Abstract
Background
OnabotulinumtoxinA (onabotA) has shown its efficacy over placebo in chronic migraine (CM), but clinical trials lasted only up to one year.
Objective
The objective of this article is to analyse our experience with onabotA treatment of CM, paying special attention to what happens after one year.
Patients and methods
We reviewed the charts of patients with CM on onabotA. Patients were injected quarterly during the first year but the fifth appointment was delayed to the fourth month to explore the need for further injections.
Results
We treated 132 CM patients (mean age 47 years; 119 women). A total of 108 (81.8%) showed response during the first year. Adverse events, always transient and mild-moderate, were seen in 19 (14.4%) patients during the first year; two showed frontotemporal muscle atrophy after being treated for more than five years. The mean number of treatments was 7.7 (limits 2–29). Among those 108 patients with treatment longer than one year, 49 (45.4%) worsened prior to the next treatment, which obliged us to return to quarterly injections and injections were stopped in 14: in 10 (9.3%) due to a lack of response and in four due to the disappearance of attacks. In responders, after an average of two years of treatment, consumption of any acute medication was reduced by 53% (62.5% in triptan overusers) and emergency visits decreased 61%.
Conclusions
Our results confirm the long-term response to onabotA in three-quarters of CM patients. After one year, lack of response occurs in about one out of 10 patients and injections can be delayed, but not stopped, to four months in around 40% of patients. Except for local muscle atrophy in two cases treated more than five years, adverse events are comparable to those already described in short-term clinical trials.
Introduction
Chronic migraine (CM), that is the presence of headache 15 or more days per month in patients with migraine history, significantly decreases quality of life and determines an unquestionable morbidity in around 2% of the general population (1–3). Treatment of CM is not easy. If analgesic overuse is present, especially due to opiates or ergots, withdrawal is compulsory. Most CM patients also need preventive treatment, but with the exception of topiramate (4,5), there are no serious controlled trials for the remaining preventives used in episodic migraine. A relevant proportion of CM patients do not respond effectively to withdrawal and to any of these preventives, alone or in combination.
OnabotulinumtoxinA (onabotA) was approved in 2010 for the preventive treatment of CM based on results from Phase III Research Evaluating Migraine Prophylaxis Therapy (PREEMPT) trials in North America and Europe, which enrolled 1354 individuals to evaluate its efficacy and safety as headache prophylaxis in CM patients. Patients were treated every 12 weeks for up to one year (6–8). Data regarding durability of benefit beyond one year for the treatment of CM are limited (9). Our goal was to analyse our experience with the use of onabotA in the treatment of patients with CM refractory or intolerant to oral preventives, focusing on the evaluation of its sustained effectiveness after at least one year of treatment in a real-world setting.
Patients and methods
All participants included in this study were adult patients from our Headache Clinic who met the following requirements: 1) fulfil the 2006 criteria for CM with or without analgesic overuse (10); 2) show insufficient response (over a minimum of six weeks), absence of tolerability or contraindications to beta-blockers, topiramate, amitriptyline, flunarizine and valproic acid; and 3) give their informed consent for treatment with onabotA injections. All patients with criteria for analgesic overuse had failed to at least one withdrawal attempt for a minimum of two months. Patients were allowed to continue with preventive oral medication during treatment with onabotA. Exclusion criteria for the use of onabotA were pregnant or breast-feeding women, excessive use of alcohol and serious, active somatic or psychiatric diseases. Patients with comorbidities usually seen in CM, such as anxiety, depression or fibromyalgia or with common vascular risk factors were not excluded. A detailed medical history was available for all patients. Patients kept a conventional headache diary regularly during onabotA treatment and data were compared with those coming during the quarter prior to the first onabotA treatment. During the first year patients were treated with onabotA every three months. All patients received a minimum of two treatments. In the last five years we followed the PREEMPT protocol, that is 155–195 U onabotA U in 31–39 sites. Before 2010, patients received a minimum of 100 U and a maximum of 200 U. A patient was considered as a moderate responder to onabotA when both endpoints were met compared to the data reflected in the diary filled in in the quarter prior to onabotA treatment: 1) according to the diary, moderate-severe headache episodes longer than four hours (or shorter if treated with acute medications) were reduced at least 50%; and 2) a subjective benefit according to a visual scale of 0–100 was also recorded by the patient between 50% and 100%. Patients without improvement of at least 50% in the two items after two treatment cycles were considered as nonresponders and onabotA was stopped. To be considered a responder after one year, these endpoints had to be met in at least three-quarters per year. To analyse the actual need of further onabotA injections after the first four treatments, patients (all responders) were given the next appointment one month later (four months). This onabotA protocol was approved by our ethics committee and all patients signed an informed consent.
Results
Main characteristics of the CM population included in this study.

CM: chronic migraine; NSAIDs: nonsteroidal anti-inflammatory drugs.
The mean number of treatments was 7.7 (median 6, range 2–29). A total of 108 patients received at least four treatments quarterly during one year and one more treatment after four months, 54 at least eight treatments during two years, 32 a minimum of 15 treatments during three years, 12 at least 15 treatments during four years and eight at least 19 treatments for a minimum five years (maximum 8.5 years).
A total of 108 patients (81.8%) responded to onabotA during the first year (Figure 1). Ninety-eight (74.2% of the total series) responded during the second year of treatment. After this period, 49 (50% of those with five or more treatments) reported clear worsening both in frequency and intensity of their headaches after injections were postponed to four months, which obliged us to resume the previous quarter frequency. In 43 (43.9% of those with five or more treatments) out of the remaining 49 patients, onabotA injections were administered every four months as they were still experiencing between three and six migraine attacks per month, but there were six patients with no attacks in the four-month period in whom we decided to wait and see. Two patients still need the injections on demand twice per year as frequent migraine attacks reappear after about six months regularly, and four have not needed more injections as they have had no more than two attacks per quarter after a follow-up of a minimum of 18 months. All responders treated for at least one year kept subjective and objective response to onabotA in at least three-quarters of treatment cycles. There were 12 patients (11.1% of those who received at least five treatments) who showed response during the first year of treatment who did not meet these response criteria after this period: 10 during the second year of treatment, two during the third year and none in those 35 patients treated for more than three years. Variables such as presence versus the absence of overuse of acute medications, aura, fibromyalgia, depression, taking one versus several oral preventives and kind of symptomatic treatment did not significantly influence the rate of response to onabotA. Adverse events during the first year were reported by 19 (14.4%) patients and included palpebral ptosis (seven cases, 5.3%), cervical pain (six, 4.5%), dysphagia (one, 0.8%) and a mixture of these (five, 3.8%). These adverse events lasted between five and 45 days, were considered as mild or moderate and were not a reason to stop onabotA injections. In these patients we either reduced to half or totally avoided the injections in the sites with adverse events (neck muscles in 12 cases and corrugator in eight cases). In addition, two (25%) of the eight patients treated more than five years showed local muscle atrophy in the frontotemporal region, but both had not expressed any complaint on this regard.
Summary of our long-term results with onabotA.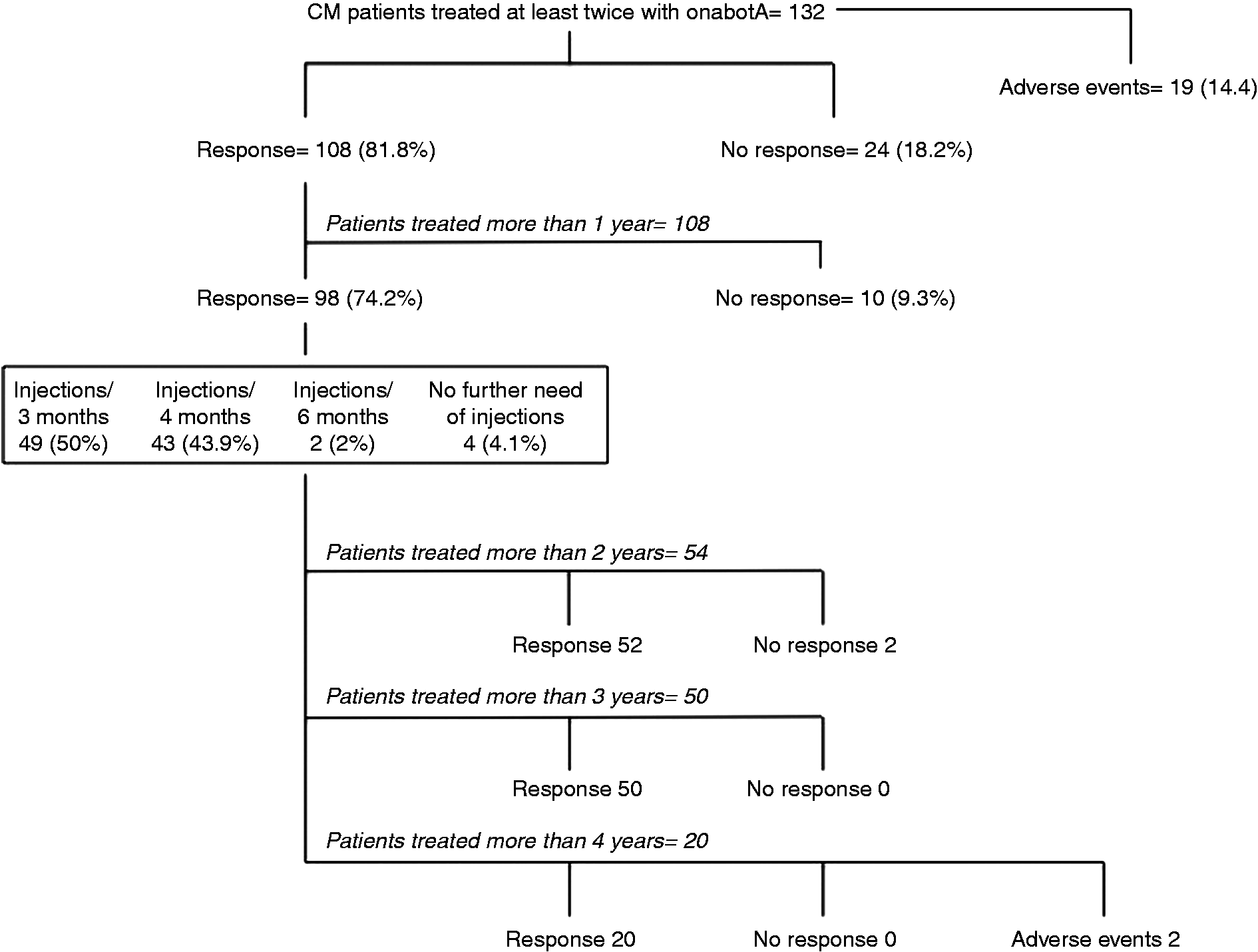
The consumption of any kind of analgesics in responders went from an average of 17 per month in the diary filled in immediately before the first treatment with onabotA to eight per month (−53%) in the most recent quarter. The group of 38 patients who were specifically overusing triptans before receiving onabotA injections went from an average of 16 intakes (either tablets, nasal spray or sumatriptan injections) per month in the diary filled in the quarter before the first treatment to six doses per month (−62.5%) in the last quarter. In responders, the number of visits to casualty departments (either their health centre or emergency services) for parenteral treatment of severe migraine attack fell from an average of 2.3 the quarter before the first onabotA treatment to 0.9 in the diary of the last quarter (−61%).
Discussion
Our data, showing that around 80% of CM patients respond to pericranial injections of onabotA during the first year of treatment, concur with those coming from PREEMPT trials (6–8) and also from short-term open trials (11–13). The main objective of this work, however, was to analyse our long-term experience with onabotA. To our knowledge, this is the biggest series analysing the clinical effectiveness of onabotA after one year. Our results can be of help for clinicians when making their decisions regarding what to do with this treatment after the PREEMPT period and confirm that seven out of 10 CM patients maintain a sustained response to onabotA for years. In fact, onabotA lost its clinical response after one year in 9.3% of patients. This secondary failure was seen in most cases in the second year of treatment as none of the 20 patients treated for more than three years lost their response to onabotA. There are several potential explanations for this loss of response after one year, such as an initial placebo response, an intrinsic worsening of migraine or a theoretical development of antibodies. As in previous studies, we found no correlation among a number of clinical variables, such as overuse, aura, depression, fibromyalgia or kind of medications used for the treatment of migraine, with the presence or absence of response (6–8).
We tried to analyse the real need for further onabotA injections after one year by routinely delaying the fifth treatment to the fourth month. There were just four CM patients in whom onabotA injections could be stopped due to a dramatic relief of migraine attacks, two patients in whom injections were needed just twice per year and 43 (43.9% of those who received at least five treatments) in whom onabotA could be postponed to every four months. However, from our experience it seems that around 90% of patients need to continue with onabotA injections after the first year of treatment to keep migraine frequency under reasonable control and that in at least half of the cases these injections are needed quarterly as patients clearly worsen after this period of time. This confirms the chronic nature of CM, at least for the worst patients attending specialised headache clinics who need prolonged preventive treatments.
Adverse events – both in profile and rate – concurred with those observed in PREEMPT clinical trials; they were easily explained by the local effects of the toxin and did not reappear just by avoiding injections or by reducing to half the dose in these sites (mostly corrugator and cervical muscles) in future treatments. In none of our patients were adverse events the reason for treatment discontinuation. There were two patients treated for more than five years in whom atrophy of both temporal and frontal muscles was evident, though both patients had made no complaint, neither aesthetic nor in terms of weakness, and we decided to continue with onabotA injections every four months.
Previous data suggest that the effect of onabotA in CM patients lies especially in a downward regulation of severe migraine attacks, which would then be less invalidating and easier to deal with (12). This is endorsed by two of our results measured after an average period of treatment of two years: the reduction by 60% both in visits to emergency for parenteral treatment and in triptan consumption in triptan overusers. The latter already make this treatment approach cost effective in responders in a condition such as CM, which costs an average of 2700 euros per patient per year in Spain (14). Our results show again that there can be a clinical response to onabotA regardless of the presence of analgesic overuse (15). The long-term response of onabotA in this study, however, would be valid only for patients without medication-overuse headache according to the 2006 criteria (10), as all our patients still meeting overuse criteria had tried withdrawal at least once for a minimum of two months.
Limitations of our work are obvious. First, it comes from a single headache centre which specialises in the management of difficult cases; consequently our experience cannot necessarily be extrapolated to the reality of other clinics. Second, these results must be interpreted with great caution as our observational study did not have a control group. Finally, the number of patients studied here and their follow-up period are still low and should be extended in the future. Therefore, multicentric long-term studies are necessary to extrapolate evidence-based recommendations to routine clinical practice. With these limitations in mind, our results confirm the long-term response to onabotA injections in three quarters of cases in clinical practice, which is maintained by almost 90% of patients for periods longer than two years, and the need for keeping injections every three or, less frequently, four months after the one-year PREEMPT period in most CM patients.
Clinical implications
In clinical practice, long-term response of onabotulinumtoxinA (onabotA) injections is kept in three-quarters of chronic migraine (CM) patients. Only in around one out of 10 patients did onabotA injections lose their clinical efficacy after more than one year of treatment. There was no failure after the third year of treatment. In about 40% of cases, onabotA injections can be delayed to every four months after the first year of treatment. Except for asymptomatic local frontotemporal muscle atrophy after at least five years of use, the long-term rate and profile of adverse events were comparable to those observed in controlled clinical trials.
Footnotes
Author contributions
Authors responsible for conception and design (J.P.), acquisition of clinical data (J.P., C.R., D.L., R.A., N.R.) and analysis and interpretation of data (E.C., J.P.), drafting the manuscript (J.P.), obtaining funding (E.C., J.P.) and supervision (J.P.).
Funding
This study was supported by the PI11/00889 FISSS grant (Fondos Feder, ISCIII, Ministry of Economy, Spain).
Conflicts of interest
J. Pascual reports consultancy for Allergan. The rest of the authors have nothing to declare.
