Abstract
Objective
To determine whether serum glial fibrillary acidic protein (GFAP) and neurofilament light chain (NfL) concentrations differ between adults with migraine and healthy controls.
Methods
This single-center, cross-sectional study was conducted from September 2020 to June 2022. Adults diagnosed with migraine and healthy controls were enrolled. Serum GFAP and NfL concentrations were quantified using an ultra-sensitive single-molecule array platform. Analyses were adjusted for potential confounders, including age, sex, body mass index and smoking status.
Results
We assessed serum samples from 603 participants with migraine and 154 controls. Serum GFAP concentrations were 6.8% higher in the overall migraine group compared to controls (95% confidence interval = 0.4–13.5%; P = 0.036). Conversely, serum NfL concentrations did not differ between participants with migraine and controls (difference: −1.4%; 95% confidence interval = −7.9 to 5.6%; p = 0.68). Neither serum GFAP, nor NfL concentrations varied according to migraine subtype or headache status at the time of blood sampling.
Conclusions
Our findings demonstrate a modest yet statistically significant increase in serum GFAP among adults with migraine, independent of migraine subtype, whereas serum NfL levels were comparable to those of controls. Further research is needed to clarify the neurobiological mechanisms underlying elevated serum GFAP in migraine.
This is a visual representation of the abstract.
Keywords
Introduction
The neurobiological basis of migraine remains incompletely understood (1), although emerging evidence suggests that neuroinflammatory processes might play a key role (2). This growing body of knowledge has fueled interest in identifying blood-based biomarkers that reflect underlying neuronal and glial alterations (3,–5), with the aim of advancing our understanding of the disease. Among the candidate biomarkers under consideration are glial fibrillary acidic protein (GFAP) and neurofilament light chain (NfL) (3,–5). GFAP is predominantly expressed in astrocytes, and elevated levels in serum have been observed in other neurological conditions such as multiple sclerosis and traumatic brain injury (6), reflecting glial activation and potential blood–brain barrier (BBB) compromise (6,7). Likewise, NfL, comprising a structural component of myelinated axons, has emerged as a sensitive indicator of axonal injury, demonstrating elevated serum concentrations in several neurological disorders, including Alzheimer's disease and stroke (8,9).
Despite growing interest, the role of GFAP and NfL in migraine remains unclear (3,–5). It is plausible that elevated serum GFAP and NfL might signal underlying astroglial activation and subtle axonal damage, respectively. Of particular interest is also whether these biomarkers differ among migraine subtypes, such as migraine with aura vs. without aura, and episodic vs. chronic migraine. Such insights might, in turn, shed light on distinct pathophysiological process that remain largely unknown.
To address these knowledge gaps, we conducted a large, cross-sectional study comparing serum GFAP and NfL concentrations between adults with migraine and healthy controls (HCs). Furthermore, we explored potential associations of these biomarkers with sociodemographic and clinical characteristics within the migraine population. By offering new insights into the involvement of glial and axonal elements in migraine pathophysiology, our results might provide directions for future research efforts.
Methods
Design
The cross-sectional data analyzed in this manuscript were derived from the prospective, single-center Registry for Migraine (REFORM) study (10). The parental protocol was approved by the relevant ethics committee, and enrollment was conducted from September 2020 through June 2022. All procedures complied with the principles of the Declaration of Helsinki and its subsequent amendments (11). Before undertaking any study-related tasks or procedures, written informed consent was obtained from every participant with migraine and HC. This work adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (12).
Participants
The participants with migraine were primarily recruited from the outpatient clinic of a national referral hospital, Copenhagen University Hospital – Rigshospitalet, Denmark. Eligible individuals were adults (≥18 years) who had a diagnosis of migraine without aura, migraine with aura or chronic migraine in accordance with the International Classification of Headache Disorders, 3rd edition (ICHD-3) (13). Participants were required to have experienced migraine attacks for at least one year prior to enrollment and to report an average of four or more monthly migraine days during the three months preceding screening. The main exclusion criteria were any history of hemiplegic migraine, cluster headache or secondary headache disorders other than medication-overuse headache (10). Furthermore, participants with a history of erenumab use or who had received another monoclonal antibody against calcitonin gene-related peptide (CGRP) signaling within three months of enrollment were excluded. Ongoing use of other preventive migraine medications were allowed, provided the dosage had remained stable for at least two months prior to enrollment and remained unchanged for the duration of the study. For the present analysis, all participants with migraine who reported comorbid neurological disorders were excluded. A complete listing of inclusion and exclusion has been published elsewhere (10).
HCs were recruited through online advertisements and had to be free from any current or past clinically significant medical conditions, psychiatric disorders or headache disorders, except for infrequent episodic tension-type headache, as defined by the ICHD-3 criteria (13). Furthermore, potential HCs were excluded if they had any first-degree relatives with a history of primary headache disorders, except for those with ≤5 monthly days of tension-type headache. A complete list of inclusion and exclusion is available elsewhere (10).
Procedures
At enrollment, site investigators conducted standardized, semi-structured interviews to record sociodemographic information, incl. age, sex, body mass index and smoking status, as well as relevant medical histories. In the migraine population, the number of monthly headache days (MHDs), monthly migraine days (MMDs), monthly days of acute headache medication use and the presence of aura were determined through retrospective self-report. Current and past use of preventive migraine medications were also recorded. For HCs, similar sociodemographic and medical data were collected to verify eligibility and to establish a baseline comparator.
Venous blood was drawn from the cubital fossa into 8-ml serum gel-separator clot-activator tubes (Vacuette®; Greiner Bio-One, Kremsmünster, Austria). Following collection, the tubes were left at room temperature for 30 minutes to allow for complete clot formation before centrifugation at 2200 g for 10 min at 4°C to separate serum from cellular components. The resulting serum was aliquoted into cryotubes and stored at −80°C until analysis.
Biomarker measurement
Serum GFAP concentrations were measured at the Department of Biochemistry and Immunology, Lillebaelt Hospital (Vejle, Denmark) and serum NfL concentrations were measured at the Neuroimmunology Laboratory, Danish Multiple Sclerosis Center, Rigshospitalet (Glostrup, Denmark). Both GFAP and NfL were quantified using the ultra-sensitive single-molecule array (SiMoA) platform (Quanterix, Billericia, MA, USA) with manufacturer-supplied reagents. Assays were conducted by independent laboratory personnel blinded to participants’ clinical status and headache features. The SiMoA technique is a well-established method for biomarker quantification, offering high sensitivity and reliability for both research and clinical applications (14,–16). The analytical lower limit of quantification was 0.686 pg/ml for GFAP and 0.174 pg/ml for NfL. Assay performance was monitored over time using quality controls (two provided by the manufacturer and one in-house prepared serum pool) included in each run. Analytical variations remained below 13%. To enhance data reliability, extreme outliers identified through visual inspection were excluded from the final analyses.
Participant classification and subgroups
A tiered classification system was used to characterize the migraine population. This system comprised four primary tiers, each further divided into specific subgroups. First, participants were classified by migraine frequency as having episodic or chronic migraine. Second, participants were categorized based on aura status, as either having migraine with aura or migraine without aura. Third, participants were classified according to their headache status at the time of blood sampling: those experiencing a probable or definite migraine attack according to ICHD-3 criteria (i.e. ictal participants), those reporting a non-migraine headache or those experiencing no headache at all. Lastly, participants were classified according to their current use of preventive medication for migraine, as either using or not using preventive medication.
This classification system allowed for non-mutually exclusive assignments, enabling a single participant to be placed in multiple subgroups simultaneously. For example, one participant could be categorized as having episodic migraine, migraine with aura, experiencing a migraine attack at the time of sampling and using preventive medication. This approach was applied to capture a more comprehensive range of clinical presentations within our migraine population.
Outcomes and measures
The primary outcomes were the differences in serum GFAP and NfL concentrations between participants with migraine and HCs. The secondary outcomes extended the comparison to specific migraine subgroups, including (i) episodic migraine vs. chronic migraine vs. HCs; (ii) migraine without aura vs migraine with aura vs HCs; (iii) ictal participants vs headache-free participants vs HCs and (IV) current preventive medication use vs no preventive medication use vs HCs. The exploratory outcomes included evaluating potential associations between serum GFAP and NfL concentrations and various sociodemographic and clinical variables, such as age, sex, body mass index and hypertension.
Statistical analysis
The number of participants with migraine and HCs was determined by enrollment into the REFORM study. The pre-specified plan included analyzing at least 600 samples from participants with migraine and at least 150 from HCs.
Continuous variables are presented as the mean ± SD or median with interquartile range (IQR), depending on the data distribution, assessed visually using Q-Q plots and histograms. Categorical variables were presented as counts and percentages. To align with common practice in migraine research, MHDs, MMDs, monthly aura days and monthly days of acute headache medication use were reported as the mean SDs. For continuous data, unpaired t-tests or Mann–Whitney U-tests were used as appropriate to assess differences in sociodemographic and clinical characteristics between participants with migraine and HCs. For categorical data, Pearson's chi-squared tests were used.
Differences in serum GFAP and NfL concentrations between participants with migraine and HCs, and across migraine subgroups, were examined using analysis of covariance with serum biomarker as the outcome variable. Because residuals were not normally distributed, serum GFAP and NfL values were log-transformed, and results were thus presented as estimated percentual differences with 95% confidence intervals (CIs). Three models were applied: (i) an unadjusted model using only participant groups as covariate; (ii) a model additionally adjusted for age, sex, body mass index and smoking status; and (iii) a fully adjusted model that also included hypertension and other cardiovascular diseases. Our selection of potential confounders was guided by a review of the literature. 8
To further explore the associations between serum biomarkers and sociodemographic and clinical variables, univariate linear regression analyses were performed in the migraine population. Variables with a p ≤ 0.10 were then included in a multivariable regression model. For all regression analyses, we assessed regression diagnostics, assumptions and variance inflations factors. We additionally assessed the correlation between GFAP and NfL levels using Spearman's coefficient.
Statistical significance was considered at p ≤ 0.05 (two-sided) after applying Bonferroni correction for multiple comparisons. The correction was applied based on the total number of pairwise comparisons within each analysis (e.g. migraine with aura vs. migraine without aura, migraine with aura vs. healthy controls and migraine without aura vs. healthy controls, resulting in three pairwise comparisons). Complete case analysis was used, as missing covariate data accounted for less than 5%. All analyses were conducted using R Statistical Software, version 4.3.3 (R Foundation, Vienna, Austria).
Results
Participants
In total, 654 participants with migraine and 154 HCs were enrolled and provided blood samples. Among these, serum GFAP concentrations were measured in 641 participants with migraine and all 154 HCs, whereas serum NfL levels were measured in 644 participants with migraine and 138 HCs. Due to the presence of comorbid neurological disorders, we excluded 49 participants with migraine. Furthermore, we excluded two participants with extreme outlier values for either serum GFAP or NfL measurements (Figure 1). No serum levels were below the limit of quantification. The sociodemographic comparisons revealed that participants with migraine were slightly older than HCs (43.8 ± 12.2 vs 41.2 ± 11.8 years; p = 0.010) (Table 1). Sex distribution, body mass index and smoking status did not differ between groups (Table 1).
Study flow. GFAP = glial fibrillary acidic protein; MS = multiple sclerosis; NfL = neurofilament light chain. Demographics and clinical characteristics of the study participants and health controls. Abbreviations: BMI = body mass index; MHD = monthly headache days; MMD = monthly migraine days. *p-value for between-group differences.

In the migraine group, 385 participants (63.8%) had chronic migraine and 180 (29.9%) experienced migraine with aura. In total, 299 participants (49.6%) reported current use of preventive migraine medications. On average, participants with migraine experienced 18.2 ± 7.3 MHDs, 13.4 ± 6.5 MMDs and used acute headache medication on 11.8 ± 6.5 days/month. Among those with migraine with aura, the mean ± SD number of monthly aura days was 3.8 ± 5.0. Comorbid conditions were prevalent among participants with migraine, with 63 (10.4%) reporting hypertension, 36 (6.0%) other cardiovascular diseases, 57 (9.5%) anxiety disorders and 56 (9.3%) major depressive disorder. The mean ± SD disease duration of migraine was 24.5 ± 13.1 years.
Serum GFAP and NfL measurements
Figure 2 and Table 2 summarize serum GFAP concentrations in participants with migraine, including various subgroups, and in HCs. Serum levels of GFAP and NfL were moderately correlated (Spearman's rho = 0.47; p < 0.0001). The results of the linear regressions adjusted for age, sex, body mass index and smoking status are reported below. Further adjustments for history of hypertension and other cardiovascular conditions did not substantially alter the results for comparisons across any migraine subgroups. Our analyses revealed that participants with migraine had significantly higher serum GFAP levels than HCs (6.8%; 95% CI = 0.4–13.5%; p = 0.036). No significant differences were identified when comparing migraine with aura and migraine without aura (−0.7%; 95% CI = −7.7% to 6.9%; p > 0.99). Serum GFAP levels also did not differ when comparing migraine with aura to HCs (6.2%; 95% CI = −3.0% to 16.3%; p > 0.99) or migraine without aura to HCs (7.0%; 95% CI = −1.0% to 15.6%; p = 0.34). Moreover, no differences were found between those with episodic and chronic migraine (−2.7%; 95% CI = −9.3% to 4.4%; p > 0.99). Likewise, serum GFAP levels did not differ when comparing episodic migraine with HCs (8.6%; 95% CI =−0.4% to 18.5%; p = 0.21) or chronic migraine to HCs (5.7%; 95% CI = −2.3% to 14.4%; p = 0.82). The timing of blood collection relative to headache status did not affect serum GFAP levels because samples taken during a migraine attack showed no significant differences from those of headache-free participants or healthy controls (all p > 0.05). Furthermore, serum GFAP levels were not influenced by preventive medication use because no significant differences were found between participants who used preventive medication, those who did not, and HCs (all p > 0.05). Detailed results on the analysis of headache status at the time of blood sampling and preventive medication use are provided in the supplemental material (Tables S1a and S1b).
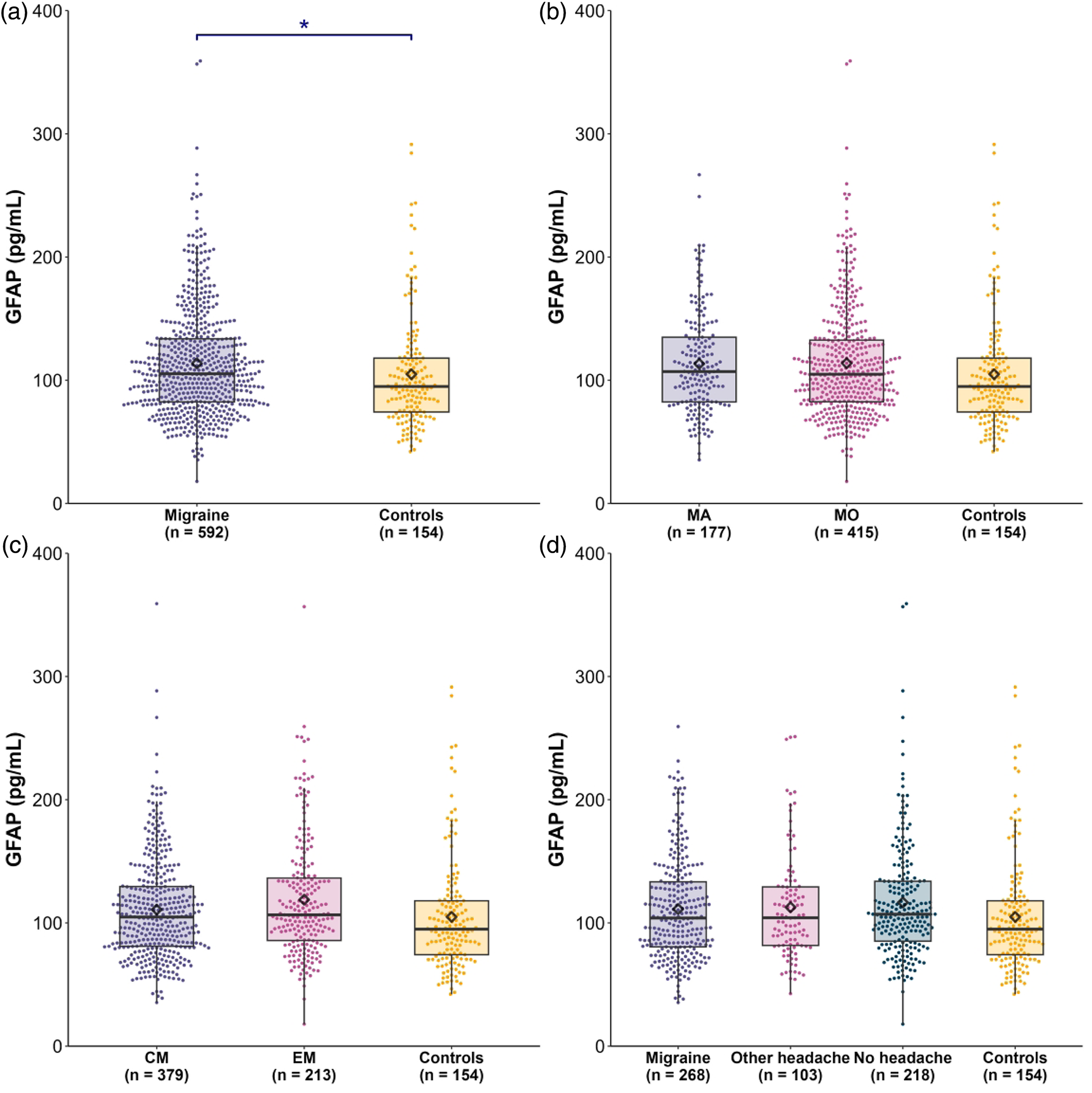
Serum glial fibrillary acidic protein (GFAP) levels in (a) participants with migraine and healthy controls (HCs), (b) migraine with aura (MA), migraine without aura and HCs, (c) chronic migraine (CM), episodic migraine (EM) and HCs and (d) migraine headache, other headache, no headache and HCs. The boxplot displays the median as a horizontal line with the interquartile range (Q1 and Q3) as the upper and lower edges of the box, and the mean as a diamond. Each dot represents an individual measurement. The asterisk indicates a significant difference (p < 0.05).
Serum GFAP levels in participants with migraine and healthy controls.
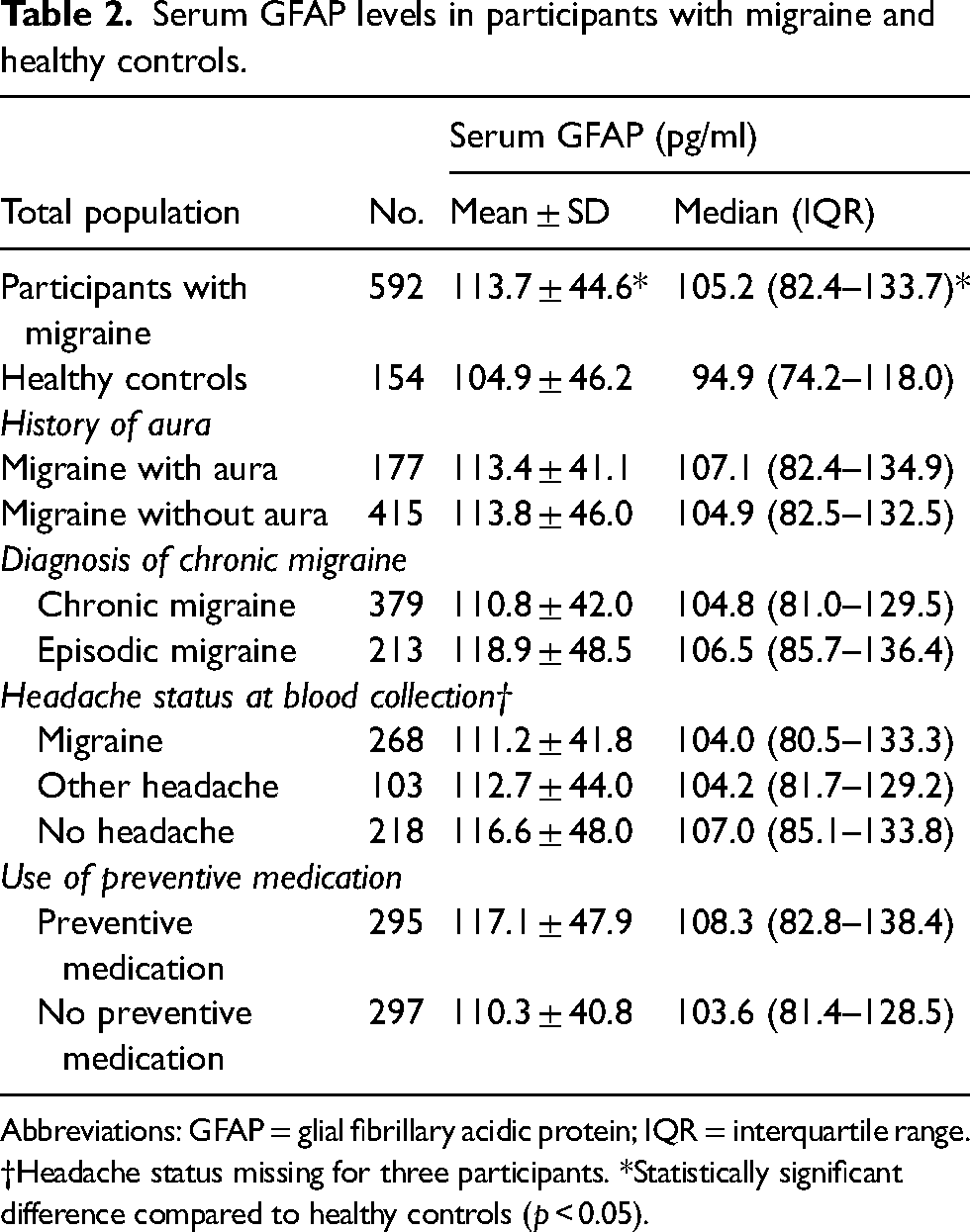
Abbreviations: GFAP = glial fibrillary acidic protein; IQR = interquartile range.
†Headache status missing for three participants. *Statistically significant difference compared to healthy controls (p < 0.05).
Serum NfL levels were similar in participants with migraine and HCs (−1.4%; 95% CI = −7.9% to 5.6%; p = 0.68). Furthermore, no significant differences were identified comparing migraine subgroups to HCs nor between migraine subgroups (Figure 3 and Table 3).
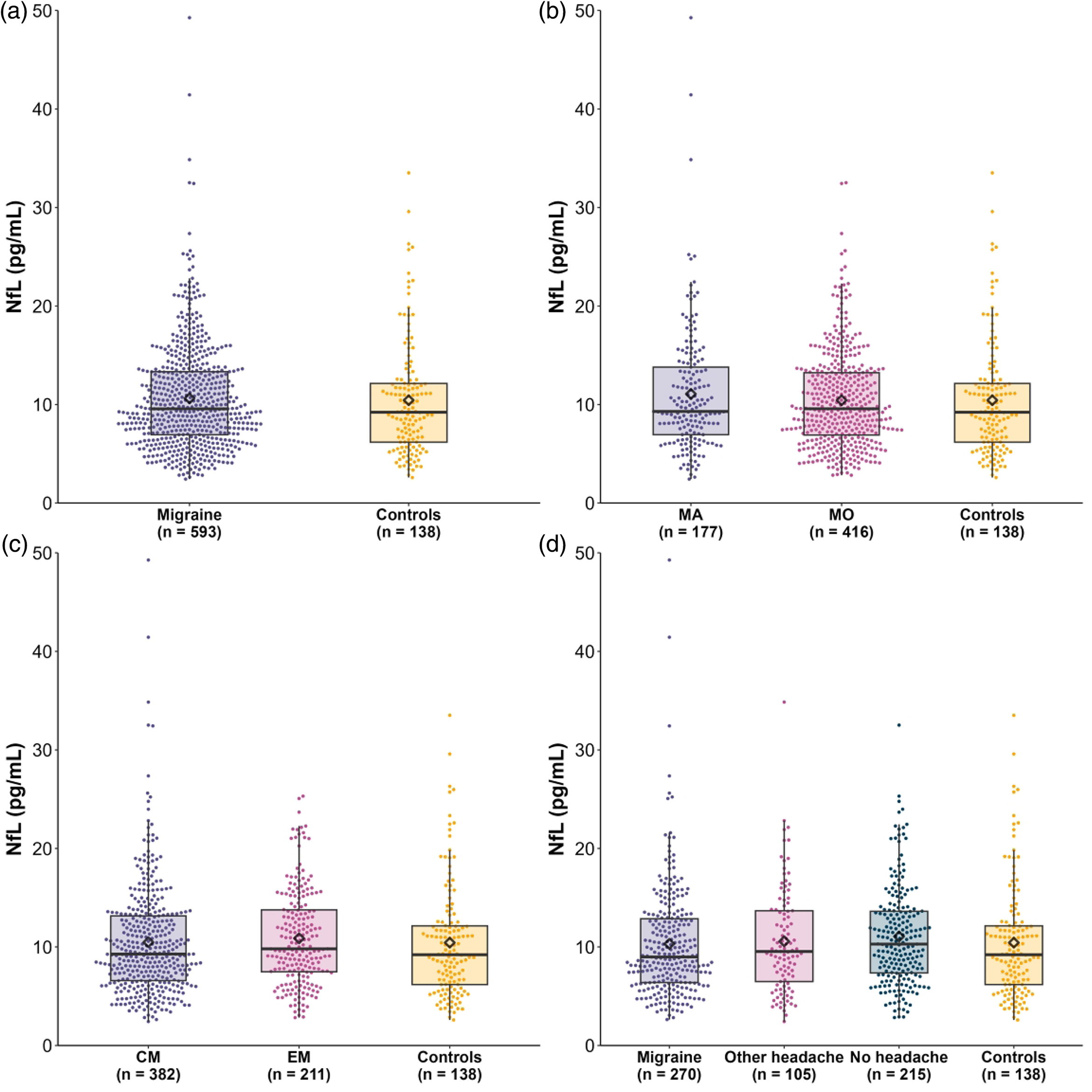
Serum neurofilament light chain (NfL) levels between (a) participants with migraine and healthy controls (HC), (b) migraine with aura (MA), migraine without aura and HC, (c) chronic migraine (CM), episodic migraine (EM) and HC and (d) migraine headache, other headache, no headache and HC. The boxplot displays the median as a horizontal line with the interquartile range (Q1 and Q3) as the upper and lower edges of the box, and the mean as a diamond. Each dot represents an individual measurement.
Serum NfL levels in participants with migraine and healthy controls.
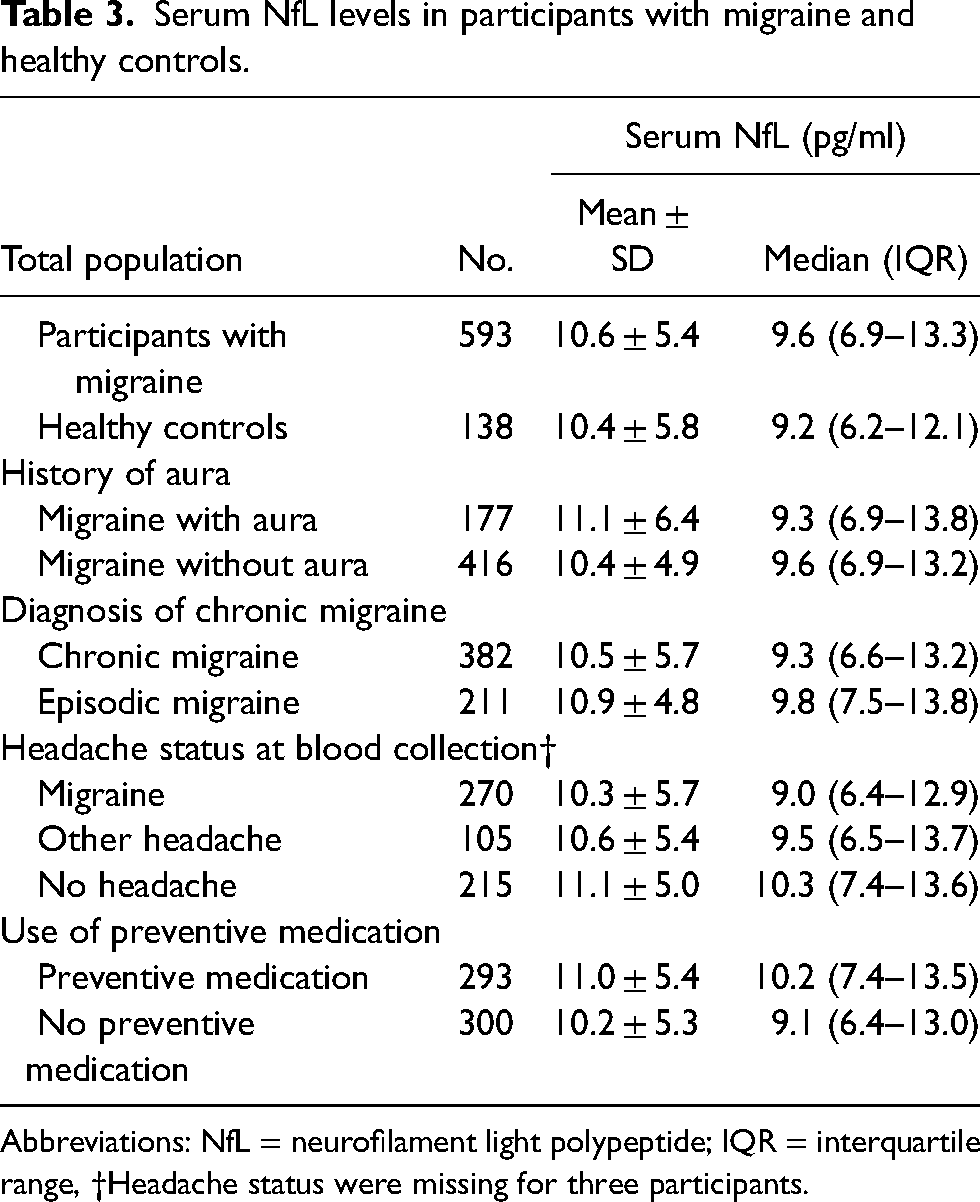
Abbreviations: NfL = neurofilament light polypeptide; IQR = interquartile range, †Headache status were missing for three participants.
Univariate and multivariable linear regression
Results of the univariate and multivariable linear regression are available in the supplemental material (Table S2a and Table S2b). The multivariable linear regression model revealed that each additional year of age was associated with a 1.3% increase in serum GFAP (95% CI = 1.0–1.6%; p < 0.0001) and a 2.6% increase in serum NfL (95% CI = 2.3–3.0%; p < 0.0001). By contrast, each 1 kg/m2 increase in body mass index corresponded to a 1.3% decrease in serum GFAP (95% CI = −1.8% to −0.7%; p < 0.0001) and a 1.3% decrease in serum NfL (95% CI = −1.9% to −0.7%; p < 0.0001). No other clinical variables, including migraine disease duration, were associated with serum GFAP or NfL levels in the multivariable analyses (see supplemental material, Table S2a and Table S2b).
Discussion
In this cross-sectional study, we demonstrated that serum GFAP concentrations were modestly but significantly elevated in adults with migraine compared to HCs. By contrast, serum NfL levels did not differ between groups. We had hypothesized that elevated serum GFAP and NfL might reflect underlying astroglial activation and subtle axonal damage, respectively. We also anticipated that these could vary according to migraine subtype or clinical status. Our findings only partially supported these expectations.
Although serum GFAP levels were higher in the migraine population as a whole, we observed no significant differences between the subgroups. These included episodic vs. chronic migraine, migraine with aura vs. without aura, ictal participants vs. headache-free participants and current preventive medication use vs. no preventive medication use. Likewise, serum NfL concentrations showed no discernable differences across these subgroups. Taken together, our results suggest that discrete astroglial alterations might be present in migraine, yet they appear not to depend on migraine frequency or aura diagnosis.
Few studies have previously evaluated GFAP levels in migraine, and notably, these investigations found no elevations in GFAP among migraine participants (3,4), potentially limited by their smaller sample sizes. Furthermore, prior studies reported no association with NfL in migraine participants (3,–5), consistent with our findings.
Elevated Serum GFAP in migraine
Our findings fit into a broader understanding of migraine as a complex neurological disorder involving multiple overlapping mechanisms (17). Previous research has underscored the roles of cortical hyperexcitability, altered sensory processing, and neuropeptidergic signaling (17,18). More recently, attention has turned toward neuroinflammation (2), including the contributions of glial cells in migraine pathophysiology (19,20). Astrocytes are critical for maintaining central nervous system (CNS) homeostasis and supporting synaptic function by clearing potassium and glutamate at the same time as preserving neuronal excitability (6). Elevated serum GFAP levels are commonly observed in disorders involving astrocytic activation or injury, such as multiple sclerosis and traumatic brain injury. 6 Our finding of modestly elevated GFAP suggests that astroglial alterations might be feature of migraine, though not as pronounced as in disorders with overt glial damage (6,7).
Preclinical experiments indicate that disruption of astrocytes might contribute to hypersensitivity to molecular migraine triggers by promoting hyperexcitability within the cingulate gyrus (21), a key pain-processing region implicated in migraine (22). Additional evidence from in vitro rodent experiments supports the role of astrocytic dysfunction (23,24). For example, injection of CGRP, comprising a well-established molecular migraine trigger, (17), into the trigeminal ganglion increased c-Fos expression in sensory neurons within the spinal trigeminal nucleus. This finding is notable because c-Fos serves as a surrogate marker of cellular activation, and GFAP expression in astrocytes within the spinal trigeminal nucleus was also reported (23). These observations suggest that astrocyte-to-neuron interactions may facilitate nociceptive signaling at the level of second-order trigeminal neurons. Thus, it is possible that astrocyte-to-neuron interactions might facilitate nociceptive messaging at the level of second-order trigeminal neurons.
Beyond the CNS, satellite glial cells (SGCs) in the peripheral nervous system also appear integral to migraine-related processes (24). SGCs enwrap sensory neurons in the trigeminal ganglion, providing structural and metabolic support, as well as potassium buffering (25). Under inflammatory conditions, SGCs can upregulate GFAP, mirroring astrocytic reactivity in the CNS (25).
Recent rodent studies have demonstrated that CGRP injection into the trigeminal ganglion induces GFAP upregulation in SGCs and thermal hyperalgesia (24). Likewise, GFAP upregulation is linked to impaired potassium buffering (26), which, in the trigeminal ganglion, contributes to pain behavior (27). These findings might therefore suggest that peripheral glial activation amplify nociceptive messaging at the level of first-order trigeminal neurons.
Considering all the above, it seems reasonable to propose that the modestly elevated serum GFAP levels might reflect persistent, low-grade glial activation at multiple sites of nociceptive messaging. This activation might exacerbate migraine pain by modulating the excitability of peripheral and central pain-processing neurons (28). Further work is needed to determine the full extent of glial involvement in the pathophysiological underpinnings of migraine. However, the lack of significant differences between participants with episodic and chronic migraine calls into question a direct relationship between elevated GFAP levels and pain signaling. It might be more plausible that this elevation reflects trait of migraine, rather than being dynamically influenced by disease progression. Translational research bridging clinical and preclinical studies will be important to understanding how astrocytes and SGCs interact with known molecular migraine triggers. Ultimately, a deeper understanding of glial contributions to migraine might open novel therapeutic avenues that target not only neural circuits but also the glial cells that modulate them.
NfL and the absence of detectable axonal injury in migraine
By contrast to GFAP, serum NfL levels were not elevated in participants with migraine compared to HCs. NfL is a sensitive marker of axonal injury and neuronal degeneration, and elevated levels are often observed in conditions characterized by substantial axonal damage, such as neurodegenerative diseases and inflammatory CNS disorders (9). The normal serum NfL levels found in people with migraine reinforces the premise that this disorder does not involve widespread or ongoing axonal damage.
Evidence from a recent diffusion tensor imaging study provides additional support for this perspective (29). The analysis showed no significant changes in microstructural white matter integrity between adults with migraine and age- and sex-matched HCs. That investigation evaluated fractional anisotropy, mean diffusivity, axial diffusivity and radial diffusivity across several major white matter tracts. No group differences were detected, and the investigators also observed no correlations between these diffusion tensor imaging parameters and clinical measures of migraine severity. This absence of detectable microstructural alterations aligns with normal NfL levels because elevated NfL often signals diffuse or progressive axonal injury. By contrast to these findings, some studies have reported microstructural white matter alterations in people with migraine (30,31). These discrepancies across neuroimaging studies can reflect variability in sample sizes, population heterogeneity and differences in analytical approaches.
The clinical implications of these findings are important. Our results, showing normal serum NfL levels, align with neuroimaging studies reporting preserved white matter microstructure. This suggests that migraine does not promote lasting damage to axonal fiber tracts. This observation bolsters the argument that axonal damage is not a central aspect of migraine pathophysiology. Normal serum NfL also reassures clinicians that migraine does not promote neurodegenerative processes. Future studies should examine whether distinct migraine subgroups predispose to axonal vulnerability. Longitudinal imaging investigations accompanied by detailed biomarker panels might offer further insights into the axonal landscape of migraine.
Influence of age and body mass index on biomarker levels
Under normal physiological conditions, GFAP and NfL enter the cerebrospinal fluid via the glymphatic system and cross into the bloodstream through the arachnoid villi (32,33). Disruptions in the blood–cerebrospinal fluid barrier, which become more common with older age (34), can promote this passage. Furthermore, biomarker concentrations in serum reflect the ratio of the analyte to the fluid volume, and this ratio can be influenced by physiological factors such as blood volume or analyte release. Elevated body mass index, for example, increases blood volume, diluting GFAP and NfL concentrations.
Our results align with well-known findings that older age is associated with higher serum GFAP and NfL levels, while higher body mass index corresponds to lower levels of these biomarkers (6,8,16,35). These expected associations support the validity of our data, and adjustments for these variables were included to ensure accurate interpretation.
Strengths and limitations
A major strength of the present study is the large sample size, which represents, to our knowledge, the most extensive investigation to date of GFAP and NfL biomarkers in people with migraine. Furthermore, the study benefits from a well-characterized participant cohort and the use of the ultra-sensitive SiMoA platform.
Despite these strengths, several limitations warrant consideration. The cross-sectional design hinders the assessment of within-person variations in serum GFAP and NfL levels during ictal and interictal phases of migraine. Minor age differences between the migraine group and HCs, with the migraine group being slightly older, were observed. To mitigate potential confounding effects, we adjusted for age in the analyses, particularly because age is known to correlate with elevated GFAP and NfL levels. However, it is possible that residual confounding may be present, and it is necessary to consider the possibility that the healthy controls could be healthier on parameters beyond migraine pathophysiology, such that the absence of other non-migraine comorbidities may contribute to their lower GFAP levels. Although we systematically inquired about all comorbidities, biochemical screening, including renal function assessment, was not performed. This is a potential limitation because previous studies have shown an association between serum GFAP, NfL and kidney function (36). Moreover, serum biomarker measurements via antecubital blood sampling might provide results that differ from other biofluids, such as the cerebrospinal fluid. Lastly, the predominance of white participants recruited from a single tertiary care clinic, characterized by high disease burden, introduces the potential for selection bias. Although this cohort may be well-suited for identifying migraine pathophysiology, similar GFAP elevations may not be generalizable to the broader migraine population with less severe disease.
Conclusions
Among adults with migraine, serum GFAP levels were modestly elevated compared with HCs, whereas serum NfL concentrations showed no significant differences. These findings suggest that low-grade glial activation, likely involving astrocytes within the CNS and SGCs in the peripheral nervous system, might contribute to the pathophysiological underpinnings of migraine. This insight underscores the need for further research to investigate the implications of elevated serum GFAP, particularly its role in the underlying mechanisms of the disease.
Serum GFAP concentrations were modestly elevated in adults with migraine compared to HCs, whereas serum NfL levels showed no significant difference. Neither serum GFAP, nor NfL levels were associated with migraine subtype or clinical status. Low-grade glial activation may contribute to the underlying pathophysiology of migraine.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251338178 - Supplemental material for Serum GFAP and NfL levels in migraine: A registry for migraine (REFORM) study
Supplemental material, sj-docx-1-cep-10.1177_03331024251338178 for Serum GFAP and NfL levels in migraine: A registry for migraine (REFORM) study by Emil Gozalov, William K. Karlsson, Rune H. Christensen, Haidar M. Al-Khazali, Malene B. Hansen, Dorte A. Olsen, Helle B. Søndergaard, Finn Sellebjerg, Jonna S. Madsen, Messoud Ashina and Håkan Ashina in Cephalalgia
Footnotes
Acknowledgements
We thank all the study participants and the staff at the Human Migraine Research Unit, Danish Headache Center.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: WKK has received a speaker's honorarium from Pfizer and Lundbeck outside of the submitted work. RHC has received personal fees from Teva, The Lundbeck Foundation and research travel funding from Augustinus Fonden. HMA reports receiving personal fees from Pfizer, outside of the submitted work. FS has served on scientific advisory boards for, served as consultant for, received support for congress participation or received speaker honoraria from Biogen, Bristol Myers Squibb, Lundbeck, Merck, Novartis, Roche and Sanofi Genzyme. His laboratory has received research support from Biogen, Merck, Novartis, Roche and Sanofi. MA is a consultant, speaker or scientific advisor for AbbVie, Amgen, Astra Zeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva, and a primary investigator for ongoing AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis and Teva trials; has no ownership interest and does not own stocks of any pharmaceutical company; and serves as associate editor of the Journal of Headache and Pain, and associate editor of Brain. HA reports having received personal fees from AbbVie, Lundbeck, Pfizer, and Teva, outside of the submitted work. The remaining authors report no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from the Lundbeck Foundation (R403-2022-1352 to HA and R310-2018-3711 to MA).
Data availability
Upon reasonable request, the corresponding author will provide the necessary data and materials to interested researchers for the purpose of academic scrutiny, reproducibility and further scientific investigation.
Ethical statement
Before undertaking any study-related tasks or procedures, written informed consent was obtained from every participant with migraine and HC.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
