Abstract
Background
Perimenstrual migraine attacks in women with menstrual migraine is difficult to treat. This post-hoc analysis evaluated the efficacy of lasmiditan, a high affinity and selective 5-HT1F receptor agonist, for perimenstrual attacks.
Methods
Patients from two randomized, double-blind, placebo-controlled clinical trials (MONONOFU and CENTURION) were instructed to treat an attack with a single dose of study medication within four hours of pain onset. After dosing, the proportion of patients who achieved freedom from migraine-related head pain, most bothersome symptom, and disability was reported at baseline up to 48 hours after dose and pooled data were evaluated.
Results
A total of 303 patients (MONONOFU N = 78; CENTURION N = 225) treated perimenstrual migraine attacks with lasmiditan 50 mg (N = 24), 100 mg (N = 90), 200 mg (N = 110), and placebo (N = 79). More patients achieved migraine-related head pain freedom with lasmiditan 200 mg versus placebo at all time points assessed. At 2 hours, 33.6% of patients in the 200-mg group (p < 0.001), and 16.7% of patients in the 100-mg (p = 0.11) and 50-mg (p = 0.19) groups were pain free, compared with 7.6% in the placebo group.
Conclusions
Lasmiditan treatment of perimenstrual migraine attacks was associated with freedom from migraine-related head pain at two hours, early onset of efficacy, and sustained efficacy.
Keywords
Introduction
After puberty, migraine affects females disproportionately more than males and is most burdensome in females between 15 and 49 years (1). The prevalence ratio of migraine (female:male) is highest during a female’s reproductive years, suggesting that hormonal changes related to that period in life may play a role in migraine expression (2).
Menstrual migraine can be classified as a) pure menstrual migraine, defined as having migraine attacks that occur exclusively during the 5-day perimenstrual interval (−2 to +3 days, where the first day of bleeding is defined as +1) (3) or b) menstrually-related migraine, defined as above but occur in women who also experience migraines at other times during their menstrual cycle (3).
Various studies have suggested that triptans can be effective for the treatment of migraine associated with menstruation in some women (4–12), however perimenstrual attacks (occurring during the 5-day perimenstrual interval) in women with menstrual migraine is still considered difficult to treat and is characterized by relatively severe and prolonged attacks as well as frequent relapse (13–16). In addition, it is associated with considerable disability and overall disease burden (17).
Lasmiditan, a novel, high affinity and selective 5-HT1F receptor agonist, is effective for the acute treatment of migraine (18–22). However, information regarding the efficacy of lasmiditan for the treatment of perimenstrual migraine is unknown. Thus, the objective of this post-hoc analysis was to evaluate the efficacy of lasmiditan in perimenstrual migraine.
Methods
Patient populations for these post-hoc analyses were pooled and derived from the intent-to-treat (ITT) populations from two randomized, double-blind, placebo-controlled clinical trials, assessing lasmiditan for the treatment of migraine attacks: MONONOFU (NCT03962738), a Phase 2 study conducted in Japan, and CENTURION (NCT03670810), a Phase 3 study conducted in Europe, North America, and Asia.
All patients gave written informed consent prior to participation in these studies. The studies were conducted with the approval of the Independent Ethics Committees of the participating institutes and in accordance with the Declaration of Helsinki, the International Conference on Harmonization Good Clinical Practice guidelines, and local regulations.
Study Design and Treatment
Details of the two randomized clinical trials have been published (18,22). In both trials, study treatment was self-administered orally within four hours of migraine pain onset. Medications for rescue or recurrence of migraine attack were allowed beginning at two hours after taking study drug. Menstruation data were also collected in these two studies, but not in other studies of lasmiditan efficacy. Females were asked to record dates and durations of menstrual period in a paper journal, and investigative staff were to transcribe this information into the database at each visit.
The MONONOFU single migraine attack study design included a screening visit, randomization visit (7:3:7:6 to placebo, lasmiditan 50 mg, lasmiditan 100 mg, or lasmiditan 200 mg), treatment period of ≤8 weeks, and an end of study visit between three and 28 days after treating a migraine attack. Patients recorded their response to treatment prior to dosing and for 48 hours post-dose.
The CENTURION multiple attack study, designed to address efficacy, consistency, and safety, included a screening visit, randomization visit (1:1:1 to one of three treatment groups for four attacks: lasmiditan 100 mg, lasmiditan 200 mg, or control group, which received placebo for three attacks and lasmiditan 50 mg for either attack 3 or attack 4 [1:1]). The treatment period continued until the four attacks had been treated or until four months after randomization, whichever came sooner. In the CENTURION trial, pain freedom at two hours in the first attack was a primary endpoint, and an exploratory objective related to assessing the effects of lasmiditan in premenopausal women during attacks in the perimenstrual interval.
Key inclusion criteria for both MONONOFU and CENTURION included age ≥18 years, migraine with/without aura per International Headache Society diagnostic criteria 1.1 or 1.2.1 (3), disabling migraine (Migraine Disability Assessment Test [MIDAS] (23,24) score ≥11) of ≥one year, onset before age of 50 years, and three to eight migraine attacks per month, but <15 headache days per month during the past three months.
Key exclusion criteria in both trials included a history of chronic migraine or other chronic headache disorders with ≥15 headache days/month within the past 12 months, hemorrhagic stroke, epilepsy, or any other condition placing the participant at increased risk of seizures, recurrent dizziness, and/or vertigo; diabetes mellitus with complications, orthostatic hypotension with syncope; significant renal or hepatic impairment, and patients who, in the investigator’s judgment, were actively suicidal and deemed to be at significant risk for suicide.
Efficacy Endpoints
Patients recorded efficacy-related data in an electronic diary at baseline and at 0.5, 1, 1.5, 2, 3, 4, 24, and 48 hours after dosing for MONONOFU and 0.5, 1, 2, 4, 6, 24, and 48 hours after dosing for CENTURION. During a five-day window starting two days before the onset of menstruation, the proportion of migraine attacks in temporal proximity to menstruation in females was assessed. Endpoints included pain freedom, pain relief, and most bothersome symptom (MBS) freedom, and disability freedom at two hours, 24-hour sustained headache pain freedom, and Patient Global Impression of Change (PGIC) (25) responder status of much better or very much better (based on response to the question “How do you feel after taking study medication?”, rated on a seven-point scale from “very much better” to “very much worse”).
Pain freedom was defined as a reduction in pain severity from mild, moderate, or severe at baseline to none at indicated time point. Pain relief was defined as a reduction in pain severity from moderate or severe at baseline to mild or none, or a reduction in pain severity from mild at baseline to none, at the indicated assessment time. MBS freedom was measured as the absence of pre-defined MBS of migraine (nausea, phonophobia, or photophobia) at an indicated time point. Finally, disability freedom was no disability recorded post-dose based on response to the question “How much is your migraine interfering with your normal activities?”, rated on a four-point scale from “not at all” to “completely needs bed rest” where those self-rating as “not at all” were considered responders.
Statistical Analyses
A perimenstrual attack was defined as an attack that was treated any time from day −2 to day +3 of menstruation (with first day of bleeding designated as day +1). First perimenstrual migraine attack efficacy analyses were conducted using data from the ITT female population (all randomized patients who used at least one dose of study drug with any post-dose pain severity assessments at or before two hours post-dose) who treated ≥1 perimenstrual attack.
Additionally, data from the ITT population among females who treated ≥1 perimenstrual attack were included in the subgroup analysis. Data from patients treated with placebo and lasmiditan 50 mg comprised a pooled control group and all treated perimenstrual and non-perimenstrual attacks were evaluated. Comparisons between treatments were made within subgroups while comparisons between subgroup (the perimenstrual attack vs. non-perimenstrual attack) levels were conducted within treatment.
A logistic regression model with treatment group and region (Asia, Europe, North America) as covariates was used for all efficacy analyses. Patients with missing outcome data at a time point were imputed as non-responders. Patients who took medication for rescue or recurrence were considered as non-responders at the time of taking medication and every time point afterward. Statistical evaluation was performed using SAS® version 9.4 or higher (SAS Institute Inc., Cary, NC).
Results
Characterization of patients experiencing perimenstrual migraine
Among 2099 patients (MONONOFU, N = 687; CENTURION, N = 1412) who treated at least one migraine attack in the original cohort studies, 303 patients (N = 79 placebo, N = 24 lasmiditan 50 mg, N = 90 lasmiditan 100 mg, and N = 110 lasmiditan 200 mg) had at least one perimenstrual attack and were considered the pooled ITT population for this study. In general, baseline demographics and clinical characteristics were similar across treatment groups (Table 1). Overall, the mean age of women was 39 years. Over half (52.8%) of patients were from Europe. Across the pooled patient population, the average duration of migraine history was 17 years (21.8% with, 68.0% without aura, and 10.2% with and without aura), the baseline attack frequency was 5.1 per month, the number of migraine headache days in past three months was 19.7, and the degree of migraine-related disability was severe (mean MIDAS score 31.3). For treated migraine attacks, the highest proportion of patients treated on day −1 (23.8%) compared to 14.9% on day 3. Most treated attacks were of moderate severity (72.6%) with photophobia the most prevalent symptom at baseline (61.7%), and most prevalent MBS (43.2%). Median time to dosing was 1.3 hours.
Baseline patient demographics and clinical characteristics.
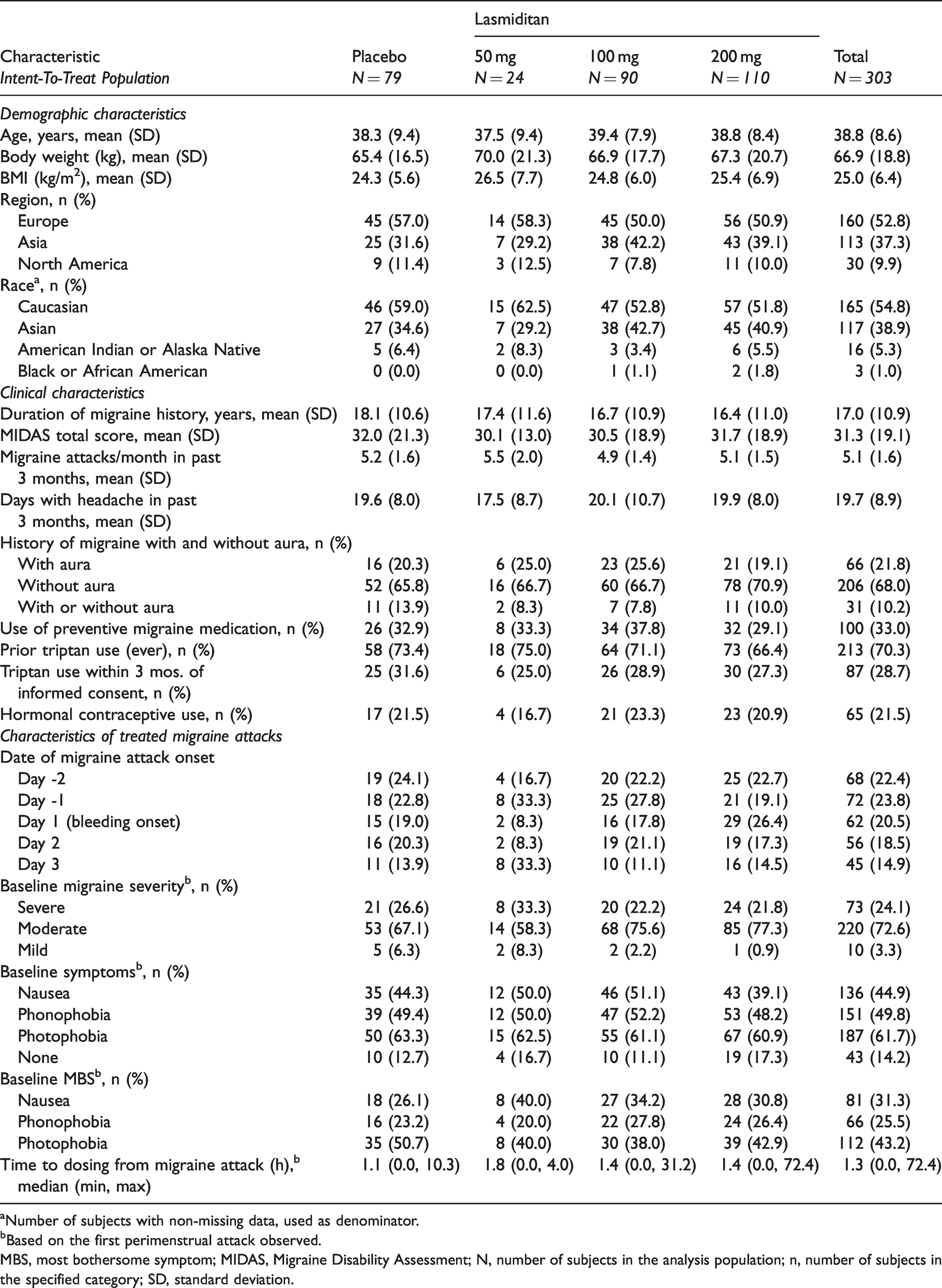
aNumber of subjects with non-missing data, used as denominator.
bBased on the first perimenstrual attack observed.
MBS, most bothersome symptom; MIDAS, Migraine Disability Assessment; N, number of subjects in the analysis population; n, number of subjects in the specified category; SD, standard deviation.
Efficacy of lasmiditan vs placebo for the treatment of first perimenstrual attacks
Table 2 displays a summary of efficacy endpoints by treatment group and online Supplemental Table 1 displays a summary of select efficacy endpoints (pain freedom, pain relief, and MBS freedom) by treatment group for up to four hours post-dose; data presented are based on the first menstrual-related migraine attack. Figure 1 shows a graphical representation of the proportion of patients achieving pain freedom up to four hours after dosing. Lasmiditan dose groups appeared to separate from placebo at one hour in the 200-mg dose group and at 2 hours in the 100-mg and 50-mg dose groups. Statistically significant results were achieved beginning at one hour with 200 mg and three hours with 100 mg (not shown). At two hours post-dose, the lasmiditan groups showed odds ratios of 2.5–6.04 compared with placebo, with statistically significant (p < 0.001) results achieved for lasmiditan 200 mg.
Summary of efficacy endpoints by treatment group.

aPain freedom was defined as a reduction in pain severity from mild, moderate, or severe at baseline to none at indicated time point.
bPain relief was defined as a reduction in pain severity from moderate or severe at baseline to mild or none, or a reduction in pain severity from mild at baseline to none, at the indicated assessment time.
cMBS freedom was defined as the absence of pre-defined MBS of migraine (nausea, phonophobia, or photophobia) at indicated time point.
dNo disability recorded post-dose based on response to the question “How much is your migraine interfering with your normal activities?”, rated on a 4-point scale from “0 = not at all” to “3 = completely, needs bed rest,” where those self-rating as “0” were considered responders).
eHeadache recurrence is defined as pain-free at 2 hours post-dose becoming mild or worse before or at 24/48 hours post-dose.
fBased on response to the question “How do you feel after taking study medication?”, rated on a 7-point scale from “very much better” to “very much worse”.
CI, confidence interval; MBS, most bothersome symptom; N, number of subjects in the analysis population; nx, number of patients with assessment, nt, number of patients with non-missing assessment at given timepoint; PGIC, Patient Global Impression of Change.

Proportion of patients with pain freedom among women treating attack during perimenstrual attack.
For pain relief, the lasmiditan 200-mg and 100-mg dose groups appeared to separate from placebo at one hour, while the 50-mg group appeared to separate from placebo at three hours. Statistically significant results were achieved beginning at one hour for both the 200 mg and 100 mg dose groups. At two hours post-dose, the lasmiditan groups showed odds ratios of 0.70–3.74 compared with placebo, with statistically significant results achieved for lasmiditan 100 mg (p < 0.001) and lasmiditan 200 mg (p = 0.007).
For MBS freedom, the lasmiditan 200-mg, 100-mg, and 50-mg dose groups appeared to separate from placebo at one hour. Statistically significant results were achieved beginning at four hours for the 100-mg dose group. At two hours post-dose, the lasmiditan treatment groups showed odds ratios 0.94–1.81 that were not statistically significant.
For sustained pain freedom at 24 hours, the lasmiditan groups showed odds ratios of 4.57–11.67 compared with placebo, with statistically significant results achieved for the lasmiditan 50-mg (p = 0.022) and 200-mg (p = 0.001) dose groups. For pain recurrence at 24 hours, the lasmiditan groups showed odds ratios of 0.07–0.25 (directionally suggesting less pain recurrence with lasmiditan than placebo) that were not statistically significant.
For disability freedom at two hours post-dose, the lasmiditan groups showed odds ratios of 0.71–1.91 that were not statistically significant from placebo. For PGIC responder status of much better or very much better at 24 hours, the lasmiditan groups showed odds ratios of 2.1–3.9 compared with placebo, with statistically significant results achieved for all lasmiditan treatment groups (lasmiditan 50 mg [p = 0.005], lasmiditan 100 mg [p = 0.042], and lasmiditan 200 mg [p = 0.003]).
Subgroup analyses: Efficacy of lasmiditan vs placebo for the treatment of perimenstrual vs non-perimenstrual attacks
In the subgroup analysis conducted for the ITT population among women who treated ≥1 perimenstrual attack, 98 received placebo, 60 received lasmiditan 50 mg, 90 received lasmiditan 100 mg, and 110 received lasmiditan 200 mg. Figure 2 highlights the proportion of perimenstrual migraine attacks with pain freedom compared to non-perimenstrual migraine attacks with pain freedom. Separation from placebo appeared to occur at one hour in the 200-mg dose groups and at two hours in the 100-mg and 50-mg dose groups. Statistically significant results vs placebo were achieved beginning at one hour for non-perimenstrual migraine attacks treated with all doses and for perimenstrual migraine attacks treated with 200 mg lasmiditan. At two hours post-dose, the lasmiditan groups showed odds ratios of 2.10–6.09 (perimenstrual) and 2.30–4.17 (non-perimenstrual) compared with placebo, with statistically significant results achieved for the lasmiditan 100-mg and 200-mg treatment groups (lasmiditan 100 mg non-perimenstrual [p < 0.001], and lasmiditan 200 mg perimenstrual [p < 0.001] and lasmiditan 200 mg non-perimenstrual [p = 0.015]).

Proportion of attacks with pain freedom in women who treated at least 1 perimenstrual migraine attack vs women who treated at least one non-perimenstrual migraine attack. Note: Solid lines correspond to women who treated perimenstrual migraine attacks and dotted lines correspond to women who treated non-perimenstrual migraine attacks.
Supplemental Figure 1 shows the proportion of perimenstrual migraine attacks with pain relief compared to non-perimenstrual migraine attacks with pain freedom. The lasmiditan 200-mg and 100-mg dose groups appeared to separate from placebo at one hour with statistically significant results achieved beginning at the same time. At two hours post-dose, the lasmiditan groups showed odds ratios of 0.94–3.09 (perimenstrual) and 0.89–2.23 (non-perimenstrual) compared with placebo, with statistically significant results achieved for the lasmiditan 100-mg and 200-mg treatment groups (lasmiditan 100 mg perimenstrual [p < 0.001] and lasmiditan 100 mg non-perimenstrual [p = 0.003] as well as lasmiditan 200 mg perimenstrual [p = 0.006].
Supplemental Figure 2 presents the proportion of perimenstrual migraine attacks with MBS freedom compared to non-perimenstrual migraine attacks with MBS freedom. The lasmiditan 200-mg and 100-mg dose groups appeared to separate from placebo at one-two hours with statistically significant results achieved beginning at the same time. At two hours post-dose, the lasmiditan groups showed odds ratios of 1.01–1.78 (perimenstrual) and 1.29–1.82 (non-perimenstrual) compared with placebo, with statistically significant results achieved for the lasmiditan 100-mg treatment group (non-perimenstrual [p = 0.043]).
Supplemental Table 2 summarizes pain freedom, pain relief, and MBS freedom efficacy endpoints by treatment group for perimenstrual vs non-perimenstrual attacks. At two hours post-dose, the proportion of pain freedom was similar in perimenstrual and non-perimenstrual attacks with lasmiditan 200 mg, but not with lower doses; the lasmiditan groups showed perimenstrual vs non-perimenstrual odds ratios ranging from 0.31–1.81 for pain freedom with statistically significant results achieved with lasmiditan 100 mg [p = 0.001], and lasmiditan 200 mg [p = 0.037]).
Discussion
Lasmiditan was superior to placebo for pain freedom, pain relief, and MBS freedom in patients treating perimenstrual migraine. Similar efficacy was observed in this study regardless of whether attacks were classified as perimenstrual or non-perimenstrual (Figure 2) and supported by previously reported efficacy in the original cohorts (18,22). In general, the 200-mg lasmiditan dose was the most efficacious of the doses tested in this study for achieving pain freedom, pain relief, and MBS freedom, especially at earlier time points, as well as sustained efficacy. Improved PGIC and disability free ratios were also observed for lasmiditan-treated perimenstrual migraine, when compared to placebo.
Lasmiditan showed efficacy for pain freedom and pain relief as early as one hour after dosing in patients who treated perimenstrual migraine attacks. This was similar to the overall study cohorts, in which efficacy was observed as early as 0.5 hours for pain freedom and pain relief in MONONOFU (22) and 0.5 hours (pain relief) and one hour (pain freedom) in CENTURION (18). Sustained efficacy was also observed with lasmiditan treatment (odds ratio 4.57–11.67) with a lower pain recurrence ratio observed at 24 hours (odds ratio 0.07-0.25).
The early and sustained efficacy observed with lasmiditan-treated perimenstrual migraine attacks may potentially provide a new therapeutic option for perimenstrual migraine considered to have severe attack characteristics. For example, 16.6% and 8.4% of placebo-treated patients achieved two-hour pain freedom in the original cohort studies (18,22) compared to 7.6% of placebo-treated patients with perimenstrual attacks in this study. Additionally, when comparing pain freedom, pain relief, and MBS freedom between the perimenstrual and non-perimenstrual subgroups of the placebo treatment group, a lower percentage of patients with pain freedom, pain relief, and MBS freedom who treated perimenstrual attacks was observed compared to the percentage who treated non-perimenstrual treated attacks. This supports the observation that perimenstrual migraine is characterized by being relatively more difficult to treat acutely (13–16). In comparison to the original cohort studies in which 10.4% and 4.3% of placebo-treated patients achieved 24-hour sustained headache pain freedom (18,22), only 2.5% of patients with perimenstrual attacks in the placebo group achieved 24-hour sustained headache pain freedom, supporting previous observations that perimenstrual migraine attacks are associated with a higher risk of recurrence (16,26–28). While some countries limit lasmiditan to a single dose every 24 hours, other countries allow a second dose for relapse, to a maximum of 200 mg per 24 hours. Where needed, other treatment options can be considered for rescue or relapse after lasmiditan treatment.
Limitations
These studies did not actively recruit women with a diagnosis of menstrual migraine. Since there is no objective diagnostic biomarker available to characterize a true association, attacks and menstruation may have been coincidental, rather than causally related. The analysis included a small number of patients, especially for the 50-mg dose. A weakness of this study is that the relatively small sample size of females with menstrual migraine population limits the interpretation of the analyses and may account for the lack of significance seen in some comparisons.
Conclusions
In patients with perimenstrual migraine attacks from two clinical trials, treatment with lasmiditan compared with placebo was associated with freedom from migraine-related head pain at two hours, early onset of efficacy, and sustained efficacy. These findings were consistent with the overall population, which suggests that lasmiditan is a potential option to treat perimenstrual migraine attacks.
Key Findings
Lasmiditan treatment of perimenstrual migraine attacks in patients from the MONONOFU and CENTURION clinical trials was associated with freedom from migraine-related head pain at two hours.
Early onset of efficacy and sustained efficacy were observed in these patients.
Lasmiditan is a potential option to treat perimenstrual migraine attacks in those with menstrual migraine.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221118929 - Supplemental material for Efficacy of lasmiditan for the acute treatment of perimenstrual migraine
Supplemental material, sj-pdf-1-cep-10.1177_03331024221118929 for Efficacy of lasmiditan for the acute treatment of perimenstrual migraine by E Anne MacGregor, Mika Komori, John Henry Krege, Simin Baygani, Maurice Vincent, Jelena Pavlovic and Hisaka Igarashi in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024221118929 - Supplemental material for Efficacy of lasmiditan for the acute treatment of perimenstrual migraine
Supplemental material, sj-pdf-2-cep-10.1177_03331024221118929 for Efficacy of lasmiditan for the acute treatment of perimenstrual migraine by E Anne MacGregor, Mika Komori, John Henry Krege, Simin Baygani, Maurice Vincent, Jelena Pavlovic and Hisaka Igarashi in Cephalalgia
Supplemental Material
sj-jpg-3-cep-10.1177_03331024221118929 - Supplemental material for Efficacy of lasmiditan for the acute treatment of perimenstrual migraine
Supplemental material, sj-jpg-3-cep-10.1177_03331024221118929 for Efficacy of lasmiditan for the acute treatment of perimenstrual migraine by E Anne MacGregor, Mika Komori, John Henry Krege, Simin Baygani, Maurice Vincent, Jelena Pavlovic and Hisaka Igarashi in Cephalalgia
Supplemental Material
sj-jpg-4-cep-10.1177_03331024221118929 - Supplemental material for Efficacy of lasmiditan for the acute treatment of perimenstrual migraine
Supplemental material, sj-jpg-4-cep-10.1177_03331024221118929 for Efficacy of lasmiditan for the acute treatment of perimenstrual migraine by E Anne MacGregor, Mika Komori, John Henry Krege, Simin Baygani, Maurice Vincent, Jelena Pavlovic and Hisaka Igarashi in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MK, JHK, SB, and MV are employees and minor stockholders of Eli Lilly and Company. EAM: Asarina Pharma (Other Activities) (Scientific Advisory or Data Safety Monitoring board), Bayer Healthcare (Other Activities) (Scientific Advisory or Data Safety Monitoring board), Eli Lilly and Company, Novartis, Theramex (Other Activities) (Scientific Advisory or Data Safety Monitoring board), Teva, and Theramex Australia (Speaking and Teaching) (Speakers Bureau). JP has received consulting honoraria from Allergan Inc., Alder, Biohaven Pharmaceuticals, and Promius Pharma. She is funded by the NIH/NIA K23AG049466-01A1. HI: Amgen (Consulting), Eli Lilly Japan KK (Consulting), Kyowa Hakko Kirin (Speaking and Teaching), Otsuka Pharmaceutical Co. Ltd. (Consulting), Pfizer (Speaking and Teaching), Takeda Pharmaceutical Company Limited (Speaking and Teaching), Lundbeck Japan KK (Teaching, Daiichi Sankyo Co. Ltd. (Speaking), Eli Lilly Japan KK (Consulting and Speaking).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Eli Lilly and Company. Medical writing support was provided by Shannon E Gardell (Evidera) and paid for by Eli Lilly and Company.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
