Abstract
Background
Damage of the optic nerve is the major complication of idiopathic intracranial hypertension. A biomarker indicative for optic nerve damage would help identifying high-risk patients requiring surgical procedures. Here, we studied the potential of cerebrospinal fluid neurofilament to predict idiopathic intracranial hypertension-induced optic nerve damage.
Methods
In two centers, serum and cerebrospinal fluid of 61 patients with clinically suspected idiopathic intracranial hypertension were prospectively collected. Neurofilament concentrations were measured and related to ophthalmological assessment.
Results
The average cerebrospinal fluid neurofilament concentration in patients with moderate and severe papilledema was increased compared to patients with minor and no papilledema (1755 ± 3507 pg/ml vs. 244 ± 102 pg/ml; p < 0.001). Cerebrospinal fluid neurofilament concentrations correlated with the maximal lumbar puncture opening pressure (r = 0.67, p < 0.001). In patients fulfilling the Friedman criteria for idiopathic intracranial hypertension with or without papilledema (n = 35), development of bilateral visual field defects and bilateral atrophy of the optic nerve were associated with increased average age-adjusted cerebrospinal fluid neurofilament concentrations. At last follow-up (n = 30), 8/13 of patients with increased, but only 3/17 with normal, cerebrospinal fluid neurofilament had developed bilateral visual field defects and/or bilateral optic nerve atrophy resulting in a sensitivity of 72.7% and a specificity of 73.7% of cerebrospinal fluid neurofilament to detect permanent optic nerve damage.
Conclusions
Cerebrospinal fluid neurofilament is a putative biomarker for optical nerve damage in idiopathic intracranial hypertension.
Introduction
Idiopathic intracranial hypertension (IIH) is a rare disease, which is characterized by elevated pressure of the cerebrospinal fluid (CSF). The reported incidence is 1:100,000 in the general population and up to 20-fold higher in obese women of childbearing age (1–4). However, over the last years, an increase of the incidence in the general population has been reported due to pandemic obesity (1,2).
Major symptoms comprise headache and impairment of the optic nerve function by pressure-induced papilledema causing blurred vision, photopsia, transient visual obscuration, diminishing visual fields, and blindness – if not treated in time – as well as nausea, vomiting, dizziness, pulsatile tinnitus, and abducens nerve palsy (5,6). Outcome is often characterized by persistent severe headache as well as visual (7) and cognitive impairment (8).
The diagnosis is made based on the revised Friedman Criteria (9), basically requiring i) normal neurological examination (aside from abducens nerve palsy); ii) normal CSF composition; iii) elevated lumbar puncture opening pressure (defined as ≥25 cm H2O in adults); and iv) normal brain parenchyma and venography. Depending on the presence of papilledema or abducens nerve palsy, IIH is classified as definite IIH, probable IIH, IIH without papilledema (IIHWOP) or suggested IIHWOP (9). Female patients with body mass indices >30 are referred to as “typical IIH”, patients with “atypical IIH” have a lower body mass index and/or male sex (5,10). However, about 6% of the patients present clinically with symptoms of IIH, including some neuro-imaging features of IIH, but without papilledema or abducens nerve palsy and thus do not meet the current diagnostic criteria (11). So far, it is unknown if these patients suffer from a clinical pre-stage of IIH/IIHWOP and thus are at risk to develop impairment of the optic nerve.
Therapy comprises treatment with acetazolamide (12) or topiramate (13) in association with weight loss and is adapted according to the ophthalmological findings (papilledema, visual field defect, abducens nerve palsy). If there is a risk for optic nerve damage with severe and irreversible visual field defects or loss of vision, surgical interventions (particularly optic nerve sheath fenestration or shunting) are used (5,14).
The clinical presentation of the disease is heterogeneous and often not correlating with the objective findings such as lumbar puncture opening pressure and papilledema. Currently, it is not possible to predict if a patient will respond to medical treatment, or which patients may develop severe permanent visual loss. Papilledema, the only non-invasive objective measurable treatment response, develops and disappears with substantial delay compared to intracranial pressure. Therefore, an objective tool indicating permanent optic nerve damage is sorely needed and will help guide treatment and predicting disease outcome.
High-grade papilledema (15), optic disc hemorrhages (16), visual field mean deviation (17), and decreased visual acuity at presentation (18) are proposed clinical predictors for worse visual outcome. However, as the permanent damage of the optic nerve may not only depend on the severity of the papilledema but also on its duration, biomarkers indicating permanent optic nerve damage and the need for immediate surgical intervention are required. So far, few potential biomarkers have been evaluated (19,20). However, no biomarkers with diagnostic, prognostic or predictive value have been established for routine use.
Neurofilament light chain (NfL) is part of the axonal cytoskeleton, providing structural support for the axon and regulating the axon diameter. It is released in case of cellular damage or neuronal degeneration and became an important biomarker in different neurological disorders such as amyotrophic lateral sclerosis or multiple sclerosis (21–23). Further, NfL has been recently proposed as a biomarker of visual outcome after optic neuritis (24,25). After release due to neuronal damage, it slowly normalizes when neuronal damage ceases (26).
So far, NfL has not been investigated in the context of IIH. We have studied 61 patients from two different centers with clinically suspected IIH aiming at defining the potential of NfL as a biomarker for permanent optic nerve damage.
Methods
Patients, protocol approvals and patient consents
Sixty-one consecutive patients with clinically suspected IIH (a combination of headache, subjective visual disturbances and obesity) were recruited from January 2018 at the Department of Neurology, Odense University Hospital (OUH, n = 38) and since November 2018 in the Danish Headache Center (DHC), Rigshospitalet-Glostrup (n = 23). All patients were ≥18 years old and gave written informed consent. The study was approved by the Ethics Committee of Southern Denmark (VEK S-20170058) and the Danish Data Protection Agency (nr. 16/20512, 06.06.2016 and 07.04.2017). Data reporting was in line with the STROBE (Reporting of Observational Studies in Epidemiology) recommendations (27). All patients received neurological and ophthalmological examination, routine blood collection, lumbar puncture including routine CSF status, cerebral MRI and cerebral venography (CT or MRI) (9). CSF and serum samples were collected following international guidelines for biobanking, aliquoted in 500 µl Sarstedt polypropylene tubes and stored at −80°C until analysis. Details of headache and visual disturbances, medical history, medication, and vital signs (including weight and height) were recorded (5,9). Table 1 provides an overview of the demographics and the clinical patient characteristics. Supplemental Figure 1 provides an overview of the diagnostic workflow used.
Demographics and clinical characteristics of the patients.

Ophthalmological assessment
All patients were assessed in the Department of Ophthalmology, OUH or Rigshospitalet- Glostrup by senior consultants. Visual acuity, visual fields (by automated perimetry), ophthalmoscopy, and papillary optical coherence tomography (OCT) were performed.
Heidelberg Spectralis OCT (Heidelberg Engineering, Heidelberg, Germany) was used to record the posterior segment of the eye. The macula and optic nerve head were imaged with Heidelberg Spectralis eye tracking and automated real-time averaging features. Scans were repeated until a good quality – as assessed by the responsible ophthalmologist – was obtained. Furthermore, the signal-to-noise ratio had to be >25 dB. The scanning protocol included one 20° foveal-centered scan (512 × 496, 5.9 mm). The scans were recorded with 49 frames to ensure a high resolution. Circle scan 12.0° (3.5 mm) was used for retinal nerve fiber layer (RNFL) measurement. The ganglion cell layer (GCL) measurement and RNFL were measured using the instrument manufacturer software Heidelberg Eye Explorer (Heidelberg Engineering, Heidelberg, Germany). The semi-automatic segmentation method for GCL measurement was used. Optic nerve atrophy was determined based on optical coherence tomography and ophthalmoscopy.
Papilledema was graded according to Frisen’s scale and transformed into mild (Frisen Scale grade 1), moderate (grade 2,3) and severe (grade 4,5) for the analysis (28). If the eyes were differentially affected, the grading of the most severely affected eye was used. At the OUH, visual fields were performed using Humphrey 24-2 (Carl Zeiss) computer perimetry. At the DHC, Haag-Streit 30-2 (Octopus) computer perimetry was used. Visual field defects were defined as an average perimetric mean deviation of less than −2 dB. Moreover, the perimetric results were always interpreted by the assessing ophthalmologist differentiating real visual field defects from defects caused by insufficient cooperation to the investigation. In patients with complete remission of papilledema at last follow-up, permanent optic nerve damage was assessed and defined as persistent bilateral optic nerve atrophy and/or bilateral visual field defects.
Lumbar puncture
Lumbar puncture was performed in line with in-hospital standard operation procedures with the patients lying in lateral decubitus position with extended legs. Lumbar puncture opening pressure was determined using a manometer after at least 1 min relaxation. Lumbar puncture was performed as part of the diagnostic procedure and as therapeutic supplement in case of risk for optic nerve damage until sufficient medical or surgical treatment was achieved. Maximal pressure was defined as the maximal lumbar puncture opening pressure measured in the course of disease of the respective patient. Lumbar puncture opening pressure at sampling was defined as the opening pressure measured at the time of sample collection. Lumbar puncture was always supplemented by ophthalmological assessment at the same stage of disease.
NfL measurement
Analyses were carried out at the Department of Biochemistry and Immunology, Lillebaelt Hospital, Vejle, a routine laboratory accredited by the Danish Accreditation Fund (DANAK) according to the ISO 15189 standard, which specifies requirements for quality and competence in medical laboratories. A commercially available NfL kit (Quanterix©, Lexington, MA, USA) for the Single Molecule Array (Simoa) HD-1 Analyzer (Quanterix) was used to quantify NfL light chain in serum and CSF according to the manufacturer’s procedure. Quality control was performed using two controls prepared from commercially available control material provided by the manufacturer. Moreover, in-house serum and CSF pools were used as internal controls and included in each assay for evaluating assay performance. The total coefficient of variation was <12%. Age adjusted NfL values in CSF were determined according to Vagberg et al. (29), corresponding to the local reference intervals (30). Increased cNfL concentrations were defined as >50% increase as compared to age-adjusted normal values. Normal cNfL was defined as <150% of age-adjusted normal values.
Statistical analysis
The groups were compared using the Mann-Whitney test for continuous variables and Chi-square test or Fisher’s exact test for categorical data. For correlation analyses, Pearson correlation was used, the significance level used for all analysis was 0.05. All statistical analyses were performed using SPSS 24 (IBM, Aarhus, Denmark).
Results
NfL concentration, lumbar puncture opening pressure and papilledema
A total of 61 patients with clinically suspected IIH were included (OUH: n = 38, DHC: n = 23). The patients’ characteristics are given in Table 1 and in Supplemental Table 1. The NfL concentration in the CSF (cNfL) correlated with NfL concentration in the serum (r = 0.93, p < 0.001, Figure 1(a)) and with the maximal lumbar puncture opening pressure (r = 0.67; p < 0.001, Figure 1(b)). Further, a less significant correlation was seen with the lumbar puncture opening pressure at sampling (r = 0.6, p = 0.001, data not shown).
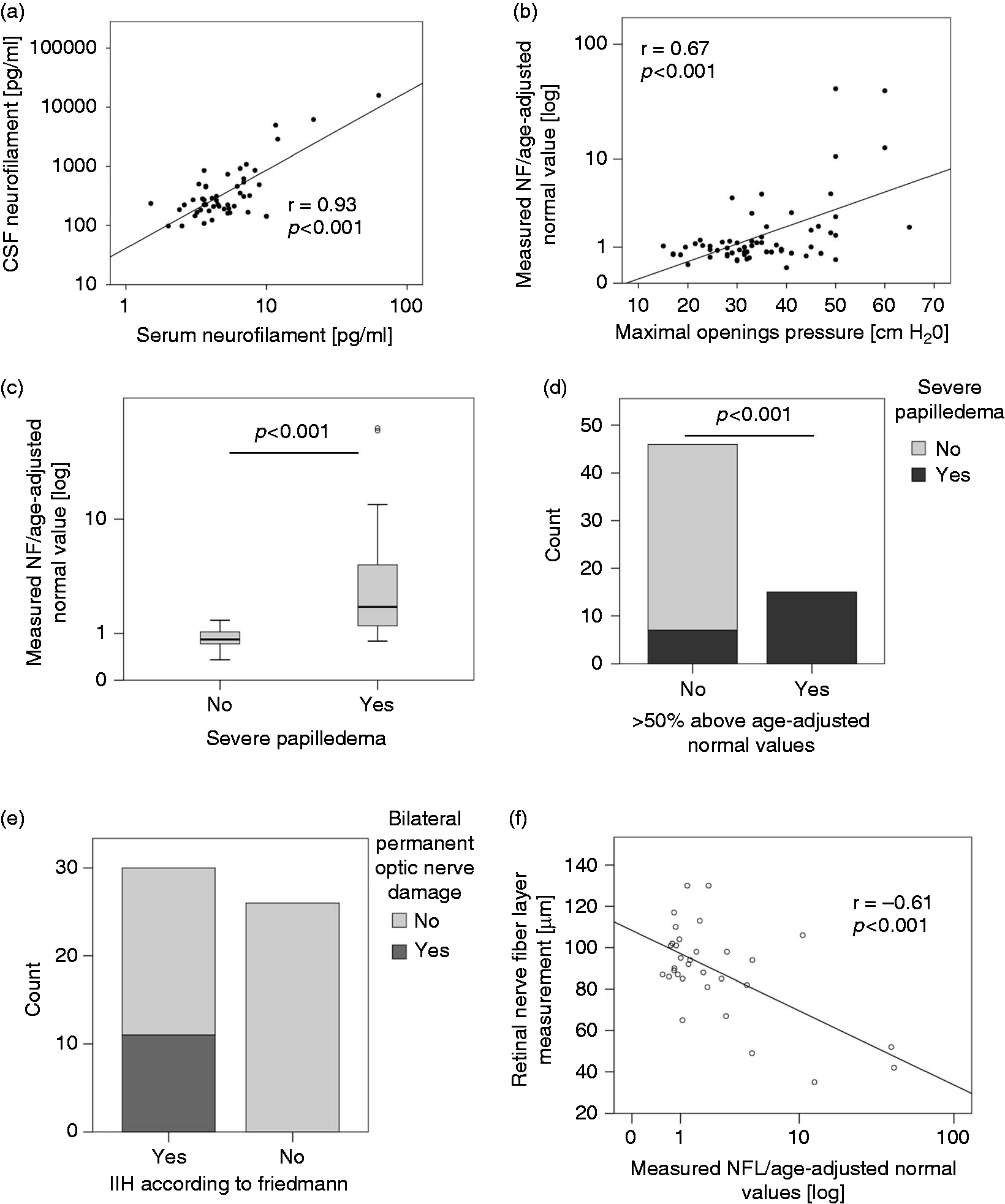
Neurofilament in the cerebrospinal fluid. The results given were based on the analysis of all patients with clinically suspected IIH from both centers (n = 61). (a) Correlation of serum NfL and cNfL (Pearson correlation). (b) Correlation of cNfL and maximal opening pressure at any time point during the disease (Pearson correlation). (c) Median ratio of measured cNfL and age-adjusted normal values in patients without or mild papilledema and in patients with moderate-severe papilledema (Mann-Whitney U test). (d) Absolute number of patients with and without moderate-severe papilledema according to neurofilament concentrations. Papilledema of patients with normal or increased cNfL concentrations is given. Normal cNfL was defined as < 150% of age-adjusted normal values (χ2 test). (e) The absolute number of patients fulfilling the Friedman criteria for IIH/IIHWOP with and without bilateral permanent optic nerve damage is given. (f) Correlation of RNFL on OCT and the median ratio of measured cNfL and age-adjusted normal values (Pearson correlation).
Association of papilledema and cNfL
In patients with clinically suspected IIH, the average ratio of cNfL concentrations to age-adjusted normal values was 6.55 in patients with moderate to severe papilledema as compared to 0.87 for patients with mild or no papilledema (p < 0.001, Mann-Whitney U test, Figure 1(c)). An increase of cNfL concentrations of >50% compared to age-adjusted normal values was found in 15 out of 61 patients and associated with the presence of at least moderate papilledema in the majority of patients (p < 0.001, Figure 1(d)).
Ophthalmological long-term outcome and cNFL
Permanent optic nerve damage only developed in patients fulfilling the Friedman criteria for IIH/IIHWOP (Figure 1(e)). Thickness of the RNFL measured by OCT and the average-ratio of cNfL concentrations to age-adjusted normal values showed a significant inverse correlation (r = 0.61; p < 0.001, Figure 1(f)). In patients fulfilling the Friedman criteria for IIH/IIHWOP (n = 35), ophthalmological long-term outcome (i.e. permanent optic nerve damage) was assessed. At timepoint of data closure, five patients still suffered from persistent papilledema; data on long-term outcome were available for 30 patients (Table 2). The maximal lumbar puncture opening pressure correlated significantly with the occurrence of bilateral visual field defects (p = 0.02, Figure 2(a)) but not with bilateral atrophy of the optic nerve (p = 0.51, Figure 2(b)). In contrast, the average-ratio of cNfL concentrations to age-adjusted normal values correlated significantly with the occurrence of bilateral visual field defects (p = 0.01, Figure 1(c)) and was associated with bilateral atrophy of the optic nerve (p = 0.06, Figure 1(d)). At last follow-up, 8/13 of patients with increased but only 3/17 with normal cNfL had developed bilateral visual field defects and/or bilateral optic nerve atrophy, resulting in a sensitivity of 72.7% and a specificity of 73.7% (positive predictive value: 61.5% / negative predictive value: 82.4%) of cNfL to detect permanent optic nerve damage (Figure 2(e),(f)).
NfL and permanent optic nerve damage in patients fulfilling the Friedman criteria for IIH/IIHWOP (n = 35).


Association of acute and chronic optic nerve damage with neurofilament concentrations in the CSF. The results given were based on the analysis of patients with complete remission of papilledema that fulfilled the Friedman criteria for IIH/IIHWOP (n = 30). (a)–(d) Permanent optic nerve damage defined as bilateral atrophy of the optic nerve and/or bilateral visual field defects at last follow up in all patients with available follow-up data (p-values: Mann-Whitney test). (a,b) Mean maximal opening pressure (±standard deviation) in patients with and without bilateral visual field defects (a) and bilateral atrophy of the optic nerve (b) (Mann-Whitney U test). Mean ratio of measured cNfL and age-adjusted normal values (± standard deviation) in patients with bilateral visual field defects (c) and bilateral atrophy of the optic nerve (d) (Mann-Whitney U test). (e,f) Proportion of patients with cNfL concentrations > 50% above age-adjusted normal values depending on the presence of bilateral visual field defects (e) or bilateral atrophy of the optic nerve (f) (Fisher’s exact test).
Discussion
Here we show for the first time a strong association of cNfL concentrations with increased lumbar puncture opening pressure, moderate or severe papilledema and permanent optic nerve damage in patients with IIH. Even though the overall number of patients studied was relatively low, the statistical associations were highly significant indicating a substantial and clinically relevant link between IIH-induced optic nerve damage and cNfL concentrations.
The release of NfL into the CSF after axonal damage or degeneration was observed in different neurological disorders such as amyotrophic lateral sclerosis or multiple sclerosis (31–33). We did not study the pathophysiological mechanisms behind the observed associations. However, both the association of permanent optic nerve damage and cNfL concentrations, and the significant correlation of cNfL with the maximal lumbar puncture opening pressure, support the concept of pressure-induced axonal damage of the optic nerve resulting in cNfL release in the CSF and permanent optic nerve damage.
We found a highly significant association between NfL concentrations in the serum and the CSF. However, the serum concentrations hardly exceeded the previously described normal values (25). Similar to other diseases (23), serum NfL concentrations are not sensitive enough for diagnostic purposes in IIH. Further prospective studies monitoring the serum NfL are needed to evaluate whether the increase of serum NfL in patients not yet suffering from papilledema can be used as a non-invasive biomarker for progressive papilledema and risk of optic nerve damage.
One major limitation of the study was the timing of CSF sampling. Samples were taken at different time points during the disease; some samples were taken at initial diagnosis, some samples later in the disease course; for example, when reoccurrence was suspected. Another problem is the vague definition of IIH onset, some patients might have symptoms of IIH for many months before diagnosis, others were diagnosed rapidly. Further, some patients received very aggressive therapy due to rapid referral to a highly specialized center, while others were treated insufficiently for months. This inter-individual variability likely explains why some patients with increased NF did not suffer from permanent optic nerve damage, while others had long-term sequelae despite normal cNfL. We expect the observed effects to become even more evident and increasingly accurate, when looking at selected subgroups with ongoing pressure-induced optic nerve damage only. The size of our prospective cohort is, however, not sufficient for these subgroup analyses and additional studies are needed to support this assumption.
Although all patients in both cohorts received a lumbar puncture due to suspicion of IIH, not all patients were diagnosed with IIH at final assessment. However, these patients were optimal controls given that all of them received the lumbar puncture due to clinical suspicion of IIH. The comparison of these patients with those in whom the IIH diagnosis was finally confirmed shows the value of cNfL to support an IIH diagnosis. Interestingly, none of the controls had increased cNfL despite increased lumbar puncture opening pressure being observed in several patients. This suggests that the increase of cNfL resulted from damage to the optic nerve only, but not from a diffuse cortical neuronal damage caused by the elevated intracranial pressure.
Conclusion
In summary, we conclude that cNfL is a promising marker for the risk of permanent optic nerve damage in patients with probable or established IIH/IIHWOP. Still, several open questions (e.g. time course of release) remain and additional prospective studies are certainly required to establish NfL as a usable biomarker for assessing the severity of IIH and optimizing its treatment.
Clinical implications
Neurofilament light chain is associated with intracranial opening pressure and acute papilledema in IIH. Neurofilament light chain is a promising biomarker for permanent optic nerve damage in IIH. Neurofilament light chain might help in identifying patients in need of immediate surgical interventions.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420944866 - Supplemental material for Neurofilament light chain as biomarker in idiopathic intracranial hypertension
Supplemental material, sj-pdf-1-cep-10.1177_0333102420944866 for Neurofilament light chain as biomarker in idiopathic intracranial hypertension by Dagmar Beier, Johanne Juhl Korsbæk, Jonna Skov Madsen, Dorte Aalund Olsen, Laleh Dehghani Molander, Snorre M Hagen, Charlotte Teunissen, Christoph P Beier and Rigmor Højland Jensen in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_0333102420944866 - Supplemental material for Neurofilament light chain as biomarker in idiopathic intracranial hypertension
Supplemental material, sj-pdf-2-cep-10.1177_0333102420944866 for Neurofilament light chain as biomarker in idiopathic intracranial hypertension by Dagmar Beier, Johanne Juhl Korsbæk, Jonna Skov Madsen, Dorte Aalund Olsen, Laleh Dehghani Molander, Snorre M Hagen, Charlotte Teunissen, Christoph P Beier and Rigmor Højland Jensen in Cephalalgia
Footnotes
Acknowledgements
The authors would like to thank Dr. Helle Hvilsted Nielsen, Cathrine Brødsgaard Nielsen, Kaja Buchholt Lund and the doctors and nurses of both departments for their precious support of this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by a grant of the Odense University Hospital/Danish National Hospital to DB and RHJ (25-A1320 (2016) and 69-A3346 (2019)) and by the Region of Southern Denmark to CPB (17/18517).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
