positive loop. Breaking this cycle might permit otherwise intractable symptoms and pain to subside.
This is a visual representation of the abstract.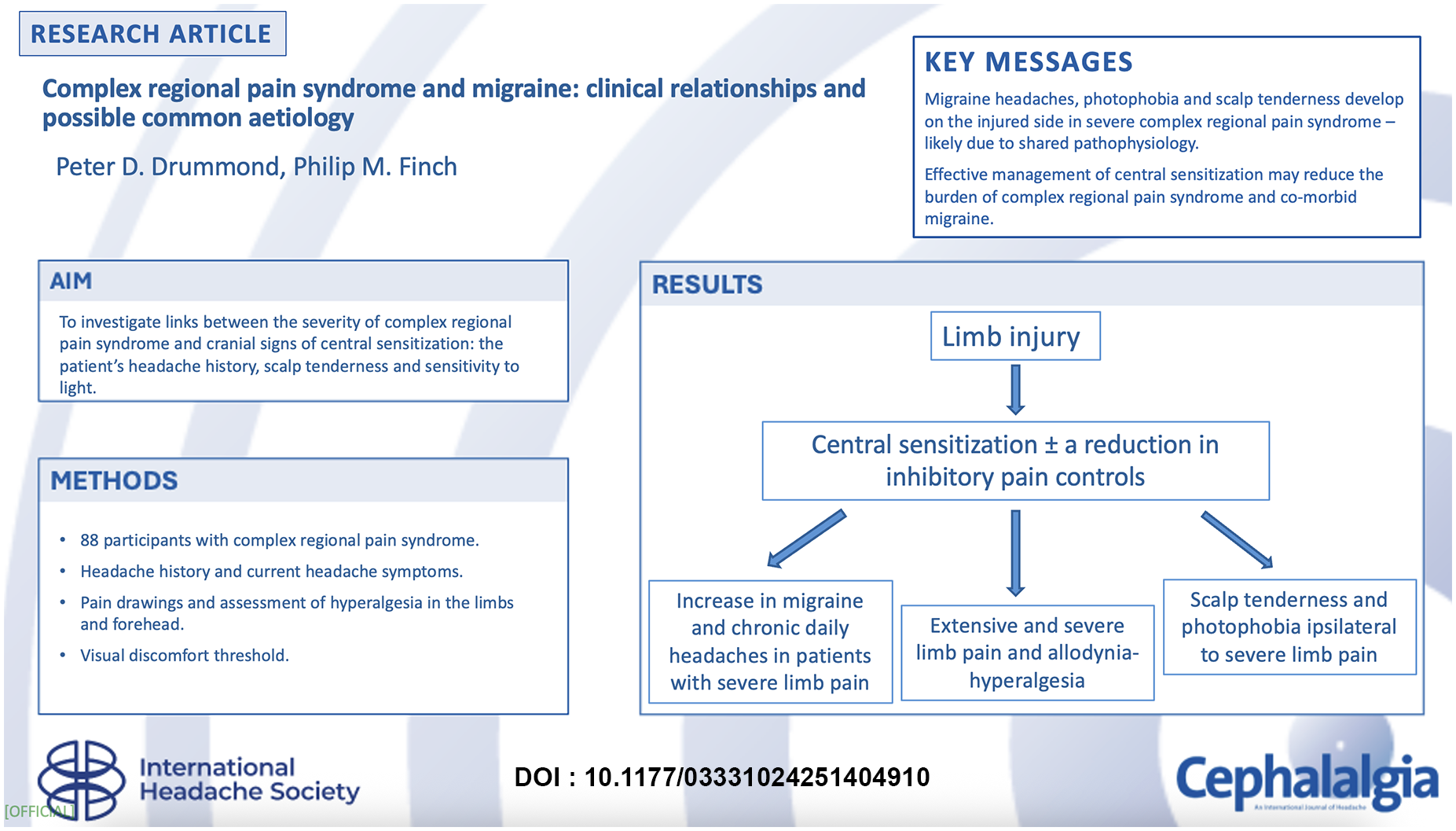
Keywords
Introduction
Complex regional pain syndrome (CRPS) usually develops after a limb fracture or sprain (1). In place of normal recovery, pain often builds and spreads away from the site of injury (2–4). Similarly, hyperalgesia to mechanical and thermal stimulation spreads to more distant ipsilateral sites (4,5), in tandem with lateralized discomfort to light and noise (6,7). Applying a 2°C stimulus to the ipsilateral forehead can aggravate pain in the CRPS-affected limb (8). Furthermore, in some cases, sudden loud noises intensify pain, more so when administered to the ipsilateral than contralateral ear (9). Together, these observations suggest that supraspinal changes in pain processing contribute to sensory disturbances in CRPS and exacerbate pain.
Migraine and chronic daily headache are risk factors for CRPS (10–12), possibly due to mechanisms shared with CRPS. At the molecular level, release of cytokines from activated microglia may contribute to neuroinflammation in migraine (13) and CRPS (14,15); furthermore, central sensitization and loss of inhibitory pain controls may exacerbate pain in both conditions (16–20). Potentially, these shared mechanisms could increase susceptibility to recurrent headaches in patients with CRPS; and, conversely, exacerbate CRPS in patients with co-morbid headaches.
Although plausible, evidence to support these proposals is scarce. In one of the few studies to explore links between CRPS and headaches, the prevalence of migraine and chronic daily headache was 1.9 to 3.6 times higher in patients with CRPS than in the general population (11). CRPS began at an earlier age, and allodynia was more common in patients with migraine or chronic daily headache than in headache-free patients or those with tension-type headache. Similar findings were reported recently (21). Recurrent headaches typically pre-dated CRPS onset in these studies (11,21) but whether the headache pattern changed thereafter is unclear.
To address this point, links between symptoms of CRPS and the patient's previous and current headache experience were investigated in the present study. As headaches are a risk factor for CRPS (10–12), we postulated that mechanisms that underlie this risk would exacerbate CRPS. Specifically, we hypothesized that the pain of CRPS would be more severe in patients with than without co-morbid headaches. Within the group with headaches, we explored whether the pain of CRPS differed between patients whose headaches had worsened versus those with stable headaches; and whether headaches were most frequent in patients with the most persistent or painful CRPS (21,22).
Cranial symptoms such as hyperalgesia, photophobia and phonophobia/hyperacusis are associated both with migraine and CRPS (4,6,7,23–26). These symptoms are more pronounced in migraine than tension-type headaches (27). As shared mechanisms may exacerbate symptoms of migraine and CRPS (21), we hypothesized that CRPS pain and hyperalgesia would be more severe in patients with migraine than tension-type headache. Photophobia and forehead hyperalgesia are more intense ipsilateral than contralateral to symptoms of CRPS (4,5,7,8,28–30), as are headaches (30). Thus, an additional aim was to determine whether these ipsilateral cranial symptoms predominated in patients whose headaches worsened after CRPS onset, and whether cranial symptoms arose both in patients with co-morbid migraine and in others with tension-type headache.
Methods
STROBE guidelines for observational studies were followed in this report.
Participants
Participants were recruited through a small private pain management practice in Perth, Western Australia. Each patient was assessed for symptoms and signs of CRPS by a pain specialist (PMF). Diagnoses that could better explain these symptoms (e.g., arthritic diseases or peripheral neuropathy from non-traumatic causes) were identified during a medical examination, and patients with these diagnoses were excluded from the study. Patients under the age of 18 years, who were pregnant or breastfeeding, with bilateral CRPS, or with hearing loss, epilepsy or a pacemaker were also excluded. Apart from these exclusions, patients who met Budapest and Valencia criteria for CRPS (31,32) were invited to participate.
Each patient provided their written informed consent for the procedures, which were approved by the University's human research ethics committee (approval number 2021/208).
Headache history
To explore the headache history, patients were interviewed in a laboratory setting by an experienced headache researcher (PDD) who presented a standard series of questions about the time course of headaches and associated symptoms. As none of the patients described symptoms of trigeminal autonomic cephalalgias or other uncommon headache types, the focus was on symptoms of migraine and tension-type headache. Specifically, patients were asked whether they experienced headaches and, if so, whether the headaches had changed after the injury that triggered CRPS. The current frequency, duration, site and severity of the headaches, and symptoms of migraine (whether the headache was preceded by an aura, was unilateral, associated with nausea or vomiting, photophobia or phonophobia or throbbing pain, and whether the headache was aggravated by physical activity) were recorded. These details were used to determine whether headaches met International Classification of Headache Disorders (third edition) criteria for migraine or other forms of headache (27).
CRPS history
Patients were asked about the injury that triggered CRPS and symptoms in the affected limb: allodynia, hyperalgesia or other sensory disturbances; autonomic and inflammatory changes such as swelling, sweating, temperature or colour; motor disturbances such as muscle weakness, decreases in range of motion, tremor, dystonia, muscular cramps or spontaneous limb movements; and dystrophic disturbances such as changes in hair, skin and nail growth. Vasomotor changes were assessed by measuring skin temperature from the dorsal surface of the proximal phalanx of each digit on the affected and contralateral limb with an infrared thermometer. A mean asymmetry of 1°C or more was regarded as a sign of vasomotor disturbance. The cutaneous distribution of abnormal sensations evoked by lightly brushing the affected limb (e.g., paraesthesia or allodynia) was also assessed. Patients with “probable” or “definite” evidence of peripheral nerve injury (i.e., sensory disturbances were identified in an anatomically-plausible nerve distribution given the site and nature of the triggering event, or a nerve injury was verified during surgical examination or by a confirmatory electrophysiological test) (33) were considered to have CRPS type II.
Quantifying the spatial distribution and intensity of pain
As part of their assessment, regions of chronic pain were marked on drawings depicting the dorsal and ventral profiles of the body (“pain drawings”). The area of pain in the affected limb was quantified from scanned drawings using the polygon selection tool in ImageJ software (34), and was expressed as a percentage of total body surface area. The pain areas were shaded and the scanned/digitized drawings were uploaded into a validated online platform (Sketch Your Pain, https://syp.spslab.ch/) (35,36) to produce “heat maps” of the average pain distribution. Patients also rated the intensity of pain in the affected limb over the past week between 0 (no pain) and 10 (extremely intense pain).
Psychophysical assessment
Patients who took analgesic medication for CRPS or headaches were asked to delay taking this medication on the day of the assessment until after the procedures were completed. Sensitivity to pressure, pinprick, cold, heat and repeated application of a monofilament, was assessed on the dorsal surface of the affected and contralateral extremity and on each side of the forehead using previously described procedures (5). To assess sensitivity to pressure-pain, an algometer (FDX, Wagner Instruments, USA) with an 8 mm diameter hemispheric rubber tip was applied at 100 g/s until the patient reported that pressure changed to pain or to a maximum of 5 kg. Next, patients rated the intensity of sensations evoked by the 2-s application of a spring-loaded metal pin at a force of 40 g (Neuro-pen, Owen Mumford, USA) on a verbal rating scale ranging from 0 (not intense) to 10 (extremely intense). To assess “wind-up” (i.e., an incremental increase in sensitivity with repeated application of a weak stimulus, regarded as a sign of central sensitization), the sensation evoked by a single 1-s application of a thin nylon monofilament with a bending force of 10 g (Neuro-pen, Owen Mumford, USA) was then rated on the same scale. This was followed by five further 1-s applications with rests of 1 s between each application. “Wind-up” was calculated as the difference in ratings between the first and final application. Next, a cylindrical brass rod (1.5 cm diameter), cooled to 4°C, was applied at each body site for 7 s and patients rated cold-pain between 0 (not painful) and 10 (extremely painful). A servo-controlled thermode (3.1 cm2 contact area), heated to 40°C, was then applied at each site for 7 s and patients rated heat-pain on the same scale.
Visual discomfort threshold
Participants sat in front of a screen illuminated evenly with white light. For each eye in turn, screen luminance increased from 500 lux at 100 lux/s until the participant reported discomfort (the visual discomfort threshold) or to a maximum of 3600 lux. The side tested first was chosen randomly. In addition to exposing the full visual field, the visual discomfort threshold was identified for the nasal then temporal portion of the visual field while masking other parts of the field. The visual discomfort threshold was averaged across two trials for each type of exposure.
Data reduction
Individual scores in each sensory modality in the affected limb were expressed as the number of standard deviations away from the mean score in the group as-a-whole in the contralateral limb (equivalent to a z-score). These normalized scores were averaged across sensory modalities (pressure-pain thresholds, pinprick, wind-up, cold-pain and heat-pain) for each patient such that positive scores represented hyperalgesia. A similar approach was used to calculate an index of hyperalgesia in the ipsilateral forehead in relation to scores in the contralateral forehead.
Statistical approach
As scores for some variables differed from a normal distribution, differences between headache categories in hyperalgesia and in the intensity and distribution of limb pain were investigated using the Kruskal-Wallis and Mann-Whitney U tests. To control for multiple comparisons, significant differences among categories were further explored with Bonferroni tests. Asymmetry of cranial symptoms was investigated using Wilcoxon's matched pairs signed ranks test. For frequency data, differences among categories were investigated using chi-square (χ2) tests.
Results
Clinical characteristics
The sample consisted of 56 women and 32 men aged between 23 and 72 years (median age 48 years) (Table 1). Pain had developed in an arm (42 patients, 47.7%) or leg (46 patients, 52.3%) between three and 421 months previously (median CRPS duration 30.5 months) (Table 1). Twelve patients had early CRPS (symptoms for <12 months); 36 patients had intermediate CRPS (12–36 months); and 40 patients had persistent CRPS (>36 months) (Table 1). The majority had CRPS I (54 patients, 61.4%) but, in 34 cases (38.6%), the history of injury and physical signs were consistent with CRPS II (Table 1). The affected limb was at least 1°C warmer than the contralateral limb in 10 patients (11.4%), and cooler by this margin in another 20 patients (22.7%) (Table 1).
Clinical characteristics.

All but 13 patients (14.8%) took one or more forms of medication daily to manage pain. Common treatments included NSAIDS (37.5%), opioids (26.1%) and/or tapentadol (26.1%), anticonvulsants such as pregabalin, gabapentin or topiramate (21.6%), antidepressant medication such as tricyclic antidepressants, selective serotonin reuptake inhibitors and serotonin norepinephrine reuptake inhibitors (19.3%), and medicinal cannabis (18.2%) (Table 1).
Changes in the headache pattern after CRPS onset
Twenty-four of 88 patients (27.3%) recalled experiencing recurrent headaches before CRPS onset (14 with symptoms of migraine and 10 with tension-type headaches). Headaches remained unchanged after the onset of CRPS in 17 patients (19.3% of the total group) but worsened in the other seven patients (8%). In an additional 51 patients (58.0%), recurrent headaches developed after the onset of CRPS (Table 1). Another 13 patients (14.8%) reported that they rarely or never experienced mild headaches and had never experienced severe headaches either before or after the onset of CRPS. No patient reported that headaches had lessened after CRPS began.
Patients whose headaches had increased were younger at CRPS onset (mean ± standard deviation, 37.9 ± 12.1 years) than patients whose headaches remained stable (45.3 ± 12.5 years) or who rarely or never experienced headaches (45.3 ± 12.8 years) [F (2,85) = 3.55, p = .033] (Table 2). Post hoc Bonferroni tests did not further clarify this effect. Forty-seven of the 58 patients whose headaches had increased (81%) experienced symptoms of migraine compared with only nine of the 17 patients whose headaches remained stable (52.9%) [χ2 (1) = 5.48, p = .019] (Table 2). In addition, headaches were chronic (i.e., recurred on 15 or more days/month) in 19 of the 58 patients (32.8%) whose headaches had increased but were episodic in all 17 patients whose headaches remained stable [χ2 (1) = 7.46, p = .006] (Table 2). Whether headaches had worsened after CRPS onset was unrelated to the patient's current age, sex, painful limb, CRPS I versus II, thermal subtype or CRPS duration category (Table 2). Eleven of 17 patients whose headaches remained stable after CRPS onset (64.7%) took daily medication compared with 53 of 58 patients whose headaches had increased (91.4%) and 11 of 13 patients with few or no headaches (84.6%) [χ2 (2) = 7.43, p = .024]. The percentage of patients who took a particular type of medication (non-steroidal anti-inflammatory drugs; tapentadol; other opioids; anticonvulsants; antidepressants; medicinal cannabis; systemic or topical ketamine; or opioid antagonists) was similar across the three headache categories.
Association between features of CRPS and headaches after CRPS onset.
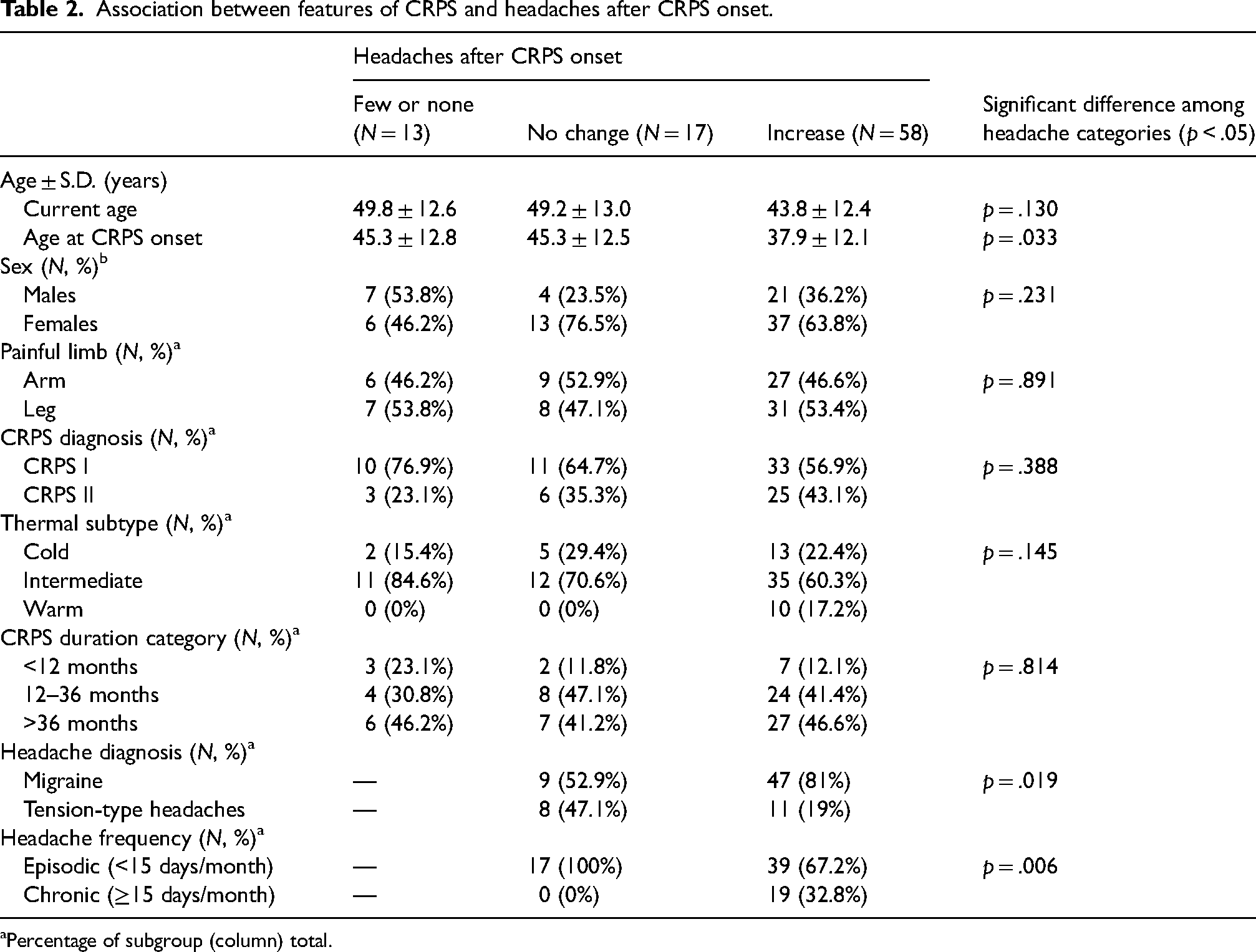
Percentage of subgroup (column) total.
Association between limb pain and changes in the headache pattern after CRPS onset
More of the affected limb was painful in patients whose headaches had increased than in patients whose headaches remained unchanged or who rarely or never experienced headaches [Kruskal-Wallis test statistic across all three groups (N = 88, 2 degrees of freedom) = 6.20, p = .045; Bonferroni test for the difference between patients whose headaches had increased versus those whose headaches remained unchanged, p = .045] (Figure 1A). Regions of chronic pain often extended beyond the site of injury in the CRPS-affected limb, sometimes to involve the entire forequarter or hindquarter. Notably, in 10 of the 58 patients whose headaches had increased, pain associated with CRPS included the ipsilateral neck (nine patients with upper-limb CRPS and one patient with ipsilateral upper and lower limb CRPS); in addition, one other patient with lower-limb CRPS developed neck pain in association with headaches several months after the onset of CRPS (Figure 1B). In contrast, neck pain was absent in all 17 patients with stable headaches and in the 13 patients who rarely or never experienced headaches [difference in the prevalence of neck pain across the three headache categories, χ2 (2) = 6.50, p = .039] (Figure 1C).

Pain distribution in CRPS in relation to headache history. (A) Area of pain in the affected limb of patients who rarely or never experienced headache (0, clear dots, N = 13), whose headaches did not change after CRPS onset (=, pink dots, N = 17) or whose headaches increased (+, red dots, N = 58). Horizontal lines represent the median. Difference between groups statistically significant (Bonferroni's test, * p < .05). (B) Heat map of the pain distribution in 58 patients whose headaches worsened, constructed from the percentage of patients with pain in each pixel of the pain drawing. For purposes of illustration, drawings in patients with left-sided CRPS were transposed to illustrate the distribution of pain in the affected limb in the group-as-a-whole. To form heat maps, pain drawings were overlaid. Sites of maximal superimposition of pain are represented in black, and sites of minimal superimposition are shown in light yellow. Uncoloured sites represent areas that were free from pain in every patient. (C) Heat map of the pain distribution in 30 patients whose headaches did not change after CRPS onset (N = 17) or who rarely or never experienced headaches (N = 13).
The intensity of limb pain was greater in patients whose headaches had increased than in patients whose headaches remained unchanged (p = .004, Bonferroni test) or who rarely or never experienced headaches (p = .018, Bonferroni test) [Kruskal-Wallis test statistic across all three groups (N = 88, 2 degrees of freedom) = 14.81, p < .001] (Figure 2A). Additionally, hyperalgesia in the affected limb was greatest in patients whose headaches had increased [Kruskal-Wallis test statistic across all three groups (N = 88, 2 degrees of freedom) = 7.93, p = .019; Bonferroni test for the difference between patients whose headaches had increased versus those whose headaches remained unchanged, p = .016] (Figure 2B). Similarly, allodynia evoked by brushing the affected limb was more prevalent in patients whose headaches had increased (42 of 58 patients, 72.4%) than in patients whose headaches remained stable (6 of 17 patients, 35.3%) or who rarely or never experienced headaches (6 of 13 patients, 46.2%) [χ2 (2) = 9.13, p = .010].

Association between headache history and indices of pain in CRPS. Intensity of pain (A) and hyperalgesia (B) in the affected limb of patients who rarely or never experienced headache (0, clear dots, N = 13), whose headaches did not change after CRPS onset (=, pink dots, N = 17) or whose headaches increased (+, red dots, N = 58). To calculate the index of hyperalgesia, individual scores in each sensory modality were expressed as the number of standard deviations away from the mean score in the contralateral limb (equivalent to a z-score). These normalized scores were averaged across sensory modalities within each patient such that positive scores represented hyperalgesia. In A and B, horizontal lines represent the median. Bonferroni test: *p < .05; **p < .01.
Features of CRPS in relation to headache category and frequency
Most patients reported that they experienced headaches on two or more days per month (Table 1). Headaches met International Classification of Headache Disorders (third edition) (27) criteria for migraine in 58 patients (65.9% of the total sample) and tension-type headache in 17 patients (19.3% of the total sample) (Table 1). Notably, 16 patients reported daily headaches associated with symptoms of migraine when the headache was severe (referred to as chronic migrainous headache in Table 1 rather than “chronic migraine” as it was not possible to establish retrospectively whether headaches fulfilled criteria for migraine on eight or more days/month) (27); and three others met International Classification of Headache Disorders (third edition) criteria for chronic tension-type headache. In all but one case, headaches had become chronic after the onset of CRPS.
Pain and hyperalgesia associated with CRPS were unrelated to headache chronicity or diagnosis; furthermore, headache frequency was unrelated to CRPS duration. However, both for migraine and tension-type headaches, headaches recurred more frequently in patients who first experienced headaches after CRPS onset than in patients with a history of stable headaches [for migraine, Mann-Whitney standardized test statistic = 2.811, p = .005; for tension-type headaches, Mann-Whitney standardized test statistic = 2.747, p = .005] (Figure 3A and 3B).

Days/month of (A) migraine in patients with stable (N = 9) or worsening headaches (N = 47); and (B) tension-type headaches in patients with stable (N = 8) or worsening headaches (N = 11). Mann-Whitney U test: ** p < .01.
Cranial symptoms
The headache was unilateral in 42 of 75 patients with recurrent headaches (56%) and usually or always recurred on the CRPS-affected side in all but five of these cases [side concurrence, χ2 (1) = 24.3, p < .001]. This association held both for upper and lower limb CRPS: 17 of the 37 patients with headache ipsilateral to CRPS (46%) and 19 of the 38 patients with bilateral or contralateral headaches (50%) had upper-limb CRPS (difference not significant). In one of the five patients with headaches contralateral to CRPS, headaches in the right occipital region began after a right-sided whiplash injury. In a separate, more recent incident, CRPS developed after a laceration of the left forearm. In the other four patients, headaches contralateral to CRPS developed with no clear cause after the onset of CRPS.
Hyperalgesia in the forehead to mechanical and thermal stimuli was more severe ipsilateral than contralateral to CRPS in patients whose headaches remained stable [Wilcoxon standardized test statistic (N = 17) = −2.44, p = .015] or worsened after CRPS onset [Wilcoxon standardized test statistic (N = 58) = −5.10, p < .001]; in contrast, forehead hyperalgesia was symmetrical in patients with few or no headaches [Wilcoxon standardized test statistic (N = 13) = −.38, p = .701] (Figure 4A–4C). In further exploratory analyses, hyperalgesia in the forehead was more severe ipsilateral than contralateral to CRPS both in patients with migrainous [Wilcoxon standardized test statistic (N = 56) = −4.61, p < .001] and tension-type headaches [Wilcoxon standardized test statistic (N = 19) = −3.34, p < .001] (Figure 4D and 4E). In addition, forehead hyperalgesia was lateralized in patients with ipsilateral [Wilcoxon standardized test statistic (N = 37) = −4.46, p < .001] or bilateral headaches [Wilcoxon standardized test statistic (N = 33) = −4.37, p < .001] but not in patients with contralateral headaches [Wilcoxon standardized test statistic (N = 5) = 1.21, p = .225] (Figure 4F–4H). In a sensitivity analysis that excluded patients with chronic daily headaches, forehead hyperalgesia remained greater ipsilateral than contralateral to CRPS [Wilcoxon standardized test statistic (N = 69) = −4.91, p < .001]. In another sensitivity analysis, forehead hyperalgesia was greater ipsilateral than contralateral to CRPS in patients with CRPS onset within the past 12 months (i.e., acute CRPS) [Wilcoxon standardized test statistic (N = 12) = −3.06, p = .002].
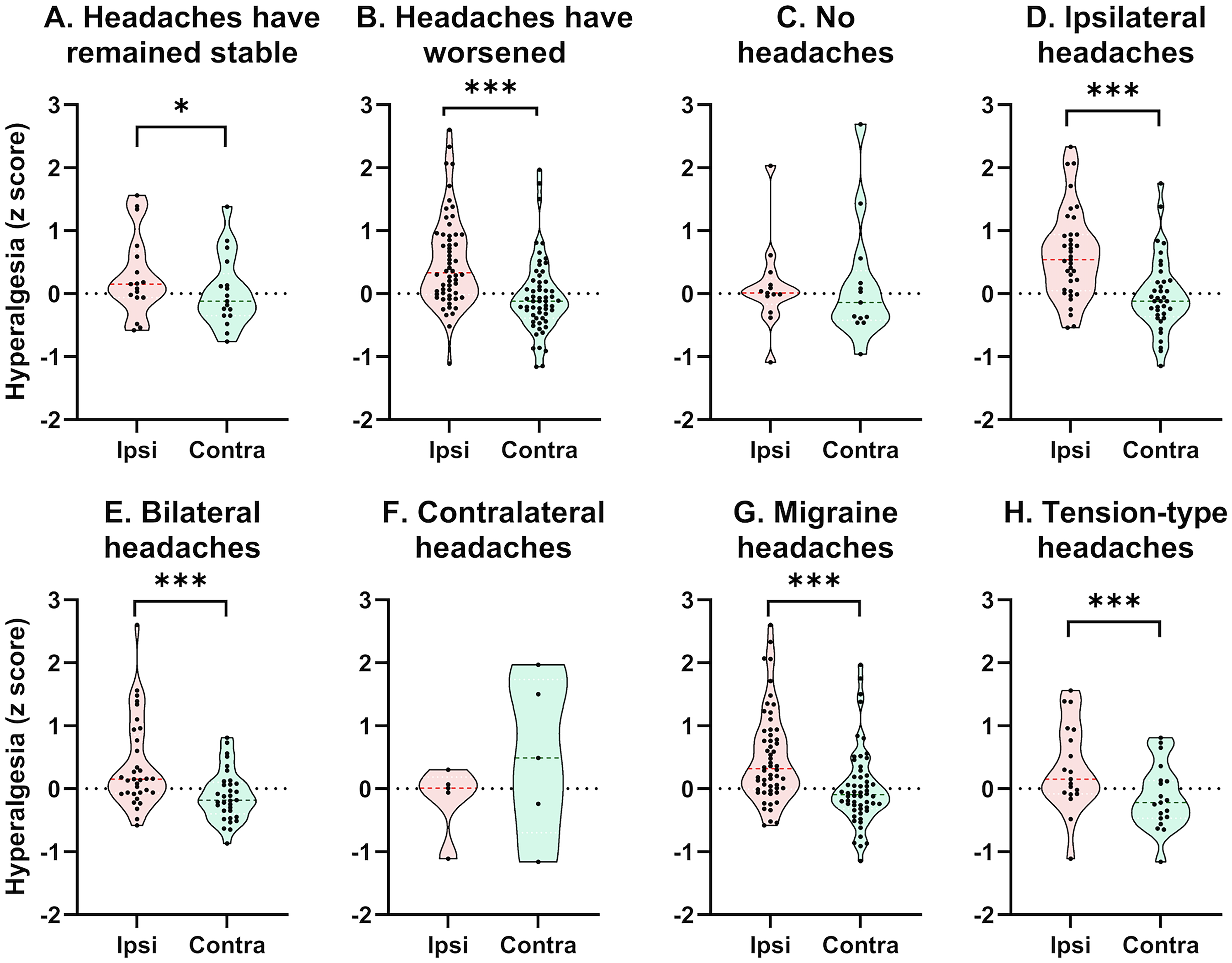
Hyperalgesia in the forehead ipsilateral (ipsi, pink) and contralateral (contra, green) to the affected limb in patients with CRPS. The index of hyperalgesia, averaged across sensory modalities, was calculated as the number of standard deviations away from the mean score in the contralateral forehead. Each dot represents the hyperalgesia score of an individual patient, and dashed lines represent the median. The ipsilateral side of the forehead was more sensitive to mechanical and thermal stimulation in patients whose headaches (A) remained stable (N = 17) or (B) worsened after CRPS onset (N = 58) but not in patients with (C) few or no headaches (N = 13). The ipsilateral side of the forehead also was more sensitive in patients with (D) ipsilateral (N = 37) or (E) bilateral headaches (N = 33) but not in patients with (F) contralateral headaches (N = 5). Sensitivity was greater ipsilateral than contralateral to CRPS in patients with (G) migraine (N = 56) and (H) tension-type headaches (N = 19). Wilcoxon test: *p < .05; ***p < .001.
Results were similar for visual stimulation. Specifically, the visual discomfort threshold was lower ipsilateral than contralateral to CRPS in patients whose headaches remained stable [Wilcoxon standardized test statistic (N = 17) = 2.16, p = .031] or worsened after CRPS onset [Wilcoxon standardized test statistic (N = 57) = 5.06, p < .001]; in contrast, the visual discomfort threshold was symmetrical in patients with few or no headaches [Wilcoxon standardized test statistic (N = 13) = 1.85, p = .064] (Figure 5A–5C). In further exploratory analyses, the visual discomfort threshold was lower ipsilateral than contralateral to CRPS in patients with migrainous headaches [Wilcoxon standardized test statistic (N = 55) = 5.45, p < .001] but was similar on both sides in patients with tension-type headaches [Wilcoxon standardized test statistic (N = 18) = 1.48, p = .139] (Figure 5D and 5E). Photophobia was lateralized in patients with ipsilateral [Wilcoxon standardized test statistic (N = 36) = 5.06, p < .001] and bilateral headaches [Wilcoxon standardized test statistic (N = 33) = 3.39, p < .001] but did not differ between sides in patients with contralateral headaches [Wilcoxon standardized test statistic (N = 4) = −1.83, p = .068] (Figure 5F–5H). These effects did not change when only the monocular nasal or temporal portion of the visual field was exposed to light. In a sensitivity analysis that excluded patients with chronic daily headaches, the visual discomfort threshold remained lower ipsilateral than contralateral to CRPS [Wilcoxon standardized test statistic (N = 67) = 4.69, p < .001]. In addition, the visual discomfort threshold was lower ipsilateral than contralateral to CRPS in patients with acute CRPS [Wilcoxon standardized test statistic (N = 11) = 2.13, p = .033].

Visual discomfort threshold (which reflects photophobia) for the full visual field ipsilateral (ipsi, pink) and contralateral (contra, green) to the affected limb in patients with CRPS. Each dot represents the visual discomfort threshold of an individual patient, and dashed lines represent the median. The visual discomfort threshold was lower ipsilateral than contralateral to CRPS in patients (A) whose headaches remained stable (N = 16) or (B) worsened after CRPS onset (N = 57) but was symmetrical in patients (C) with few or no headaches (N = 13). The visual discomfort threshold also was lower ipsilateral than contralateral to CRPS in patients with (D) ipsilateral (N = 36) or (E) bilateral headaches (N = 33) but not in patients with (F) contralateral (N = 4) headaches. In addition, the visual discomfort threshold was lower ipsilateral than contralateral to CRPS (G) in patients with migraine (N = 55) but (H) symmetrical in patients with tension-type headaches (N = 18). Wilcoxon test: ***p < .001.
Discussion
The aims of this study were to investigate whether susceptibility to recurrent headaches increased after CRPS onset and whether this was associated with heightened pain or lateralized cranial symptoms. In many patients, headaches developed or worsened after the onset of CRPS and met criteria for migraine. This applied equally for CRPS I and CRPS II, the warm and cool subtypes, patients with upper and lower limb pain, and acute and longstanding CRPS. CRPS area, pain intensity, hyperalgesia and allodynia were greater in patients whose headaches had worsened than in the minority of patients with stable headaches or who reported few or no headaches. Most headaches either were exclusively ipsilateral to CRPS or had an ipsilateral component. By-and-large, photophobia and forehead hyperalgesia were greater ipsilateral than contralateral to symptoms of CRPS except in the few patients with contralateral headaches or who remained headache-free. Notably, however, photophobia was symmetrical in patients with co-morbid tension-type headaches. In sum, the findings suggest that a mechanism linked with CRPS initiated a steep rise in headaches, primarily migrainous, together with lateralized cranial symptoms.
Central sensitization in CRPS may provoke headaches
Almost two-thirds of patients reported that headaches had worsened after CRPS onset. At the time of assessment, months or years after CRPS began, 48 percent of patients had symptoms of episodic migraine compared with an estimated population prevalence of 12 percent (37); headaches resembled chronic migraine in a further 18 percent of patients, far exceeding the population prevalence of 1–2 percent (37). Whether disrupted sleep patterns, psychological stress or medication overuse contributed to the increase in headaches is unknown; notwithstanding this, the link with pain parameters – CRPS area, pain intensity, hyperalgesia and allodynia – is consistent with a mediating influence of pain. Importantly, the association with allodynia implies involvement of central sensitization (i.e., facilitation of nociceptive signalling within the central nervous system) (11,21,38). Central sensitization develops in a wide array of acute and chronic pain conditions, including CRPS, migraine and chronic tension-type headaches (23,39–41). Importantly, pain and allodynia may develop in the ipsilateral upper limb during attacks of migraine (42–45), possibly due to progressive sensitization of trigemino-vascular and thalamic nociceptors (46,47). Thus, we speculate that supraspinal nociceptive sensitization, triggered by CRPS, increased vulnerability to headaches in our clinical cohort. Central sensitization is stronger in patients with frequent than infrequent tension-type headaches (41). This link with headache frequency might explain why the pain of CRPS was greater in patients with worsening headaches than in patients with stable, infrequent headaches.
In a subgroup of patients, CRPS pain extended from the upper limb to the back of the neck. Cervical nociceptive afferents converge onto second-order neurons in the trigeminocervical complex, thereby facilitating referral of pain from the occipital region to the forehead and temple (48–50). In a rodent model of headache, chemical and supramaximal electrical stimulation of the greater occipital nerve sensitized trigeminocervical neurons and increased their excitability to dural stimulation, a probable mechanism of headache (50). A similar process might have exacerbated headaches after CRPS onset in patients with neck pain. However, additional mechanisms must be involved as ipsilateral headaches also increased in patients with lower limb CRPS.
Patients whose headaches worsened were younger at CRPS onset than patients with stable headaches or who remained headache-free; most of these headaches met criteria for episodic migraine or recurred daily. In previous studies, migraine and chronic daily headache were identified as risk factors for CRPS (11,21). Our findings additionally suggest that CRPS onset at a younger age increases the risk of more painful CRPS and co-morbid migraine, in line with the peak in migraine prevalence in early adulthood (51).
Do headache mechanisms intensify CRPS?
It was not possible in this cross-sectional study to determine whether an increase in headaches foreshadowed an increase in the pain of CRPS, or vice versa. However, it is worth noting that a worsening trajectory of headaches was associated with the most severe CRPS. In our previous work, limb pain and headache cycled together on a daily basis in two patients with CRPS and co-morbid trigeminal autonomic cephalalgias (7). More broadly, it is tempting to speculate that a labile influence on supraspinal nociceptive activity aggravates CRPS and increases vulnerability to headaches in previously resilient patients. What this influence might be is unclear but anxiety (a risk factor for progression from episodic to chronic migraine) (52,53) could be important as chronic psychological stress induces neuroinflammatory and neuroplastic changes that intensify central sensitization in brain areas involved in pain processing (54).
Intriguingly, heartbeat detection accuracy is compromised both in migraine (55) and CRPS (56), possibly reflecting a reduced capacity to accurately monitor interoceptive sensations such as pain. Potentially, this deficit exacerbates symptoms in both disorders by facilitating recurrent “allostatic resets” (i.e., drastic shifts in physiological state accompanied by unpleasant sensations that compel the body to rest and recover from perceived harm) (57).
Laterality of headaches and cranial symptoms in CRPS
Photophobia and forehead hyperalgesia typically were more intense on the CRPS-affected side than contralaterally. Bright light triggered visual discomfort in the entire monocular visual field, implying interaction between visual and nociceptive signals in the brainstem – a likely source of photophobia in migraine (30,58). Cranial symptoms were stronger on the CRPS-affected side than contralaterally in patients with an ipsilateral component of headache, notably in patients with stable or worsening headaches, but were symmetrical in patients with few or no headaches. Moreover, lateralization of cranial symptoms was stronger for migraine (which frequently is unilateral) than tension-type headaches (usually bilateral), as the visual discomfort threshold was symmetrical in patients with recurrent tension-type headaches. Whether CRPS mechanisms trigger ipsilateral cranial symptoms generally associated with migraine requires further study. Nonetheless, it is intriguing that administering ultraviolet B radiation to the forearm not only triggers local inflammation but also induces hyperalgesia to pressure-pain in the ipsilateral forehead (59,60), thus simulating the development of ipsilateral cranial hyperalgesia in CRPS.
In a murine model of posttraumatic chronic pain, α1D-adrenoceptors were upregulated in the ipsilateral dorsal horn of the spinal cord and contralateral locus coeruleus after distal tibia facture in elderly female mice (61). This was associated with a reduction in descending noradrenergic inhibitory influences on pain. Together with an upregulation of α1-adrenoceptors on primary nociceptive afferents (62,63), these changes might facilitate ascending nociceptive traffic in a positive loop (64). The reduction in inhibitory noradrenergic influences on pain on the affected side of the body could account for the presence of ipsilateral cranial symptoms and headache after CRPS onset. More generally, this disruption might result in spread of pain/hyperalgesia and other nociplastic symptoms as CRPS progressively worsens (4).
In a few patients, headaches and other cranial symptoms developed contralateral to CRPS. Apart from this anomaly, CRPS and headache profiles resembled those of patients with ipsilateral or bilateral headaches. CRPS pain occasionally spreads to the contralateral limb, possibly due to cross-spinal or inter-hemispheric spread of sensitization (2,3). Whether a similar mechanism mediated contralateral cranial symptoms in this study is unknown. However, it is important to note that photophobia and hyperalgesia to pinprick and electrical stimulation of the forehead are greater bilaterally in CRPS patients than in pain-free controls (30,65), consistent with widespread sensitization.
Limitations
A power calculation to determine the sample size was not possible as this was the first study to examine changes in the headache pattern after CRPS onset. As the sample was 2.7 times larger than in a report of ipsilateral headaches and photophobia in CRPS (30), it was considered sufficient to explore individual differences in these attributes. However, the sample most likely was underpowered to assess links between CRPS and headache symptoms in subgroups with low numbers (e.g., with headaches contralateral to symptoms of CRPS); thus, findings relating to these subgroups should be interpreted cautiously.
Sufficient information was obtained during the headache interview to assign patients to broad diagnostic categories; however, in the absence of a headache diary, it was not possible to determine whether patients with daily headaches had chronic migraine. Although many patients described headaches that met International Classification of Headache Disorders (third edition) criteria for migraine (27), the side-locked nature of the headache and photophobia in CRPS suggest that biological mechanisms may differ from migraine. Furthermore, cranial autonomic symptoms such as lacrimation and rhinorrhoea were not assessed. It would be important in future studies to prospectively document headache and associated sensory and autonomic symptoms, together with the sensory and autonomic disturbances of CRPS, to clarify links between these symptoms and to determine whether headaches associated with CRPS are phenotypically distinct from migraine.
Whether the patient's headache trajectory had changed after CRPS onset was necessarily assessed retrospectively. This might have introduced recall bias regarding whether headaches had remained stable, developed or worsened after CRPS onset.
Most of our patients had longstanding CRPS in a single limb and were recruited from a small pain management practice that specialized in treating patients after an industrial accident. This may restrict the generalizability of findings to patients with similar characteristics. In addition, restricted range or ceiling effects might explain why neither the pain (21) nor the duration of CRPS (22) was associated with headache frequency. This needs to be investigated further in other research centres with different referral pipelines.
The visual discomfort threshold was often associated with squinting, looking away or closing the exposed eye, consistent with ocular pain. It would be interesting in future studies to determine whether visual stimulation also aggravates headache or limb pain, similar to the effect of auditory stimulation on limb pain in CRPS (9).
To minimize effects of medication on study outcomes, patients delayed taking analgesic medication on the day of assessment until after the tests had been completed. Despite this precaution, analgesic effects probably persisted to some extent, particularly for drugs with a long half-life. This might have weakened the association between CRPS pain severity and headaches. Nevertheless, the association remained strong regardless of any lingering analgesia. The percentage of patients who took medication daily to manage the pain of CRPS was greater in the subgroup with worsening headaches than in patients whose headaches remained stable. Whether medication overuse contributes to a rise in headaches after CRPS onset, or delays resolution of CRPS, requires further investigation. Whether CRPS symptoms improve with effective treatment of migraine is also unknown.
Lastly, it is important to establish whether ipsilateral cranial symptoms develop in other forms of chronic unilateral limb pain or are unique to CRPS.
Conclusions and clinical implications
Migraine and chronic daily headache developed in previously headache-free patients after the onset of CRPS, in association with ipsilateral cranial symptoms. Recognising this link is crucial for effective management of these co-morbid conditions. Our findings suggest that mechanisms shared by CRPS and migraine, such as central sensitization and loss of inhibitory pain controls, contribute to headache and other cranial symptoms.
The findings also imply that CRPS and migraine aggravate each other, possibly in a reciprocal cycle. If so, breaking this cycle could allow otherwise intractable symptoms and pain to subside.
Article highlights
Migraine and chronic daily headache develop in previously headache-free patients after the onset of CRPS
Photophobia and forehead hyperalgesia are more intense ipsilateral than contralateral to CRPS in patients with co-morbid migraine
Mechanisms shared by CRPS and migraine, such as central sensitization and loss of inhibitory pain controls, may contribute to headache and other cranial symptoms
Treatments that relieve one of these co-morbid conditions might also help the other
Footnotes
Acknowledgments
We wish to thank Dr Di Ye and Dr Marco Barbero for their assistance. The conducted research and statistical analysis plan were not preregistered.
Author contributions
PDD: conceptualization, data curation, data analysis, funding acquisition, investigation, methodology, project administration, visualization, writing – original draft, writing – review and editing
PMF: conceptualization, funding acquisition, investigation, supervision, writing – review and editing
Data availability statement
The data will be made available upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
The study was approved by the University's human research ethics committee (approval number 2021/208).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grant 22-011 from the Australian and New Zealand College of Anaesthetists.
