Abstract
Background
OnabotulinumtoxinA is widely used for patients with chronic migraine, but its safety and efficacy in pregnancy remain unclear. In 2020, we published a report of 45 subjects who became pregnant on OnabotulinumtoxinA treatment for chronic migraine. Although numbers were small, the results suggested that OnabotulinumtoxinA treatment was effective in pregnancy without evidence of adverse pregnancy outcome. We now have an additional 81 subjects to add to the database.
Method
This is a tertiary centre (Hull) prospective study between 2010–2024. Subjects who became pregnant during treatment with OnabotulinumtoxinA for chronic migraine were recruited. Subjects were provided informed consent whether to continue treatment with OnabotulinumtoxinA. All subjects were followed up 12-weekly with headache and pregnancy outcomes collected.
Results
Between 2010–2024, 126 subjects became pregnant during OnabotulinumtoxinA treatment for chronic migraine. Of these, 97/126 (77.0%) opted to continue treatment with OnabotulinumtoxinA. All 97/97 (100.0%) subjects who continued OnabotulinumtoxinA treatment remained in migraine remission, compared to only 9/29 (31.0%) who discontinued treatment. 2/97 (2.1%) subjects who continued treatment suffered miscarriages whereas 1/29 (3.4%) subjects who discontinued treatment suffered a miscarriage. No foetal malformations were reported.
Conclusion
In our small descriptive study, subjects who continued OnabotulinumtoxinA in pregnancy continued to have migraine remission with no adverse pregnancy outcomes in our cohort.
This is a visual representation of the abstract.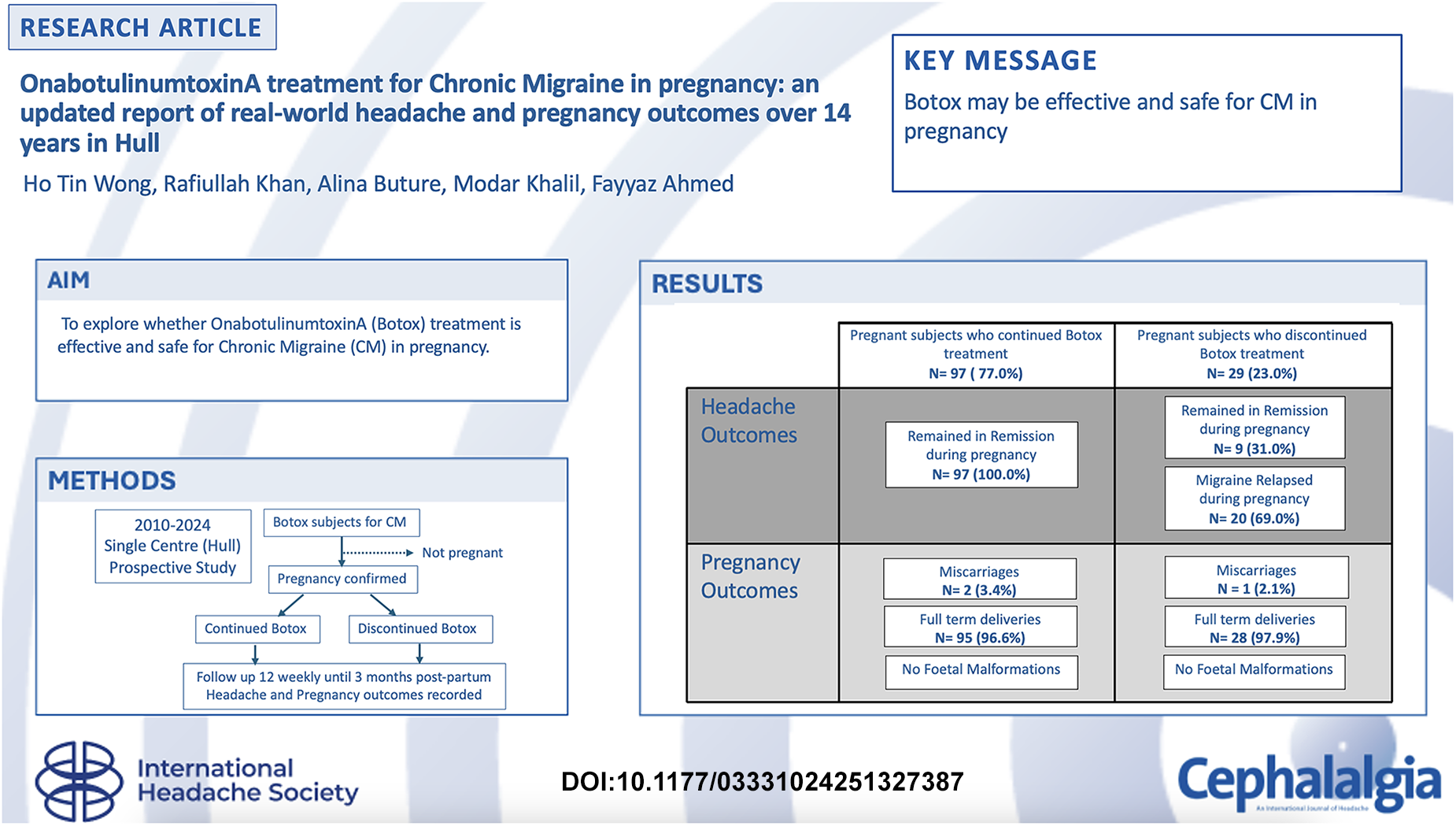
Introduction
OnabotulinumtoxinA treatment is approved for prophylaxis of chronic migraine (CM) in the UK, where three or more oral preventative treatments have been unsuccessfully trialled (1). In more recent years, further prophylactic therapies have become available in the UK, including the CGRP monoclonal antibodies (2–4) and the small molecule CGRP receptor antagonists, also known as Gepants (5,6).
Despite the increasing range of therapies available, the only prophylactic options considered safe in pregnancy remain limited to low dose amitriptyline or propranolol (7). The original PREEMPT trials for OnabotulinumtoxinA treatment in CM did not include any pregnant subjects (8). Due to animal studies citing pregnancy complications and insufficient human data, the official guidance from the UK Medicines and Healthcare products Regulatory Agency (MHRA) and the US Food and Drug Administration (FDA) is that Botulinum Toxin is not recommended in pregnancy (9,10).
Headaches and migraine are common in pregnancy and the post-partum period (11). It is estimated that about half to three quarters of women experience an improvement of their migraine in pregnancy (11). There is, however, a lack of data for migraine outcomes if OnabotulinumtoxinA is discontinued abruptly in pregnancy.
We previously reported our real-world tertiary centre experience (Hull) of OnabotulinumtoxinA treatment for CM in pregnancy (12). Although this was a relatively small cohort of 45 subjects, the results provided an early indication that OnabotulinumtoxinA treatment is effective in pregnancy without adverse pregnancy outcomes. Since the original report in 2020, we have recorded an additional 81 cases of OnabotulinumtoxinA treatment for CM during pregnancy.
We provide an updated total report of 126 subjects who became pregnant on treatment with OnabotulinumtoxinA for CM, with their migraine and pregnancy outcomes.
Method
The methodology for the first 45 cases has been described in our previous report (12). The current study continues the same methodology and extends the recruitment period from 2010 to 2024. This was a prospective study where all subjects who received OnabotulinumtoxinA for CM in a single Tertiary centre (Hull) and became pregnant during treatment were recruited. No subjects were initiated on OnabotulinumtoxinA treatment for the first time after pregnancy was confirmed.
All subjects followed the PREEMPT paradigm (8) for OnabotulinumtoxinA treatment with 12 weekly OnabotulinumtoxinA injections. Women of childbearing potential who received OnabotulinumtoxinA for CM were routinely informed about the uncertain risks on pregnancy and teratogenicity. Contraceptive advice was provided.
This study is a descriptive analysis. Subjects who did not report pregnancy during their treatment were excluded from further analysis. When subjects reported pregnancy during their treatment with OnabotulinumtoxinA, they were offered a further informed discussion about the unknown risks of OnabotulinumtoxinA on pregnancy. Those who reported pregnancy after our previous publication (12) had the results of the study explained to them. All subjects were provided the options to either continue or discontinue OnabotulinumtoxinA. Subjects who opted to continue had to sign a consent form and a disclaimer. All subjects provided written consent for the data collection and access to their medical and pregnancy records.
For headache outcomes, a good response is defined either by the UK National Institute of Health and Care Excellence (NICE) criteria of 30% reduction in headache days or the Hull criteria of 50% reduction in migraine days (13). Those who continued to fulfil this on further follow up are deemed to be in remission. A relapse is defined as the response falling below the NICE criteria of <30% reduction in headache days or <50% reduction in migraine days as per Hull criteria on follow up.
For pregnancy outcomes, ‘full term’ is defined as over 37 weeks gestation. Deliveries were recorded as spontaneous vaginal, assisted, or caesarean section. Caesarean sections may be elective or emergency. A miscarriage is defined as a spontaneous loss of pregnancy before the foetus reaches viability which means from conception until 24 weeks gestation.
All pregnant subjects were followed up 12-weekly with headache diaries irrespective of their decision to continue or discontinue OnabotulinumtoxinA treatment. Subjects who consented to continue OnabotulinumtoxinA treatment continued to receive the same 12 weekly protocol. All pregnant subjects consented for their pregnancy outcomes to be accessed.
All pregnant subjects were reassessed after delivery in the first three months post-partum and were asked to continue headache diaries. Those who suffered from a relapse in their migraine were offered the opportunity to restart OnabotulinumtoxinA (if this was discontinued in pregnancy) or an alternative preventative.
Subjects who became pregnant on OnabotulinumtoxinA treatment more than once were counted as a single case with separate records for each pregnancy and headache outcomes.
Results
There were 126 subjects who became pregnant during treatment with OnabotulinumtoxinA for CM in Hull between 2010 and 2024. The subject demographics are illustrated in Table 1. There were three subjects who became pregnant on more than one occasion on OnabotulinumtoxinA; one subject became pregnant three times and two subjects became pregnant twice on OnabotulinumtoxinA. At confirmation of pregnancy, 80/126 (63.5%) subjects received their most recent treatment with OnabotulinumtoxinA during the first trimester. The remaining 46/126 (36.5%) of subjects had their most recent OnabotulinumtoxinA treatment in the 10 weeks prior to conception.
Subject demographics.

Values are in years unless stated otherwise. Botox, OnabotulinumtoxinA; CM, chronic migraine.
97/126 (77.0%) subjects consented to continue OnabotulinumtoxinA treatment, and 29/126 (23.0%) subjects decided to discontinue. The subject disposition is illustrated in Figure 1. The number of cycles of OnabotulinumtoxinA received before pregnancy is illustrated in Table 2.

A diagram to illustrate the patient disposition in the pregnancy and post-partum periods. Botox = OnabotulinumtoxinA, CGRP = Calcitonin Gene Related Peptide, mAb = Monoclonal antibody
Number of prior treatment cycles with OnabotulinumtoxinA for chronic migraine before pregnancy.

Subjects have been followed up with longest follow-up of 13 years (range 1–13 years).
Headache outcomes
All 97/97 (100.0%) subjects who consented to continue OnabotulinumtoxinA treatment showed a good response, as defined either by the NICE criteria of 30% reduction in headache days or the Hull criteria of 50% reduction in migraine days (13).
In contrast, for those who discontinued treatment, 20/29 (69.0%) subjects relapsed during pregnancy 4–6 months after their last OnabotulinumtoxinA treatment. They were given greater occipital nerve block. 17/20 (85.0%) subjects chose to restart OnabotulinumtoxinA and 3/20 (15.0%) opted to switch to oral preventative after pregnancy.
The remaining 9/29 (31.0%) who discontinued treatment had continued remission of migraine during pregnancy but 6/9 (66.7%) suffered a relapse in the first three months post-partum. Of these post-partum subjects who relapsed, 3/6 (50.0%) recommenced OnabotulinumtoxinA treatment and 3/6 (50.0%) chose a CGRP monoclonal antibody.
Pregnancy outcomes
Of the 97/126 (77.0%) subjects who continued OnabotulinumtoxinA, 2/97 (2.1%) subjects suffered miscarriages. The first case was already described in the previous study (12). This was a miscarriage at 9 weeks and 1 day gestation and had no pre-existing medical problems. The subject then went on to have two further pregnancies continuing with OnabotulinumtoxinA treatment for CM and delivered two full term healthy babies. The second case of miscarriage had a previous history of unexplained miscarriage prior to the current pregnancy with OnabotulinumtoxinA treatment. She went on to have a further pregnancy, on OnabotulinumtoxinA treatment, and delivered at full term.
The remaining 95/97 (97.9%) subjects who continued OnabotulinumtoxinA treatment had full term deliveries. There were 10 forceps assisted deliveries and seven caesarean sections.
Of the 29/126 (23.0%) subjects who discontinued OnabotulinumtoxinA treatment, 1/29 (3.4%) suffered a miscarriage. She had received OnabotulinumtoxinA four weeks before conception. The remaining 28/29 (96.6%) subjects had full term deliveries. There were four forceps assisted deliveries and four caesarean sections. There were no foetal malformations reported in this study.
All caesarean sections in this cohort were elective, and the reasons for them were not available. There were no obvious causes found for the miscarriages reported.
Subject's choice
The subject demographics for those who continued and discontinued OnabotulinumtoxinA treatment are illustrated in Table 1. The majority 80/97 (82.5%) of subjects who chose to continue had one or more children from previous pregnancies while the majority 24/29 (82.8%) who stopped were pregnant for the first time. Thus, subjects were more inclined to stop in their first than the subsequent pregnancies.
Safety data
There were no adverse events reported in both groups. All subjects who stopped treatment and relapsed received greater occipital nerve blocks every three months for the remainder of their pregnancy.
Breastfeeding
80/97 (82.5%) subjects who continued to receive OnabotulinumtoxinA in pregnancy were breastfeeding. 20/29 (69.0%) who discontinued OnabotulinumtoxinA chose to breastfeed, of which 14/20 (70.0%) resumed OnabotulinumtoxinA during breastfeeding. Subjects were asked about the child on every visit until they were a year old to report if there were any adverse outcomes and none were reported.
Discussion
The PREEMPT trials demonstrated the efficacy of OnabotulinumtoxinA treatment in CM (8), but there were no pregnant subjects. Our previous prospective study in Hull with real-life data also supported the effectiveness of OnabotulinumtoxinA in CM, but there were again no pregnant subjects (13).
There is still no official recommendation to use OnabotulinumtoxinA in pregnancy. An updated MRHA summary of product characteristics specifies that Botulinum Toxin type A ‘is not recommended during pregnancy’ (9). The FDA's prescribing information states ‘there are no studies or adequate data from post marketing surveillance on the developmental risk associated with the use of Botox in pregnant women’ (10). These comments are due to insufficient data in human subjects. There are also animal studies in rats and rabbits demonstrating adverse outcomes associated with ‘clinically relevant doses’ in pregnancy, as summarised in the FDA's prescribing information (10). Despite these comments, there is an increasing number of women exposed to OnabotulinumtoxinA during pregnancy reported in the literature.
Previously we reported our real-world experience of 45 subjects over nine years who became pregnant on treatment with OnabotulinumtoxinA for CM (12). We provided an informed discussion regarding continuing or discontinuing treatment. Although the number of cases were small, there were no concerning adverse effects in pregnancy observed in this study. We continued our recruitment over the following five years and now have an additional 81 subjects to add to the database. This is a total of 126 subjects who have become pregnant during OnabotulinumtoxinA treatment for CM at Hull between 2010–2024. There were two subjects who became pregnant on OnabotulinumtoxinA twice and one subject who became pregnant on OnabotulinumtoxinA three times.
Despite the unknown risks of OnabotulinumtoxinA in pregnancy, our real-world experience shows most subjects (97/126, 77.0%) chose to continue treatment after informed discussion. This was because of previous treatment failures and fear of disabling migraine returning upon stopping the treatment. All subjects (97/97, 100%) who chose to continue treatment continued to have good migraine control as defined either by the NICE criteria of 30% reduction in headache days or the Hull criteria of 50% reduction in migraine days (13). In contrast, 20/29 (69.0%) who discontinued OnabotulinumtoxinA suffered a relapse of their migraine control 4–6 months later in pregnancy. For the 20/29 (69.0%) who discontinued OnabotulinumtoxinA and suffered a relapse in their migraine control later in pregnancy, 17/20 (85.0%) decided to restart OnabotulinumtoxinA in the post-partum period. The 6/9 (66.7%) subjects who discontinued OnabotulinumtoxinA and continued to have good migraine control during pregnancy then relapsed in the postpartum period. For these 6/9 (66.7%) subjects, half (3/6, 50.0%) chose to restart OnabotulinumtoxinA in the post-partum period and half (3/6, 50.0%) chose a CGRP monoclonal antibody. Despite an estimated half to three quarters of women reporting an improvement of migraine during pregnancy (11), our data suggests a high risk of migraine relapse (26/29, 90.0%) in pregnancy and in the early post-partum period if OnabotulinumtoxinA treatment is discontinued.
There were no foetal malformations reported in the 126 cases. There were three miscarriages reported, with 2/97 (2.1%) in the group who chose to continue OnabotulinumtoxinA treatment and 1/29 (3.4%) in the group who chose to discontinue treatment. The overall rate of miscarriage here (3/126, 2.4%) is not higher than the range of rates described by NICE for clinically recognised pregnancies, which ranges from 8% to 24% (14).
Since our previous publication in 2020 (12), there have been some updates in literature surrounding OnabotulinumtoxinA in pregnancy. The Allergan Global Safety Database has been updated recently with 29 years of data in total (15), There are 357 reported cases of OnabotulinumtoxinA exposure in pregnancy over 29 years with known pregnancy outcomes. Although this was a large sample size, the population is heterogenous, with location (i.e., indication of treatment) and dosage of OnabotulinumtoxinA treatment varying significantly between subjects. In that study, 103 subjects received OnabotulinumtoxinA for ‘Migraine’ or ‘Headache’ (15). Whilst the database has been generated over 29 years, OnabotulinumtoxinA for CM has only been widely used since the PREEMPT trials in 2010 (8). The rate of major foetal defect amongst live births and the rates of spontaneous foetal loss in OnabotulinumtoxinA treated women were comparable to the general population (15). One major (diaphragmatic hernia) and one minor (talipes) foetal defect in live births was reported in subjects who received OnabotulinumtoxinA for CM in this updated database (15).
Although this updated Allergan Global Safety Database is not directly comparable with our study, it also reports no evidence of adverse pregnancy outcomes with Botulinum Toxin treatment (15). The headache outcomes of OnabotulinumtoxinA in pregnancy were however not reported and it is unclear in that report how many migraine subjects continued treatment in pregnancy. The subjects in our study have not been reported to the Allergan Global Safety Database and so there is no overlap in the subjects included in the two studies.
The main concerns with Botulinum Toxin in pregnancy have been due to the adverse pregnancy outcomes observed in rats and rabbits (10). When pregnant rats and rabbits were administered intramuscular OnabotulinumtoxinA daily during organogenesis there have been reports of reduced birth weight, abortions, early deliveries as well as maternal deaths (10). It has been suggested that the spread of injected Botulinum Toxin may be affected by several variables, including the total dose and concentration of toxin, the size of the needle, number of injections as well as the fluid volume (16). There are significant differences in anatomy, physiology as well as the regime of injections used in these animal studies compared to our pregnant human subjects who received OnabotulinumtoxinA for CM. Separately, in a small sample report of human pregnancy with botulism, there were no cases of neonatal loss or congenital botulism but there were reports of preterm deliveries in six out of 12 cases that had neonatal data (17). The clinical circumstances of botulism are clearly very different to therapeutically administered OnabotulinumtoxinA for CM.
There are some limitations of this study. The subjects who were recruited after our 2020 study (12) had the results of that study explained to them, which may have influenced their decision to continue or discontinue OnabotulinumtoxinA. The study population have refractory CM, having failed or not tolerated at least three oral preventatives, and may therefore be more likely to continue the treatment which has worked well for them. To our knowledge, this is the largest database of OnabotulinumtoxinA treatment for CM in pregnancy, but the size of the cohort is still relatively small. Additionally, this study is only observational and not placebo controlled.
Conclusion
In our small descriptive study, subjects who continued OnabotulinumtoxinA in pregnancy continued to have migraine remission. There is no evidence of adverse effects on human pregnancy outcomes in this cohort. Additionally, our data, albeit small, suggests that discontinuation of OnabotulinumtoxinA for CM in pregnancy carries a high risk of migraine relapse later in pregnancy or the early post-partum period. In our experience, most women chose to continue treatment with OnabotulinumtoxinA even with the uncertain effects on pregnancy explained to them.
Clinical implications
OnabotulinumtoxinA treatment for chronic migraine is effective in pregnancy
There is no evidence of pregnancy related adverse effects of OnabotulinumtoxinA treatment for chronic migraine in pregnancy in this cohort
Abrupt discontinuation of OnabotulinumtoxinA treatment for Chronic Migraine in pregnancy is associated with a high risk of Migraine relapse
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethic approval and patient consent
The study was an observational audit for which no ethics approval was required. All subjects provided written consent for the data collection and access to their medical and pregnancy record. All subjects who continued OnabotulinumtoxinA signed a consent form/disclaimer. They had chosen to continue/stop the treatment after they were informed on the pros and cons of the current evidence.
