Abstract
Background
Pain freedom after 2 hours is the recommended primary endpoint by the International Headache Society in randomized trials investigating drug treatment of acute migraine attacks. In order to demonstrate an early effect of a drug, some drug companies, however, have promoted headache relief (improvement from severe or moderate pain to mild or no pain) at earlier time points than 2 hours as outcome parameter.
Methods and results
We analyzed the relationship between pain freedom and headache relief in acute migraine trials and observed that persistent mild headache constituted 90% of headache relief after 0.5 hour and 40% of headache relief after 2 hours.
Conclusion
Headache relief at 2 hours should in our view only be used as an outcome measure for comparison with historic data. Prior to 2 hours, headache relief varies with time from intake and the therapeutic gain is very small. Therefore, pain freedom should be used at these early time points.
Introduction
The International Headache Society (IHS) recommends pain freedom after 2 hours as the primary efficacy parameter in randomized, controlled trials (RCTs) investigating drug treatment of acute migraine attacks (1). In two studies, migraine patients expressed complete relief of headache within 30 min after drug treatment as their highest priority (2,3). Thirty to 20 years ago when the triptans, 5-HT1B/1D receptor agonists, were developed, headache relief (improvement from severe or moderate pain to mild or no pain) at 2 hours was used as the primary efficacy parameter (4). In the current guidelines for acute clinical trials in migraine of the IHS, headache relief is recommended as a secondary efficacy parameter, mainly for comparison with previous results in RCTs with acute migraine drugs (1).
In some recent, large RCTs of migraine attacks, treatment headache relief (5,6) has been used to claim early onset of effect. It was thus reported that for lasmiditan 200 mg, headache relief was significantly higher starting as early as 30 minutes (lasmiditan 19% vs. placebo 13%, p < 0.001) (5), but the therapeutic gain (active drug minus placebo) for pain freedom was 2.4–1.4% = +1% (95% CI: −0.2 to +2%) (5).
The no headache of headache relief is the same as pain freedom, and the very low pain freedom in the above example with lasmiditan 200 mg vs. placebo suggests that headache relief at 0.5 hour mainly consists of mild headache, whereas headache relief at 2 hours often consists of approximately 50% no headache and 50% mild headache; for example, headache relief for sumatriptan 100 mg in the large Cochrane review on sumatriptan is 61% at 2 hours (32% pain freedom and 29% mild headache) (7).
The purpose of the present study was to evaluate whether headache relief, apart from its use at 2 hours as a historical control, is a clinically useful and consistent efficacy measure, also when used before the 2-hour time-point.
Methods
We screened by hand searching the headache literature for papers on randomized, controlled trials (RCTs) of oral drug treatment of migraine attacks presenting both results for headache relief and pain freedom, at 0.5 h, 1.0 h, 1.5 h, and 2.0 h. The percentage results with active drugs were used in the calculations for each active treatment. Percentage of headache relief of a drug minus percentage of pain freedom for the drug resulted in “% of persistent mild headache” and this value was divided by percentage of headache relief for each time point. Similar calculations were performed when placebo results were available.
We also included three RCTs with known similar early effect on headache relief and with three different drugs and administration forms (8–10).
In addition, the therapeutic gain for headache relief was calculated for headache relief at 30 min and earlier time points, if early results were available.
Results
The screening was terminated after 19 consecutively retrieved papers of RCTs with sequential results from 0.5 h to 2 h. We retrieved one RCT with aspirin 1000 mg (11), ibuprofen 400 mg (11), sumatriptan 100 mg (13), lasmiditan 50 mg (5), zolmitriptan 2.5 mg (16), naratriptan 2.5 mg (17), almotriptan 12.5 mg (19), and ergotamine 2 mg (18); and two RCTs with lasmiditan 100 mg (5), lasmiditan 200 mg (5), and sumatriptan 50 mg (11,12). Finally, we retrieved seven RCTs with rizatriptan 10 mg, (12–18]. All trials showed the same development of the headache relief versus pain freedom ratio over time. The pattern for percent of persistent mild headache/percent of headache relief, last row in Table 1, was in all cases: % 0.5 h > % 1.0 h > % 1.5 h > % 2.0 h. For percentage of persistent mild headache/percentage of headache relief, the median was 90% (range, 78–96%) after 0.5 h, 72% (range, 63–88%) after 1.0 h, 63% (range, 49–72%) after 1.5 h, and 43% (range, 34–55%) after 2 h. For placebo, similar calculations of persistent mild headache could be performed in eight time-effect curves. The pattern for percentage of persistent mild headache/percentage of headache relief in time was generally: % 0.5 h > % 1.0 h > % 1.5 h > % 2.0 h, except in two cases in which the percentages at 0.5 h were lower than those at 1 h. For percentage of persistent mild headache/percentage of headache relief, the median was 90% (range, 75–100%) after 0.5 h, 85% (range, 72–94%) after 1.0 h, 77% (range, 71–85%) after 1.5 h, and 66% (range, 59–80%) after 2 h.
Distribution of headache responses at 2 h, headache relief (HR) percentage and pain freedom (PF) percentage, and “persistent mild headache” (HR% – PF%) divided by HR% after 0.5 h, 1.0 h, 1.5 h and 2 h, after four oral drugs (in bold), and three other drugs administered intranasally, as oral inhalation and subcutaneously. Results for placebo are presented in parentheses and in italics.
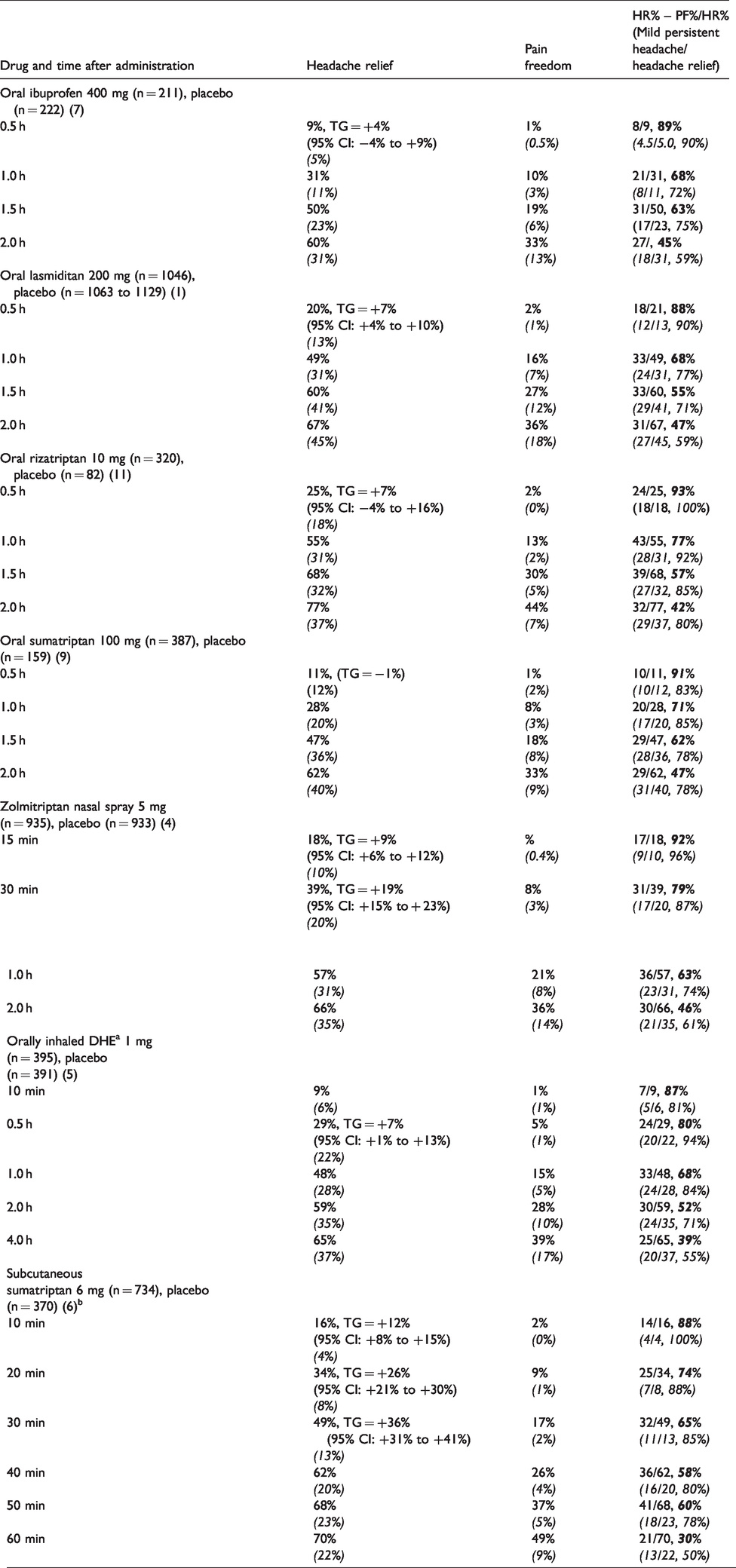
In 16 RCTs with oral drugs, the TG for headache relief was statistically significant for five drugs at 30 min (+6 to +9%) (5,11,12,19), but the TG for pain freedom at 30 min was not statistically significant in any of the 16 RCTs.
Similar decreases of the ratio for percentage of persistent mild headache/percentage of headache relief from 0.5 h to 2 h were observed for zolmitriptan nasal spray 5 mg (8), orally inhaled dihydroergotamine 1 mg (9), and subcutaneous sumatriptan 6 mg (10), as shown in Table 1.
Discussion
Pain freedom at 2 hours after intake of an oral drug has been recommended as the primary efficacy parameter in RCTs in all guidelines on clinical trial design for migraine trials by the International Headache Society (IHS) from 1991, 2000, 2012, and 2019 (1). In contrast, some pharmaceutical companies who developed triptans deviated from this recommendation and used headache relief as the primary outcome parameter.
For example, Glaxo introduced headache relief at 2 hours as the primary efficacy when sumatriptan was developed. Headache relief at 2 hours was also used in the development of several other triptans, see meta-analysis by Ferrari et al. (4). In the most recent IHS Guidelines on Drug Trials, headache relief at 2 hours is recommended only as a secondary effect measure, and it is stated: “Headache relief should be used as an outcome measure, but only as a secondary endpoint and mainly to facilitate comparison of the results of new clinical trials with those of previous programmes” (1).
General use of headache relief at other time points than 2 hours is thus not recommended by the IHS (1). Analysis of headache relief at early time-points has been used in “demonstrating” very early effects, see for example headache relief at 15 min after nasal zolmitriptan, therapeutic gain (TG) = +9%, at 30 min after orally inhaled DHE, TG = +7% (Table 1). The TGs for pain freedom are considerably lower: +1% (95% CI; +0.4 – +2%) for nasal zolmitriptan (8) and +4% (95% CI: +1 – +7%) for orally inhaled DHE (9). These therapeutic gains for early pain freedom are in our view not clinically relevant. An effect on headache relief at 30 min after oral lasmiditan 100 mg and 200 mg (5), and at 60 min after oral rimegepant 75 mg (6), has been used to claim early onset of effect despite the fact that pain freedom was not significant at this time point.
The current analysis of the distribution at the 4 time-points (0.5 h, 1.0 h, 1.5 h, and 2 h) of pain freedom vs. headache relief calculated as “persistent mild headache”/headache relief shows a value of 90% at 0.5 hour (thus 90% of the headache relief is mild headache and 10% is pain freedom), and a value of 40% at 2.0 h (thus 40% of the headache relief is mild headache and 60% is pain freedom). There is a similar tendency in the placebo groups: “persistent mild headache”/headache relief shows a value of 90% at 0.5 hour and 66% at 2 hours.
We doubt, whether a small therapeutic gain for headache relief at 0.5 hour is clinically relevant. Among 16 RCTs analysed, the TG for headache relief was statistically significant for five drugs at 30 min (+6 – +9%) (5,11,12,19), but TG for pain freedom was not statistically significant in any of these 16 RCTs.
In our view it is therefore unlikely that there is a clinically relevant effect of oral triptans already after 30 min.
Conclusion
Headache relief at 2 hours should in our view only be used as an outcome measure for comparison with historic data. Prior to 2 hours headache, relief varies with time from intake and the therapeutic gain is relatively small early after intake. Therefore, pain freedom should, if possible, be used at these early time points.
Clinical implications
In acute migraine trials, headache relief at 2 hours can be used to compare results with historic data. At earlier time points, we suggest that only pain freedom, if possible, should be used.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
