Abstract
Background
Atogepant is an oral, small-molecule, calcitonin gene-related peptide receptor antagonist for the preventive treatment of episodic migraine.
Methods
In this 52-week, multicenter, randomized, open-label trial, adults with 4–14 monthly migraine days received atogepant 60 mg once-daily or standard care. Health outcome endpoints collected from participants randomized to atogepant included change from baseline in Migraine-Specific Quality of Life Questionnaire version 2.1 (MSQ v2.1) Role Function-Restrictive (RFR), Role Function-Preventive (RFP) and Emotional Function (EF) domain scores, change in Activity Impairment in Migraine-Diary (AIM-D) Performance of Daily Activities (PDA) and Physical Impairment (PI) domain scores, and change in Headache Impact Test-6 (HIT-6) total score.
Results
Of 744 randomized participants, 521 received atogepant 60 mg in the modified intent-to-treat population. Least-squares mean changes from baseline in MSQ-RFR score were 30.02 (95% confidence interval = 28.16–31.87) at week 12 and 34.70 (95% confidence interval = 32.74–36.66) at week 52. Improvements were also observed in other MSQ domains, AIM-D PDA, PI and HIT-6 total scores. A ≥5-point improvement from baseline in HIT-6 score was observed in 59.9% of participants at week 4 and 80.8% of participants at week 52.
Conclusion
Over 52 weeks, atogepant 60 mg once-daily was associated with sustained improvements in quality of life and reductions in activity impairment and headache impact.
Introduction
Migraine attacks can substantially impact health-related quality of life (HRQoL), including performance of daily activities and both physical and emotional functioning (1–3). The impact of migraine on physical, social and emotional function can be experienced during attacks, as well as in the interictal period (i.e. the time between attacks) (4). Preventive treatment of migraine should not only reduce the frequency and intensity of attacks, but also improve HRQoL and restore the person’s ability to function (5).
Atogepant is an oral, small-molecule, calcitonin gene–related peptide (CGRP) receptor antagonist approved by the US Food and Drug Administration for the preventive treatment of episodic migraine in adults (6). Atogepant is the first oral CGRP receptor antagonist developed specifically for the preventive treatment of migraine (7). The efficacy, safety and tolerability of atogepant for the preventive treatment of migraine have been demonstrated in phase 2b/3 and phase 3 (ADVANCE) placebo-controlled clinical trials (8,9). In the ADVANCE trial, significant and clinically meaningful improvements were observed throughout the 12-week double-blind treatment period in several patient-reported outcomes (PROs).
In a recent long-term trial, atogepant 60 mg once-daily was demonstrated to be safe, well tolerated and efficacious across 52 weeks of open-label treatment (10). This open-label trial also included an assessment of PROs over the 52-week period of atogepant therapy, which is the subject of the present study.
The objective of this analysis was to evaluate PROs in adults receiving once-daily atogepant throughout the 52-week long-term safety trial. Outcomes reported included the Migraine-Specific Quality of Life Questionnaire version 2.1 (MSQ v2.1), the Activity Impairment in Migraine-Diary (AIM-D) and the Headache Impact Test-6 (HIT-6).
Methods
Trial design
Details of this long-term safety trial have been reported previously. The trial was conducted from 8 October 2018 through 29 May 2020 at 111 centers in the USA. Eligible participants were randomized to receive atogepant 60 mg once-daily or standard care (SC) migraine preventive medication in a 5:2 ratio. Randomization was performed with an interactive web response system to minimize bias. Participants randomized to the SC arm were treated with a medication recognized as safe and effective for the preventive treatment of migraine. The study sponsor supplied atogepant 60-mg tablets but did not supply SC medications. The trial consisted of a 4-week screening and baseline period, a 52-week treatment period and a 4-week safety follow-up period (Figure 1). Study visits occurred every 4 weeks during the 52-week treatment period. Efficacy and PRO measurements were collected only from participants who were randomized to the atogepant 60-mg treatment arm. The SC arm was used to contextualize hepatic laboratory data, and no efficacy measures of participants randomized to this arm were assessed. The protocol and all amendments were approved by an institutional review board or independent ethics committee for each study center. All participants provided written informed consent prior to the initiation of any study-related activity.
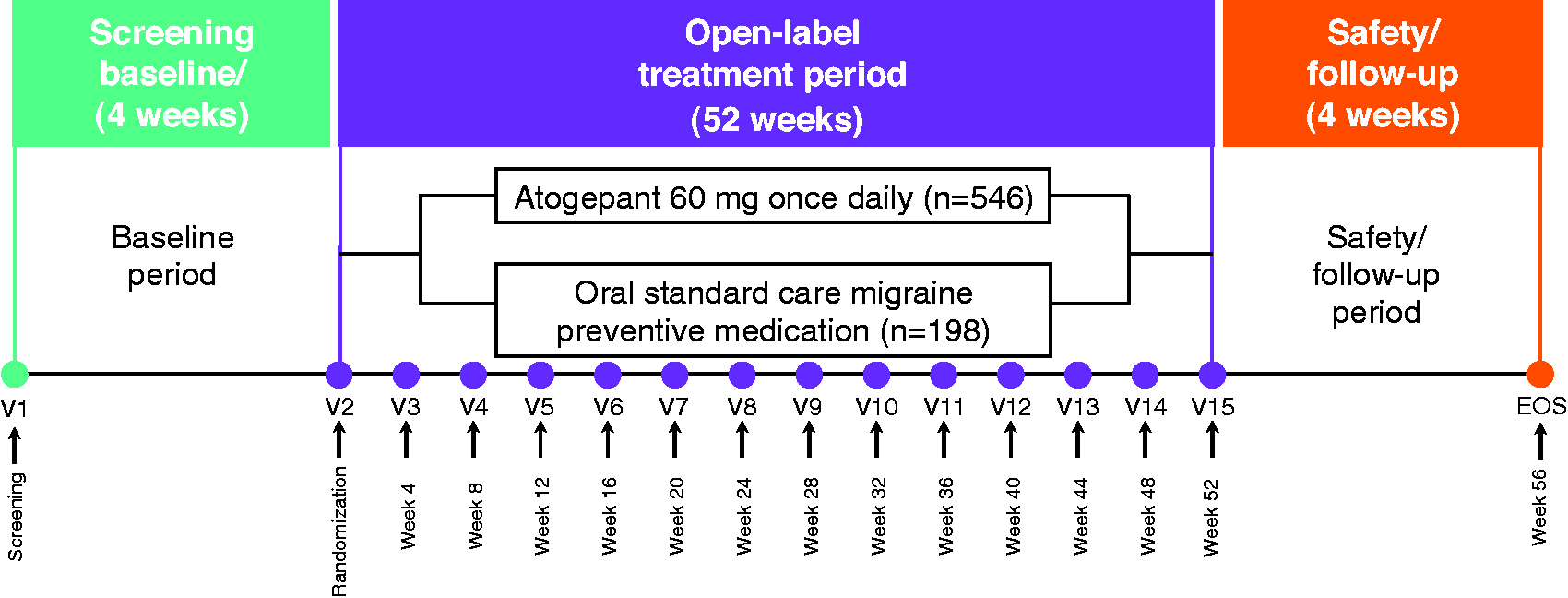
Study design. EOS, end of study; V, visit.
Participants
Eligible participants were adults aged 18–80 years with a ≥1-year history of migraine with or without aura consistent with a diagnosis according to the International Classification of Headache Disorders, 3rd edition (11), migraine onset prior to 50 years of age and 4–14 monthly migraine days on average in the 3 months prior to screening. Participants had either completed a prior phase 2b/3 trial (NCT02848326) of atogepant or were newly enrolled in the current trial. Participants from the prior phase 2b/3 trial were eligible to participate in this trial if they completed the previous trial without significant protocol deviations or an adverse event that may have indicated an unacceptable safety risk. Participants were excluded if they had difficulty distinguishing migraine from other tension-type headaches, had a current diagnosis of chronic migraine or had a mean of 15 or more headache days per month in the previous 3 months.
Outcome measures
MSQ v2.1
Changes from baseline in the MSQ v2.1 Role Function-Restrictive (RFR), Role Function-Preventive (RFP) and Emotional Function (EF) domain scores at weeks 12, 24, 36, 48 and 52 were prespecified endpoints. The MSQ v2.1 was administered via an electronic tablet at visits to the study clinics. The MSQ v2.1 is a 14-item questionnaire that is divided into three domains and measures HRQoL impairments attributed to migraine in the previous 4 weeks (12). The RFR domain consists of seven items that assess limitations imposed by migraine on the performance of an individual’s daily social and work-related activities. The RFP domain consists of four items that assess how migraine attacks interrupt or prevent the performance of daily social and work-related activities. The EF domain consists of three items that assess feelings of frustration and helplessness as a result of migraine. Each MSQ v2.1 domain uses a 6-point scale that is used to respond to the items and ranges from “none of the time” to “all of the time.” Raw domain scores are computed as a sum of item responses and rescaled to a scale of 0–100; higher scores indicate better daily function (13). The MSQ v2.1 has been shown to be a valid and reliable tool for measuring the impact of migraine among people with episodic migraine (12). Within-group minimally important differences (MIDs) for RFR, RFP and EF domains of the MSQ v2.1 have been reported as 5 points, 5–8 points and 8–10 points, respectively; between-group MIDs were 3.2, 4.6, and 7.5 points, respectively (14).
AIM-D
Changes from baseline in monthly AIM-D Performance of Daily Activities (PDA) and Physical Impairment (PI) domain scores were assessed every 4 weeks and were prespecified efficacy endpoints. AIM-D scores were collected daily via an electronic diary (eDiary). The AIM-D is an 11-item daily diary that measures the impact of migraine in 2 domains. The PDA domain includes seven items (difficulty with household chores, errands, leisure activities at home, leisure or social activities outside the home, strenuous physical activities, concentrating, and thinking clearly) and the PI domain includes four items (difficulty walking, moving body, bending forward, and moving head). A 6-point scale ranging from “not difficult at all” to “I could not do it at all” is used to rate the difficulty experienced in the previous 24-hour period. Three items also include a response of “I did not …”; for example, “I did not have errands planned.” Each daily domain score and raw daily total score are transformed to a scale of 0–100; higher scores indicate a greater impact of migraine. The AIM-D has shown robust evidence of validity, reliability and ability to detect meaningful change in people with episodic migraine (15,16). Threshold values for within-group and between-group MIDs for AIM-D domains have not been established.
HIT-6
Participants completed the HIT-6 on an electronic tablet every 4 weeks. Change from baseline in HIT-6 total score and a ≥5-point improvement from baseline in total HIT-6 score were prespecified efficacy endpoints. HIT-6 is a six-item assessment that measures the impact of headaches on normal daily life and the ability to function on the job, at school, at home and in social situations. The 5-point scale used to respond to the items ranges from “never” to “always” and is assigned a score from 6 to 13 points, respectively. Total scores range from 36 to 78, interpreted as little to no impact (≤49), some impact (50–55), substantial impact (56–59) and severe impact (60–78); lower scores indicate improvement (17). HIT-6 has been validated in people with episodic migraine (18). The within-group MID for HIT-6 is a ≥5-point change, and the between-group MID has been reported as −1.5 points in episodic migraine (19).
Statistical analysis
Efficacy analyses were conducted using the modified intent-to-treat (mITT) population, which consisted of all randomized participants who received ≥1 dose of atogepant, had an evaluable baseline period of eDiary data and had ≥1 evaluable post-baseline 4-week period of eDiary data. Changes from baseline for each MSQ v2.1 RFR, RFP and EF domain score, AIM-D PDA and PI score, and HIT-6 total score were analyzed using the mixed-effects model for repeated measures (MMRM). The model included visit as a fixed effect, baseline score and baseline-by-visit interaction as covariates. Restricted maximum likelihood method was used. The within-participant correlation was modeled using the unstructured covariance matrix. The analyses were conducted based on all post-baseline values using only the observed cases without imputation of missing values. Summary statistics were provided for the proportion of HIT-6 responders at each time point. A HIT-6 responder was defined as a participant who achieved a ≥5-point decrease from baseline in HIT-6 total score. Post-hoc sensitivity analyses were completed for MSQ v2.1 RFR, AIM-D PDA and HIT-6 using the MMRM previously described. Additional analyses were also performed on the completer subset, which consisted of participants who had MSQ v2.1, AIM-D and HIT-6 data at every assessment for each respective outcome during the 52-week open-label treatment period.
Results
Participant characteristics
Of 546 participants randomized to receive atogepant 60 mg, 521 were included in the mITT population. A total of 79 of the 546 (14.5%) participants randomized to atogepant 60 mg had completed the prior phase 2b/3 trial, whereas all other participants had no exposure to atogepant prior to this long-term open-label trial. Overall, 173 of the 546 (31.7%) participants randomized to atogepant discontinued the study, most commonly because of subject withdrawal (n = 75; 13.7%), protocol deviation (n = 31; 5.7%) or adverse events (n = 31; 5.7%; of these, 13 participants discontinued because of a serious adverse event). Only five participants (0.9%) discontinued because of lack of efficacy. Demographic and baseline characteristics of the mITT population are shown in Table 1. The mean (SD) age of participants who received atogepant was 42.5 (12.0) years, and most participants were female (88.3%) and White (76.8%). During the baseline period, participants had a mean (SD) of 7.3 (2.6) monthly migraine days and 8.4 (3.0) monthly headache days.
Demographics and baseline characteristics of the mITT population.

Age is relevant to date of informed consent.
N = 516.
AIM-D, Activity Impairment in Migraine-Diary; HIT-6, Headache Impact Test-6; mITT, modified intent-to-treat; MSQ v2.1, Migraine-Specific Quality of Life Questionnaire version 2.1.
MSQ v2.1
Mean (SD) MSQ v2.1 RFR, RFP and EF scores at baseline were 48.1 (20.3), 63.0 (23.4) and 57.1 (28.3), respectively (Table 1). The least-squares (LS) mean change from baseline in all MSQ v2.1 domain scores indicated an improvement at week 12 that was sustained throughout the 52-week treatment period. The LS mean change from baseline in MSQ v2.1 RFR domain score was 30.02 [95% confidence interval (CI) = 28.16–31.87] at week 12 and 34.70 (95% CI = 32.74–36.66) at week 52 (Figure 2a). Similar early and sustained improvements were observed with the MSQ v2.1 RFP and EF domain scores (Figure 2b and 2c). Change scores with 95% CIs excluding zero were present at all post-baseline time points assessed within each MSQ v2.1 domain, indicating a significant improvement throughout the 52-week trial. Similar results were observed in a post-hoc sensitivity analysis of respondents who completed all 52 weeks of open-label treatment (Supplementary Figure 1).

Changes from baseline in Migraine-Specific Quality of Life Questionnaire version 2.1 Role Function-Restrictive (a), Role Function-Preventive (b) and Emotional Function (c) domain scores (mITT population). EF, Emotional Function; LS, least-squares; mITT, modified intent-to-treat; MSQ v2.1, Migraine-Specific Quality of Life Questionnaire version 2.1; RFP, Role Function-Preventive; RFR, Role Function-Restrictive; SE, standard error. n, number of participants with evaluable data at both baseline and a specific time point in the mITT population.
AIM-D
Mean (SD) AIM-D PDA and PI domain scores at baseline were 15.0 (8.8) and 11.4 (8.9), respectively (Table 1). The LS mean changes from baseline in the AIM-D PDA and PI scores demonstrated an early and consistent reduction in impairment as a result of migraine throughout the 52-week trial. The LS mean change from baseline in AIM-D PDA domain score at weeks 1 to 4 was −7.61 (95% CI = −8.26 to −6.96) and, at weeks 49–52, was −10.17 (95% CI = −10.90 to −9.44) (Figure 3a). At weeks 1 to 4, the LS mean change from baseline in AIM-D PI was −5.56 (95% CI = −6.13 to −4.99) and, at weeks 49–52, was −7.20 (95% CI = −7.91 to −6.50) (Figure 3b). The 95% CIs for AIM-D PDA and PI change scores were below zero at all time points assessed, indicating a significant improvement (i.e., reduction) throughout the 52-week trial. The results of the analysis of completers were similar (Supplementary Figure 2).

Changes from baseline in Activity Impairment Migraine-Diary monthly Performance of Daily Activities (a) and Physical Impairment (b) domain scores (mITT population). AIM-D, Activity Impairment in Migraine-Diary; LS, least-squares; mITT, modified intent-to-treat; PDA, Performance of Daily Activities; PI, Physical Impairment; SE, standard error. n, number of participants with evaluable data at both baseline and a specific time point in the mITT population.
HIT-6
The mean (SD) HIT-6 total score at baseline was 64.1 (4.9) for atogepant 60-mg participants, indicating severe impact from migraine (Table 1). The LS mean change from baseline in HIT-6 total scores demonstrated rapid improvement at week 4 (−7.63; 95% CI = −8.31 to −6.95) and showed continued improvement up to 52 weeks (−12.10; 95% CI = −12.98 to −11.22) (Figure 4). Similar results were observed with the 95% CIs of the change scores in HIT-6 total score, indicating a significant reduction in impairment. Similar results were observed in the completer analysis (Supplementary Figure 3). At week 4, 59.9% of participants receiving atogepant 60 mg achieved a ≥5-point improvement from baseline in HIT-6 total score (HIT-6 responder) (Figure 5). The percentage of HIT-6 responders increased with continued atogepant treatment, with 80.8% of participants achieving a HIT-6 response at week 52.
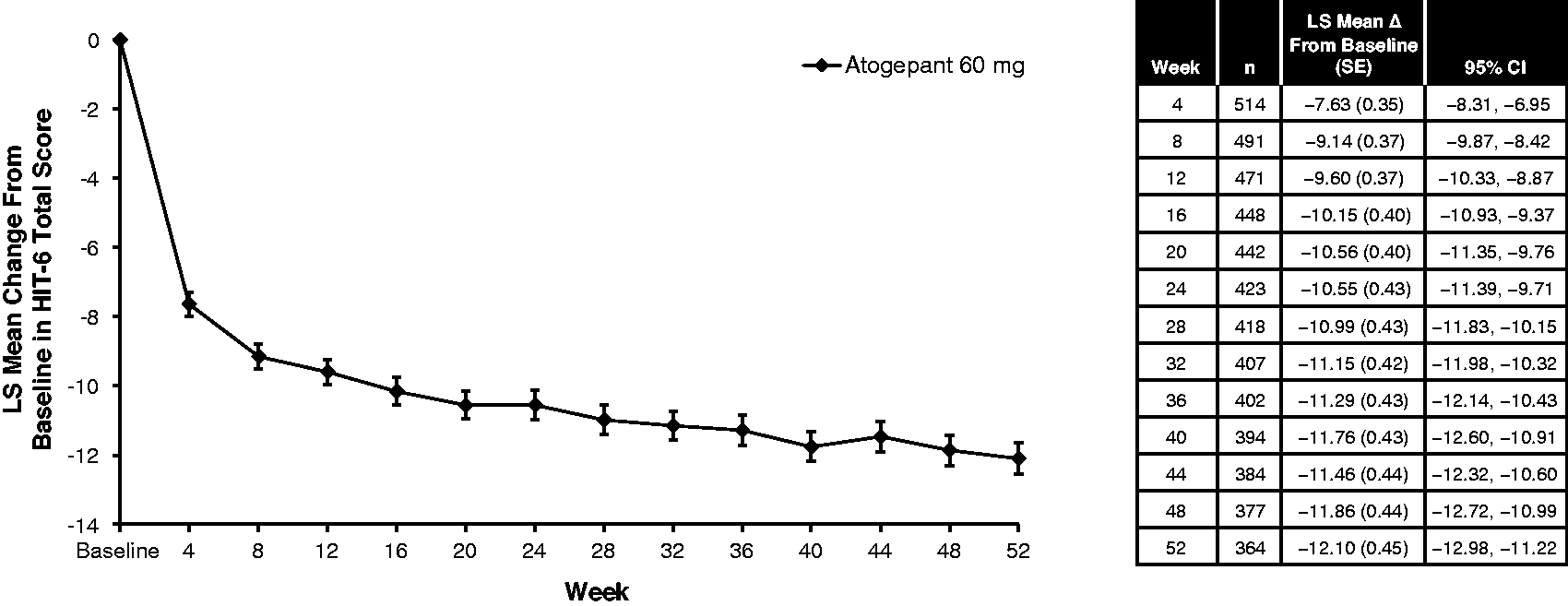
Changes from baseline in Headache Impact Test-6 total score (mITT population). HIT-6, Headache Impact Test-6; LS, least-squares; mITT, modified intent-to-treat; SE, standard error. n, number of participants with evaluable data at both baseline and a specific time point in the mITT population.

Headache Impact Test-6 total score responder rates (mITT population). mITT, modified intent-to-treat. Total (N), number of participants with nonmissing values at the postbaseline analysis visit.
Discussion
The results reported here demonstrate significant and clinically meaningful improvements in PROs with long-term use of atogepant 60 mg once-daily in people with episodic migraine. Improvements in MSQ v2.1 RFR, RFP and EF domain scores were noted at the first post-baseline assessment (12 weeks) and these improvements were maintained throughout the 52 weeks of open-label treatment. Similar early and sustained improvements were observed with AIM-D PDA, AIM-D PI and HIT-6 total scores. The proportion of participants with a clinically meaningful improvement in HIT-6 score (e.g. ≥5-point improvement) steadily increased throughout the trial, with 80.77% of participants achieving a HIT-6 response at 52 weeks (Figure 5).
Primary efficacy results from this open-label trial demonstrated corresponding reductions in the number of monthly migraine days over time in participants receiving atogepant. The proportions of participants achieving a ≥50%, ≥75% and 100% reduction in monthly migraine days from baseline were 60.4%, 37.2% and 20.7%, respectively, within the first 4 weeks of atogepant treatment. At the end of the trial (weeks 49–52), 84.2%, 69.9% and 48.4% of participants achieved a ≥50%, ≥75% and 100% reduction, respectively, in monthly migraine days (10). In the 12-week, randomized, double-blind, placebo-controlled ADVANCE trial, once-daily atogepant 60 mg was associated with statistically significant improvements in MSQ v2.1, HIT-6 and AIM-D scores compared to those of placebo recipients at 4 weeks, and these improvements were maintained throughout the 12-week treatment period. These findings are consistent with those of the ADVANCE trial, which showed early improvement in PROs in the group treated with atogepant 60 mg relative to the placebo group. These results are also consistent with the rapid efficacy of atogepant relative to placebo in the ADVANCE trial; the proportion of participants with migraine declined with active treatment versus placebo on the first full day after treatment initiation (20). Overall, these data further support the beneficial effects of atogepant as an effective treatment for the prevention of migraine.
In previous studies, several preventive treatments have been associated with improvements in daily function and HRQoL in people with migraine (21–24). In addition, several recent studies have shown long-term improvements in function and quality of life with CGRP-targeted preventive treatments in people with migraine (25–27). In an open-label trial of erenumab, improvements from baseline in all MSQ v2.1 domains and HIT-6 scores were maintained throughout 5 years of treatment. A ≥5-point reduction in HIT-6 score was achieved by 66% of participants at week 64 and by 73% of participants at week 268 (25). Similar sustained improvements in HIT-6 total scores were observed throughout a 2-year open-label trial of eptinezumab (26). During 12 months of open-label treatment with galcanezumab, improvements in all MSQ v2.1 domains were observed during the first month and maintained throughout all 12 months. The largest improvement was observed in the RFR domain, with LS mean changes from baseline of −31.6 for galcanezumab 120 mg and −33.4 for galcanezumab 240 mg (mean RFR domain scores at baseline were 47.4 and 47.7, respectively) (27). Altogether, these results suggest that long-term use of an oral or injectable CGRP-targeted preventive treatment for migraine is associated with sustained improvements in function and overall quality of life. Although assessments of function and quality of life are common across clinical trials in the preventive treatment of migraine, comparisons across trials should be made with caution because of differences in enrollment criteria, study designs and methodologies.
Other preventive treatments for migraine, such as galcanezumab, include MSQ v2.1 RFR in labeling (28). Atogepant includes AIM-D PDA and PI as well as MSQ v2.1 RFR in the prescribing information (6). However, the prescribing information for both medications was based on trials with relatively short durations (6,28). The present study used validated PRO instruments to measure changes in functional ability, activity impairment and headache impact over 52 weeks of treatment with atogepant. In addition, the use of the MSQ v2.1 and HIT-6 in headache studies is recommended by the National Institutes of Health (29).
The MSQ v2.1 and HIT-6 have 4-week recall periods, whereas the AIM-D has a same-day recall period (12,18,30). The use of AIM-D PDA and PI allowed for further understanding of the effects of atogepant on daily function and physical impairment. MSQ v2.1, HIT-6 and AIM-D contribute valuable information regarding the impact of functioning, headache disability and performance of daily activities for people with migraine at baseline as well as the patient-centered benefits of treatment over the course of a year. AIM-D evaluates aspects of physical and cognitive functioning using a daily diary and provides clinically relevant information regarding the level of difficulty experienced by people over a 24-hour period. This daily and potentially more precise evaluation of the individual’s experience complements the monthly assessment provided by MSQ v2.1 and HIT-6 (16). This long-term trial provided additional support for the effects of atogepant on PRO measurements, with the completer analysis producing results similar to those of the mITT population. The duration of this trial was 52 weeks, which enabled a long-term assessment of the impact of atogepant on daily functioning and quality of life. Because this was an open-label trial that lacked a placebo group, statistical comparisons are against the pretreatment baseline and there are no comparisons against placebo. Because participants could discontinue the trial at any time, dropouts may preferentially include people with tolerability issues or limited efficacy. Although the trial included an SC treatment arm, efficacy was not assessed and PROs were not collected from the SC group; the SC arm was included to contextualize hepatic safety results. Because only 0.9% of participants discontinued because of lack of efficacy and the overall pattern of treatment effect was similar in the completer analysis, we consider that preferential loss of non-responders is likely a minor factor.
Conclusions
Long-term daily use of atogepant 60 mg for the preventive treatment of migraine improved daily functioning (MSQ v2.1), reduced activity impairment (AIM-D) and reduced headache impact (HIT-6) over 52 weeks of open-label treatment. Improvements in PROs were observed at the earliest time point assessed (4 weeks) and increased over the 52-week trial. These results reinforce the beneficial effects of atogepant as an effective treatment for the prevention of migraine.
Clinical implications
In this long-term safety trial, once-daily atogepant was found to improve daily functioning, reduce activity impairment and reduce headache impact throughout the 52-week treatment period. The sustained improvements in patient-reported outcomes further support the beneficial effects of long-term atogepant use as a preventive treatment for migraine.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231190296 - Supplemental material for Patient-reported migraine-specific quality of life, activity impairment and headache impact with once-daily atogepant for preventive treatment of migraine in a randomized, 52-week trial
Supplemental material, sj-pdf-1-cep-10.1177_03331024231190296 for Patient-reported migraine-specific quality of life, activity impairment and headache impact with once-daily atogepant for preventive treatment of migraine in a randomized, 52-week trial by Richard B. Lipton, Rashmi B. Halker Singh, Laszlo Mechtler, Jennifer McVige, Julia Ma, Sung Yun Yu, Jonathan Stokes, Brett Dabruzzo, Pranav Gandhi and Messoud Ashina in Cephalalgia
Footnotes
Acknowledgements
We wish to express our appreciation to the patients who participated in the study, as well as the clinical and research personnel who were involved with conducting the trial at each of the participating centers. Without their support, we would not be able to further our understanding of migraine and develop new treatments.
Author contributions
PG, RHS and RBL were responsible for the study concept and design. LM was responsible for acquisition of data. PG, RHS and JM were responsible for analysis and interpretation of data. PG and RHS were responsible for drafting the manuscript. All authors were responsible for revising the manuscript for intellectual content. All authors were responsible for final approval of the completed manuscript.
Data availability
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual and trial-level data (analysis data sets), as well as other information (e.g. protocols, clinical study reports, or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications.
These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent scientific research, and will be provided following review and approval of a research proposal and Statistical Analysis Plan (SAP) and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the US and Europe and after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit the following link: ![]() .
.
Declaration of conflicting interests
Richard B. Lipton, MD, serves on the editorial boards of Neurology and Cephalalgia and is a senior advisor for Headache. He has received research support from the National Institutes of Health. He also receives support from the Migraine Research Foundation and the National Headache Foundation. He has reviewed for the National Institute on Aging and National Institute of Neurological Disorders and Stroke; serves as consultant, advisory board member, or has received honoraria or research support from AbbVie, Amgen, Biohaven, Dr. Reddy’s Laboratories, electroCore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Novartis, Teva, Vector and Vedanta Research. He receives royalties from Wolff’s Headache, 8th edition (Oxford University Press, 2009), and Informa. He holds stock options in Biohaven and Manistee.
Rashmi B. Halker Singh, MD, serves on the editorial board of Current Neurology and Neuroscience Reports, serves as deputy editor for Headache, reports honoraria for advisory boards from Impel, Supernus and Teva, and grants for research support from Amgen and Eli Lilly.
Laszlo Mechtler, MD, reports advisory board appointment, speakers bureau participation and/or research support from the following: AbbVie, Amgen, Avanir Pharmaceuticals, Biohaven Pharmaceuticals, Boston Biomedical Inc., CellDex Pharmaceuticals, Delmar Pharmaceuticals, electroCore, Novartis, Orbis Pharmaceuticals, Promius Pharma, Teva Pharmaceuticals and The Harry Dent Family Foundation, Inc.
Jennifer McVige, MD, MA, has received research support from Alder, Allergan/AbbVie, Amgen/Novartis, Avanir, Biohaven, Currax, Eli Lilly, Lundbeck, Teva and Theranica. She serves as a speaker for Allergan/AbbVie, Amgen, Avanir, Biohaven, Eli Lilly, Neurelis, Teva and Theranica.
Julia Ma, PhD; Jonathan Stokes, MBA; Brett Dabruzzo, PhD; and Pranav Gandhi, PhD, are employees of AbbVie and may own AbbVie stock.
Sung Yun Yu, BA, was an employee of AbbVie at time of the study, and may own AbbVie stock.
Messoud Ashina, MD, PhD, DMSc, reports personal fees from AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis, Pfizer, and Teva; has received research funding from Lundbeck Foundation, Novo Nordisk Foundation, and Novartis; and serves as an associate editor for Cephalalgia, an associate editor for The Journal of Headache and Pain, and an associate editor for Brain.
Ethical statement
All participants provided written informed consent prior to the initiation of any study-related activity.
Funding
This study was sponsored by Allergan (prior to its acquisition by AbbVie); they contributed to the study design; to the collection, analysis and interpretation of data; and to the writing, reviewing and approval of the final version. No honoraria or payments were made for authorship. Medical writing support was provided by Cory R. Hussar, PhD, of Peloton Advantage, LLC, an OPEN Health company, Parsippany, NJ, and was funded by AbbVie.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
