Abstract
Background
Sustained pain freedom is an important attribute of acute migraine therapies for patients and physicians. Here we report efficacy of the centrally penetrant, highly selective, 5-HT1F agonist lasmiditan on sustained pain freedom and other outcomes at 24 and 48 hours post-dose.
Study design and methods
Data from the similarly designed, Phase 3, double-blind studies SAMURAI (NCT02439320) and SPARTAN (NCT02605174) were pooled to more precisely estimate efficacy effects in these post-hoc analyses. In both studies, inclusion criteria were 3–8 migraine attacks per month and Migraine Disability Assessment Score of ≥ 11 (at least moderate disability). Patients were randomized equally to lasmiditan 200 mg, 100 mg, 50 mg (50 mg only in SPARTAN), or to placebo. The study drug was to be taken within 4 hours of onset of pain for non-improving headache of at least moderate severity. Sustained pain freedom was defined as being pain free at 2 hours and at the given time point (24 or 48 hours) post-dose without use of additional study drug or migraine medications. Sustained responses were assessed similarly for most bothersome symptom-free, total migraine-free, and disability-free outcomes. For comparisons with previously published data on other acute medications, an additional endpoint of modified sustained pain freedom at 24 hours was defined as being pain free at 2 hours and no moderate-to-severe headache at 24 hours post-dose without use of additional study drug or migraine medications.
Results
Significantly higher proportions of patients treated with lasmiditan versus placebo achieved headache pain freedom at 2 hours post-dose: 200 mg: 35.6%; 100 mg: 29.9%; 50 mg: 28.6%; placebo: 18.3% (all p < 0.001). Sustained pain freedom was significantly higher in patients treated with lasmiditan versus placebo at 24 hours: 200 mg: 21.2%; 100 mg: 16.9%; 50 mg: 17.4%; placebo: 10.3% (all p < 0.01); and at 48 hours: 200 mg: 18.4%; 100 mg: 15.2%; 50 mg: 14.9%; placebo: 9.6% (all p < 0.05). Similar sustained benefits of lasmiditan versus placebo at 24 and 48 hours were noted for most bothersome symptom-free, total migraine-free and disability-free responses. Modified sustained pain freedom at 24 hours was also observed in significantly higher proportions of lasmiditan-treated patients versus placebo: 200 mg: 27.0%; 100 mg: 21.7%; 50 mg: 21.7%; placebo: 12.9% (all p < 0.01).
Conclusion
Sustained responses at 24 and 48 hours were noted in significantly more patients treated with lasmiditan versus placebo for several efficacy outcomes including pain freedom, most bothersome symptom-free, total migraine-free and disability-free responses.
Clinicaltrials.gov identifier numbers
SAMURAI: NCT02439320; SPARTAN: NCT02605174.
Introduction
Migraine, a debilitating chronic intermittent neurologic disease, was ranked second for years lost to disability in the most recent Global Burden of Disease study (1). Patients with migraine suffer from attacks of moderate-to-severe head pain that are often accompanied by symptoms such as photophobia, phonophobia and nausea (2). The management of acute migraine attacks is complex. While several acute treatments for migraine are available, they do not meet the needs of all patients. In a survey of > 5000 patients with migraine, 40.7% identified with at least one of five defined unmet need categories (3). Among these patients, 47.0% had moderate-to-severe headache-related disability, 37.4% were dissatisfied with their acute migraine treatment, 32.0% had excessive opioid or barbiturate use and/or probable dependence and 26.2% had a history of cardiovascular (CV) events (3). Inadequate management of migraine can lead to increasing migraine attack frequency, which in turn can lead to medication overuse headache or to chronification of migraine (4,5).
Triptans, selective 5-HT1B/1D agonists, are the most widely used drugs for the acute treatment of migraine (6). Some patients do not respond to triptans and others do not tolerate side effects (6). Also, because triptans cause vasoconstriction through 5-HT1B receptors and have been temporally associated with rare CV events, they are contraindicated in patients with certain CV conditions (7–9); such conditions are common among people with migraine (10–12). Lasmiditan is a high-affinity, centrally penetrant, selective 5-HT1F receptor agonist without vasoconstrictive activity and is being developed specifically for the acute treatment of migraine (13–15). Evidence suggests that lasmiditan exerts its therapeutic effects in the treatment of migraine by decreasing neuropeptide release and inhibiting pain pathways, including at the trigeminal nerve (14,15).
The efficacy of lasmiditan in patients with migraine has been demonstrated in pivotal Phase 3 studies, SAMURAI and SPARTAN (16,17). In these studies, significantly higher proportions of patients treated with lasmiditan were free from headache pain at 2 hours following dosing compared to placebo. Additionally, significantly higher proportions of patients treated with lasmiditan were also free from their most bothersome symptom (MBS) at 2 hours post-dose compared to placebo. The most common adverse events for lasmiditan were dizziness, paresthesia, somnolence, fatigue and nausea. The adverse events were mostly mild to moderate in severity and self-limiting with a short duration (16–18).
Important goals of acute treatments for migraine attacks are rendering a patient pain free as quickly as possible without recurrence of headache (19–21). Therefore, it is important to consider the proportion of patients who achieve sustained responses following dosing with acute medication. Sustained pain freedom has been described by the International Headache Society (IHS) as the “ideal migraine treatment response” (22). Sustained pain freedom is easy to explain to patients as “the proportion of patients who require only a single dose to abort their attack by 2 hours and for at least 24 hours” (20). In these post-hoc analyses of pooled data from Phase 3, placebo-controlled studies of lasmiditan, we describe the proportions of patients treated with lasmiditan versus placebo who achieved sustained responses to various migraine outcomes, including pain freedom, MBS-free, total migraine-free and disability-free responses.
Methods
Patients and study design
SAMURAI (NCT02439320) and SPARTAN (NCT02605174) were similarly designed Phase 3, randomized, double-blind, placebo-controlled studies in patients with migraine with or without aura. SAMURAI enrolled patients from across 99 centers in the US. SPARTAN enrolled patients from 125 centers in the US, UK and Germany. Common inclusion criteria included men or women aged ≥ 18 years with a history of disabling migraine with or without aura for ≥ 1 year fulfilling the IHS diagnostic criteria 1.1 or 1.2.1 (23). Patients were required to have a Migraine Disability Assessment (MIDAS) total score of ≥ 11 (24,25) and a history of 3–8 migraine attacks per month (< 15 headache days per month). Common exclusion criteria included: History (within the past 12 months) of chronic migraine, medication overuse headache, or other forms of chronic headache disease where headache frequency was > 15 headache days per month; increased risk of seizures; and initiation of, or change in, migraine preventive treatments within 3 months prior to screening. In addition, while SAMURAI excluded patients with known coronary artery disease, clinically significant arrhythmia or uncontrolled hypertension, SPARTAN did not exclude these patients.
At the screening visit, patients were randomized using Interactive Response Technology equally to lasmiditan 200 mg, 100 mg, 50 mg (50 mg in SPARTAN only), or to placebo. Patients were stratified for use of concomitant migraine preventive treatments. Patients were asked to treat a single migraine attack with study drug on an outpatient basis within 8 weeks of enrollment. For rescue or recurrence, if needed, patients were randomized to a second dose of study drug; however, information regarding the second dose is not described here. Alternatively, patients were allowed to take their own non-excluded medication for rescue or recurrence between 2 and 24 hours post-dose.
The study protocols were approved by an independent ethics committee or institutional review board at each study site. All patients provided written informed consent prior to the start of the studies and enrollment. The studies were conducted in accordance with good clinical practice as required by the International Conference on Harmonisation guidelines, and in accordance with local laws and regulations governing clinical studies of investigational products. Compliance with these requirements also constituted conformity with principles of the Declaration of Helsinki.
Outcome measures and assessments
Patients were asked to treat a single migraine attack with the study drug on an outpatient basis and capture data using an electronic Diary (eDiary). Baseline data included date and time of migraine onset, date and time of first dose of study drug, level of headache pain, presence or absence of associated symptoms (nausea, vomiting, phonophobia, photophobia), identification of most bothersome symptom (from nausea, phonophobia, photophobia), and level of disability. Data captured at additional time points (0.5, 1, 1.5, 2, 3, 4, 24 and 48 hours) included level of headache pain, presence or absence of associated symptoms, and level of disability.
In SAMURAI and SPARTAN, the primary efficacy endpoint was a comparison between lasmiditan and placebo on the proportion of patients who were headache pain free at 2 hours after the first dose of study drug (16,17). The headache pain-free response was defined as a reduction in severity of headache from mild, moderate or severe pain at baseline to none. Sustained pain freedom was defined as being pain free at 2 hours following the first dose of study drug and at the indicated time point, without having used a second dose of study drug or other migraine medications following the first dose. At times through 2 hours, a subject was not counted as pain free if he or she used rescue medication at or before those times. Further, patients with missing data at 24 or 48 hours were assumed to not be pain free at those time points.
The key secondary endpoint was MBS freedom at 2 hours. Other exploratory efficacy endpoints captured at 2 hours in the studies and reported here are total migraine-free and disability-free responses. Patients were defined as having total migraine freedom at a given time point if they were pain free and were not experiencing any other migraine symptoms (i.e. nausea, photophobia, phonophobia or vomiting). Severity of migraine-associated disability was recorded at specific time points in the eDiary. Disability was measured on a four-point scale: Not at all (none [0]), mild interference (mild [1]), marked interference (moderate [2]), and need complete bed rest (severe [3]). Freedom from disability was defined as having no disability at the specified time point.
For the outcomes described above, response at 2 hours, and sustained responses at 24 hours and 48 hours post-dose, were defined in a similar manner to the sustained pain-free response. An additional modified sustained pain freedom outcome at 24 hours was analyzed based on the sustained pain freedom endpoint described by Ferrari et al. in a meta-analysis of triptans (19,20). Ferrari et al. describe 24-hour sustained pain freedom as being pain free at 2 hours, no use of rescue medication, and no moderate to severe headache 2–24 hours post-dose. Ferrari et al. allowed patients to experience recurrence of mild headache if they did not take rescue medication since such a recurrence was unlikely to be clinically significant. To match this endpoint, modified sustained pain freedom was defined for patients with available data at 24 hours as being pain free at 2 hours following the first dose of study drug, no moderate or severe pain at 24 hours, without having used a second dose of study drug or other migraine medications following the first dose.
Statistical analyses
SAMURAI and SPARTAN were similar in study design, which allowed data to be pooled. Efficacy analyses used a two-sided logistic regression model (Wald chi-squared test), with treatment group, study, and background use of migraine preventive medications as covariates. Additionally, analyses were performed to assess for study-by-treatment interaction effects; if any such interactions were found, these interactions were planned to be included in the model. For comparisons between individual lasmiditan doses and placebo, an estimate of the odds ratio of achieving a response, the corresponding confidence interval (CI) and p-value (Wald test), were computed. p-values were not corrected for multiplicity.
The safety population included all randomized patients who took at least one dose of study drug. The intent-to-treat (ITT) population included patients in the safety population who recorded any headache severity or symptom assessments in the eDiary. The modified intent-to-treat (mITT) population included patients in the ITT population who treated their migraine attack within 4 hours of onset of pain. The primary and key secondary analyses of pain freedom and MBS freedom were conducted in the mITT population as prespecified in the statistical analysis plan (SAP) for these studies. The sustained pain-free endpoint was prespecified in the SAP of the SPARTAN study, but was not prespecified in the SAP of the SAMURAI study. All other sustained endpoints and the modified sustained pain free endpoint were specified post-hoc. All endpoints, except for the modified sustained pain free endpoint at 24 hours, were conducted in the mITT population to allow for comparison with the prespecified outcomes. Modified sustained pain-free response at 24 hours was conducted in the ITT population for comparison of results with published meta-analyses of triptans (19,20).
Results
Baseline demographics and disease characteristics
Baseline demographics and disease characteristics in SAMURAI and SPARTAN Studies in mITT population.

BMI: body mass index; CV: cardiovascular; MBS: most bothersome symptom; MIDAS: Migraine Disability Assessment; N: number of patients with at least one dose of study drug; n: subgroup of patients; SD: standard deviation.
Sustained pain freedom
In this pooled analysis of SPARTAN and SAMURAI studies, significantly higher proportions of patients treated with lasmiditan 200 mg, 100 mg or 50 mg versus placebo reported being pain free at 2 hours post-dose (all doses p < 0.001). The proportion of patients who achieved pain freedom at 2 hours was 35.6% with lasmiditan 200 mg (OR: 2.5 [95% CI: 2.0, 3.0]), 29.9% with lasmiditan 100 mg (OR: 1.9 [95% CI: 1.5, 2.3]) and 28.6% with lasmiditan 50 mg (OR: 1.6 [95% CI: 1.2, 2.0]) versus 18.3% with placebo. Results of sustained pain freedom are shown in Figure 1. At 24 hours, significantly higher proportions of patients treated with lasmiditan versus placebo had a sustained pain-free response: 200 mg: 21.2% (OR: 2.3 [95% CI: 1.8, 3.0]); 100 mg: 16.9% (OR: 1.8 [95% CI: 1.4, 2.3]); 50 mg: 17.4% (OR: 1.6 [95% CI: 1.2, 2.1]); placebo 10.3% (all doses p < 0.01). Similar results were observed for the proportions of patients with sustained pain-free response at 48 hours: 200 mg: 18.4% (OR: 2.1 [95% CI: 1.6, 2.7]); 100 mg: 15.2%, (OR: 1.7 [95% CI: 1.3, 2.2]); 50 mg: 14.9%, (OR: 1.5 [95% CI: 1.1, 2.1]); placebo (9.6%) (all doses p < 0.05).
Proportions of patients in mITT population experiencing sustained pain freedom at 24 and 48 hours, following the first dose of placebo, lasmiditan 50 mg, 100 mg or 200 mg. from post-hoc analyses of SAMURAI and SPARTAN studies. Denominators for calculating percentages are the counts of patients experiencing mild, moderate or severe headache pain at baseline: Placebo: n = 1063; LTN 50 mg: n = 556; LTN 100 mg: n = 1035; LTN 200 mg: n = 1046. Patients in SAMURAI did not take LTN 50 mg.
Sustained most bothersome symptom-free response
At 2 hours post-dose, significantly higher proportions of patients reported being MBS free in lasmiditan dose groups: 200 mg: 44.7%; 100 mg: 42.6%; 50 mg: 40.8%; versus placebo: 31.5% (all doses p < 0.05). Sustained MBS-free results are shown in Figure 2. At 24 hours post-dose of study drug, significantly higher proportions of patients treated with lasmiditan achieved sustained MBS-free response: 200 mg: 24.5%; 100 mg: 21.4%; 50 mg: 21.9%; versus placebo: 14.2% (all doses p < 0.05). Similar results were noted at 48 hours post-dose for sustained MBS-free response: 200 mg: 20.5%; 100 mg: 19.2%; 50 mg: 18.8%; versus placebo: 12.8% (all doses p < 0.05).
Proportions of patients in mITT population experiencing sustained most bothersome symptom freedom at 24 and 48 hours, following the first dose of lasmiditan 50 mg, 100 mg, 200 mg or placebo. From a post-hoc analyses of SAMURAI and SPARTAN studies.
Sustained total migraine-free and disability-free responses
Proportions of patients in mITT population experiencing total freedom from migraine and freedom from disability at 2 hours, and sustained responses at 24 and 48 hours, following the first dose of lasmiditan 50 mg, 100 mg, 200 mg or placebo from post-hoc analyses of SAMURAI and SPARTAN studies.

For total migraine freedom, denominators are patients with ≥ mild headache recorded at baseline: Placebo: n = 1063; LTN 200 mg: n = 556; 100 mg: n = 1035; 50 mg: n = 1046. Total migraine-free and disability-free responses were assessed as described in Methods. Patients in SAMURAI did not take LTN 50 mg.
p < 0.001,**p < 0.01, *p < 0.05 for comparisons versus placebo. aOdds ratio and confidence interval ranges are provided within parentheses.
hrs: hours; LTN: lasmiditan.
Modified sustained pain freedom at 24 hours
Figure 3 shows side-by-side results for modified sustained pain freedom and sustained pain freedom at 24 hours. Significantly higher proportions of patients treated with lasmiditan versus placebo had modified sustained pain freedom at 24 hours: lasmiditan 200 mg: 27.0% (OR: 2.5 [95% CI: 2.0, 3.2]); lasmiditan 100 mg: 21.7% (OR: 1.9 [95% CI: 1.5, 2.4]); lasmiditan 50 mg: 21.7% (OR: 1.6 [95% CI: 1.2, 2.1]); placebo: 12.9% (all doses p < 0.01).
Side-by-side comparison of modified sustained pain freedom at 24 hours (ITT population) and sustained pain freedom at 24 hours (mITT population) from post-hoc analyses of SAMURAI and SPARTAN studies. For modified sustained pain-free response, patients had to be pain free at 2 hours, have no/mild pain at 24 hours and should not have used acute medications in between; patients with missing data at 24 hours were excluded from analysis. For the sustained pain-free response, patients had to be pain free at 2 and 24 hours, and should not have used acute medications in between; patients with missing data at 24 hours were assumed to not be pain free at 24 hours. Denominators (n) for calculating percentages in both analyses are counts of patients experiencing pain of any severity at baseline.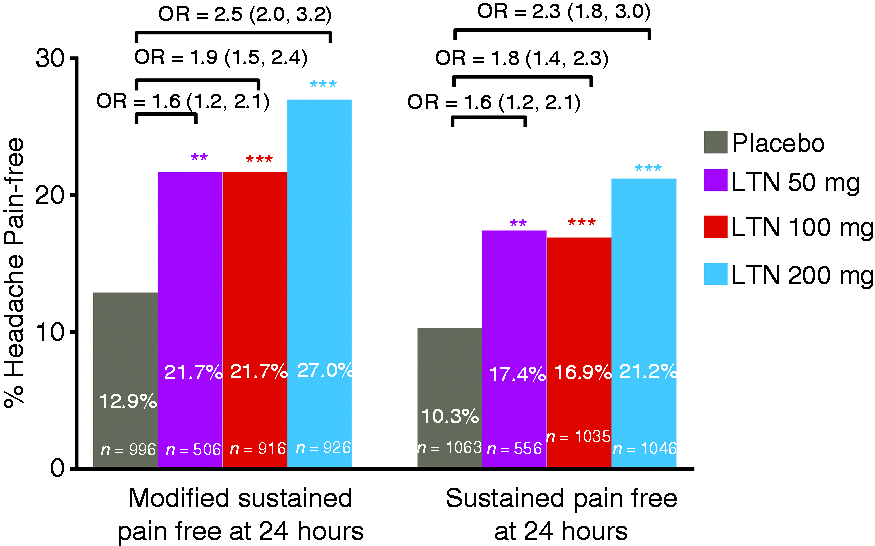
Discussion
In these post-hoc analyses of pooled data from Phase 3 randomized controlled trials (RCTs), sustained pain freedom was achieved by significantly higher percentages of patients treated with lasmiditan 200 mg, 100 mg or 50 mg versus placebo. The results for sustained pain freedom at 24 and 48 hours are notable and clinically significant as they indicate a sustained effect of a single dose of lasmiditan and decreased requirement for additional interventions for acute treatment of migraine. In the latest IHS guidelines for controlled trials of acute treatment of migraine attacks, sustained pain freedom is described as an important measure of response to acute treatment (26). Sustained pain-free rate is described as more scientifically robust than the relapse or recurrence rate as it integrates initial response, use of rescue medication, and relapse. Further, the guidelines describe the usefulness of sustained pain-free rate for comparisons with other acute treatments including triptans (26).
Comparing our sustained pain freedom results to the results with other acute treatments is difficult because of differing definitions of sustained pain freedom. To address this issue, we also performed a modified sustained pain freedom analysis to compare our results based on definitions used in meta-analyses of 53 trials with oral triptans (19,20). In this indirect comparison of modified sustained pain-free response at 24 hours with sumatriptan 100 mg (20% vs. 6% with placebo) to that of lasmiditan 200 mg (27% vs. 13% with placebo), the therapeutic gains were similar (14% vs. 14%). From the Ferrari et al. data, we also calculated an odds ratio of 3.9 for sumatriptan 100 mg compared to 2.5 for lasmiditan 200 mg from current analysis; this numeric difference in odds ratios is likely a result of the disparate placebo response rates. The findings from this analysis suggest that lasmiditan 200 mg has a similar sustained effect to that of sumatriptan 100 mg (19,20). We chose to make this comparison to sumatriptan 100 mg as it is the standard reference triptan and the most commonly used triptan. Thus, the similarity of sustained effect demonstrated by lasmiditan 200 mg is clinically significant.
Lasmiditan also showed a sustained response at 24 and 48 hours on a number of other end points, including MBS-free, total migraine-free, and disability-free responses. These results indicate that lasmiditan has a sustained benefit on all aspects of migraine attacks including associated symptoms and effects on activities.
The strengths of our findings include that they are based on two similarly designed large multi-center RCTs, which allowed us to pool the data to increase estimate precision. Limitations include that this was a post-hoc analysis and that the studies were not designed or powered to assess recurrence or sustained responses. In addition, multiple analyses of efficacy were conducted without correction for multiplicity. Also, the original studies were not powered to compare efficacy differences between lasmiditan doses. Another limitation is that lasmiditan 50 mg was taken by patients only in one of the two studies (SPARTAN).
It is possible that our analyses may underestimate the sustained responses to lasmiditan, as patients with missing data at 24 and 48 hours were treated as not being pain free. The eDiary required data entry at eight post-dose time points, and approximately 16% of trial participants had missing data at 24 hours. The lack of compliance with the eDiary at later time points may be due to the significant burden associated with migraine attacks and the multiple time points at which data entry was required. The assumption that patients with missing data at 24 and 48 hours were not pain free is likely to be over-conservative, and the modified sustained pain freedom endpoint that assesses patients who had available data at 24 hours may be a more correct approach.
In conclusion, the results of these post-hoc analyses suggest that lasmiditan is effective for the acute treatment of migraine attacks with sustained efficacy on all aspects of acute migraine attacks at 24 and 48 hours.
Clinical implications
Migraine is a chronic, debilitating neurological disease that is associated with multiple unmet needs for patients, including dissatisfaction with acute migraine treatment. In a pooled analysis of two Phase 3 randomized clinical trials of lasmiditan, sustained pain freedom at 24 and 48 hours was significantly greater for patients treated with lasmiditan versus placebo. Sustained benefits of lasmiditan versus placebo were also noted for other efficacy outcomes including most bothersome symptom-free, total migraine-free and disability-free responses. A significantly higher proportion of patients treated with lasmiditan versus placebo achieved a modified sustained pain freedom response (utilizing a meta-analysis definition). The reported results suggest that lasmiditan is effective for the acute treatment of migraine attacks, with sustained efficacy on all aspects of acute migraine attacks at 24 and 48 hours.
Footnotes
Acknowledgements
Sriram Govindan, a full-time employee of Eli Lilly, provided medical writing assistance for this manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EGD, JHK and JR are employees of Eli Lilly and Company, and/or its subsidiaries, and hold company stock. RBHS has served on an advisory board for Biohaven, has received honoraria from Medlink, Medscape/WebMD, and serves as the headache section editor for Current Neurology & Neuroscience Reports. KK has served on the advisory board for Promius Pharma, and has received honoraria from Assertio Therapeutics, Amgen, Allergan, Teva Pharmaceuticals and Eli Lilly and Company.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Studies were funded by CoLucid Pharmaceuticals, Inc., a wholly owned subsidiary of Eli Lilly and Company.
