Abstract
Background
Chronic migraine (CM) is highly disabling, and many patients fail to respond to monotherapy with approved preventive treatments. OnabotulinumtoxinA (BoNTA) and atogepant act on distinct but complementary targets within the trigeminovascular system and may exert additive or synergistic effects when used together. Real-world data on their combination remain scarce.
Methods
We prospectively analyzed adult patients with CM who had received at least three prior BoNTA cycles and initiated atogepant 60 mg/day for a minimum of 24 weeks as add on to BoNTA. Co-primary outcomes were changes in monthly migraine days (MMDs) and ≥50% response rate at 24 weeks. Secondary outcomes included disability, medication use, tolerability and subgroup comparisons by prior monoclonal antibodies exposure.
Results
Among 101 patients, 82 completed 24 weeks of co-treatment. Mean MMDs decreased by 6.5 days (p < 0.001) and 45.1% of patients achieved a ≥50% reduction. Acute medication days decreased by 6.0 (p < 0.001) and Headache Impact Test-6 scores improved significantly (mean change: −4.0; p < 0.001). Patient’s Global Impression of Change scores indicated moderate-to-great improvement. Anti-calcitonin gene-related peptide naïve patients experienced larger reductions in MMDs (−7.75 vs. −5.87) and disability scores compared to non-naïve patients. Multivariable analysis identified only baseline acute medication use as predictor of response. Adverse events were mild and consistent with known safety profiles for both drugs separately; no novel safety concerns emerged.
Conclusions
The addition of atogepant to BoNTA might be effective and well tolerated in real-world setting, including CM patients previously exposed to multiple preventives. Prospective controlled trials and health-economic evaluations are warranted to validate these observations and inform future clinical guidelines.
Introduction
Chronic migraine (CM) is a debilitating neurological disorder characterized by substantial impact on quality of life, daily functioning, and healthcare utilization (1). Despite the growing number of available preventive therapies (2,3), a significant proportion of patients are non-responsive or partially responsive to monotherapy, continuing to experience a high migraine burden (4,5), thus necessitating new strategies that improve efficacy. Another rationale for exploring combination therapy is that improved outcomes may also be desirable in patients formally classified as responders to a single preventive. As highlighted by the International Headache Society position statement (5), even partial responders often continue to experience a substantial migraine burden.
Along with the development of novel targets (6), combining treatments with distinct and potentially additive/synergistic pharmacological and non-pharmacological mechanisms of action represents an option in difficult-to-treat migraine (7).
OnabotulinumtoxinA (BoNTA) and calcitonin gene-related peptide (CGRP)-targeted therapies, monoclonal antibodies (mAbs) and small molecules CGRP receptor antagonists (gepants) are characterized by two different pharmacological mechanisms and are approved for the prevention of CM (8–10).
BoNTA inhibits the release of different pro-nociceptive neurotransmitters (including CGRP) from peripheral nerve terminals and the translocation of transient receptor channels (TRP), mainly TRPA1 and TRPV1, primarily within unmyelinated C-fibers of the trigeminovascular system (10), but exactly how this translates to the therapeutic effect in migraine is not yet fully understood.
In parallel, gepants have demonstrated efficacy in both acute (rimegepant, ubrogepant and zavegepant) and preventive (rimegepant and atogepant) treatment of migraine (8). Atogepant (10 mg or 60 mg) was recently approved in Europe for the preventive treatment of episodic migraine and CM. Randomized clinical trials (RCTs) (11) have extensively demonstrated its efficacy and tolerability, recently confirmed by real-word evidence (12–14).
Preclinical studies have proposed that BoNTA and CGRP receptor antagonists may act on complementary components of the trigeminovascular system (15,16). While BoNTA modulates chronic sensitization processes via C-fiber inhibition (16,17), atogepant modulates nociceptive transmission by attenuating cortical spreading depression–induced activation of both C- and Aδ-fiber meningeal nociceptors, with temporally distinct effects, an early inhibition of C-fiber activity and a delayed inhibition of Aδ-fiber responses (18). This compartmentalized targeting suggests the potential for pharmacodynamic additive/synergy, particularly relevant in treatment-refractory CM, where monotherapy may not be sufficient to obtain clinically meaningful outcomes.
Despite this mechanistic rationale, real-world evidence supporting the clinical benefit of combining BoNTA with CGRP receptor antagonists is limited (7). Most studies to date have focused on the combination of anti-CGRP mAbs targeting CGRP or its receptor with BoNTA, whereas data on gepants are scarce (7).
In this context, the present study aims to evaluate the real-world effectiveness, tolerability and patient-reported outcomes associated with the addition of atogepant to a stable regimen of BoNTA in adults with CM over a 24-week follow-up, including patients with prior failure to anti-CGRP mAbs.
Methods
Study design
We performed a real-world, prospective, single-center, investigator-initiated and independent study (Synergy study), considering all consecutive outpatients treated with at least three consecutive cycles of BoNTA (nine months of treatment) for CM and subsequently initiated atogepant 60 mg orally as an add-on therapy for at least 24 weeks. All patients were recruited at the NeuroClinic Norway (Lillestrom, Norway).
In this study, we included, and followed, all patients on stable BoNTA starting add-oral atogepant 60 mg tablet with a potential 24-week follow-up period, regardless of whether they discontinued treatment for any reason from August 2024 to March 2025. We analyzed two different cohorts:
The effectiveness cohort, comprising patients who continued treatment for the full first 24 weeks; The overall/safety cohort, comprising all enrolled patients regardless of treatment discontinuation.
All medications have been administered in accordance with standard clinical practice in Norway and aligned with the International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria for CM (18,19).
Data were collected using the open online database Research Electronic Data Capture (REDCap) (https://project-redcap.org). The Ethics committee approved the data collection with the code REK 604900. All patients provided their written informed consent prior to initiating treatment.
Patient features
Participants were enrolled regardless of the number of previously interrupted preventive treatments due to ineffectiveness or lack of tolerability. Ineffectiveness was defined as the absence of meaningful improvement in migraine-related variables following the administration of drugs for at least weeks at an appropriate dose, as specified by the European Headache Federation criteria (19). In practice, if a patient did not experience a reduction in migraine frequency, intensity or disability after at least six weeks of continuous use at the recommended dose, the treatment was recorded as ineffective.
Inclusion criteria were: (i) patients aged 18 years or older; (ii) diagnosis of CM, with- or without aura, according to ICHD-3 criteria (20); (ii) availability of electronic headache diaries over at least three months before enrollment; (iii) clinical indication for the prescription of BoNTA and atogepant 60 mg; (iv) stable treatment with other preventive treatments for migraine at least three months prior to and throughout the study period; (v) at least three cycles of BoNTA prior to initiation of atogepant; and (vi) need to improve the treatment effectiveness according to Clinicians evaluation.
Exclusion criteria were: (i) patients with any contraindications to atogepant according to the summary of product characteristics; (ii) unstable treatments with other preventive migraine treatments before or during the study; and (iii) pregnancy and breastfeeding.
Clinical assessment and data collection
Migraine diagnosis along with clinical and demographic data was obtained by clinicians during a face-to-face interview using a semi-structured questionnaire. Collected information included: concomitant and previous preventive treatments, monthly headache days (MHDs), monthly migraine days (MMDs) and days with at least one analgesic use (AMDs) prior to the first intake of atogepant (defined as baseline). The information prior to starting add-on treatment was defined as “BoNTA only”. All patients were instructed to complete an electronic diary. Data were averaged as month values, (e.g. weeks 21–24). A “headache day” was defined as any day on which the patient reported experiencing a headache, including those with tension-type features. A “migraine day” was defined as any day on which the headache exhibited migraine characteristics or responded to triptan administration. Headache and migraine days were further categorized into mild, moderate or severe headache days based on reported pain intensity. Additional clinical variables, including comorbidities, were extracted from medical records and outpatient interviews during routine clinical practice.
Patient-reported outcomes and safety monitoring
The Headache Impact Test (HIT-6) (21) and the Hospital Anxiety and Depression Scale (HADS) (22) were administered at baseline and after 12 and 24 weeks of therapy. For the HIT-6 questionnaire, a change of −1.5 to −2.5 points is generally considered the minimal clinically important difference (MCID), whereas a change of −6 points or more is considered substantial and clearly clinically meaningful.
The HADS consists of 14 items, divided into two seven-item subscales: anxiety (HADS-A) for anxiety and HADSD for depression. The anxiety subscale reflects generalized anxiety symptoms, while the depression scale primarily captures features of anhedonia. Each item is rated on a four-point Likert scale ranging from 0 (not at all) to 3 (most of the time), yielding a maximum score of 21 per subscale and a total score is out of 42. The total HADS score can be interpreted as a global measure of psychological distress.
The Patient's Global Impression of Change (PGIC) was administered at weeks 12 and 24 to assess patients’ subjective perception of treatment effectiveness (23). No universal numeric MCID is reported, but a rating of 5 (“moderately better”) or above is usually interpreted as a meaningful clinical improvement.
Adverse events (AEs) were monitored and recorded throughout the study duration.
Outcomes
The co-primary outcomes were: (i) the change in MMDs from baseline (four weeks before adding atogepant) to week-24 (weeks 21–24) and (ii) the proportion of responders (namely patients who presented a reduction of MMDs ≥50% (RR50%) compared to baseline) at week 24.
Secondary outcomes:
Changes in MHDs at week 24 compared to baseline; Changes in AMDs at week 24 compared to baseline; Change in MMDs, MHDs, AMDs and RR50% at week 12; Changes in HIT-6 questionnaire score (36–78 scale) at week 12 and 24; Changes in HADS questionnaire score (0–42 scale) at week 12 and 24; Descriptive evaluation of the patient satisfaction measured with the PGIC questionnaire; Comparing clinical features of responders and non-responders; Comparing patients naïve to monoclonal antibodies anti-CGRP (anti-CGRP mAbs, naïve group) and those previously treated with anti-CGRP mAbs (non-naïve group). Evaluation of potential variables related to RR50% at 24 weeks including the overall population; Descriptive evaluation of adverse events and dropouts.
Statistical analysis
Given the limited real-world data on the use of gepants, particularly in combination with other migraine preventive treatments, no formal sample size calculation was performed. Instead, analyses were conducted on a convenience sample, including all consecutive outpatients.
Normality of distribution was assessed using the Shapiro–Wilk test, which indicated non-normality of several variables. Thus, non-parametric tests were employed for statistical comparison. Continuous variables were summarized as mean with 95% confidence interval (CI), interquartile range (IQR) or mean ± standard deviation (SD) as appropriate. Categorical data were reported as counts and percentages. No imputation was performed for missing data; these were explicitly reported in both tables and the text. To evaluate within-group pre–post treatment differences in continuous variables, we used the Wilcoxon signed-rank test. For paired categorical data, the exact McNemar's test was applied. Comparison between independent groups were conducted using the Mann–Whitney U test for continuous variables and the chi-squared test for categorical variables.
To explore potential factors of clinical response, both univariate and multivariable logistic regression analyses were performed. The dependent variable was binary and defined as achieving a ≥50% reduction in MMDs from baseline, assessed at 24 weeks. All patients (overall cohort, n = 101) were included, with patients who discontinued treatment categorized as “non-responders”. Independent variables were selected a priori based on clinical relevance. Univariate logistic regression was first conducted for each variable separately, using response status at six months as the dependent variable. Odds ratios (Exp(B)), 95% CIs and p-values were reported. Variables with p < 0.05 in univariate models, or of clinical interest, were considered for inclusion in the multivariable analysis. Multivariable logistic regression was then performed to identify independent predictors of response at 24 weeks. Multicollinearity was assessed prior to inclusion. Model fit was evaluated using the omnibus chi-squared test and Nagelkerke R2 statistics. Missing data were not imputed and the number of available patients per variable are as stated, where appropriate. No missing data were present for primary outcomes.
A sensitivity analysis, using the conservative baseline carried forward approach (for MMDs) was performed to assess the impact of patients who discontinued (regardless of the reason) on the co-primary outcomes in the overall cohort.
A two-tailed p < 0.05 was considered statistically significant for all variables, with a Bonferroni's correction where appropriate. All data were analyzed using SPSS, version 29.0 (IBM Corp., Armonk, NY, USA) and graphs were designed using Prism, version 10.00 (GraphPad Software Inc., San Diego, CA, USA).
Results
The study enrolled 101 consecutive patients with CM fulfilling the criteria for inclusion (female 93.1%, mean ± SD age was 42.1 ± 10.9 years). Nausea/vomiting was reported by all patients, and a first-degree family history for migraine was present in 76.2%. Some 65.7% had at least one comorbidity in the overall cohort Most participants had a middle socioeconomic status (54.5%) and were employed full-time (52.0%). All cohort features are reported in Table 1.
Clinical and demographic features of the overall/safety and effectiveness cohorts.

Percentages are expressed on the column total. aCalculated only in female patients. BMI = body mass index; MHDs = monthly headache days; AMDs = days with analgesics use per month; AMD = days with at least one analgesics use per month; HIT-6 = Headache Impact Test-6.
When BoNTA was first introduced (BoNTA-only timepoint), the cohort reported a mean ± SD of 25 ± 4.9 MHDs and 21.1 ± 5.9 MMDs. At study baseline (i.e. introducing add-on atogepant during treatment with BoNTA), the overall population reported a mean ± SD of 23 ± 5.9 MHDs and 16.4 ± 6.7 MMDs. AMDs, HADS-A and HIT6 scores are reported in Table 1.
Patients had previously an average of 5.0 prior preventive treatments before starting BoNTA, increasing to 6.1 when including BoNTA. The mean number of BoNTA cycles before introducing atogepant was 12.3. Data regarding specific molecules/classes are reported in the supplementary material (Table S1).
Overall, 19/101 (18.8%) patients dropped out from the study for different reasons (see below) after 12 weeks of treatment and therefore 82/101 (81.2%) were included in the effectiveness analysis (Figure 1). Baseline details subdivided between the overall and effectiveness cohort are reported in Table 1 and the supplementary material (Table S1).
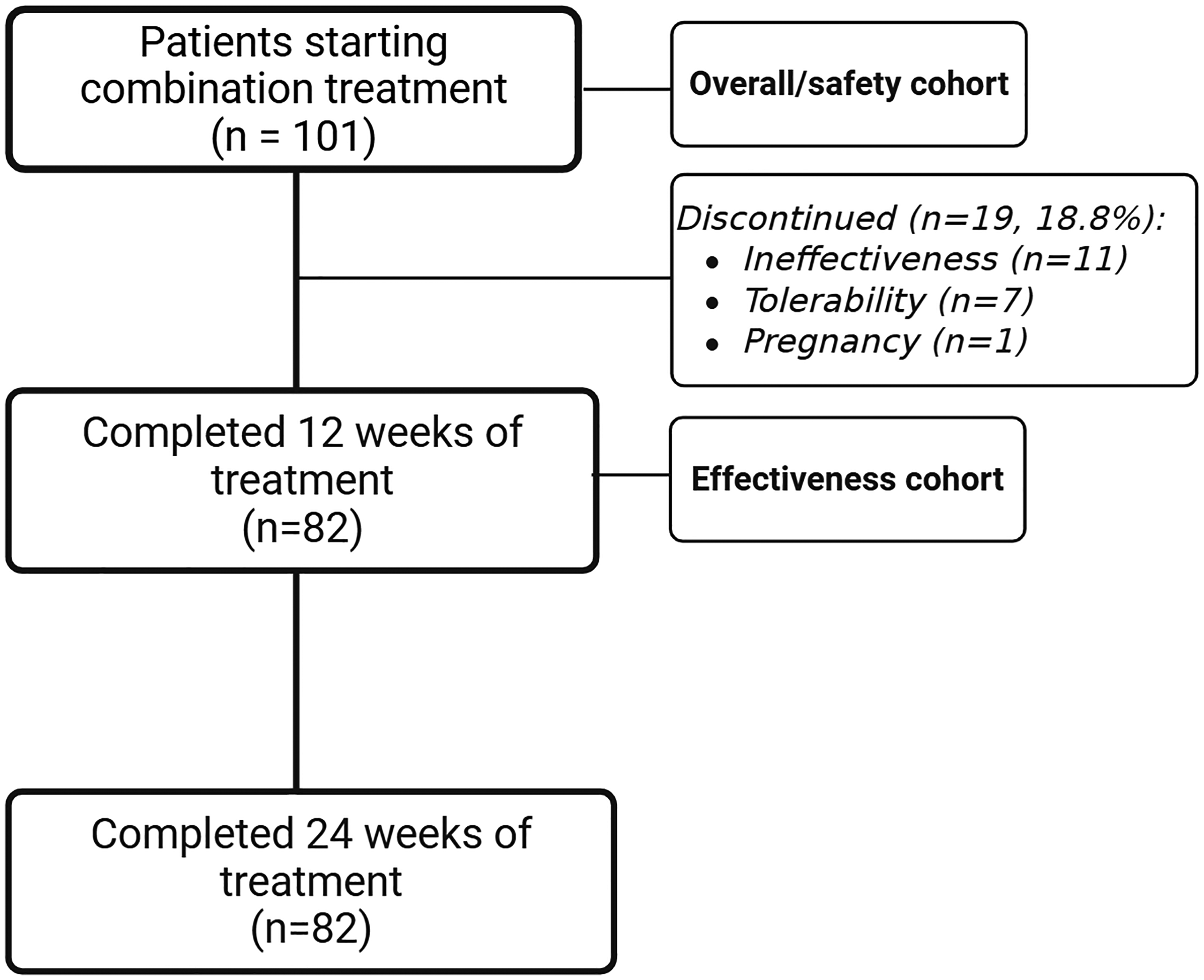
Flow chart with patients treated with combination treatment at each follow up, with patients included in the effectiveness and overall/safety analysis.
Effectiveness of BoNTA before introducing add-on atogepant
To evaluate whether BoNTA was effective before the introduction of add-on atogepant, we compared the study baseline (i.e. before starting atogepant during BoNTA treatment) with the month prior to the initiation of BoNTA (BoNTA only).
MMDs were significantly reduced after treatment with BoNTA (−4.78; 95% CI = −5.79 to −3.76; p < 0.001), as were MHDs (−2.78; 95% CI = −3.80 to −1.75; p < 0.001), suggesting a modest yet significant and consistent effectiveness of BoNTA alone. Baseline MMD values (before starting BoNTA and atogepant) are reported in the supplementary material (Table S2).
Based on the 50% reduction threshold in MMDs (RR50%), 22.0% of patients (18/82) in the BoNTA-only cohort achieved a ≥50% response, while the remaining 78.0% (64/82) did not meet this responder criterion.
Effectiveness outcomes at 24 weeks in the effectiveness population
At 24 weeks of co-treatment (mean weeks 21–24), MMD decreased to 10.0 (6.9), with a median reduction of −6.5 (95% CI = −7.46 to −5.20; p < 0.001) (co-primary outcome). Similarly, MHDs dropped to a mean ± SD of 16.0 ± 8.3 (p < 0.001) and AMDs decreased at 6.3 ± 1.5 (p < 0.001) (Figure 2 and Table 2).
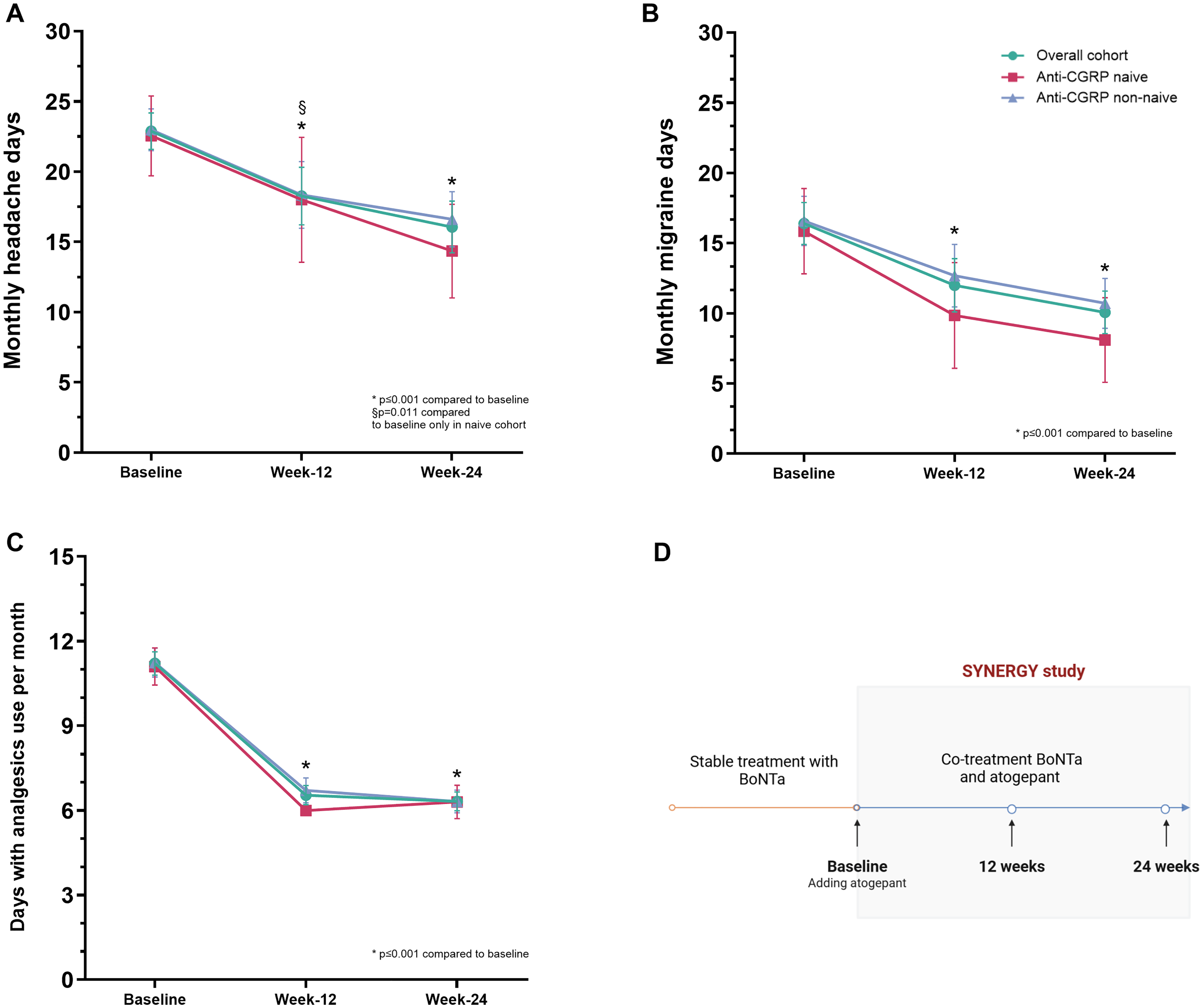
Migraine-related variables monthly headache days (A), monthly migraine days (B) and days with analgesic use (C) at baseline and after 12 and 24 weeks with cotreatment in the effectiveness cohort and sub-cohorts of patients with or without prior failure to anti-CGRP. (D) The study design. p-values compared to baseline assessed with the Wilcoxon rank test and corrected with Bonferroni. No statistical comparison among group was performed. BoNTa = onabotulinumtoxinA; CGRP = calcitonin gene-related peptide.
Patient reported outcomes and migraine-related variables at baseline and after 12- and 24-weeks with atogepant in combination in the effectiveness cohort.

Significant values are in bold. Percentages are expressed on column total. Baseline before atogepant refers to the 4 weeks before the introduction of atogepant as add-on treatment with BoNTA; Weeks refer to add-on treatment with BoNTa and atogepant. MHDs, monthly headache days; AMDs, days with analgesics use per month; HIT6, headache impact test; SD, Standard deviation. Wilcoxon test with Bonferroni for multiple correction was applied (p<0.0167).
The results on headache severity subdivided in mild, moderate and severe headache days are reported in Table 2. Regarding disability, HIT-6 scores decreased to a mean ± SD of 59.11 ± 6.03 (p < 0.001). To note, a change of −1.5 to −2.5 points is generally considered the minimal MCID. Anxiety symptoms showed a not significant reduction, with a median change of −0.0 (95% CI = −1.76 to 0.11; p = 0.185) (Table 2). The PGIC had a mean ± SD of 5.6 ± 1.1, reflecting a “moderate to great improvement” perception from patients. In detail, 87.8% reported at least a “moderately better” change, with over half reporting “a definite worthwhile difference” (52.4%) and 13.4% “a great deal better and considerable improvement”. Very few patients (≤6.0%) reported only minimal or no meaningful change”.
The proportion of MMD 50% responders was 45.1% (37/82 patients) at 24 weeks of co-treatment (co-primary outcome). According to sub-categories, a ≥75% response was achieved by 17.1% of patients, while complete migraine freedom (RR100%) was achieved by one patient (1.2%) (Figures 3 and 4A and Table 3).

Response rate 50% (reduction in monthly migraine days, RR50%) in the effectiveness cohort and naïve and non-naïve patients to anti-CGRP mAbs during co-treatment. CGRP = calcitonin gene-related peptide.
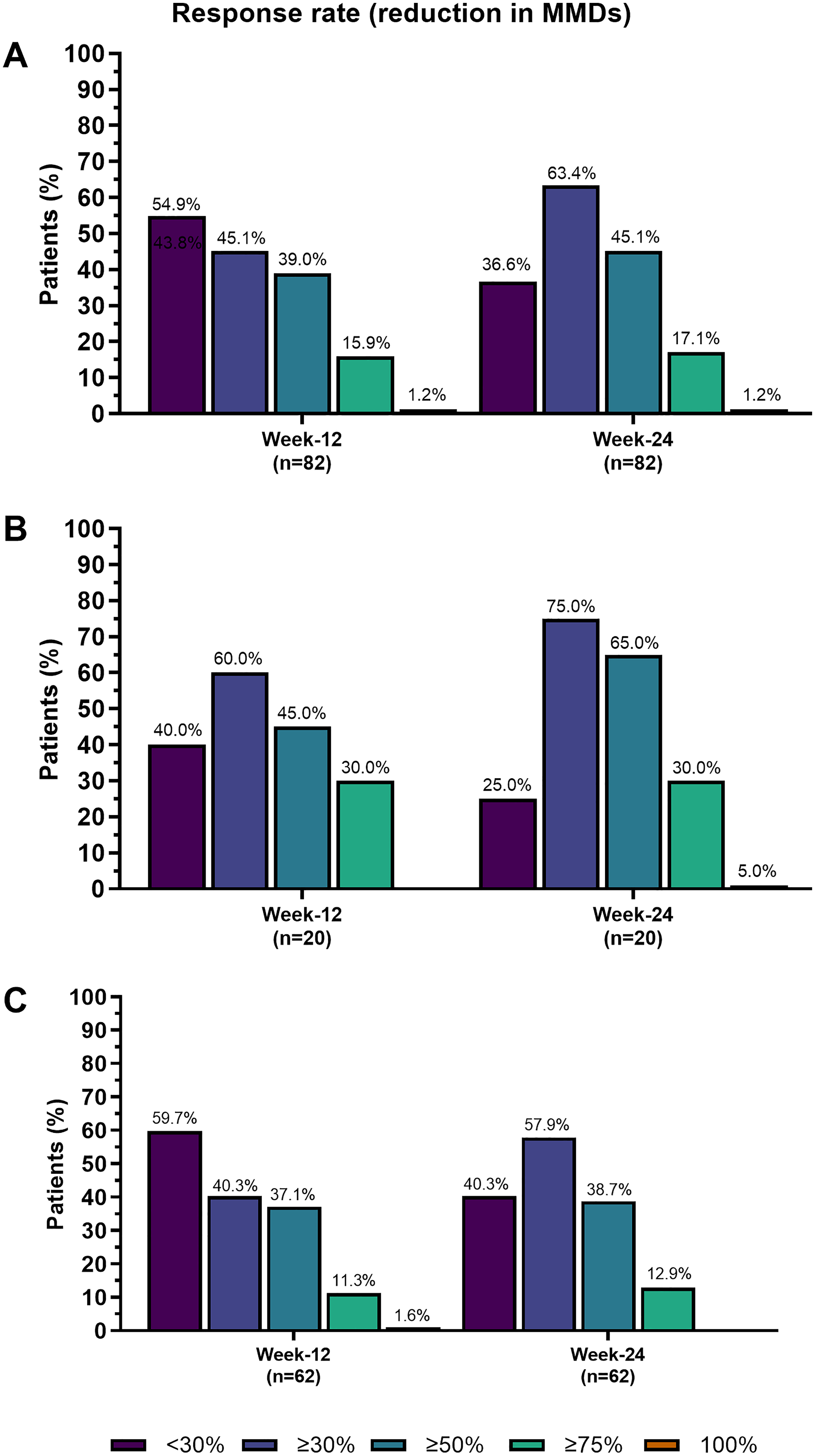
Response rates (reduction in monthly migraine days, RR50%) in the effectiveness cohort (A) and naïve (B) and non-naïve (B) patients to anti-CGRP mAbs during co-treatment. CGRP = calcitonin gene-related peptide.
Response rate (reduction in MMDs) after 12 and 24 weeks with atogepant in co-treatment in the effectiveness cohort and subdivided in anti-CGRP naïve and non-naïve cohorts.

Weeks refer to add-on treatment with BoNTa and atogepant. Percentages are not cumulative (nested subsets). CGRP = calcitonin gene-related peptide; MMDs = monthly headache days.
Based on the ≥50% responder rate (RR50%) for MMDs, among the 64 patients who were non-responders to BoNTA alone, 30 (46.9%) achieved responder status after 24 weeks of add-on atogepant. Conversely, among the 18 patients who had already achieved responder status with BoNT-A, 7/18 (38.9%) maintained their response, while 11/18 (61.1%) lost the responder status after 24 weeks of combination therapy.
The baseline observation carried forward (BOCF) sensitivity analysis, which also includes patients who discontinued treatment, is presented in the Supplementary Data and confirms a progressive, clinically meaningful reduction in migraine frequency over time.
Effectiveness outcomes at 12 weeks of treatment
The clinical benefit was already reported after 12 weeks of treatment. In particular, MHDs decreased from a baseline mean ± SD of 22.9 ± 5.92 to 18.2 ± 9.3 (p < 0.001). MMDs were similarly reduced from 16.4 ± 0.75 to 11.9 ± 8.6 (p < 0.001) and AMDs dropped from 11.2 ± 1.7 to 6.5 ± 1.5 (p < 0.001) (Figure 2 and Table 2).
Headache severity is reported in Table 2. The HIT-6 scores improved from 63.5 ± 3.7 to 60.3 ± 6.2 (p < 0.001). Anxiety symptoms showed a small and non-significant improvement, with a mean reduction of −0.56 (95% CI = −1.46 to 0.34; p = 0.277) (Table 2). The PGIC had a mean ± SD of 5.4 ± 1.0, reflecting a “moderately better” perception from patients. In detail, 88.9% of patients reported at least a “moderately better” change, with the majority rating their improvement as “a definite worthwhile difference” (52.4%) or “a great deal better” (7.3%).At 12 weeks, 32 out of 82 patients (39.0%) achieved a ≥50% reduction in MMDs. According to sub-categories, a ≥75% response was achieved by 15.9% of patients, while complete migraine freedom (RR100%) was achieved by only one patient (1.2%) (Figures 3 and 4A and Table 3).
Differences in responder versus non-responder (≥50% reduction in MMDs) subgroups
In the effectiveness cohort (n = 82), a comparative analysis of baseline characteristics between responders and non-responders at 24 weeks revealed several statistically significant differences (Table 4). Responders exhibited a higher baseline use of AMDs (11.7 ± 1.3 vs. 10.8 ± 2.2; p = 0.035) and reported lower baseline headache-related disability as measured by HIT-6 score (62.5 ± 3.0 vs. 64.8 ± 3.1; p < 0.001). Additionally, responders had undergone fewer prior treatment attempts, including both the number of BoNTA cycles (10.9 ± 6.6 vs. 14.8 ± 7.8; p = 0.018) and the total number of previously ineffective preventive therapies (4.8 ± 1.4 vs. 5.3 ± 1.1; p = 0.049). Prior exposure to anti-CGRP mAbs was also significantly less frequent among responders (64.9% vs. 84.4%; p = 0.040). No statistically significant differences were observed between groups with respect to age, sex, migraine onset, BMI, migraine with aura or the presence of comorbid conditions, although sleep disturbances showed a non-significant trend toward greater prevalence among non-responders (31.1% vs. 13.5%; p = 0.060) (Table 4).
Comparison of baseline characteristics between responders and non-responders based on reduction of monthly migraine days (MMDs) at 24 weeks.

Percentages are expressed on the column total. BMI = body mass index; CGRP = calcitonin gene-related peptide; MHDs = monthly headache days; AMD = days with at least one analgesics use per month; HIT-6 = Headache Impact Test-6. Chi-squared or Fisher’s exact test were applied for categorical data and the Mann–Whitney U-test for continue data.
Variables related of response status at 24 weeks
A series of univariate logistic regressions were performed to evaluate potential variables related to the 50% response at 24 weeks including all patients (safety cohort, n = 101). Patients who discontinued treatment were reported as non-responders (see supplementary material, Table S3). Several baseline clinical features were evaluated, selected based on previous studies and clinical relevance. However, only higher acute medication use (odds ratio (OR) = 1.379, p = 0.047) and HIT-6 score at baseline (OR = 0.884, p = 0.037) were predictive of achieving ≥50% response at 24 weeks. Other demographic and clinical variables were not significantly associated with treatment response in univariate analyses (see supplementary material, Table S3).
Therefore, a multivariable logistic regression was performed including variables with p > 0.1 at univariate analyses, corrected with sex and age. A Nagelkerke R2 of 0.155 indicates modest explanatory power. Among the variables included, only higher baseline use of acute medications (OR = 1.492, 95% CI = 1.053–2.114, p = 0.024) was significantly associated with response. Other variables did not reach statistical significance (see supplementary material, Table S4).
Anti-CGRP naïve and not naïve subgroups
In the between-group comparisons at baseline, anti-CGRP naïve (n = 20) and non-naïve patients (n = 62) showed similar clinical, demographic and migraine-related parameters (Table 5).
Comparison of baseline characteristics between anti-CGRP mAbs naïve and non-naïve.

Percentages are expressed on the column total. BMI = body mass index; CGRP = calcitonin gene-related peptide; MHDs = monthly headache days; AMNs = number analgesics per month; AMD = days with at least one analgesics use per month; HIT-6 = Headache Impact Test-6. Chi-squared or Fisher’s exact test were applied for categorical data and Mann-Whitney U test for continue data.
However, significant differences were observed in migraine onset age, which was later in naïve patients (23.15 ± 11.92 vs. 14.8 ± 6.7 years; p = 0.007) and in treatment history. Naïve patients had received fewer prior preventive treatments (4.45 ± 0.61 vs. 6.63 ± 0.91; p < 0.001) and fewer BoNTA cycles before starting atogepant (7.70 ± 5.70 vs. 14.77 ± 7.19; p < 0.001).
Although not statistically significant, comorbidities were numerically less frequent in the naïve group. Details are reported in Table 5.
Regarding effectiveness during combination treatment, both anti-CGRP naïve and non-naïve patients reported significant clinical improvements in multiple migraine-related and patient-reported outcomes following 12 and 24 weeks of treatment with atogepant in combination (Table 6). Among anti-CGRP naïve patients, the mean number of MHDs significantly decreased from a mean ± SD of 22.5 ± 6.06 at baseline to 18.0 ± 9.49 at week 12 (p = 0.011) and further declined to 14.3 ± 7.12 at week 24 (p < 0.001), with a statistically significant reduction between week 12 and week 24 (p = 0.013). MMDs followed a similar trajectory, dropping from 15.8 ± 6.48 at baseline to 9.8 ± 8.04 at week 12 (p < 0.001) and to 8.1 ± 6.43 at week 24 (p < 0.001), although the additional change between weeks 12 and 24 was not significant (p = 0.115) (Figure 2 and Table 6).
Patient reported outcomes and migraine-related variables at baseline and after 12- and 24-weeks with atogepant in combination in the effectiveness cohort between anti-CGRP naïve and non-naïve (switch).
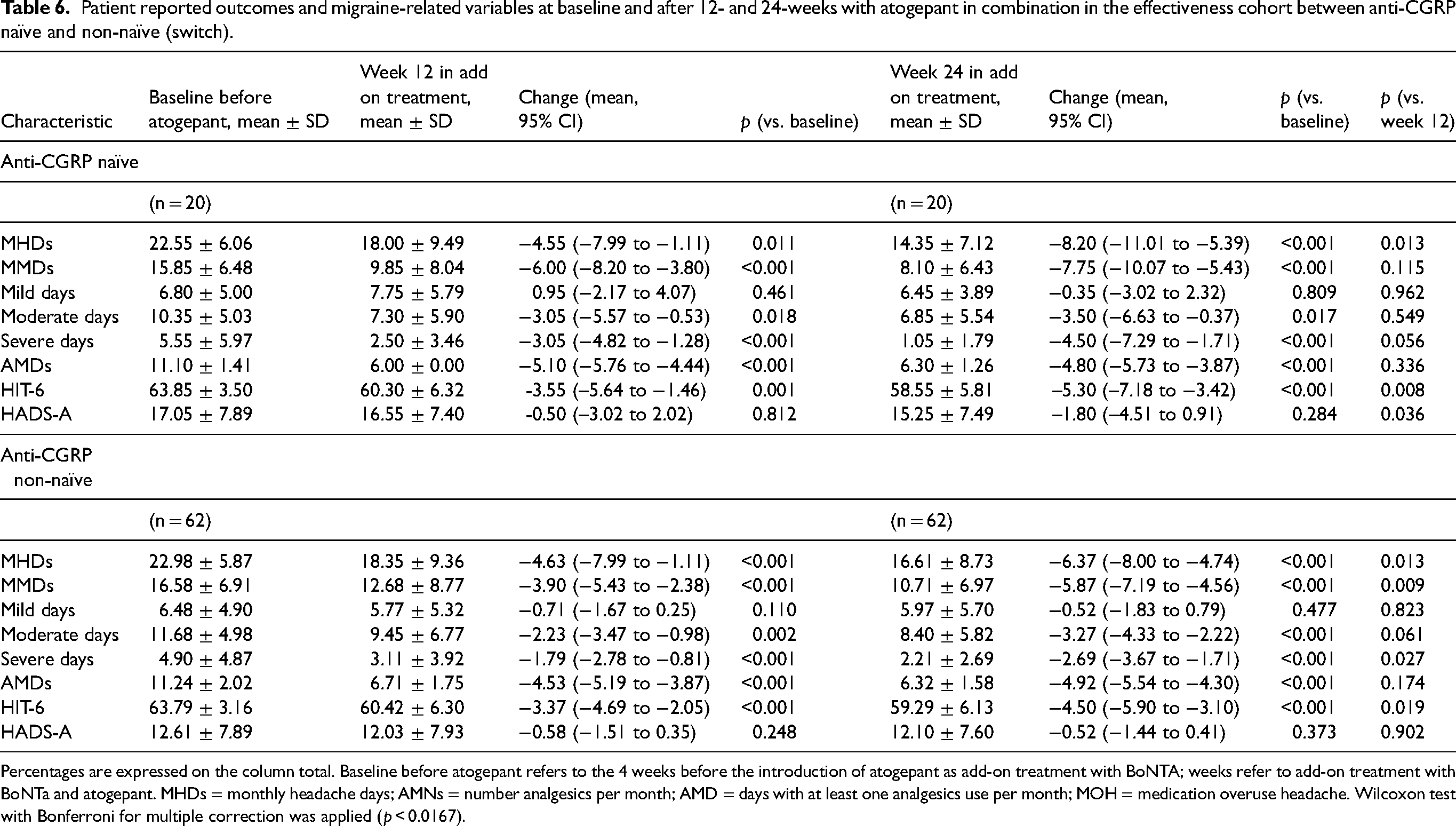
Percentages are expressed on the column total. Baseline before atogepant refers to the 4 weeks before the introduction of atogepant as add-on treatment with BoNTA; weeks refer to add-on treatment with BoNTa and atogepant. MHDs = monthly headache days; AMNs = number analgesics per month; AMD = days with at least one analgesics use per month; MOH = medication overuse headache. Wilcoxon test with Bonferroni for multiple correction was applied (p < 0.0167).
Differences in mild, moderate and severe pain days are reported in Table 6.
The AMDs showed a substantial and rapid decrease at week 12 (from 11.1 to 6.0; −5.1; p < 0.001), which was largely maintained at week 24 (6.3; −4.8; p < 0.001). The disability, measured via HIT-6 scores, significantly declined from 63.8 to 60.3 at week 12 (−3.5; p = 0.001) and to 58.5 at week 24 (−5.3; p < 0.001), with a further significant improvement between week 12 and 24 (p = 0.008). Anxiety symptoms assessed with the HADS-A scale showed only a non-significant trend toward improvement over time (Table 6).
In patients who had previously received anti-CGRP mAbs (non-naïve group), similar but generally more modest patterns of improvement were observed. MHDs were reduced from a mean ± SD of 22.98 ± 5.87 to 18.3 ± 9.36 at week 12 (p < 0.001) and to 16.61 ± 8.73 at week 24 (p < 0.001). MMDs followed a similar trend, decreasing from 16.5 at baseline to 12.6 at week 12 (−3.9; p < 0.001) and to 10.7 at week 24 (−5.8; p < 0.001), with a significant change from week 12 to 24 (p = 0.009) (Figure 2 and Table 6). Reductions in moderate and severe days were also significant at both time points, as well as AMDs (Table 6). The HIT-6 score improved from 63.7 to 60.4 at week 12 (−3.3; p < 0.001) and further to 59.2 at week 24 (−4.5; p < 0.001), while HADS-A scores remained unchanged throughout the study period. Detailed differences are reported in Table 6.
Finally, according to response rates, in the naïve group at 12 weeks, 9 patients (45.0%) achieved ≥50% reduction in MMDs and 6 (30.0%) reached a response of ≥75%. At 24 weeks, the distribution shifted with 13 (65.0%) and 6 (30.0%) patients reaching the ≥50%, and ≥75% thresholds, respectively. One patient (5.0%) achieved a complete response. In non-naïve patients, at 12 weeks, 23 (37.1%) patients achieved ≥50%, 7 (11.3%) reached ≥75% and one (1.6%) complete response. At 24 weeks, response distribution was that 24 patients (38.7%) reached ≥50% and eight (12.9%) achieved ≥75%. No patients reached full response at 24 weeks. Response rates are reported in Table 3 and Figures 3 and 4,C).
Overall tolerability and adverse events
Among the 101 patients included in the overall analysis, 65 (64.4%) reported at least one AE during treatment. The most frequently reported was fatigue, experienced by 22 patients (21.8%), followed by constipation in 21 patients (20.8%) and nausea in 13 patients (12.9%). Other adverse events are reported in Table 7. Although AEs were common, they were generally mild to moderate.
Adverse events and dropout rates during combination treatment.

Calculated on 81 adverse events. Percentages are expressed on the column total if not otherwise specified.
Overall, 19 patients discontinued treatment for various reasons (18.8%). Specifically, 11 patients (11/101; 36.9%) discontinued due to lack of effectiveness, seven (7/101; 6.9%) due to AEs and one patient (1/101; 1.0%) for not planned pregnancy after 12 weeks of treatment (six weeks of gestation) (Table 7).
Discussion
This real-world study offers novel evidence on the combined use of BoNTA and atogepant for the preventive treatment of CM over a 24-week period. The addition of atogepant to patients already receiving for a long period BoNTA with modest but significant effectiveness, resulted in higher reductions in MMDs, MHDs, acute medication uses and patient-reported disability. These benefits were accompanied also by favorable patient-reported outcomes. Notably, the combination therapy seems particularly effective in patients who were naïve anti-CGRP mAbs and those with a lower baseline disease burden.
Our findings are consistent with and build upon those reported by Blumenfeld et al. (24) who evaluated the addition of atogepant to ongoing BoNTA treatment in a retrospective multicenter cohort in the USA. In their study of 31 patients, the combination led to a mean reduction in MHDs of 4.53 days at 12 weeks and 8.75 days at 24 weeks, with 57.1% of patients achieving a ≥50% reduction in MHDs. In our larger cohort (n = 82 in the effectiveness population), we observed a comparable ≥50% responder rate of 45.1% at 24 weeks, along with meaningful reductions in both MMDs and HIT-6 scores. These findings confirm the consistency of clinical benefit associated with the combination therapy across independent real-world settings.
Importantly, both studies demonstrated a similar tolerability profile with low dropout rates (18.8% in our study vs. 16.1% in the study by Blumenfeld et al. (24) and no serious adverse events reported. The AE profile observed was consistent with known effects of either drug alone with no novel or unexpected AES emerging AEs from the combination. This aligns with the expectations given to the different mechanisms of action and the locally administered BoNTA.
Moreover, our subgroup analyses yielded novel insights. Patients who were anti-CGRP naïve experienced numerically greater reductions in MHDs (−8.2 vs. −6.4) and disability (HIT-6: −5.3 vs. −4.5), suggesting a potential enhanced response to anti-CGRP drugs (both gepants and mAbs) when used earlier in the preventive treatment paradigm.
Our responder-level analysis provides important insights. At baseline (prior to add-on treatment with atogepant), only 22.0% of patients were responders to BoNTA. With the addition of atogepant, almost half of prior BoNTA non-responders (30/64, 46.9%) achieved a ≥50% response at 24 weeks. Conversely, among the 18 patients who had already achieved responder status with BoNTA, seven of 18 (38.9%) maintained their response, while 11/18 (61.1%) lost it after 24 weeks of combination therapy.
This suggests that the primary therapeutic advantage of the combination could be in converting BoNTA non-responders or partially responders into responders, whereas its incremental benefit in prior responders appears uncertain. However, responder status, while widely used, is not by itself a sufficiently reliable parameter to capture overall treatment effectiveness because it simplifies continuous changes in migraine days and does not account for improvements in disability or quality of life. Further studies are needed to better characterize the add-on effects of atogepant in responders, partially responders and non-responders to monotherapy.
Regarding variables related to 50% response status, it is interesting and unexpected that a higher (although overall moderate) use of acute medication at baseline is related with higher probability to be a responders to atogepant at 24 weeks. A possible explanation is that, given the high headache frequency, patients who continue to use analgesics are those who obtain at least partial relief, indicating that, even in very severe cases, the condition is not entirely refractory to treatment. However, the potential predictive value is modest, and these baseline factors could not be viewed as a reliable clinical tool for individual-level prediction, and so further studies with larger samples are needed to confirm or refine these preliminary observations.
Preclinical data support the biological plausibility of an additive, or potentially synergistic, effect of the combination therapy (15,16,18) as reported before.
Dual-targeting approaches, especially those involving different mechanistic classes such as gepants and BoNTA, appear promising for patients with persistent (residual) migraine burden. One of the advantages of BoNTA is its favorable safety profile, with side effects that are generally mild and rarely lead to treatment discontinuation. Furthermore, BoNTA has no known pharmacological interactions with other medications, making it a suitable option for combination therapy. Several clinical studies have evaluated the combination of BoNTA and anti-CGRP mAbs for migraine prophylaxis (recently reviewed in. 7 Although no RCTs have been conducted to date, 15 studies (most of them retrospective) have investigated this combination in a total of 1428 patients with CM (7). However, only the aforementioned study (24) assessed the combination of atogepant and BoNTA. Thus, data on gepant-based combinations have so far remained scarce. Our study directly addresses this knowledge gap.
The potential carry-over effect of BoNTA monotherapy should be considered. Clinical trials have shown that therapeutic benefit may continue to accumulate over the first three treatment cycles (25,26), which is why guidelines recommend assessing efficacy only after this period (27). To minimize confounding in our study, we included only patients who had already received at least three BoNTA cycles before starting atogepant, reducing the likelihood that observed improvements were due to BoNTA monotherapy alone. Furthermore, the present study does not provide direct evidence that combination therapy yields superior clinical benefit compared with optimized monotherapy. Although the observed improvements in migraine frequency, acute medication use and disability suggest a potential additive effect, the absence of direct comparison arms, specifically BoNTA or atogepant monotherapy (see also limitations), precludes firm conclusions regarding superiority and it was not the aim of this real-world observational study. Therefore, the findings should be interpreted as exploratory and hypothesis-generating, underscoring the need for RCTs directly comparing optimized monotherapy versus combination treatment to determine whether dual targeting truly confers incremental therapeutic advantage.
Another important consideration concerns the economic implications of dual preventive therapy. Both BoNTA and atogepant are high-cost treatments, and their combined use may represent a substantial financial burden for healthcare systems and patients. This issue is particularly relevant for clinicians and policymakers when weighing the clinical benefits against affordability and access. Although our study was not designed to perform cost-effectiveness analyses, the real-world evidence provided here demonstrates that a higher proportion of patients were working after 24 weeks of dual treatments, highlighting the need for future health-economic evaluations to assess whether the incremental clinical benefits of combination therapy justify its higher costs, as well as to define which patient subgroups are most likely to derive sufficient benefit to warrant dual therapy.
Finally, it is important to acknowledge that management of highly refractory CM cannot rely solely on a sequential “trial-and-error” strategy in which patients are continuously switched across preventive options. While innovation is essential, indiscriminate layering of treatments may lead to unrealistic expectations, increase frustration and risk of refractoriness. In such complex cases, a different therapeutic paradigm, including also different non-pharmacological approaches, needs to be also considered. Clinicians also have an ethical responsibility to balance the promise of novel therapies with scientific rigor and the use of appropriate resources. These need to be balanced with appropriate clinical evidence to whether dual therapy provides sufficient additional clinical benefit to justify its use compared with monotherapy, as discussed below.
The SYNERGY study presents several strengths, including a real-world population of difficult-to-treat migraine, a comprehensive 24-week follow-up, inclusion of both clinical and patient-reported outcomes, and robust subgroup analyses (also including patients non-naïve to anti-CGRP drugs). Compared to previous data, our study has enrolled a larger cohort, provided stratification by prior treatment failures and assessed predictors of response.
Nonetheless, some limitations must be acknowledged. First, its non-randomized design limits causal inference and introduces the possibility of selection bias, and no formal sample size calculation was performed.
Second, we did not include a control group treated with BoNTA alone, which precludes direct comparisons with monotherapy. We also recognize that switching to atogepant monotherapy in patients with suboptimal or waning response to BoNTA represents a clinically relevant management option. However, this study was conceived to evaluate the feasibility, tolerability and potential additive benefit of combining atogepant with ongoing BoNTA, considering also the patient's willingness and the partial response of BoNTA reported, rather than substituting one treatment for another. The rationale was based on the emerging evidence of combination therapy discussed above.
Nonetheless, the absence of an atogepant monotherapy arm limits the ability to determine whether the observed benefit reflects a true synergistic/additional effect or simply a response to atogepant alone after partial BoNTA response. Future RCTs (and real-word studies) should be specifically designed to address this gap. An ideal study design would include at least three parallel arms: (i) BoNTA monotherapy; (ii) atogepant monotherapy following BoNTA discontinuation; and (iii) combined BoNTA plus atogepant therapy. Such a comparative framework would allow evaluation of differential efficacy, onset of action, durability and safety across strategies, as well as help define evidence-based treatment algorithms, whether sequential, alternating or combined, for patients with CM inadequately controlled on monotherapy.
A further limitation is the absence of complete post-discontinuation data, which prevented a full intention-to-treat (ITT) analysis; however, a sensitivity analysis including patients who discontiued treatment for ineffectiveness was performed, confirming the consistency of the main findings. Future studies should aim to implement comprehensive ITT analyses because relying only on observed-case data may inflate treatment effects and underestimate variability in a real-world response. We did not impute for missing data, although they were not present for co-primary outcomes. Finally, the study was conducted at a single center in Norway, which may limit generalizability to other healthcare systems and populations.
Conclusions
The combination of BoNTA and atogepant seems to be an effective and well-tolerated strategy for the preventive treatment of CM, particularly in patients with a history of multiple treatment failures, including those involving anti-CGRP mAbs. Our findings contribute to the growing evidence that might support combination therapy in migraine prophylaxis, where agents with complementary mechanisms of action may offer enhanced clinical benefit, although our study does not yet confirm that dual therapy is better than monotherapy. Prospective controlled trials and health-economic evaluations are warranted to validate these observations and inform future clinical guidelines.
Adding atogepant to ongoing onabotulinumtoxinA led to meaningful reductions in migraine frequency, acute medication use and disability, with almost half of patients reaching a ≥50% response at 24 weeks. This dual-targeted strategy appears particularly useful in chronic migraine patients with prior treatment failures, offering an effective real-world preventive option. The combination was well tolerated, with adverse events consistent with the known safety profiles of each drug and no novel safety concerns observed
Supplemental Material
sj-docx-1-cep-10.1177_03331024251398011 - Supplemental material for Combination preventive therapy with onabotulinumtoxinA and atogepant for chronic migraine: A 24-week, prospective, real-world evaluation (SYNERGY study)
Supplemental material, sj-docx-1-cep-10.1177_03331024251398011 for Combination preventive therapy with onabotulinumtoxinA and atogepant for chronic migraine: A 24-week, prospective, real-world evaluation (SYNERGY study) by Luigi Francesco Iannone, Marina Romozzi, Antonio Russo, Ian Finkelstein, Dineo Seabi, Adam Ahlden, Anne Hege Aamodt, Edoardo Caronna, Patricia Pozo-Rosich, Erling Andreas Tronvik and Christina Sundal in Cephalalgia
Footnotes
Acknowledgments
Figure 1 created with Biorender.com
Author contributions
All authors critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final version of the manuscript submitted for publication.
Consent to participate
All provided their written informed consent prior to initiating treatment.
Data availability
Data supporting the findings in the present study are reported in the article and in the supplementary materials. The data collected and analyzed for the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
LFI received financial support, consulting fees for the participation in advisory boards and support for attending meetings from: Teva, Eli Lilly, Lundbeck, Pfizer and AbbVie; he is Associate Editor for Frontiers in Neurology and junior editor of Cephalalgia and Cephalalgia Reports.
MR: None.
AR: has received speaker honoraria from Allergan, Lilly, AbbVie, Pfizer, Novartis and Teva, and serves as an associate editor of Frontiers in Neurology (Headache Medicine and Facial Pain session).
IF: Grants/honorarium/Consulting fees: Allergan, Amgen, Eli Lilly, J and J, Merck, Purdue, Teva, Aralez, Novartis, Lundbeck and Pfizer.
DS: None.
AA: None.
AHA: has received personal fees for lectures/advisory boards: Novartis, Abbvie, TEVA, Roche, Lundbeck, Pfizer and Boehringer Ingelheim. Non-personal research grants from several sources, including KlinBeForsk, EU, Boehringer Ingelheim, Medtronic and BMS.
EC: has received honoraria from Novartis, Chiesi, Lundbeck, MedScape and Lilly; his salary has been partially funded by Río Hortega grant Acción Estratégica en Salud 2017–2020 from Instituto de Salud Carlos III (CM20/00217) and Juan Rodés fellowship, Subprograma Estatal de Incorporación de la Acción Estratégica en Salud 2023 (JR23/00065). He is a junior editor for Cephalalgia.
PPR: has received, in the last three years, honoraria as a consultant and speaker from AbbVie, Amgen, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis, Organon, Pfizer and Teva Pharmaceuticals. Her research group has received research grants from AbbVie, AGAUR, EraNet Neuron, FEDER RIS3CAT, Instituto Investigación Carlos III, MICINN, Novartis and Teva Pharmaceuticals, and has received funding for clinical trials from AbbVie, Amgen, Biohaven, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva Pharmaceuticals. She is the Honorary Secretary of the International Headache Society, is on the editorial board of Revista de Neurologia, is an associate editor for Cephalalgia, Headache, Neurologia and Frontiers of Neurology, and is an advisor of the Scientific Committee of the Editorial Board of The Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee and Scientific Committee of the International Headache. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() .
.
ET: has received personal fees for lectures/advisory boards: Novartis, Eli Lilly, Abbvie, TEVA, Roche, Lundbeck, Pfizer, Biogen and Organon. Consultant for and owner of stocks and IP in Man & Science. Stocks and IP in Nordic Brain Tech. Stocks in Keimon Medical. Non-personal research grants from several sources, including Norwegian Research Council, KlinBeForsk, EU. Commissioned research (non-personal): Lundbeck.
CS: has received honorarium for lectures/advisory boards: Novartis, Abbvie, TEVA and Vigil NeuroScience.
Ethical considerations
The local Ethics committee approved the data collection 16.02.2024 with code #604900.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
