Abstract
Background
The HEAD-WINd® study was designed to examine the burden, characteristics and lived experiences of headache disorders in the Danish adult population. By integrating data from surveys, a smartphone application, and national health and social registries, the study addresses limitations of prior epidemiological research.
Methods
A random sample of Danish residents aged 18–75 years were invited to participate (“base population”). Two nested cohorts were recruited using a population-based approach: (i) a survey cohort consisting of individuals reporting active headache disorders (“headache population”) and (ii) a smartphone application cohort, derived from the headache population, which was followed longitudinally for 12 weeks. Data from these cohorts were enriched with data from national health and social registries, including information on medication use, hospital records, socioeconomic status and healthcare utilization. An adapted version of the Headache-Attributed Restriction, Disability, Social Handicap and Impaired Participation (HARDSHIP) questionnaire was used to classify multiple headache disorders and assess headache-attributed burden.
Results
Of the 100,030 invited individuals, 28,617 (28.6%) completed the general survey. Among them, 15,571 (54.4%) reported experiencing headache in the preceding year; 14,074 (90.4%) completed the headache-specific survey. In total, 663 individuals (4.7%) participated in the app-based longitudinal study. The mean ± SD participant age was 53.2 ± 15.5 years, 57.4% were women and the mean ± SD body mass index was 26.8 ± 7.4 kg/m².
Conclusions
HEAD-WINd® has established a comprehensive, population-based cohort of Danish residents, including individuals both with and without headache disorders. This resource provides a framework for generating population-level insights into the burden and management of headache disorders.
Keywords
Introduction
Headache disorders are responsible for more than three-quarters of all years lived with disability (YLDs) attributable to neurological disorders, with migraine being the leading contributor (1). Their prevalence, severity and treatment are influenced by age, sex, socioeconomic status, education and family structure, comprising factors that contribute to disparities in disease burden and access to care (2,3). Addressing these inequalities requires high-quality, nationally representative epidemiological data.
Despite extensive research, epidemiological studies on headache disorders often suffer from methodological shortcomings, including small or biased samples, inconsistent diagnostic criteria, and variability in survey tools and response rates (4). These inconsistencies undermine comparability and limit our understanding of the true burden and distribution of headache disorders. In addition, limited data exist on which types of individuals are likely to participate in longitudinal, patient-reported studies, which is an important consideration for inclusive and effective research design.
The HEAD-WINd® (Headache Disorders Among Adults in Denmark: Innovating Real-World Evidence Generation Through Registry, Survey and Patient-Centric Data Integration for Migraine) study was designed to overcome these challenges through a nationwide, population-based sample. By integrating validated instruments, such as a modified Headache-Attributed Restriction, Disability, Social Handicap and Impaired Participation (HARDSHIP) questionnaire and smartphone-based daily diaries, with data from Danish national health and social registries, HEAD-WINd® provides a rich, real-world dataset. This enables precise characterization of headache disorders at the same time as allowing researchers to explore socioeconomic and health-related predictors of burden and care access.
This present study reports the design, methodology, participation rates and demographic characteristics of participants in the HEAD-WINd® study. The broader aim of the study is to advance efforts to reduce the overall burden of headache disorders and improve health outcomes through data-driven policy and practice.
Methods
Oversight
HEAD-WINd® is a public-private partnership established between the Danish Knowledge Center on Headache Disorders and H. Lundbeck A/S to advance population-based research on headache disorders. The Danish Knowledge Center on Headache Disorders, functioning as a public institution, is responsible for promoting clinical and public awareness of headache disorders and supports research initiatives that aim to improve diagnostic and treatment pathways.
The study protocol received exemption from formal ethical review by the regional ethics committee of the Capital Region of Denmark (reference: F-24003935), in accordance with Danish legislation governing non-interventional epidemiological research. The Danish Medicines Agency viewed that the longitudinal smartphone application did not meet the criteria for classification as a medical device under applicable regulatory definitions, and therefore did not require additional evaluation or certification.
For registry-based analyses, Danish law does not require individual informed consent because these studies rely exclusively on de-identified secondary data. In contrast, all participants contributing to primary data collection, either through survey responses or smartphone-based entries, were provided with detailed information about the study's purpose, scope and data handling practices, and were required to give informed consent prior to enrollment.
Design and recruitment
HEAD-WINd® applied a population-based approach to recruit two nested cohorts: (i) a survey population comprising individuals reporting active headache disorders (“headache population”) and (ii) a smartphone application population followed longitudinally. Both cohorts were drawn from a “base population”, randomly sampled from the Danish Population Register and supplemented with data from Danish national health and social registries (5).
Base population and inclusion criteria
Individuals were eligible if they were between 18 and 75 years of age at the time of selection and resided in Denmark. Eligibility also required access to the national electronic mail system, which is mandatory for all residents aged 18 years or older unless formally exempted. This system serves as Denmark's primary digital communication platform, enabling individuals to receive official documents and messages from public authorities securely and in accordance with legal requirements. Individuals exempted from this system were eligible if they could receive communications via traditional postal delivery.
From the pool of eligible residents, a random sample of 100,030 individuals was drawn to establish the study's base population. This sample was designed to be demographically and socioeconomically representative of the broader Danish adult population. Representativeness was assessed using national registry variables that included age, biologically assigned sex at birth, region of residence, country of origin, cohabitation status, family structure, educational attainment, employment status, household income and hospital contact history.
Each individual received a general health survey invitation via the national digital platform and those exempted from the system were contacted by traditional mail. Importantly, the invitation materials did not disclose the study's focus on headache disorders to avoid condition-specific participation bias. Prior to the main launch, a soft rollout involving 1000 individuals was conducted to confirm system compatibility and optimize message delivery. Survey invitations were distributed in three successive waves, and a single reminder was issued 14 days following the initial contact to non-responders.
Survey population (“headache population”)
All members of the base population were initially invited to complete a general health survey that included screening questions for twelve chronic conditions, including headache disorders. Participants who reported experiencing at least one day of headache within the preceding 12 months were classified as having an active headache disorder. These individuals were subsequently directed to a supplementary questionnaire designed to assess symptom characteristics, functional impact, and headache-attributable burden. Individuals who met this screening criterion and completed the supplementary assessment constituted the survey-defined cohort of participants with headache disorders (“headache population”).
Smartphone application population
The smartphone application cohort consisted of participants who met four sequential inclusion criteria following completion of the general and headache-specific surveys. Eligible individuals had reported an active headache disorder, completed the follow-up headache questionnaire, consented to recontact, and accepted a subsequent invitation to install and use a study-specific smartphone application.
Participants in this cohort were monitored for up to 12 consecutive weeks, during which they contributed prospective, daily data on headache characteristics, symptom variability, medication use and associated health outcomes. The enrollment sequence and study flow are summarized in Figure 1.

Study overview. ASC-12, 12-item Allodynia Symptom Checklist; DREAM, ; GAD-7, Generalized Anxiety Disorder-7; HALT-30, Headache-Attributed Lost Time; HCRU, healthcare resource utilization; PHQ-8, Patient Health Questionnaire-8; WHO-5, WHO-5 Well-Being Index.
Data collection
The HEAD-WINd® study applied an integrated data collection framework that combined self-reported questionnaire responses with real-time, smartphone-based symptom tracking to comprehensively assess headache disorders and related health outcomes. Study content was developed collaboratively by the HEAD-WINd® investigator team and operationalized through two coordinating partners, each responsible for distinct aspects of data capture. Statistics Denmark administered the registry-linked, web-based survey on behalf of the Danish Knowledge Center on Headache Disorders and H. Lundbeck A/S, targeting the base and survey populations. Monsenso, a digital health technology provider, oversaw the deployment and maintenance of the smartphone application used for longitudinal data collection in the app-based cohort.
General health survey
The general health survey was administered to all participants and included items assessing the presence of 12 chronic conditions within the preceding 12 months, along with self-reported height and weight. Participants were asked to indicate whether they had experienced any of the listed conditions, including headache, by responding “yes” or “no” to each item. A “yes” response to the headache item triggered a subsequent, condition-specific survey designed to capture detailed information on headache characteristics, impact and burden. The primary objective of this screener was to estimate the frequency of common systemic comorbidities within the general survey population. A secondary aim was to conceal the study's focus on headache disorders by embedding the relevant question within a broader set of health-related items. To enhance clarity and cultural relevance, the condition labels were adapted from the Danish National Health Profile, which also served as the source for height and weight items (6). The full general health survey instrument is provided in the suppmentary material (Appendix S1).
Headache survey
The headache survey was administered to participants who had reported at least one day of headache within the past 12 months. It was based on an adapted version of the HARDSHIP questionnaire (7). This adaptation enabled classification of multiple headache types, including migraine, tension-type headache, medication-overuse headache and post-traumatic headache, and supported the assessment of headache-attributable burden using standardized, patient-reported outcomes. Although the adapted version had not undergone formal validation, forward and backward translation procedures were applied to ensure linguistic precision and conceptual alignment across languages. To accommodate the self-completion format, modifications were made to the original face-to-face version of HARDSHIP, including adjustments to phrasing and question flow.
The survey design also incorporated Computer-Assisted Web Interviewing (CAWI) principles to enhance usability, streamline data entry and enable skip logic for conditionally irrelevant questions. While the survey instrument applied HARDSHIP-derived criteria to assign diagnostic classifications, these were not disclosed to participants and were used exclusively for population-level prevalence estimation.
To capture the functional impact of the most bothersome headache, the survey additionally included five items from a modified four-week version of the Headache-Attributed Lost Time (HALT-30) index, which quantified productivity losses across paid employment, household duties, and social or leisure activities. The full survey instrument and supporting materials are provided in the supplementary material (Appendix S2).
Smartphone application
Participants who completed the headache-specific survey were invited to enroll in the longitudinal data-collection phase via a dedicated smartphone application. To ensure linkage with registry-based data from the base population, participants were required to input their unique Danish personal identification number (i.e. CPR) during the enrollment process. After registering for the app study, participants were given instructions on how to install the app and set up their accounts. They also received introductory materials via email explaining how to use the app. When logging in for the first time, participants were guided through a short in-app tutorial that highlighted its key functions.
The core feature of the application was a structured daily diary, initiated on day 1 and continuing through day 89, which prompted participants once per day to record headache activity and general well-being. Each diary entry began with a general self-assessment of daily quality, followed by a headache screening item; if a headache was reported, a detailed follow-up module assessed headache characteristics, symptom profile and acute medication use. If the headache diary was not completed on a given day, this was recorded as missing data in the dataset. This structured logic was designed to facilitate the differentiation of migraine from other headache types. A two-day recall window, covering two calendar days regardless of the time of entry, was incorporated to accommodate variability in participant adherence and to reduce missing data related to daily availability. The complete diary instrument and its underlying structure are available in the supplementary material (Appendix S3).
Additional survey instruments were administered at baseline and at approximately four-week intervals throughout the follow-up period to assess secondary outcomes relevant to health status and headache-related burden. These instruments included the Patient Health Questionnaire-8 (PHQ-8) for depressive symptoms (8), the Generalized Anxiety Disorder-7 (GAD-7) scale for anxiety (9), the WHO-5 Well-Being Index for mental well-being (10) and the 12-item Allodynia Symptom Checklist (ASC-12) for pain sensitivity (11). Furthermore, healthcare resource utilization (HCRU) was assessed and included a modified 30-day version of the Headache-Attributed Lost Time (HALT-30) index (12). Figure 1 and Table 1 summarize the content and administration timeline for each instrument. The smartphone application did not provide any diagnostic interpretation or treatment guidance to participants and was used exclusively to collect real-time, participant-reported data on headache experience. Prior to implementation, the application was pilot-tested in a user-experience study involving six adult participants with headache disorders. Each participant completed two individual interviews: the first explored general preferences regarding health-related smartphone applications and evaluated the clarity of invitation materials for both the survey and the app components. The second interview included hands-on testing of a prototype application, with targeted feedback collected on navigation, user engagement features and language tone of content. Findings from the pilot phase informed iterative revisions to improve usability and maximize participant retention. The final smartphone application also featured lay-language articles and video content about the study to participants to support study engagement and retention. All content was co-developed with Danish migraine patient organizations.
Numbers of eligible participants and participating proportions for each study assessment.
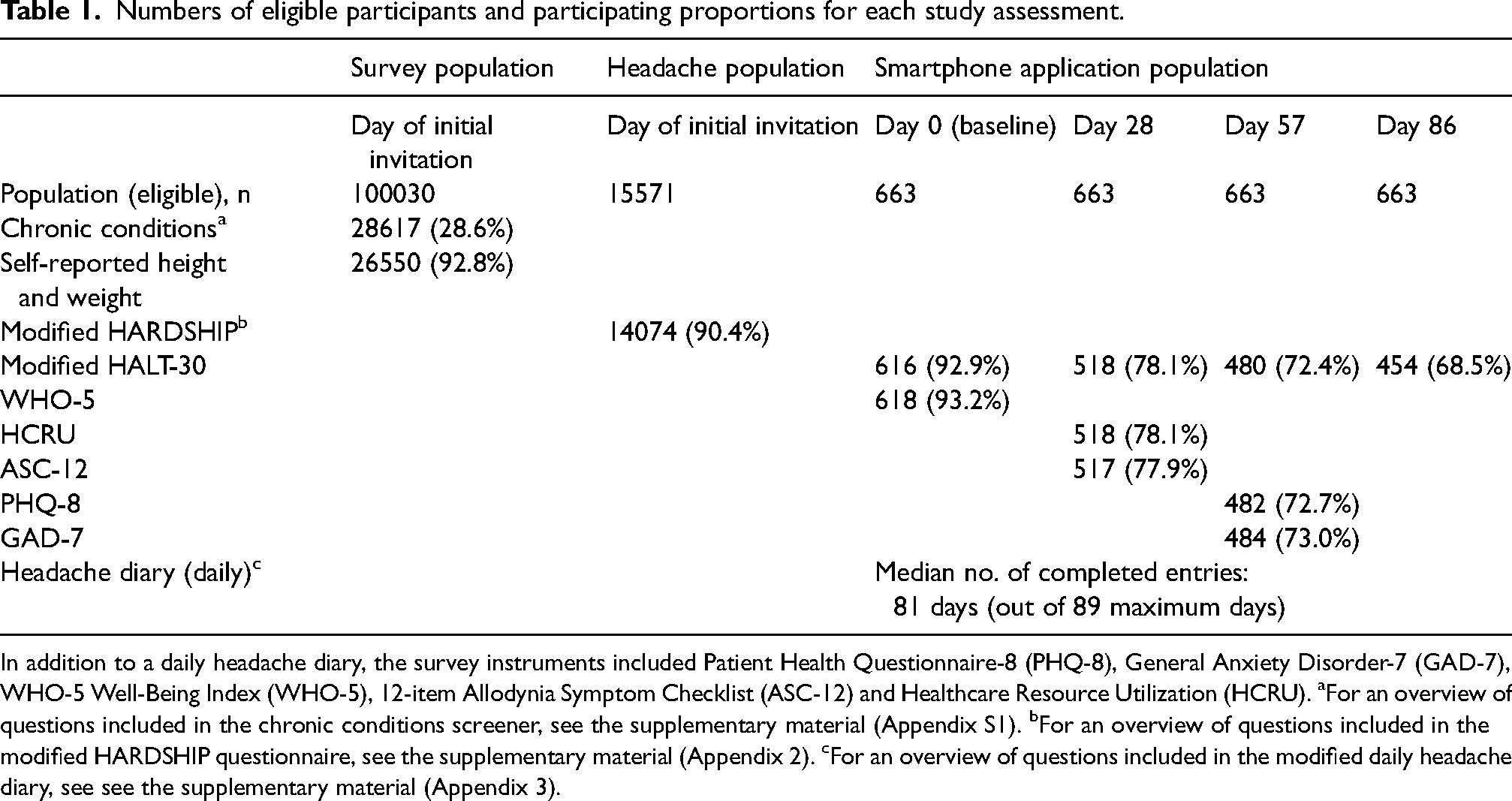
In addition to a daily headache diary, the survey instruments included Patient Health Questionnaire-8 (PHQ-8), General Anxiety Disorder-7 (GAD-7), WHO-5 Well-Being Index (WHO-5), 12-item Allodynia Symptom Checklist (ASC-12) and Healthcare Resource Utilization (HCRU). aFor an overview of questions included in the chronic conditions screener, see the supplementary material (Appendix S1). bFor an overview of questions included in the modified HARDSHIP questionnaire, see the supplementary material (Appendix 2). cFor an overview of questions included in the modified daily headache diary, see see the supplementary material (Appendix 3).
Record linkage to danish health and social registries
Since 1968, all Danish residents have been assigned a unique personal identification number (Danish: Centrale Personregister, CPR), recorded in the Danish Civil Registration System (5). This identifier enables secure, individual-level linkage of longitudinal data across national health and social registries, supporting high-resolution tracking of clinical, behavioral and socioeconomic outcomes. For the present study, registry-based data were integrated to complement participant-reported information and enhance completeness and validity across multiple domains.
The following national registries were utilized:
Extracted data from the DREAM register and the Danish National Patient Registry spanned from the first quarter of 1995 through the first quarter of 2024. In contrast, data from the Danish National Prescription Registry, the Danish National Hospital Medication Register and the Danish National Public Health Insurance Register extended through the third quarter of 2024, reflecting the full duration of the app-based study period. All clinical, pharmaceutical and healthcare utilization data were curated and maintained by the Danish Health Data Authority. To complement these datasets, sociodemographic variables, including age, sex, educational attainment, household income, cohabitation status, employment status, region of residence and country of origin, were obtained from nationwide population registries managed by Statistics Denmark. These variables were extracted based on the registry entries most proximal to the launch of the general health survey to maximize temporal alignment and data relevance.
The integration of linked national registry data enabled systematic quantification of healthcare utilization and expenditures across the entire base population and all analytically defined sub-cohorts, including individuals with headache disorders and those enrolled in longitudinal follow-up.
In addition, diagnostic and pharmacologic data from the national patient and prescription registries supported the identification of relevant comorbid conditions using established classification frameworks. All personal identifiers were encrypted and stored on secure servers within Statistics Denmark, which maintained full control over data access and upheld nationally mandated protocols for privacy and confidentiality.
Data management
Following completion of the general and headache-specific screening surveys, Statistics Denmark transferred the resulting datasets to its secure Research Services platform (Forskermaskinen). The base population dataset was then expanded with historical data from national population registries curated by Statistics Denmark and the Danish Health Data Authority. Upon completion of app-based data collection, longitudinal data were securely uploaded by Monsenso and linked by Statistics Denmark to existing survey and registry records using pseudonymized personal identification numbers. To enhance the temporal completeness of the dataset, the base population was further supplemented with prospective data from the National Prescription Registry and the National Public Health Insurance Register, capturing entries generated after the initial survey rollout.
Merged datasets were stored exclusively within Statistics Denmark's secure environment, where authorized personnel from H. Lundbeck A/S and the Danish Knowledge Center on Headache Disorders were granted controlled access. All analyses were conducted using pseudonymized microdata, and only aggregated, non-identifiable outputs were exported for reporting and publication in accordance with Danish data protection regulations.
Statistical analysis
Because participation in the general population survey was anticipated to vary across demographic strata, unadjusted results risked compromising the representativeness of the survey population relative to the base population. To address this potential selection bias, Statistics Denmark calculated survey responder weights using registry-based data on sex, age, highest level of education, household composition, employment status and frequency of physician contact. The weighting model was developed by Statistics Denmark through an iterative process that began with examining the relationships between multiple register-based variables (demographic, socioeconomic and health-related) and response propensity. While several variables were associated with response status, not all could be retained in the final model. Variables and groupings were prioritized to ensure robust estimation and to avoid small or empty categories, which can produce unstable correction factors. The final model included age, sex, education, family type, employment status and frequency of physician contact because these provided the most balanced and stable weighting structure. Other variables such as region, income and country of origin were excluded because they contributed little additional explanatory power or were highly correlated with retained variables (e.g. education and income). This approach ensured weights that effectively corrected for non-response at the same time as maintaining model stability and representativeness. These weights were generated using established methods consistent with those employed in the Danish Health and Morbidity Surveys and were applied to both the general health and headache-specific surveys (18,19). As a result, weighted estimates derived from these survey responses are expected to approximate population-level distributions within the original sampling frame.
Descriptive analyses were performed using SAS, version 9.4 (SAS Institute Inc., Cary, NC, USA) and R, version 4.2.0 (R Foundation, Vienna, Austria). For categorical variables, proportions were calculated and reported as counts and percentages. For continuous variables, summary statistics included means, standard deviations, medians and observed ranges; estimates for cells with fewer than five observations were censored. Weighted summary statistics were calculated using Taylor series variance estimation, with the SAS NOMCAR option applied to handle missing responses under the assumption that data were not missing at random. Unweighted response proportions for the headache-specific survey are presented in the main results section. Weighted estimates for both the general health survey and the headache-specific survey, including analyses of chronic conditions, are detailed in the supplementary material (Appendices S4 and S5).
Results
Cohort characteristics
Base population and survey populations
Of 100,030 individuals randomly selected and invited to complete the general health survey, 28,617 responded and submitted a fully completed questionnaire, corresponding to an overall participation rate of 28.6%. Among the 71,413 non-responders, most (n = 68,517) did not reply to either the initial invitation or the reminder, 2761 individuals formally declined participation, whereas 570 could not be reached despite repeated contact attempts. Partial survey completion was observed in 36 individuals, 25 reported language-related barriers and 74 discontinued participation for other unspecified reasons. A summary of participation status at each level of recruitment is provided in Figure 2 and Table 1.
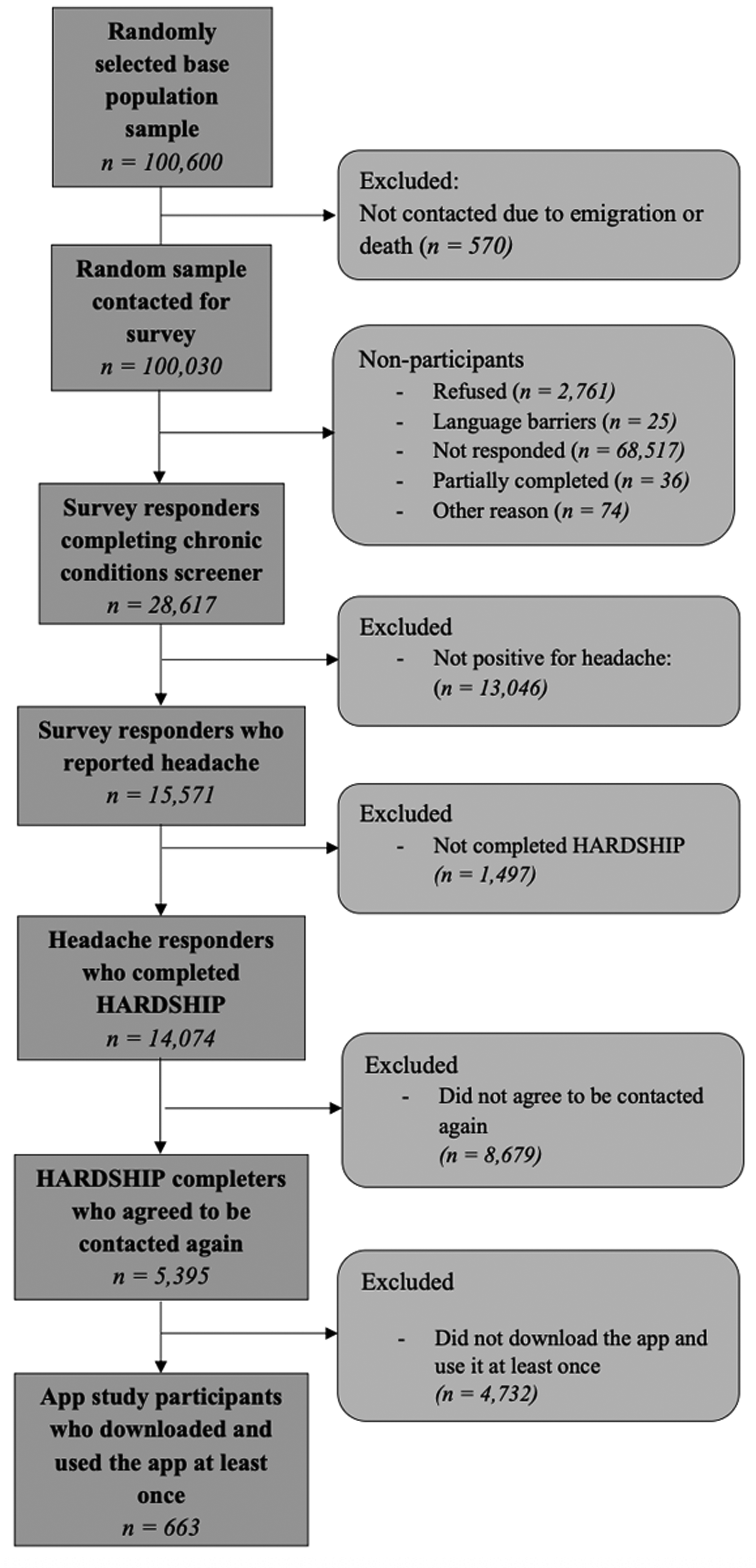
Study participation flow diagram.
Headache population
Among the 28,617 individuals who completed the general health survey, 15,571 (54.4%) reported experiencing at least one headache in the preceding 12 months and were directed to the headache-specific questionnaire. Of these, 14,074 participants (90.4%) completed the follow-up survey, forming the headache population.
Smartphone application population
Of the 14,074 individuals who completed the headache-specific survey, 5395 participants (38.3%) consented to follow-up contact. From this subset, 663 participants (4.7%) enrolled in the smartphone application study by installing the app and engaging with it at least once. Among those enrolled, 641 participants (4.6%) submitted at least one diary entry during the 12-week observation period, with a median completion of 81 diary days.
Additional survey instruments were administered through the app at baseline and approximately four-week intervals. Completion rates for these instruments are reported both for within-app users (n = 663) and for the broader group who completed the headache survey (n = 14,074). Specifically, the WHO-5 was completed by 618 participants (93% of within-app users; 4% of all headache survey respondents), the ASC-12 by 517 (78%; 4%), the HCRU by 518 (78%; 4%), the GAD-7 by 484 (73%; 3%) and the PHQ-8 by 482 (73%; 3%). The HALT-30 instrument was completed by 616 participants at baseline (93%; 4%), by 518 at day 28 (78%; 4%), by 476 at day 57 (72%; 3%) and by 454 at day 86 (68%; 3%).
Sociodemographic characteristics
Among the 28,617 individuals from the base population who completed the general health survey, the mean ± SD age was 53.2 ± 15.5 years and 16,418 participants (57.4%) were women. The mean ± SD body mass index was 26.8 ± 7.4 kg/m². When respondents were grouped by decade of age, participation was lower among younger individuals and higher among older age groups, relative to the distribution of the overall base population. Participation was also more frequent among individuals with higher household income, higher educational attainment and women. A full summary of baseline demographic and socioeconomic characteristics is provided in Table 2.
Distributions of the samples by sociodemographic factors and health factors.

Equivalized disposable income is the total income of a household, after taxes and deductions, available for spending or saving, divided by the number of household members adjusted for age using the modified Organisation for Economic Co-operation and Development (OECD) equivalence scale.
Prevalence of headache and frequencies of headache days and acute headache medication use
Among the 28,617 participants who completed the general health survey, the one-year prevalence of individuals reporting at least one day of headache in the previous year was 54.4%, with a weighted estimate of 59.6% (95% confidence interval = 59.55–59.64). Prevalence was higher among women than men, with weighted proportions of 65.4% and 43.6%, respectively, corresponding to a weighted female-to-male prevalence ratio of 1.3:1. The highest prevalence was observed in the 18–29-years age group.
Among respondents who completed the headache-specific survey, the mean ± SD number of headache days reported over the preceding four weeks was 5.6 ± 6.1, while the mean ± SD number of days with acute headache medication use was 3.6 ± 5.2.
Discussion
The HEAD-WINd® study integrates primary data from a population-based, cross-sectional survey and a nested longitudinal smartphone application study with comprehensive secondary data from national health and social registries. This multilevel design enables rigorous characterization of headache disorders across clinical, demographic and socioeconomic domains, while also supporting longitudinal tracking of symptoms and health outcomes. Methodological features central to the study include masked recruitment procedures to reduce condition-specific response bias, use of the HARDSHIP diagnostic framework in place of unstructured self-reporting and registry-based ascertainment of key sociodemographic indicators. In addition, repeated measures collected via the smartphone application allow for fine-grained analyses of within-person variation in headache burden over time.
The present report offers a baseline characterization of the study population, including sociodemographic attributes of survey participants and individuals who contributed app-based data. Among the 100,030 individuals invited to participate, 28,617 completed the general health survey (first-level participation: 28.6%). Of these, 15,571 reported at least one day of headache in the previous year, corresponding to a one-year prevalence of any headache disorder of 54.4%. Among respondents with headache, 14,074 completed the headache-specific survey (second-level participation: 90.4%) and 663 ultimately enrolled in the smartphone application study (third-level participation: 4.7%).
Existing evidence and added value
Selective participation remains a pervasive source of bias in epidemiologic studies, including those addressing headache disorders (4). In this context, a first-level participation rate of 28.6%, although comparable to other population-based health surveys, necessitates close scrutiny of representativeness. To evaluate potential participation bias, demographic comparisons were made between individuals who responded to the general health survey and the full invited base population. Participants tended to be slightly older, more often female, and more likely to report higher levels of educational attainment and household income. This demographic skew is noteworthy because headache disorders are sometimes associated with lower socioeconomic status and increased severity in socially disadvantaged groups, although the empirical evidence remains inconclusive (2). Discrepancies in age and sex distribution were particularly evident and corresponding adjustments in weighted prevalence estimates produced substantial upward corrections, most notably among younger male subgroups. These patterns suggest that selective participation could have led to underrepresentation of individuals with lower socioeconomic status or less stable living conditions, potentially attenuating observed prevalence estimates in the unweighted sample. Therefore, the application of responder weights, grounded in registry-based demographic data, was essential to enhance generalizability and mitigate bias introduced by differential participation.
Although masked recruitment was intended to reduce self-selection based on disease status, the potential for underreporting of headache remains a valid concern. Individuals with headache disorders, including those experiencing considerable disease burden, frequently underrecognize or deprioritize their symptoms, leading to reduced disclosure in health surveys (20). This tendency might have been reinforced by the design of the general health survey, which presented headache alongside other chronic conditions that could be perceived as more serious. Such framing could inadvertently diminish the perceived salience of headache disorders, thereby contributing to non-reporting. This interpretation aligns with two key findings. First, only 54.4% of participants reported experiencing headache within the preceding year, a one-year prevalence that is comparatively low for population-based studies. Second, those who did report headache indicated relatively high frequencies of headache days and acute medication use, suggesting that individuals with lower symptom burden were disproportionately less likely to report their headache disorder.
Despite these limitations, the combined use of masked recruitment and random sampling from the general population constitutes a notable methodological strength. This design enables the identification of individuals with headache disorders independently of care-seeking behavior or prior diagnostic labeling, thereby reducing two frequent sources of bias encountered in registry-based epidemiological studies. In contrast, previous Danish estimates have relied heavily on diagnostic codes from the Danish National Patient Registry (21), which is not routinely updated in primary care settings where most individuals with headache are managed. Alternative strategies, including case identification based on prescriptions of triptans (22), comprising agents that are often underutilized for migraine and generally inappropriate for other headache types, or convenience sampling from the Danish Blood Donor Study (23), which consists of a particularly healthy sub-population, have yielded samples with limited external validity. The present study's approach improves the representativeness and generalizability of findings by addressing these sampling constraints at the design level.
Older age and female sex have previously been identified as predictors of electronic diary compliance in a cohort of individuals with headaches managed at a tertiary care center (24). The findings from the HEAD-WINd® study reinforce this observation because similar sociodemographic characteristics predicted initial study participation. In the forthcoming analytical phase, the study will investigate additional factors influencing adherence to smartphone-based data collection tools for patient-reported outcomes. Planned analyses will also characterize the clinical trajectories of distinct headache disorders, including migraine and tension-type headache, and evaluate how these conditions relate to sociodemographic profiles, healthcare utilization patterns, and both individual-level and societal burden.
Strengths and limitations
A principal strength of the present study is the inclusion of 28,617 participants drawn from a national population of fewer than five million adults, enabling adequately powered subgroup analyses across demographic strata. The integration of unique personal registration numbers allowed for linkage of individual-level survey data to multiple national registries, facilitating comprehensive analysis of associations between headache disorders and outcomes such as comorbidity, healthcare utilization, mortality and social health disparities. Furthermore, masked recruitment procedures and application of post-stratification weighting helped reduce selective participation bias, although these adjustments were limited to recognized sociodemographic variables. The study also identified 13,046 individuals without self-reported headache in the prior year, enabling the construction of a contemporaneous control group for future comparative analyses. Because the smartphone application cohort was nested within the base population and linked to registry data, it was possible to characterize participant profiles across all stages of recruitment, from initial sampling (n = 100,030) to the final app-engaged cohort (n = 663). This structure supports detailed investigation of participation dynamics and can inform future strategies for recruitment in decentralized epidemiologic studies.
Nonetheless, several limitations merit consideration. Despite the use of validated instruments, including the HARDSHIP questionnaire and HALT-30 index, adaptations for self-completion and digital administration were not formally validated in this population. Moreover, the headache survey focused exclusively on participants’ most bothersome headache, typically migraine, limiting its capacity to characterize those with multiple concurrent headache disorders.
On another note, the overall participation rate was low, particularly among younger individuals and men, raising concerns about representativeness despite the use of corrective weighting. Although weighting improves the accuracy of prevalence estimates, it cannot eliminate all sources of bias, particularly those related to unmeasured behavioral or motivational factors. In addition, because the smartphone application cohort was small and self-selected, weighting adjustments were not applied to its data, and attrition during follow-up can further constrain generalizability.
Lastly, although the weighting methodology was developed for general health surveys, its application to headache-specific outcomes is limited by the absence of headache-specific calibration benchmarks. While corrective weighting improves prevalence estimates, residual biases from unmeasured behavioral and motivational factors may still limit generalizability for certain subgroups. This is particularly relevant for the smartphone application cohort, where the small, self-selected sample and attrition during follow-up further reduce representativeness. Therefore, the findings from this cohort should be interpreted with particular caution. These limitations notwithstanding, the study design, size and integration of registry data provide a framework for generating population-level insights into the burden and management of headache disorders.
Conclusions
The HEAD-WINd® study establishes a population-based cohort of individuals with and without headache disorders, drawn from a random sample of the adult Danish population and enriched through linkage to comprehensive national registry data. Using statistical adjustments based on the characteristics of the base population, HEAD-WINd® aims to provide data that are representative at the population level. While acknowledging limitations, the study design improves generalizability and enables estimation of disease prevalence, characterization of heterogeneous headache phenotypes, and assessment of health-related and sociodemographic correlates at scale. The longitudinal integration of patient-reported outcomes with secondary health and social data facilitates detailed tracking of clinical trajectories and burden across multiple time points. Subsequent publications will explore these domains in greater detail, offering novel insights into the lived experience and health system burden associated with headache disorders.
Public health relevance
The HEAD-WINd® study generates population-level evidence on headache prevalence, burden, and health disparities, supporting more equitable healthcare planning and resource allocation.
The multilevel design of HEAD-WINd®, integrating survey, smartphone and registry data, improves generalizability in epidemiologic research on headache disorders.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251404430 - Supplemental material for HEAD-WINd®: A nationwide cohort study of headache disorders in danish adults using survey, smartphone and registry data
Supplemental material, sj-docx-1-cep-10.1177_03331024251404430 for HEAD-WINd®: A nationwide cohort study of headache disorders in danish adults using survey, smartphone and registry data by Thien Phu Do, Signe Marie Aagaard, Håkan Ashina, Timothy J. Steiner, Arun Micheelsen, Lene Hammer-Helmich, Amar Mehta and Messoud Ashina in Cephalalgia
Footnotes
Acknowledgments
MA received funding support through the Lundbeck Foundation Professor Grant (R310-2018-3711). We express our sincere gratitude to all participants whose contributions made this study possible. The registry-based survey was administered with the assistance of Statistics Denmark, while Monsenso supported the deployment and maintenance of the smartphone-based data collection platform. Additional appreciation is extended to the Danish Migraine and Headache Association, the Danish Patient Association for Headache Sufferers, and Migraine Denmark for their role in co-developing content to the smartphone application.
Data availability
Access to the HEAD-WINd database can be facilitated through ancillary study proposals, which may be submitted to the HEAD-WINd steering committee via the corresponding author. Any approved proposals will also require data access applications to both Statistics Denmark and the Danish Health Data Authority.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical considerations
Danish law does not require individual informed consent because these studies rely exclusively on de-identified secondary data. The study protocol received exemption from formal ethical review by the regional ethics committee of the Capital Region of Denmark (reference: F-24003935).
Funding
This research was funded by and conducted in collaboration with H. Lundbeck A/S, Valby, Denmark. Lundbeck Foundation, H. Lundbeck A/S (grant number R310-2018-3711).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
