Abstract
Background
Treatment with onabotulinumtoxinA is effective in reducing migraine day frequency and duration in individuals with chronic migraine (CM), but given the severity of the disease, those with CM often need additional treatment to achieve optimal outcomes. Combining preventive treatments with distinctly different physiological targets may yield greater benefit than monotherapy. This study evaluated the safety, tolerability, and efficacy of adding atogepant to onabotulinumtoxinA for preventive treatment of CM.
Methods
In this 24-week, Phase 3, open-label, single arm, multicenter study (NCT05216263), 75 participants on a stable dose of onabotulinumtoxinA (155–200U) with baseline mean monthly migraine days (MMDs) of 8–23 (inclusive) received add-on atogepant 60 mg once daily. Primary safety endpoints included treatment-emergent adverse events (TEAEs), and exploratory efficacy endpoints included changes in MMDs, changes in mean monthly headache days (MHDs) and responder rates (RRs) (≥50%, ≥75%, and 100% MMDs) over Weeks 1–12, Weeks 13–24, and at each 4-week interval.
Results
The mean age of study participants was 48 (13.68) years (mean (SD)); 89% were women, and 97% were white. Participants had an established CM diagnosis for 15 (13.27) years (mean (SD)) and had been treated with onabotulinumtoxinA for 4 (3.45) years (mean (SD)). In the safety population (n = 75), the incidence of TEAEs was 65.3%. TEAEs occurring in ≥5% of participants were constipation (n = 12, 16.0%), nausea (n = 10, 13.3%), and urinary tract infection (n = 6, 8.0%). An AE was considered the primary reason for drug discontinuation in 2 (2.7%) participants. Treatment-emergent serious AEs (TESAEs) occurred in 2 participants; neither were considered treatment-related by the investigators. In the modified intention-to-treat population (n = 72), the least squares (LS) mean change from baseline of 14.34 MMDs was −6.45 MMDs (95% CI: −7.7, −5.1) at Weeks 1–4, −6.89 MMDs (95% CI: −8.1, −5.6) across Weeks 1–12, and −7.20 MMDs (95% CI: −8.4, −5.9) across Weeks 13–24. The least square mean change from baseline of 17.00 MHDs was −6.57 MHDs (95% CI: −7.8, −5.3) at Weeks 1–4, −7.33 MHDs (95% CI: −8.6, −6.0) across Weeks 1–12 and −8.15 MHDs (95% CI: −9.4, −6.8) across Weeks 13–24. A ≥ 50% RR in MMD was achieved by 54.2% and 61.9% of participants across Weeks 1–12 and Weeks 13–24 of combined treatment, respectively. A ≥ 75% RR was achieved by 30.6% and 38.1% of participants.
Conclusions
In this study, combination preventive treatment for CM with onabotulinumtoxinA and atogepant was safe and generally well-tolerated. Furthermore, the addition of atogepant to those stable on onabotulinumtoxinA resulted in clinically meaningful reductions in migraine days and improvement in responder rates, suggesting a benefit of combining treatments with distinct and complementary mechanisms of action for suppression of CM.
Trial Registration
Clinical Trials.gov NCT05216263.
This is a visual representation of the abstract.
Background
Introduction
Chronic migraine (CM) is a debilitating neurological disease, defined by the International Classification of Headache Disorders, 3rd edition (ICHD-3), as 15 or more headache days per month with at least 8 qualifying as migraine days (Headache Classification Committee of the IHS, 2018. 1 CM is associated with substantial personal, societal, and economic burden, resulting in impaired quality of life and increased healthcare resource utilization.2–8 Multiple evidence-based preventive therapies are available to treat CM. OnabotulinumtoxinA, which was approved in 2010, has demonstrated robust efficacy and an established safety profile across randomized controlled studies and real-world evidence in CM.9–13 Calcitonin gene-related peptide (CGRP) and its receptor have emerged as pivotal targets in migraine, and findings from real-world, open-label, and randomized controlled studies have demonstrated 4 monoclonal antibodies (mAbs) and an oral antagonist (gepant) to be safe and effective preventive treatments for CM.14–17
Despite the availability of these 6 therapies, many individuals with CM managed with a single preventive medication may still experience frequent migraine attacks and ongoing disability; therefore, may need adjunctive preventive therapies to achieve optimal migraine control.18,19 Identification of effective multimodal management requires an understanding of how different treatments act on distinct physiological pathways and that 20 treatments targeting multiple pathways have potential for additive or synergistic benefit.21–23
OnabotulinumtoxinA and the anti-CGRP preventive treatments act on different physiological pathways with distinct mechanisms of action,24,25 so treatment regimens that combine onabotulinumtoxinA and CGRP pathway–inhibiting therapies are increasingly recommended by international guidelines for individuals with refractory or persistent migraine.20,26,27 Supporting the safety and efficacy of this combination approach, clinical observational studies and retrospective chart reviews have demonstrated meaningful reductions in migraine burden when CGRP preventive treatments are added to onabotulinumtoxinA.18,19,28 Specifically, a recent chart review study on combination atogepant plus OnabotulinumtoxinA 19 as well as preclinical evidence on this combination 25 suggest the need to further investigate the utility of adding atogepant, an oral, once-daily CGRP receptor antagonist with onabotulinumtoxinA.15,16,29
Building upon existing real world and preliminary clinical research and recognizing the need for evidence-based guidance on combination preventive treatment, we designed this multicenter, 24-week open-label investigational study to evaluate the safety, tolerability, and efficacy of atogepant 60 mg once daily (QD) when added to onabotulinumtoxinA for adults with CM and persistent migraine burden.
Methods
Study design
This is a 24-week, Phase 3, open-label, single-arm, multicenter study to evaluate the safety, tolerability, and efficacy of atogepant when added to onabotulinumtoxinA in participants with CM (Figure 1). Although both drugs are individually approved, the combination is not, which aligns with a regulatory recommendation for Phase 3 trial classification as well as how it is posted on clinicaltrials.gov. The study screened 171 participants and 56.1% [96] participants failed screening with the top three reasons for screen failure being: Must have 8 to 23 (inclusive) migraine days in the 28-day careening/baseline period (as assessed with eDiary) (42.7%), no clinically significant laboratory values or any of the following laboratory values at visit 1: Alanine aminotransferase or aspartate aminotransferase>1 (12.5%), and previous administration of onabotulinumtoxinA prior to study entry was between 8 to 1 week prior to scheduled screening (8.3%). This study enrolled 75 participants from 23 sites in the United States (03-22-2022–05-02-2025). The screening/baseline period was up to 12 weeks, which included at least 28 days of eDiary collection of migraine days and headache days. The treatment period included 7 scheduled clinic visits: Visit 1/Visit 1a (screening/baseline), Visit 2 (Day 1), Visit 3 (Week 4), Visit 4 (Week 8), Visit 5 (Week 12), Visit 6 (Week 16), Visit 7 (Week 20), and Visit 8/Premature Discontinuation (Week 24). There was a safety follow-up period of 4 weeks that included 1 remote visit at Visit 9 (Week 28 or 4 weeks post-atogepant treatment for premature discontinuation). Participants received atogepant 60 mg once daily (QD) from Day 1 to Week 24, along with their concomitant onabotulinumtoxinA. Concomitant onabotulinumtoxinA was administered on Visits 2, 5, and 8. The final Visit of the treatment period was at Week 24 with a post-atogepant treatment follow-up at Week 28 or 4 weeks post-atogepant treatment for premature discontinuation.

Study design
Study participants
Participants were adults aged 18 to 75 years, who had at least a one-year history of CM, with or without aura, consistent with a diagnosis according to the International Classification of Headache Disorders 3rd edition (ICHD-3, 2018), and with or without acute medication overuse, defined as follows: use of triptans on 10 or more days, ergots on 10 or more days, use of simple analgesics (i.e., aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), or acetaminophen) on ≥15 days,
Participants were excluded if they had used opioid-containing products for more than 4 days for headache treatment in the 3 months prior to screening or during the baseline period; received acupuncture, TENS, cranial traction, dental splints, or anesthetic/steroid injections in target muscles within 4 weeks before screening; or require botulinum toxin treatment for any reason other than the study protocol. Individuals with hypertension (sitting systolic blood pressure >160 mmHg or diastolic pressure >100 mmHg), significant risk of self-harm, harm to others, or serious suicidal ideation (Type 4/5 on C-SSRS in the past 6 months), or significant medical or surgical conditions—including abnormal labs or imaging—that could compromise safety or participate effectively, were excluded. Exclusion also applied to those with active infections (such as varicella, herpes zoster, severe viral infections, and SARS-CoV-2), unremitting “long COVID,” or failure to meet SARS-CoV-2 viral clearance or vaccination requirements. Additional exclusions included active or recent malignancy (within the last 5 years except for adequately treated non-melanoma skin cancer or localized cervical carcinoma in situ), organ transplantation history or plans, significant drug or alcohol abuse within 2 years, allergy or sensitivity to study drug or similar agents, significant ECG abnormalities (QTcF >450 ms for males, >470 ms for females), acute hepatitis within 6 months, chronic liver disease or positive hepatitis B/HCV tests, and key lab abnormalities (ALT, AST, or total bilirubin >1× ULN except Gilbert's disease, or serum albumin <2.8 g/dL). Receipt of live virus vaccine within 4 weeks prior to enrollment or anticipated need during/within 4 weeks after the study also resulted in exclusion. Other exclusions included any female known to be pregnant, breastfeeding, or considering becoming pregnant during the study or within 30 days after the last dose of study drug, concurrent use of any preventive treatment of migraine other than onabotulinumtoxinA in the 4 weeks prior to Visit 1 or during the screening/baseline period, use of mAbs blocking the CGRP pathway within the 6 months (24 weeks) prior to screening (Visit 1), and the concurrent use of oral gepants for acute treatment of migraine attacks in the 4 weeks prior to Visit 1 or during the screening/baseline period.
Outcome measures
The primary outcomes involved an assessment of the safety and tolerability of atogepant added to onabotulinumtoxinA over a 24-week treatment period. Safety assessments included treatment-emergent adverse events (TEAEs), defined as any AE with the onset that was after the first dose of atogepant, serious adverse events (SAEs), laboratory measures (including hepatic function), vital signs, weight, and ECG parameters. Additionally, a Data Monitoring Committee (DMC) monitored safety data and advised the sponsor on study modifications, as well as a Hepatic Event Adjudication Committee (HEAC) for adjudication of cases with postbaseline elevations of ALT and/or AST ≥ 3× the upper limit of normal.
Exploratory efficacy endpoints included change from baseline in monthly migraine days (MMDs) across weeks 1 to 12, weeks 13 to 24, and at each 4-week interval, change from baseline in monthly headache days across (MHDs) weeks 1 to 12, weeks 13 to 24, and at each monthly period and responder rates (RRs) defined as achievement of ≥50%, ≥75%, and 100% reduction in mean MMDs across weeks 1 to 12, weeks 13 to 24, and at each 4-week interval.
Statistical analysis
The study was powered empirically to include 75 participants. An estimated early termination rate of 20% allowed for 60 participants to complete the 6-month study. Seventy-five participants provided a 95% probability to detect at least 1 occurrence of an AE with an incidence of ≥4%. The precision (half width of the 95% CI) of the estimate of change from baseline in mean MMDs was approximately 1.8, assuming a SD of 7.0 with an effective sample size of 60 participants.
The safety population included all participants receiving ≥1 dose of atogepant. The modified intention-to-treat (mITT) population comprised those with ≥1 dose of study drug, have an evaluable baseline period of eDiary data, and ≥1 postbaseline evaluable postbaseline 4-week period of eDiary data during the open-label treatment period. Change from baseline in MMDs and MHDs were evaluated using mixed-effects models for repeated measures (MMRM), with least squares (LS) means and 95% CI) reported. Responder rates were summarized using descriptive statistics.
The safety parameters include AEs, clinical laboratory tests (hematology, chemistry, and urinalysis), ECGs, vital signs, and the C-SSRS. For each safety parameter, the last non-missing safety assessment before the first dose of atogepant was used as the baseline for all analyses of that safety parameter. Categorical variables were summarized by number and percentage of participants. All analyses on efficacy variables were performed in the mITT population. For monthly endpoints, baseline was defined as assessments during the last 28 days of baseline period. Descriptive statistics for efficacy endpoints was based on observed cases only. Missing data were handled using MMRM without imputation for the efficacy analysis for continuous endpoints. No multiplicity adjustment for overall Type I error control was planned for this study, and multiplicity adjustments were not performed for exploratory endpoints in this study.
Results
Demographics and baseline characteristics
A total of 171 participants were screened; 75 constituted the safety population, and 72 were included in the mITT population. In the safety population (N = 75), the mean (SD) age was 47.7 (13.68) years; 89% were female and 97% were White. The mean (SD) BMI was 28.51 (6.31) kg/m2. During Visit 2, 42 participants (58.3%) received a dose of 155 units, while the remaining participants received doses ranging from 165 to 200 units (Table 1). Detailed onabotulinumtoxinA dosing reported in Supplementary table 3. Dosing information was missing for 3 participants (Table 1). Nearly half of the participants (49.3%) had a history of migraine with aura. Participants had an established CM diagnosis for 15 (13.27) years (mean (SD)) and had been treated with onabotulinumtoxinA for 4 (3.45) years (mean (SD)) (Table 1). In the open-label phase of this study, 77.3% of participants (58 out of 75) successfully completed the 24-week atogepant treatment period. Discontinuation occurred in 22.7% of participants (17 out of 75), with the primary reasons being withdrawal by participant (10.7%), other reasons (6.7%), lost to follow-up (2.7%), and AEs (2.7%). The ‘other’ category included site closure and voluntary withdrawal due to an AE (Figure 2).

Participant disposition open-label – safety/mITT population
Baseline demographics, migraine history and exposure - safety population

BMI – Body mass index, onabotA – onabotulinumtoxinA.
Safety outcomes
Mean duration of atogepant treatment was 155.2 (39.2) days, with 77.3% of participants receiving atogepant for ≥168 days. The cumulative participant-years of exposure was 31.9 (Table 1). In the safety population (n = 75), TEAEs occurred in 49 participants (65.3%). TEAEs with reasonable possibility of being related to atogepant occurred in 22 participants (29.3%), and those with reasonable possibility of being related to onabotulinumtoxinA in 1 participant (1.3%) (Table 2). Most frequent TEAEs occurring in ≥5% of participants were constipation (n = 12, 16.0%), nausea (n = 10, 13.3%), urinary tract infection (n = 6, 8.0%), decreased appetite (n = 5, (6.7%) and COVID-19 (n = 4, (5.3%)). Treatment-emergent SAEs (TESAEs) occurred in 2 participants, and neither were considered related to study treatment by the investigators. TEAEs leading to withdrawal of atogepant occurred in 7 participants (9.3%); 2 of the 7 reported AEs as the primary reason for discontinuation of atogepant, 1 reported “Other reason” (subject withdrew consent due to AE), and the remaining 4 reported “Withdrawal from Treatment by Participant” as primary reason for discontinuation (Table 3). TEAEs leading to the withdrawal of onabotulinumtoxinA occurred in 2 participants (2.7%), both of whom also discontinued atogepant. No deaths were reported (Table 2).
Overall summary of adverse events - safety population

TEAEs - Treatment-emergent adverse events, *2 out of the 7 reported AE as the primary reason for discontinuation of atogepant; 1 reported “Other Reason” (subject withdrew consent due to AE), and the remaining 4 reported “Withdrawal from Treatment by Participant” as primary reason for discontinuation.
These 2 participants were among the 7 with TEAEs leading to withdrawal of atogepant, i.e., they discontinued both background onabotulinumtoxinA treatment and study treatment of atogepant.
Most frequent (≥ 2%) TEAEs - safety population

TEAEs - Treatment-emergent adverse events.
No cases met criteria for Hy's Law and no ALT or AST elevations ≥3× the ULN were observed (Supplementary table 1). When assessing potentially clinically significant (PCS) changes occurring at any study visit in vital signs among the safety population, no participant showed PCS increases or decreases in sitting systolic blood pressure. For sitting diastolic blood pressure, 1 participant (1.4%) had a PCS increase and 1 participant (1.4%) had a PCS decrease (Supplementary table 2). Body weight measures showed that 6 out of 73 participants (8.2%) experienced a weight (kg) increase of ≥7%, and 6 out of 73 (8.2%) experienced a decrease of ≥7% (Supplementary table 2).
Efficacy outcomes
In the mITT population (n = 72), participants had a mean baseline of 14.34 MMDs. The LS mean change from baseline was −6.45 days [95% CI: −7.7, −5.1] at Weeks 1–4, −6.89 days [95% CI: −8.1, −5.6] across Weeks 1–12, and −7.20 days [95% CI: −8.4, −5.9] across Weeks 13–24 (Figure 3). Participants had a mean baseline of 17.00 MHDs. The LS mean change from baseline for atogepant was −6.57 days [95% CI: −7.8, −5.3] at Weeks 1–4, −7.33 days [95% CI: −8.6, −6.0] across Weeks 1–12 and −8.15 days [95% CI: −9.4, −6.8] across Weeks 13–24 (Figure 4). A total of 54.2% [39] participants achieved a ≥ 50% reduction from baseline in MMDs across Weeks 1–12 and 61.9% [39] during Weeks 13–24. 30.6% [22] participants achieved a ≥ 75% reduction from baseline in MMDs across Weeks 1–12 and 38.1% [24] during Weeks 13–24. 1.4% [1] participant achieved a 100% reduction from baseline in MMDs across Weeks 1–12 and 4.8% [3] during Weeks 13–24 (Table 4).
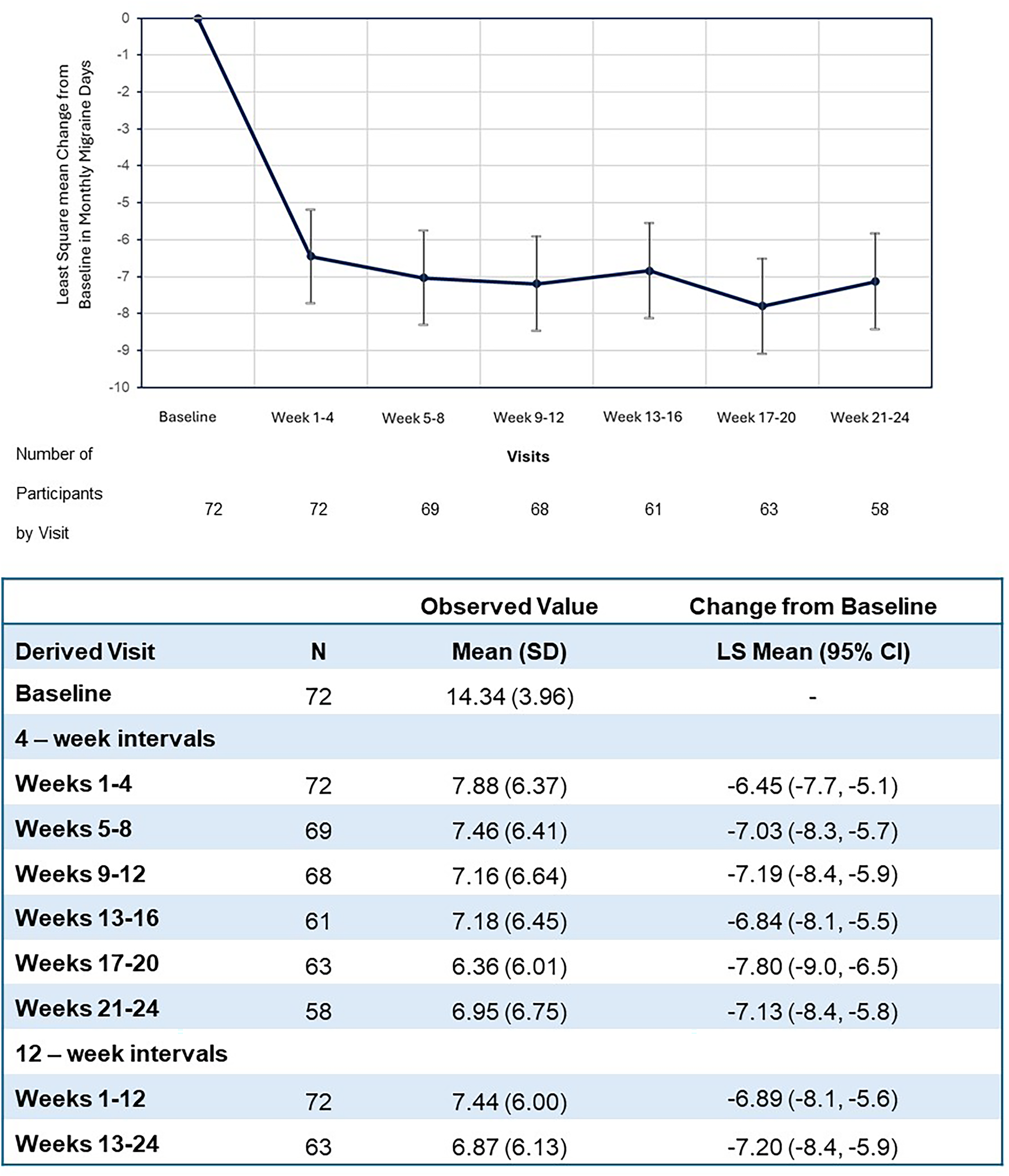
Change from baseline in MMD during 24-week open-label period - mITT population

Change from baseline in monthly headache days during 24-week ol period - mITT population
Responder rates - mITT population
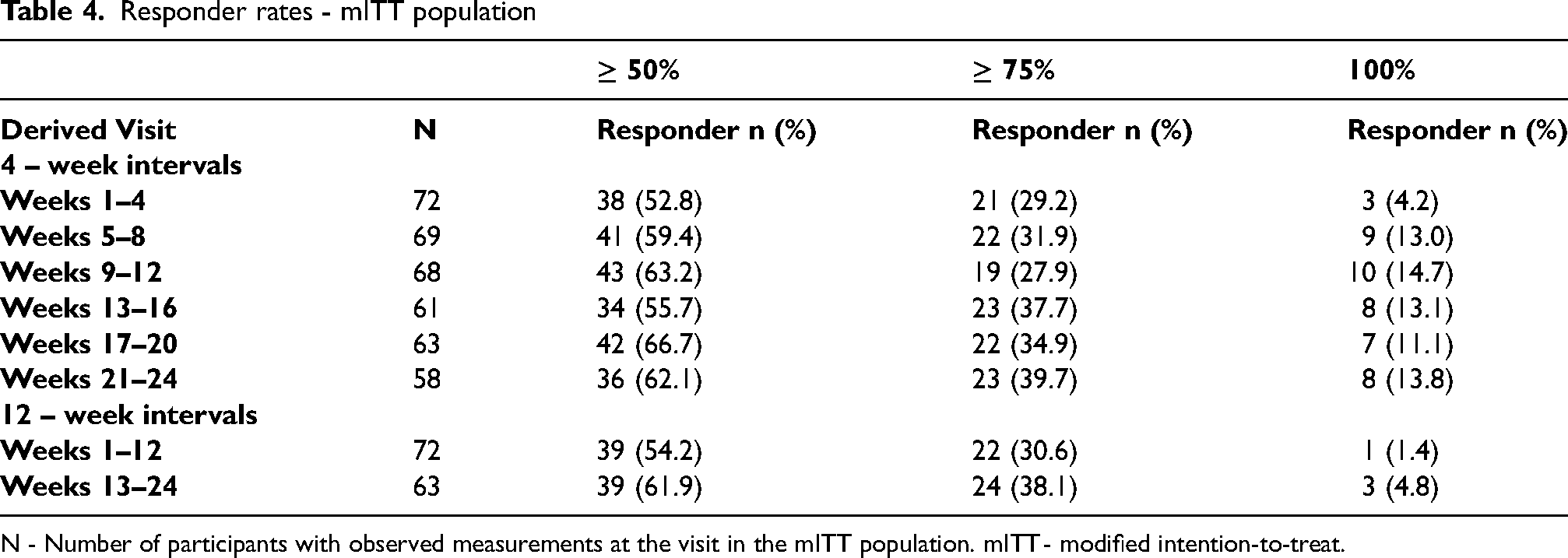
N - Number of participants with observed measurements at the visit in the mITT population. mITT - modified intention-to-treat.
Discussion
Many individuals with CM continue to experience substantial disease burden, including frequent headaches, disability, and impaired quality of life despite standard preventive monotherapy. Recent clinical guidance and position statements emphasize the importance of aiming for ambitious treatment targets, such as migraine freedom or optimal control (fewer than four migraine days per month).20,30 To achieve these more ambitious treatment goals often requires combination treatment.
This phase 3, multicenter, 24-week open-label study provides prospective evidence supporting the safety, tolerability, and efficacy of adding atogepant to onabotulinumtoxinA in adults with CM. CM participants enrolled had been on a stable and effective dose of onabotulinumtoxinA and had documented payer authorization to support continued onabotulinumtoxinA; the mean time prior to the addition of atogepant was 4 years. OnabotulinumtoxinA dosing variability (155 U to 200U) reflects real-world clinical practice and the selection of onabotulinumtoxinA dose was not based on a standard protocol. HCPs were allowed to continue to prescribe as they had done prior to entry to the study. Our findings demonstrate that this combination management regimen was generally safe and well-tolerated. The AEs were consistent with the known profiles of each agent alone and no new safety signals were observed. Importantly, the addition of atogepant resulted in early and sustained reductions in MMDs and MHDs, and improved RR supporting the additive benefits observed in prior retrospective studies and real-world practice.
The clinical rationale for combining atogepant with onabotulinumtoxinA lies in distinct and complementary mechanisms of sensory neuron modulation and CGRP pathway inhibition.24,25 OnabotulinumtoxinA effectiveness in CM is attributed to its inhibition of peripheral sensory nerve endings by blocking the fusion of synaptic vesicles with the nerve membrane and blocking the release of multiple neurotransmitters and neuropeptides involved in pain activation. 24 In contrast, atogepant specifically binds and blocks the CGRP receptor, thereby preventing CGRP activation of the migraine cascade. In addition, atogepant blocks the Amy 1 receptor which may also be implicated in migraine pathophysiology. 31 Preclinical studies also indicate that onabotulinumtoxinA and atogepant target different nociceptors implicated in migraine pathophysiology, and atogepant primarily prevents activation of CGRP receptors on thinly myelinated Aδ-fiber nociceptors with reduced activation of C-fiber nociceptors early and briefly 32 while onabotulinumtoxinA reduces sustained firing of unmyelinated C-fiber nociceptors. 33 Collectively, emerging clinical guidance, real-world and preclinical data support the rationale for combining onabotulinumtoxinA and atogepant.
Our findings are consistent with previously published real-world studies that evaluated the effectiveness of combination treatment with onabotulinumtoxinA and CGRP antagonists in the management of CM. Several studies support the use of combination treatment with CGRP mAbs and onabotulinumtoxinA in patients with CM (Supplementary table 4). As for the gepant, atogepant, a real-world retrospective chart review study in patients with CM demonstrated that adding atogepant to onabotulinumtoxinA was effective in providing reductions in MHDs of −4.53 days at 3 months and −8.75 days at 6 months. 34 Additionally, Koutsandreas et al. (2024) found that the addition of atogepant 60 mg once daily to ongoing onabotulinumtoxinA treatment yielded a mean reduction of 6.6 MHDs and a 50% or greater reduction in mean MMDs in 63% of patients over a 12 week treatment period. 35 Recently, the SYNERGY study reported that adding atogepant to ongoing onabotulinumtoxinA therapy in patients with chronic migraine led to a reduction in MMDs of 6.5 days at 24 weeks, with 45.1% of patients achieving a ≥ 50% reduction. 36 Although comparison with the PROGRESS study, 16 a randomized clinical trial in an untreated chronic migraine population, is possible, it should be noted that these two study populations are different since UNCHAINED requires co-treatment with onabotulinumtoxinA and hence baseline MMDS were higher in untreated patients (PROGRESS (18.6)) compared to our treated population (14.3). Collectively, these studies support the benefit of combining onabotulinumtoxinA with atogepant in patients with CM. In our open-label cohort, atogepant addition led to a mean change from baseline in MMDs exceeding 6 days as early as Weeks 1–4 and sustained through Week 24, and more than 50% of participants achieving a ≥ 50% reduction from baseline in MMDs.
Safety results from this study were consistent with previous combination treatment studies, with constipation, nausea, and urinary tract infection as the most common AEs; these were almost uniformly mild or moderate, and only a small number of participants discontinued treatment due to tolerability. Multiple real-world and retrospective chart reviews have demonstrated that CGRP antagonists, when added to onabotulinumtoxinA, have added clinical benefit and no new safety signals for the preventive treatment of CM.18,19,28 Safety profiles from this current study are also consistent with previous open-label extension studies and large randomized studies of atogepant.15,16,29,37 There were no hepatic safety signals or relevant laboratory or ECG changes.
Results should be interpreted in the context of study limitations. The sample size and, the homogenous demographic characteristics of the subject population (97% white and 89% female), and the study's limited regional scope may reduce the power to detect rare AEs or to assess outcomes in subpopulations of individuals with CM, including more racially/ethnically diverse populations or patients with varying histories of preventive treatment failure. The absence of baseline data related to onabotulinumtoxinA response represents a study design constraint. Additionally, the homogenous study cohort was comprised mainly of individuals with long-standing chronic migraine and pre-existing insurance authorization for onabotulinumtoxinA, both variables which may restrict the generalizability of these findings to populations and healthcare systems with varying access limitations. Finally, it should be emphasized that without the inclusion of a control group randomized to placebo vs atogepant, whether the observed reduction in headache burden reflected a synergistic effect from combining therapies or instead was attributable to atogepant alone cannot be determined from this investigation.
Despite these limitations, the trends observed in the study were consistent with those reported in a retrospective chart review and in a single-center investigation that restricted participation to a more racially diverse population of patients who had experienced a partial positive response to onabotulinumtoxinA.18,35 The results of the present study further extend those real-world observations and align with guidance for clinical practice.
This study focused on primary safety, tolerability, and clinical outcomes; future analyses of this study will provide data on clinically meaningful patient reported outcomes. Long-term safety and sustained efficacy beyond 6 months merit future investigation.
In conclusion, our results provide further evidence that atogepant added to onabotulinumtoxinA treatment for the preventive treatment of CM is safe, well tolerated, and provides clinically meaningful benefit for adults with CM. These findings extend prior real-world observations and align with guidance for clinical practice.
Article highlights
Combination preventive treatment for CM with onabotulinumtoxinA and atogepant was safe and generally well-tolerated.
Addition of atogepant to those stable on onabotulinumtoxinA resulted in clinically meaningful reductions in migraine days and improvement in responder rates, suggesting a benefit of combining treatments with distinct and complementary mechanisms of action for suppression of CM.
These findings extend prior real-world observations and align with guidance for clinical practice.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261429118 - Supplemental material for Safety, tolerability, and efficacy of atogepant added to onabotulinumtoxinA for the preventive treatment of chronic migraine: A phase 3, multicenter, 24-week, open-label study
Supplemental material, sj-docx-1-cep-10.1177_03331024261429118 for Safety, tolerability, and efficacy of atogepant added to onabotulinumtoxinA for the preventive treatment of chronic migraine: A phase 3, multicenter, 24-week, open-label study by John Rothrock, Umer Najib, Jessica Ailani, Sait Ashina, Jia Bao, Jonathan H. Smith, Aubrey M. Adams, Brett Dabruzzo, Kimberly Pfleeger and Andrew Blumenfeld in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024261429118 - Supplemental material for Safety, tolerability, and efficacy of atogepant added to onabotulinumtoxinA for the preventive treatment of chronic migraine: A phase 3, multicenter, 24-week, open-label study
Supplemental material, sj-docx-2-cep-10.1177_03331024261429118 for Safety, tolerability, and efficacy of atogepant added to onabotulinumtoxinA for the preventive treatment of chronic migraine: A phase 3, multicenter, 24-week, open-label study by John Rothrock, Umer Najib, Jessica Ailani, Sait Ashina, Jia Bao, Jonathan H. Smith, Aubrey M. Adams, Brett Dabruzzo, Kimberly Pfleeger and Andrew Blumenfeld in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024261429118 - Supplemental material for Safety, tolerability, and efficacy of atogepant added to onabotulinumtoxinA for the preventive treatment of chronic migraine: A phase 3, multicenter, 24-week, open-label study
Supplemental material, sj-docx-3-cep-10.1177_03331024261429118 for Safety, tolerability, and efficacy of atogepant added to onabotulinumtoxinA for the preventive treatment of chronic migraine: A phase 3, multicenter, 24-week, open-label study by John Rothrock, Umer Najib, Jessica Ailani, Sait Ashina, Jia Bao, Jonathan H. Smith, Aubrey M. Adams, Brett Dabruzzo, Kimberly Pfleeger and Andrew Blumenfeld in Cephalalgia
Supplemental Material
sj-docx-4-cep-10.1177_03331024261429118 - Supplemental material for Safety, tolerability, and efficacy of atogepant added to onabotulinumtoxinA for the preventive treatment of chronic migraine: A phase 3, multicenter, 24-week, open-label study
Supplemental material, sj-docx-4-cep-10.1177_03331024261429118 for Safety, tolerability, and efficacy of atogepant added to onabotulinumtoxinA for the preventive treatment of chronic migraine: A phase 3, multicenter, 24-week, open-label study by John Rothrock, Umer Najib, Jessica Ailani, Sait Ashina, Jia Bao, Jonathan H. Smith, Aubrey M. Adams, Brett Dabruzzo, Kimberly Pfleeger and Andrew Blumenfeld in Cephalalgia
Footnotes
Acknowledgements
AbbVie Inc. and authors thank all the study investigators and the patients who participated in this clinical study. Amutha Selvamani, PhD, of AbbVie provided medical writing assistance and Michael Austin, of AbbVie provided editing assistance for the development of this publication.
Author contributions
JFR, UN, JA, SA, AMB, KP, JB, JS, AMA, BD, interpreted the data and contributed to writing the manuscript. All authors read and approved the final manuscript, meeting the ICMJE authorship critria. No honoraria or payments were made to the authors.
Consent to participate
All participants provided informed consent
Consent for publishing
All authors agree to publish the article in Cephalalgia
Data availability statement
AbbVie Inc. is committed to responsible data sharing regarding the clinical studies we sponsor. This includes access to anonymized, individual, and study-level data (analysis data sets), as well as other information (e.g., protocols, clinical study reports, or analysis plans), as long as the studies are not part of an ongoing or planned regulatory submission. This includes requests for clinical study data for unlicensed products and indications.
These clinical study data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, Statistical Analysis Plan (SAP), and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the US and Europe and/or after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit the following link: ![]() then select “Home.”
then select “Home.”
Declaration of conflicting interests
Financial arrangements of the authors with companies whose products may be related to the present report are listed below, as declared by the authors.
Ethical considerations
The study was conducted in accordance with the protocol, Operations Manual, International Council for Harmonisation (ICH) guidelines, applicable regulations, and guidelines governing clinical study conduct and the ethical principles that have their origin in the Declaration of Helsinki.
Funding
AbbVie Inc. funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship.
Open practices
Not applicable.
Supplemental material
Supplemental material for this article is available online.
