Abstract
Aim
To evaluate the effect of treatment onset-time on the effectiveness of remote electrical neuromodulation (REN) for acute treatment of migraine.
Methods
A real-world evidence study on migraine patients who treated with REN . REN treatments initiated within one hour of migraine attack onset (headache or aura) were classified as “early”; those initiated after one hour were classified as “late”. Treatments with baseline and two-hour reports were termed “evaluable” and analyzed.
Results
Among 55,261 patients (37.9 ± 18.5 years, 83.4% female) who conducted 586,981 treatments, 56.5% were administered early. Effectiveness was calculated from “evaluable” treatments, varying between 6413 and 35,581 treatments depending on the outcome. Early treatments yielded higher responder-rates than late (p < 0.001, significant following Bonferroni correction for multiple comparisons) for pain relief (65.1% vs. 46.6%; Δ = 18.5%), pain freedom (28.8% vs. 14.5%; Δ = 14.3%), functional disability relief (58.1% vs. 49.3%; Δ = 8.8%), functional disability freedom (35.4% vs. 20.9%; Δ = 14.5%), and freedom from photophobia (26.9% vs. 19.0%; Δ = 7.9%), phonophobia (34.0% vs. 25.9%; Δ = 8.1%) and nausea/vomiting (51.5% vs. 38.7%; Δ = 12.8%). Similarly, patients consistently treating early with REN (in 50% or more of their treatments) experienced higher efficacy (p < 0.001). Similar effects were seen in youths.
Conclusions
Early acute treatment with REN enhanced patient outcomes by up to two-fold compared to late treatment onset. Educating providers and patients to “treat as early as possible” boosts clinical and patient-centered results.
This is a visual representation of the abstract.
Introduction
Migraine is a prevalent and debilitating neurological disease affecting an estimated one billion people worldwide (1, 2). Characterized by recurrent attacks of moderate to severe headache and disabling symptoms, migraine is ranked as the 2nd most disabling medical illness (GBD, 2016) (3), significantly impacting an individual's quality of life, productivity and overall well-being (2). Early administration of acute therapies can improve clinical outcomes and prevent headache escalation, an effect well-established for pharmacological interventions (4–6). This concept is supported by the pathophysiology of migraine which involves a complex cascade of events that, if left untreated, lead to a fully developed attack (7,8). Guidelines highlight the benefits of initiating early treatment of migraine attacks to disrupt this cascade and prevent the attack from reaching its full intensity (9,10). Inadequate and untimely acute treatment is often associated with migraine chronification, disease progression, and medication over-use headache (11).
Several neuromodulation devices offering drug-free migraine treatment are available. Remote electrical neuromodulation (REN) is a wearable device cleared by the US Food and Drug Administration (FDA) for acute and/or preventive migraine treatment in episodic and chronic migraine with or without aura, in ages 8 years and older. REN modulates nociceptive signals through peripheral nerve stimulation in the upper arm activating an endogenous pain–modulating pathway known as conditioned pain modulation (CPM) (13). REN has been found safe and effective through multiple clinical trials (14–17) and real–word evidence studies (18–20). A systematic review and metaanalysis of randomized controlled trials focusing on migraine treatment found the wearable REN to be effective (12). The objective of this study is to compare REN efficacy when administered “early” (within one hour of attack onset) to its efficacy when used “late” (more than one hour or after attack onset), with the hypothesis is that early onset will lead to higher acute efficacy than late treatment.
Methods
Study design and setting
This prospective, real-world evidence study evaluated the effect of REN treatment onset time on efficacy outcome measures, comparing early versus late treatments. Data were collected from US-based REN users between January 2019 to December 2024.
Participants and data collection
Participants were REN users who treated their migraine attacks with the device and reported treatment onset time in at least one treatment through the REN companion smartphone app (Nerivio®, Theranica). The study was reviewed and approved by the WIRB-Copernicus Group (WCG IRB tracking number 20251626). During app registration, patients consented to the collection of de-identified data for research purposes and provided demographic information. Participants could voluntarily report baseline characteristics once they initiated a REN treatment, including treatment onset time relative to attack onset, headache pain, functional disability, and presence/absence of migraine-associated symptoms (photophobia, phonophobia and nausea/vomiting), as well as treatment outcomes two-hour post-treatment. All data were stored on a Health Insurance Portability and Accountability Act (HIPAA) compliant secure server.
Study device
The REN wearable device is an FDA-cleared, drug-free, non-invasive, prescribed therapy indicated for the acute and/or preventive treatment of migraine in patients aged 8 years and older. REN users control the treatment and its intensity through a smartphone application and can perform daily activities during the 45-minute treatment session.
Time-from-onset classification
Treatments were classified as “early treatments” if REN was administered within one hour from migraine onset and classified as “late treatments” if administered more than one hour after onset. On a patient level, a patient was defined as an “early-treater” if they reported treatment onset in at least two treatments and had treated early in 50% or more of treatments. A patient was defined as a “late-treater” if they reported treatment onset in at least two treatments and treated late in more than 50% of treatments.
Efficacy outcomes
Seven prospective acute treatment efficacy outcomes were assessed:
These outcomes were evaluated both at treatment level and at patient level
“
Given the different definitions of efficacy outcomes, the number of “evaluable treatments” may vary between outcomes.
Statistical analysis
Treatments were pooled according to treatment onset time (early vs. late) for treatment-level comparisons of each efficacy outcome. Patient-level analyses compared consistent efficacy of “early-treaters” versus “late-treaters”. Continuous variables were summarized using means and standard deviations, and compared between subgroups using independent-samples t-tests. Categorical variables were summarized using frequencies and percentages, and statistical significance was assessed using chi-squared tests. To account for multiple comparisons across the seven efficacy outcomes (pain relief, pain freedom, functional disability relief, functional disability freedom, freedom from photophobia, freedom from phonophobia, and freedom from nausea/vomiting), a Bonferroni correction was applied, resulting in an adjusted significance threshold of p < 0.007 (0.05/7). Sensitivity analysis was conducted, assigning patients to “early” and “late” treaters based on their reported time-from-onset and efficacy in at least 67% of their treatments.
Results
Demographics and utilization
In total, 55,261 patients (83.4% female) with a mean ± SD age of 37.9 ± 18.5 years were included in this analysis. These patients reported the time from onset in 586,981 treatments. Of the total treatments, 56.5% (n = 331,760) were administered “early” (within one hour from attack onset).
Treatment-level efficacy
The number of treatments meeting the criteria for efficacy analysis varied between 6413 and 35,581 for the different efficacy outcomes (see Methods) (Table 1). Early administration of REN yielded higher efficacy rates for all outcome measures (p < 0.001, statistically significant following Bonferroni correction for multiple comparisons): Pain relief was reported in 65.1% of early treatments compared to 46.6% of late treatments (Δ = 18.5). Pain freedom was reported in 28.8% of early treatments versus 14.5% of late treatments (Δ = 14.3%). Functional disability relief and functional disability freedom were higher in early treatments: 58.1% versus 49.3% functional disability relief (Δ = 8.8%) and 35.4% versus 20.9% functional disability freedom (14.5%) (Figure 1a). Freedom from migraine-associated symptoms that were present at treatment onset was significantly higher in early treatments, with 26.9% versus 19.0% freedom from photophobia (Δ = 7.9%), 34.0% versus 25.9% freedom from phonophobia (Δ = 8.1%) and 51.5% versus 38.7% freedom from nausea/vomiting (Δ = 12.8%) (Figure 1b and Table 1).
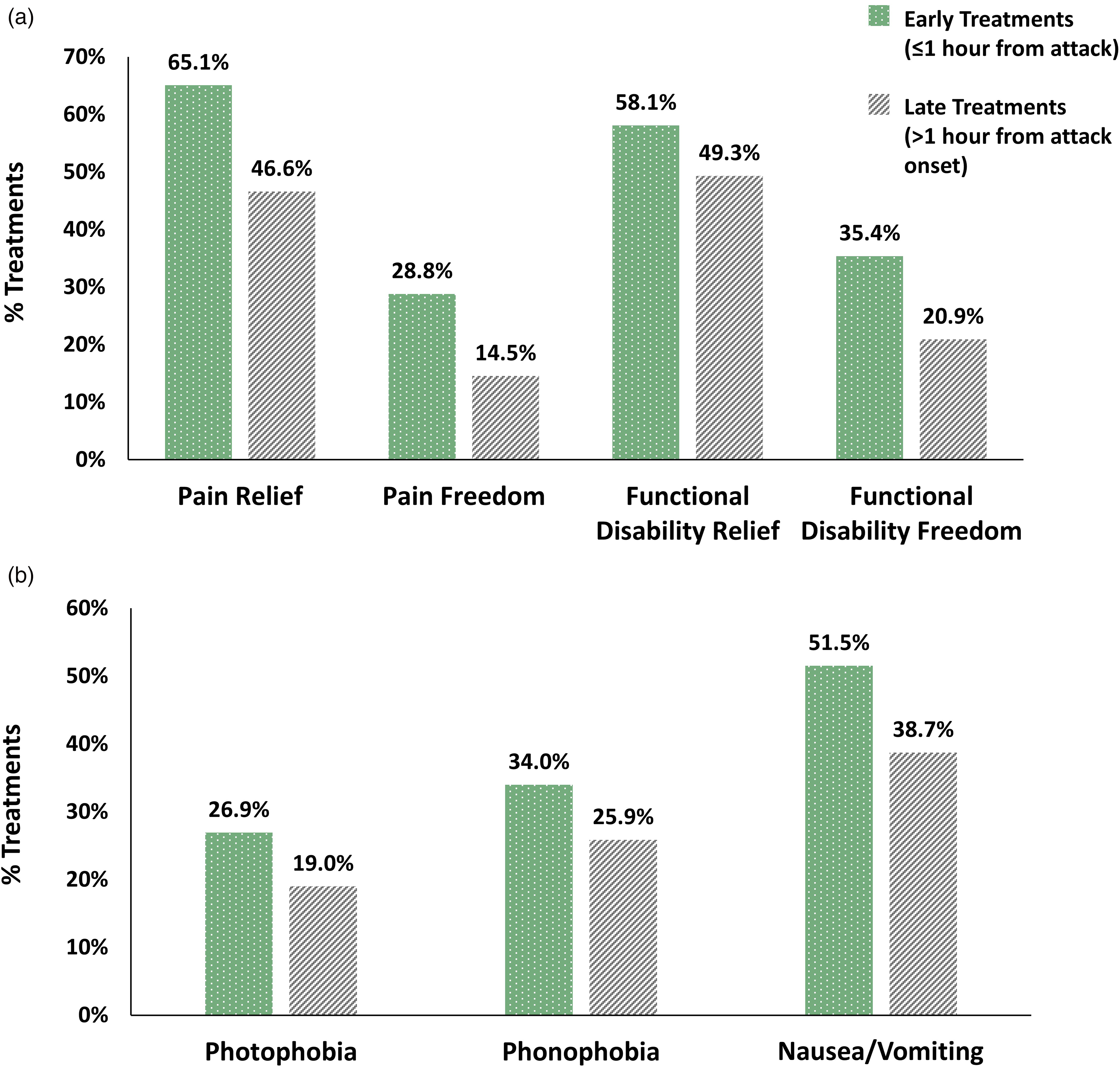
Treatment-level efficacy outcomes. (a) Pain and functional disability relief and freedom rates are higher when remote electrical neuromodulation (REN) is initiated “early” compared to “late”. (b) Symptom freedom rates are higher when REN is initiated early compared to “late”. All efficacy outcome measures are significant (p < 0.001). Early treatments (one hour or less from attack onset); late treatments (more than one hour from attack onset).
Comparing “early” initiation of remote electrical neuromodulation (REN) acute treatment (within one hour of attack onset) versus “late” treatment initiation (after one hour from attack onset), at “treatment level” and at “patient level”.

Demographic data are presented for each level of analysis.
Patient-level efficacy
Participants who administered 50% or more of their treatments early (“early treaters”) were slightly older (38.5 ± 16.3 vs. 36.0 ± 22.3; p < 0.001) and included more females (% of females: 85.4% vs. 84.1%; p < 0.001) than those who administered early treatments in less than 50% of their treatments (“late treaters”). The number of participants meeting the criteria for efficacy analysis varied between 761 and 7187 for the different efficacy outcomes (see Methods) (Table 1). “Early treaters” experienced higher success rates compared to “late treaters” in all outcome measures (p < 0.001): Pain relief was reported in 73.9% of early-treaters compared to 53.6% of late-treaters (Δ = 20.3%). Pain freedom was reported in 30.0% of early-treaters compared to 16.6% of late-treaters (Δ = 13.4%). Functional disability relief was reported in 69.4% of early-treaters versus 54.2% of late-treaters (Δ = 15.2%) and functional disability freedom was reported in 38.5% of early-treaters versus 24.2% of late-treaters (Δ = 14.3%) (Figure 2a). Freedom from migraine-associated symptoms was reported in more early-treaters than late-treaters; 33.5% versus 22.4% freedom from photophobia (Δ = 11.2%), 44.9% versus 32.3% freedom from phonophobia (Δ = 12.6%) and 62.4% versus 51.0% freedom from nausea/vomiting (Δ = 11.3%) (Figure 2b and Table 1). Sensitivity analysis in participants who administered 67% or more of their treatments early compared to those who administered 67% or more of their treatments later yielded similar results of higher response rates (p < 0.001).
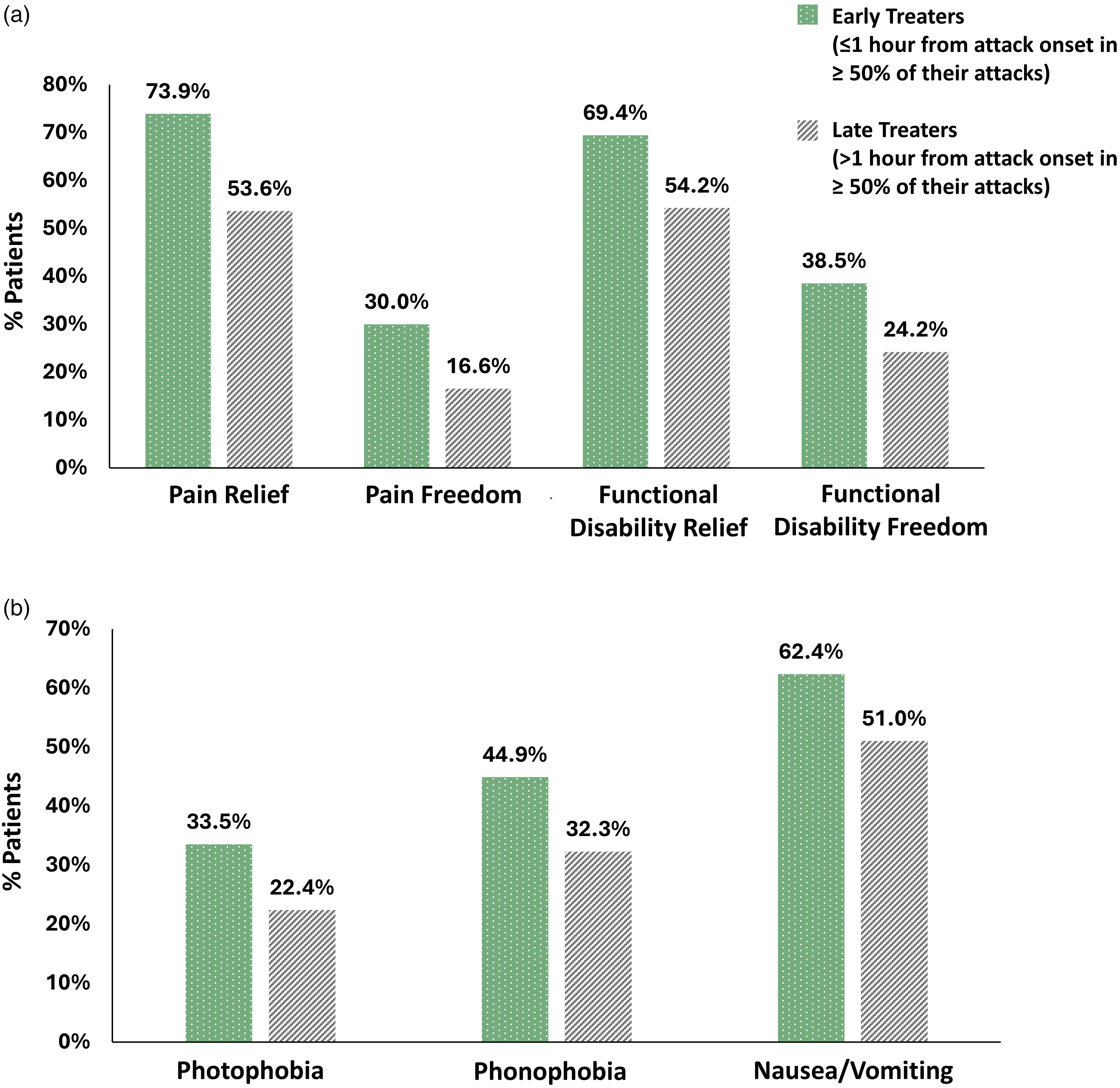
Patient-level efficacy outcomes. (a) Pain and functional disability relief and freedom rates are higher for remote electrical neuromodulation (REN) “early-treaters”. (b) Symptom freedom rates are higher for REN “early-treaters”. All efficacy outcome measures are significant (p < 0.001). “Early-treaters” (treated at one hour or less in ≥50% of attacks); “late-treaters” (treated after more than one hour in ≥50% of attacks).
Youth sub-analysis
A sub-analysis of youth patients (aged <18 years) showed that 8,886 youths (mean age 15.8 ± 1.4 years; 84.2% female) performed 82,040 treatments, of which 47.9% were administered early. Treatment-level response rates significantly improved across all treatment endpoints when treatments were initiated early (p < 0.001): pain relief was 64.4% versus 41.0% (Δ = 23.3%), pain freedom was 26.4% versus 11.2% (Δ = 15.2%), functional disability relief was 62.1% versus 46.2% (Δ = 15.9%) and functional disability freedom was 40.9% versus 21.5% (Δ = 19.4%), freedom from photophobia was 21.7% versus 15.1% (Δ = 6.5%), freedom from phonophobia was 29.8% versus 22.9% (Δ = 6.9%), and freedom from nausea/vomiting was 39.6% versus 33.6% (Δ = 6.0%). Similar effects were seen in patient-level analysis (p < 0.001).
Discussion
The findings from this large real-world study demonstrate that early treatment with REN (within 1 hour from migraine attack onset) improves acute treatment outcomes in a significant and clinically meaningful manner. Early intervention advantage persists for all efficacy measures, including pain relief, pain freedom, functional disability relief, functional disability freedom, ,and freedom from associated migraine symptoms of photophobia, phonophobia and nausea/vomiting. While positive outcomes were still observed for late administration, early intervention increased efficacy by up to 20.4% and as much as doubled (1.2 to 2.0-fold) the likelihood of reaching freedom or relief during a migraine attack, aligning with the broader principle that rapid treatment can halt migraine progression more effectively (4,6).
These results underline the importance of educating patients on using REN promptly at migraine onset. As the indication for REN states administering treatment “at the onset of migraine headache or aura”, patients should be advised to begin treatment “as early as possible” even as early as this symptom ensues, or even earlier at the onset of prodrome symptoms, yet this warrants some further study. Timely treatment may offer a key strategy to optimizing clinical results and reducing the burden of migraine, including reduction of quality of life, medication over-use headache, and disease chronification (5,21). Moreover, timely treatment is expected to increase treatment adherence as patients who treat early and experience clinical benefits are more likely to continue treatment.
The study has a few limitations, including a reliance on self-reported data of pain severity and treatment initiation time from attack onset. Yet migraine symptoms, by nature, are subjective and self-reported. Another limitation is that two-hour post-treatment data were not available for the majority of treatments, limiting efficacy analyses to approximately 10% of treatments and patients. However, the fact that baseline and post-treatment reports were voluntary provided by patients treating with the device as part of routine medical care, rather than within a structured clinical trial, offers insights into real-world treatment patterns and outcomes. While these data may be subject to reporting biases, the inclusion of a large number of treatments (6413 to 35,581) and patients (761 to 7187) in the efficacy analyses, strengthens the statistical power of the study and supports the generalizability of the findings.
Future research should explore the barriers that deter patients from applying REN early and develop targeted interventions to educate and encourage prompt REN treatment.
Clinical implications
This large real-world cohort demonstrates that early REN treatment (within one hour of attack onset) improves acute treatment outcomes by up to 20% compared to late treatment.
Higher rates of freedom from pain and functional disability from early REN treatment are increased in youth compared to adults.
Clinicians should instruct patients to “treat as early as possible”.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jessica Ailani: Consulting (Honoraria): Abbvie, Aeon, Axsome, Amneal, Dr Reddy, Eli-Lilly, Lundbeck, Linpharma, Ipsen, Merz, Pfizer, Gore, Satsuma, Vectura Fertin; Clinical Trials (Grant to institution): Parema (2024), Ipsen, Lundbeck, Pfizer, Merz; Clinical Trials (Grants to PI): Mi-Helper; Editorial Boards/Steering Committee: SELF magazine (medical editor). Daniel Noam Lax: Sub-Investigator for trial by Biohaven, honoraria from American Headache Society and MedLink Neurology for CME activities, advisory board for Theranica. Istvan Tomaschek: None. Alit Stark-Inbar: Theranica employee. Sharon Shmuely: Theranica employee. Alon Ironi: Employee and stockholder at Theranica. The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
The study was reviewed and approved by WCG IRB (tracking number 20251626).
Funding
This work was supported by Theranica .
