Abstract
Background
The anti-calcitonin gene-related peptide (CGRP), or its receptor (CGRP/R) monoclonal antibodies (mAbs), offer targeted, effective, and tolerated drugs for migraine. However, about 25% of patients fail to achieve a clinically meaningful response, usually leading to discontinuation. These patients often have a lengthy migraine history and multiple prior preventive treatment failures, resulting in limited therapeutic options. Herein, we describe the cause for and outcome of withdrawal of anti-CGRP/R mAb and evaluate the treatment course until discontinuation.
Methods
We conducted a prospective analysis on migraine patients attending the Florence Headache Center in Italy, who discontinued treatment with anti-CGRP/R mAbs. The primary objectives were to describe the reasons for anti-CGRP mAbs discontinuation and the treatment course. Secondary objectives were the evaluation of the absolute change from baseline in monthly headache days, response rates, persistence in medication overuse, absolute change from baseline of the overall number and days of analgesics use per month, change of MIDAS and HIT-6 at three, six, and 12 months, and the last month of treatment.
Results
Among 472 patients, 136 (28.8%) discontinued mAb treatment after an average of 9.0 ± 6.1 (mean ± SD) months. The majority (96/136, 70.6%) discontinued due to ineffectiveness, followed by lost to follow-up during treatment (18/136, 13.1%) and adverse events (10/136, 7.3%). In total, 77.9% of the 136 patients ceased treatment within the first year. Following discontinuation, 48.5% initiated new pharmacological treatment, 39.7% were lost to follow-up, and 11.8% opted not to start another treatment. The majority of patients that started a new pharmacology treatment switched to another anti-CGRP/R (46/68, 67.6%). The second most-used treatment was onabotulinumtoxinA (7/68, 10.2%; all patients in this subgroup were naïve to this treatment), followed by an anticonvulsive medication (7/68, 10.2%). The response status (≥50% reduction in monthly headache days) was achieved by 30.5%, 34.6%, and 40.0% of patients at month 3, 6, and 12 of treatment, respectively. Considering only the comprehensive last month of treatment before withdrawn the percentage of responders was 16.9%.
Conclusion
Although anti-CGRP/R mAbs have provided a substantial amelioration of migraine management, a relevant proportion of patients remains unresponsive and requires additional therapeutic support. Further research is required to identify non-responder features and address unmet needs in migraine treatment.
This is a visual representation of the abstract.
Introduction
The introduction of monoclonal antibodies (mAbs) against calcitonin gene-related peptide (CGRP) or its receptor (CGRP/R) has substantially changed migraine treatment, providing target-specific, effective, and well-tolerated drugs (1). Notwithstanding, about 25% of patients do not achieve a clinically meaningful response, leading to treatment discontinuation (2–4). Due to the recent introduction of these novel medicines and restrictions in the reimbursement policy, most patients treated with anti-CGRP/R mAbs present with high disability, a long migraine history, and several prior unsuccessful preventive treatment attempts. Therefore, if these patients discontinue treatment with anti-CGRP/R mAbs, it is challenging to find an adequate and promising subsequent treatment (2). In addition, there are reports that these patients may be less adherent to oral treatment, distrustful of beneficial effects of new treatments, and more likely to refuse control visits and further therapies (5–7).
Therefore, a better characterization of these patients and understanding of their unmet needs is crucial in order to optimize their clinical management and identify novel therapeutic prospectives, ultimately reducing the stigma of patient refractory to pharmacological treatments. Recent real-world studies have evaluated the response and the predictors of response for anti-CGRP/R mAbs, with ≥50% responder rates ranging from approximately 40% to 70% (8,9), with non-responders more likely to report psychiatric comorbidities or other relevant comorbidities (9). One study has specifically assessed differences between super non-responders and responders to anti-CGRP/R mAbs, evaluating the outcome of 26 patients that withdrew treatment (3), highlighting the challenge in treating patients with ineffectiveness to anti-CGRP mAbs. Indeed, these patients had limited pharmacological choices as deciding to continue treatment (due to subjective improvement), switching anti-CGRP/R mAbs, or trying drugs with lower evidence in migraine is based on clinical practice, with no consensus available so far. Although several studies evaluated the effectiveness of anti-CGRP/R mAbs and collaterally reported the percentage of non-responders (with discordant results), no studies have been performed focusing on withdrawn and subsequent treatments. These data could be useful to identify non-responder features and address unmet needs in migraine treatment. Herein, we report the outcome of a larger cohort of patients who permanently discontinued an anti-CGRP/R mAb with the aim of describing treatment effectiveness, causes of withdrawal, and subsequent course of management.
Materials and methods
Study design and variables collected
We performed a prospective, descriptive analysis on all outpatients who started treatment with an anti-CGRP/R mAb (i.e., erenumab, galcanezumab, or fremanezumab) from October 2019 to March 2023 at the Headache Center of the Careggi University Hospital in Florence, Italy. Eptinezumab was not available at the time of the study. All patients signed informed consent before treatment with an anti-CGRP/R mAb. The study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines and is part of the Registro Italiano Cefalee (RICe) study, approved by the local Ethics committee of the Careggi University Hospital (CEAVC Studio RICe, 14591_oss and subsequent amendments 2022-609).
For this analysis, we selected patients who permanently discontinued treatment from the first anti-CGRP/R mAb (erenumab 70–140 mg monthly; galcanezumab 240 mg first dose and 120 mg monthly; or fremanezumab 225 mg monthly) until March 2023. Study participants were older than 18 years with episodic migraine (EM) or chronic migraine (CM), with or without medication overuse (MO), according to ICHD-3 (10) criteria. Before the anti-CGRP/R mAb treatment, all patients were naïve to anti-CGRP agents and reported failure for ineffectiveness (no meaningful improvement in the frequency of headaches after the administration of drugs for ≥3 months at appropriate dose) or non-tolerability with at least three preventive treatments, including onabotulinumtoxinA (BTX-A). Patients who temporarily discontinued for sustained clinical response (i.e., treatment no longer needed; 3.5%, 16/472) are not included and considered as ‘persist treatment’ (Figure 1). The mandatory temporary one-month discontinuation after 12 months was not considered as withdrawn. No other specific inclusion or exclusion criteria were applied.
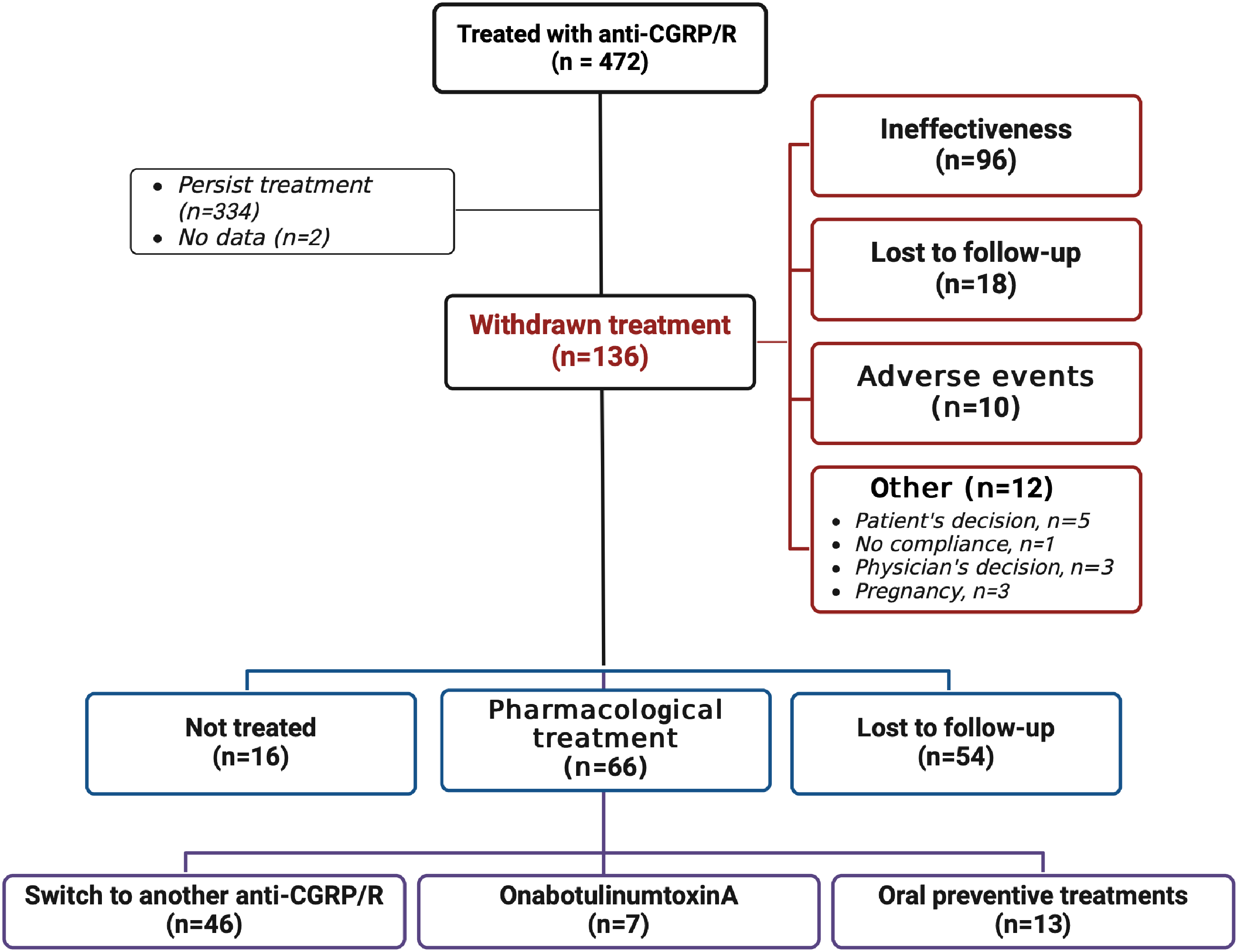
Flowchart of patients with reasons for withdrawal and subsequent treatments. CGRP/R, calcitonin gene-related protein/receptor. All values in the flow chart represent the number of patients.
During treatment with anti-CGRP/R mAbs, patients completed a paper headache diary and were trained in their correct use prior to the treatment onset, recording monthly headache days (MHDs), acute medication use (absolute number [AMNs], and days with at least one acute medication [AMDs] per month). A headache day was defined as any day on which a patient recorded any type of headache. Response rates were defined as a reduction in MHDs of ≥30%, ≥ 50% ≥ 75%, or 100% when compared to baseline. Patients compiled the Headache Impact Test (HIT-6) questionnaire monthly and the MIDAS questionnaire quarterly. Adverse events (AEs) were collected during treatment. All these questionnaires and response rate cut-offs are widely used and standardized in headache research. Demographics, migraine characteristics (presence of aura, disease duration, and time of CM onset), previous failures of drug classes, including β-blockers, tricyclic antidepressants (TCA), antiseizure medications (ASMs), BTX-A, and current concomitant preventive and acute symptomatic treatments, were collected. Moreover, we reported the reason for discontinuation and data on the subsequent treatment, including medication class and dosage. Baseline and all follow-up visits were part of the outpatient clinical routine.
Outcomes and analysis
The primary objective was to describe the reasons for anti-CGRP/R mAbs permanent discontinuation and the subsequent treatment approach. Reasons for discontinuation were singularly collected and then dichotomized between ineffectiveness and other reasons (including adverse events). Ineffectiveness was defined as reported by clinicians according to MIDAS score, MHDs, and/or combination of different variables, such as no meaningful improvement as reported by the patient. There were no predefined cut-offs and ‘withdrawn’ was decided by clinicians. However, for MIDAS, MHDs, AMNs and AMDs a < 30% or <50% cut-off were usually used. Pharmacological treatments after anti-CGRP/R were collected and categorized into: switch to another anti-CGRP/R agent; BTX-A; or other oral preventive treatments.
Secondary outcomes were evaluated by the multi-assessed response (see below) to anti-CGRP/R mAbs during treatment until the last month before discontinuation. The response included secondary endpoints as follows: i) the absolute change from baseline in MHDs; ii) response rates (≥30%, ≥ 50% ≥ 75%, and 100% reduction in MHDs); iii) persistence in MO; iv) absolute change from baseline of the overall number of AMN and AMDs; and v) the MIDAS and HIT-6 scores at three, six, and 12 months, and the last month of treatment (including all patients’ last month of treatment regardless of timepoint). The same analysis was performed in the entire cohort of patients that discontinued (i.e., withdrawal for any reason, discontinuation cohort), and then for the subgroup cohort that withdrew for ineffectiveness-only (ineffectiveness only cohort). All patients (except two with no data) have been included in the analysis (Figure 1).
Statistical analysis
All outpatients who discontinued anti-CGRP/R mAbs, regardless of reason (excluding patients that discontinued due to sustained clinical benefit or for the mandatory temporary one-month discontinuation after 12 months), were included in the study, not based on any statistical consideration or sample size calculation. Demographic and baseline characteristics were summarized descriptively, namely mean ± standard deviation [SD] or median interquartile range [IQR] for continuous variables and number (percentage) for categorical data. Normality assumption was assessed using the Shapiro-Wilk test. Considering the non-normally distributed data, a Friedman test was calculated to evaluate effectiveness variables pre-post changes in continuous variables and pairwise comparisons performed with a Bonferroni correction. A Cochrane's Q test was run for categorical dependent variables and pairwise comparisons were performed using Dunn's procedure with a Bonferroni correction for multiple comparisons. All pairwise comparisons included baseline, month-3, month-6, and month-12 for each of the analyzed variables. McNemar exact test and Wilcoxon rank test were performed to calculate pre-post changes from baseline to last month follow-up, for continuous or categorical data, respectively. No imputation was done for missing data or missing variables; no missing data for headache diary were present. The number of patients evaluated is reported in tables and graphs. Considering the withdrawal rate, not all patient data were available for every endpoint. The last month included all patients’ last month of treatment, regardless of the withdrawal timepoint.
A two-tailed p-value <0.05 was considered significant for all variables, excluding for multiple comparisons. All data were analyzed using SPSS software version 26.0 (IBM Corp. SPSS Statistics, Armonk, NY, USA) and graphs designed using GraphPad Prism version 9.00 (La Jolla, USA).
Results
Cohort characteristics
Overall, 472 patients were treated with at least one subcutaneous injection of anti-CGRP/R mAbs. In detail, 38.3% (181/472) with erenumab, 22.2% (105/472) with fremanezumab and 39.4% (186/472) with galcanezumab. Among them, 136 (28.8%) withdrew from treatment after an average of 9.0 ± 6.1 months. On the total number of anti-CGRP/R mAbs administered, 34.8% (63/181) discontinued erenumab, 19.4% (20/105) fremanezumab and 28.4% (53/186) galcanezumab. Patient flowchart is reported in Figure 1. Clinical and demographic features are summarized in Table 1 for all patients that discontinued and separately for patients that withdrew for ineffectiveness only. Comorbidities for both cohorts are reported in the Online Supplementary Table S1.
Patients’ demographic and clinical features.

Discontinuation cohort includes all patients that discontinued treatment regardless to reason. bIneffectiveness only cohort includes only patients that withdrawn for ineffectiveness, and it is part of the discontinuation cohort. HIT-6, Headache Impact Test 6; IQR, interquartile range; MIDAS, Migraine Disability Assessment; NRS, numeric rating scale; SD, standard deviation. Percentages are expressed on column total. cOnly patients with chronic migraine. dCalculated on 17 and 15 patients, respectively.
Considering the patients who withdrew from treatment, 46.3% of patients (63/136) were treated with erenumab, 39.0% (53/136) with galcanezumab, and 14.7% with fremanezumab (20/136). Almost all patients suffered from CM (125/126, 91.9%) and 84.8% (106/126) from MO. MHDs at baseline were 25.6 (IQR 6.2). The average headache pain intensity on a 0–10 scale (numeric rating scale [NRS] scale) was 7.7 ± 1.4 points. The median number of acute medications per month was 41.1 (IQR 43), and mean days with at least one analgesic use per month 21.4 (9.7). Patients presented elevated MIDAS (94.3 ± 63.0) and HIT-6 total scores (68.3 ± 6.7) at baseline. All concomitant and prior treatments at baseline are reported in Table 2. Briefly, 94.8% of patients had at least three classes of preventive treatments documented as ineffective (67.6% with onabotulinumtoxinA), with a mean of 4.4 ± 1.0 class failures. The remaining other 5.2% had at least three medication trials but one or more were due to intolerance or onset of contraindications (Table 2). All the data for the subgroup that discontinued for ineffectiveness-only are reported in Tables 1 and 2.
Concomitant and prior preventive treatments.
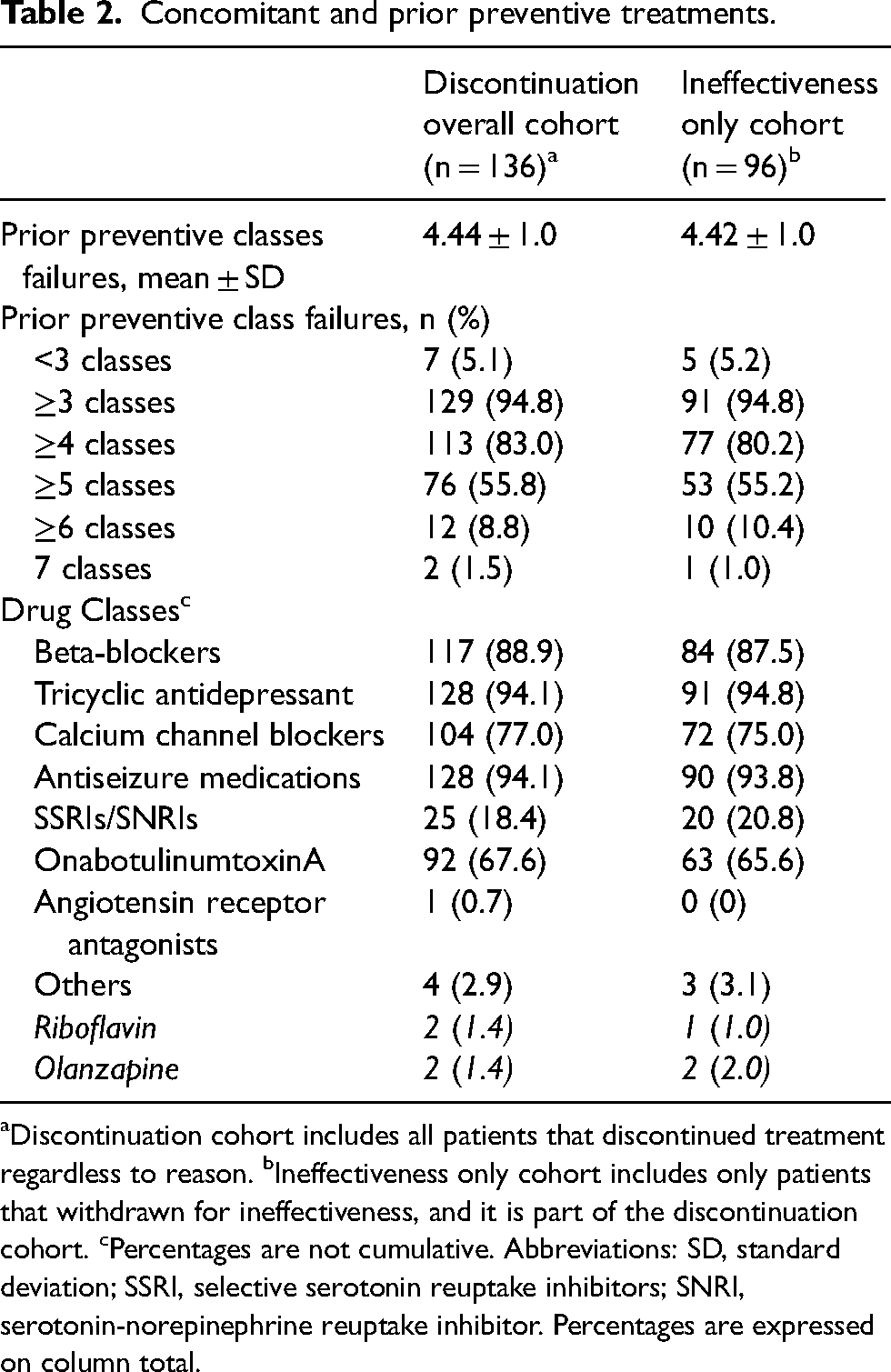
Discontinuation cohort includes all patients that discontinued treatment regardless to reason. bIneffectiveness only cohort includes only patients that withdrawn for ineffectiveness, and it is part of the discontinuation cohort. cPercentages are not cumulative. Abbreviations: SD, standard deviation; SSRI, selective serotonin reuptake inhibitors; SNRI, serotonin-norepinephrine reuptake inhibitor. Percentages are expressed on column total.
Withdrawal causes and follow-up
Among 136 patients, the majority of patients discontinued anti-CGRP/R mAbs due to ineffectiveness (96, 70.6%), followed by lost to follow-up during treatment (18, 13.2%), and adverse events (10, 7.3%). Five patients (3.6%) withdrew from treatment for personal reasons, and three (2.2%) withdrew due to pregnancy or pregnancy planning. One patient withdrew for non-compliance to treatment (1, 0.7%), and three (2.2%) for physician decision (not better accountable in other categories nor specified in patient records) (Table 3).
Reasons for treatment withdrawn.
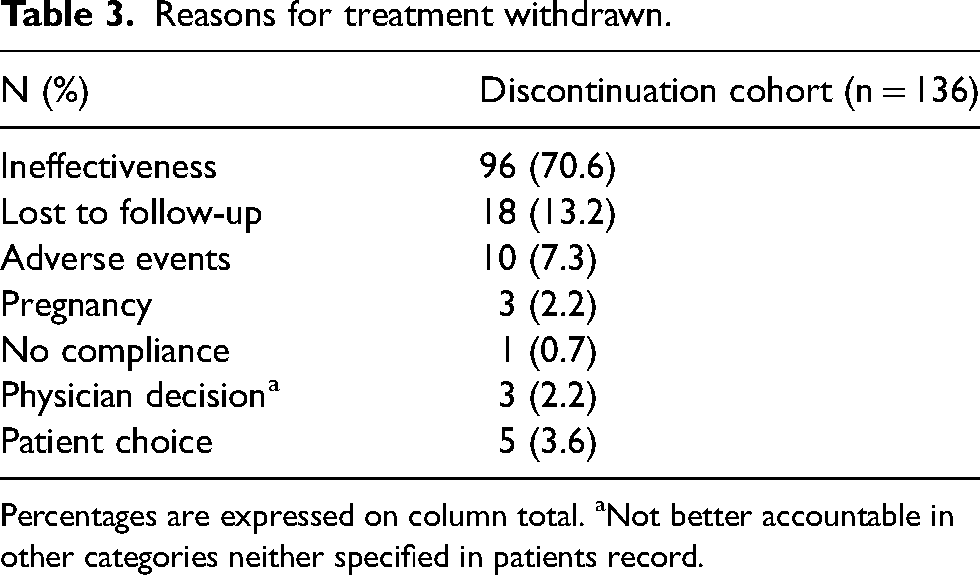
Percentages are expressed on column total. aNot better accountable in other categories neither specified in patients record.
Overall, 77.9% (106/136) of patients discontinued anti-CGRP/R mAb treatment during the first 12 months of follow-up, with 37/136 (27.2%) from month 1 to 3 (included), 39/136 (28.7%) patients from month 4 to 6 (included), and 30/136 (22.1%) in the last six months of treatment. Only 30/136 patients (22.1%) discontinued treatment after one year of treatment, ranging from 13 to 33 months (mean ± SD 17.8 ± 4.4). For patients (18/136, 13.2%) that withdrew in month-1 and −2 (before the three months suggested by guidelines), five patients (3.6%) were lost to follow-up, two patients (1.4%) each withdrew for personal reasons or physician decision, one patient (0.7%) for adverse events and eight patients (5.8%) for supposed ineffectiveness.
For the subgroup of patients that withdrew treatment for ineffectiveness, 74/96 (77.0%) patients discontinued treatment during the first 12-month follow-up, with 55/96 (57.2%) from month 1 to 3, 29/96 (30.2%) patients from month 4 to 6, and 19/96 (19.8%) in the last six months of treatment. Twenty-two patients (22.9%) discontinued treatment after one year of treatment, ranging from 13 to 26 months (mean 17.7 ± 3.3).
Among all patients who discontinued treatment, at the first follow-up, 66 (48.5%) patients started a new pharmacological treatment, 54 (39.7%) patients did not perform other visits (i.e., lost to follow-up), and 16 (11.8%) decided to not start another treatment (Table 3 and Figure 1). Considering only patients who discontinued for ineffectiveness (96), 57 (59.4%) started a new pharmacological treatment, 29 (30.2%) patients did not perform other visits (i.e., lost to follow-up), and 10 (10.4%) decided to not start another treatment. Most patients who started a new pharmacological treatment switched to another anti-CGRP/R (46/66, 69.6%). If the first treatment was a monoclonal antibody targeting the CGRP ligand, the second was an antibody targeting the CGRP receptor and vice versa. No switches were performed between galcanezumab and fremanezumab. The second most used treatment was BTX-A (7/66, 10.6%; with all patients in this subgroup naïve to this treatment) followed by antiseizure medications (6/66, 9.1%). Details of all treatments for both cohorts are reported in Table 4.
Pharmacological treatment after anti-CGRP/R mAbs discontinuation.
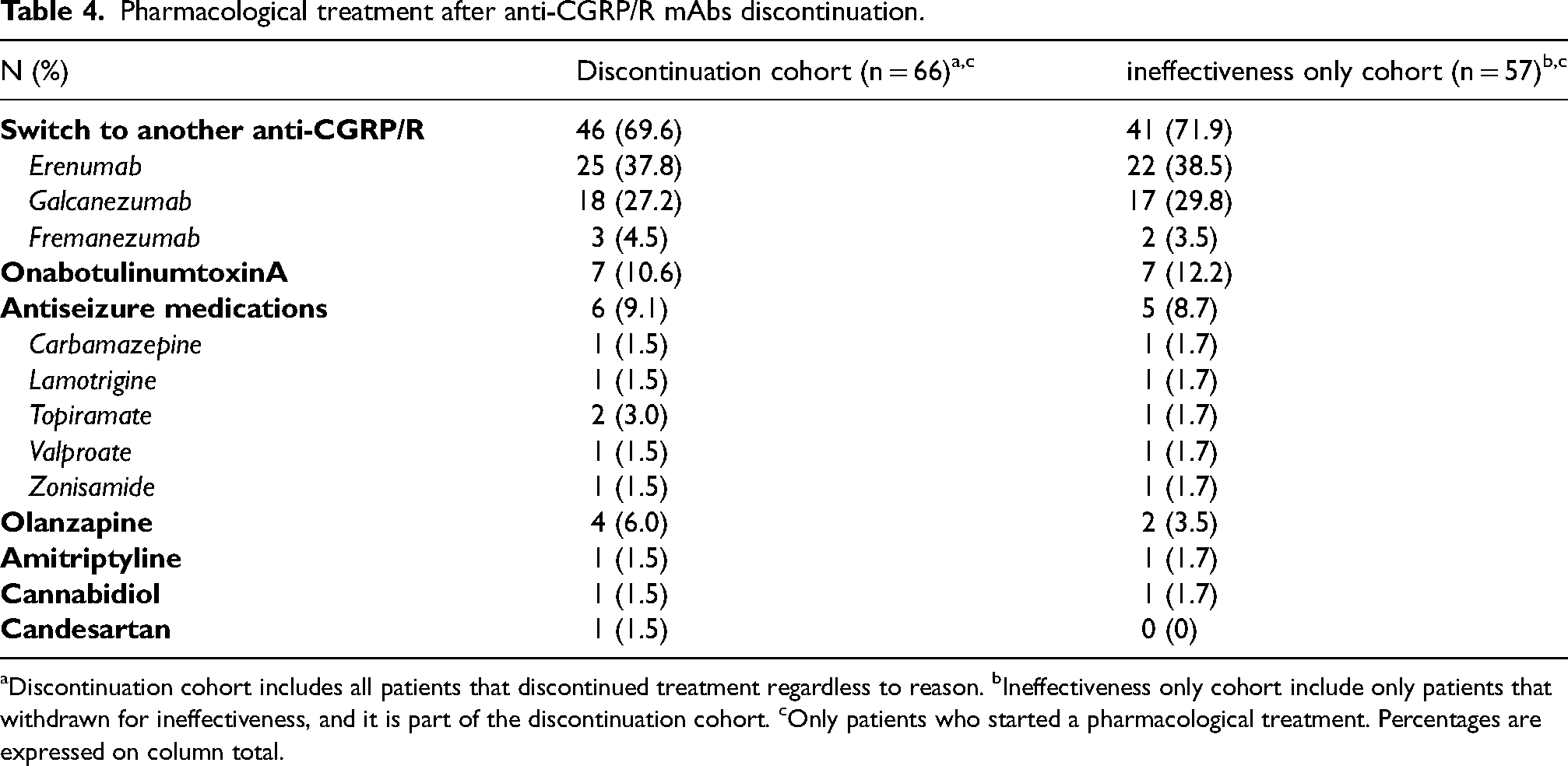
Discontinuation cohort includes all patients that discontinued treatment regardless to reason. bIneffectiveness only cohort include only patients that withdrawn for ineffectiveness, and it is part of the discontinuation cohort. cOnly patients who started a pharmacological treatment. Percentages are expressed on column total.
Treatment course
Discontinuation cohort
Although all patients were selected due to treatment discontinuation, MHDs were significantly reduced at months 1, 3, 6, and 12 compared with baseline (Figure 2 and Online Supplementary Table S2), as well in the last month of treatment (p ≤ 0.0001). A reduction in MHDs in the last month of treatment compared to baseline ranged from −2.0 [10.0] to −10.0 (12.0), with a significant decrease reported at month 12 (−2.0 [10.0], p ≤ 0.0001) (Figure 2 and Online Supplementary Table S2). It should be noted that the number of patients decreased during follow-ups, considering the withdrawal rate overtime, as expected for the selected population.
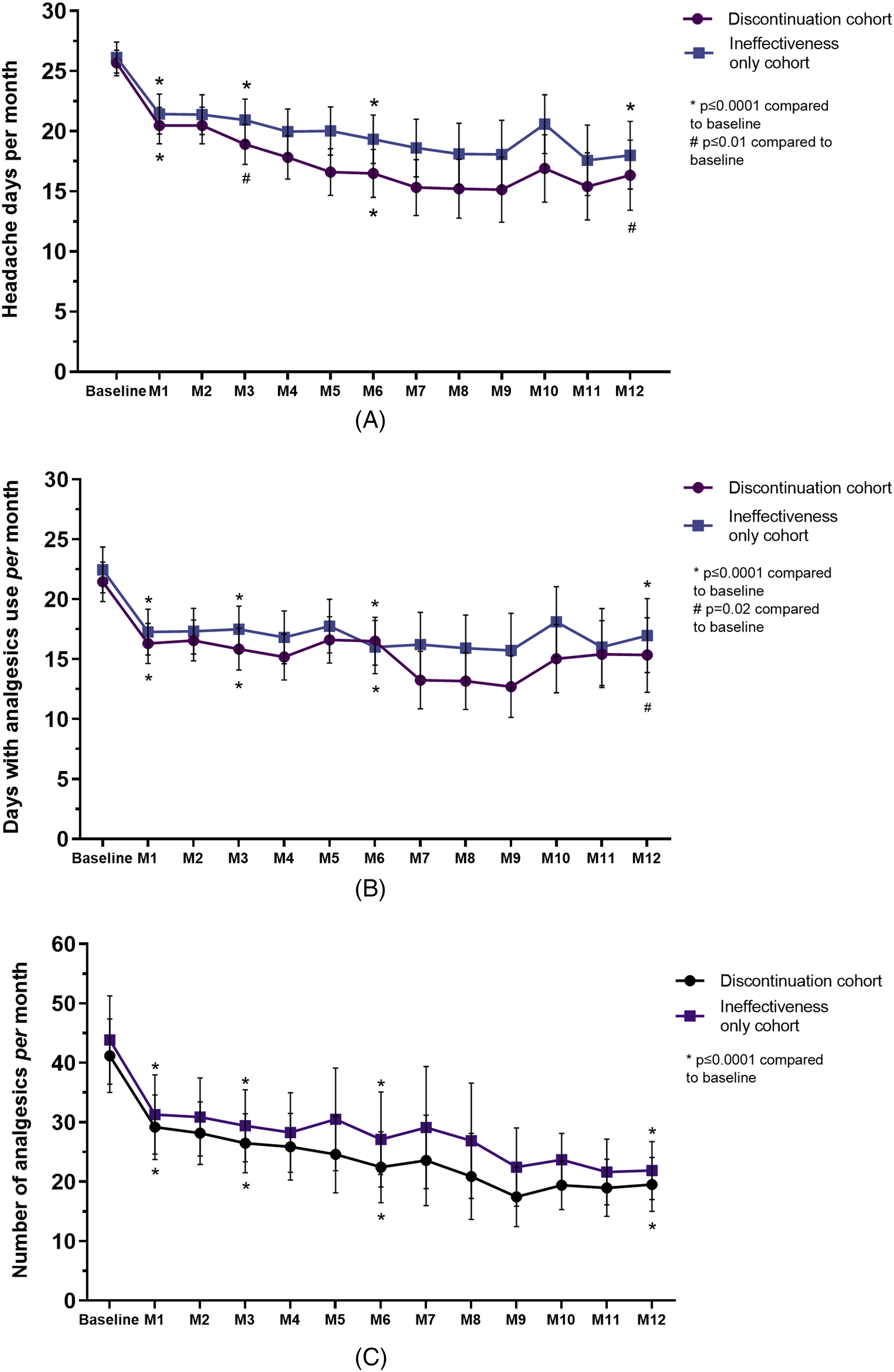
Number of monthly headache days (A), days with at least one analgesic use per month (B), and number of analgesics per month (C). M, months. Discontinuation cohort includes all patients who withdrew from treatment regardless of reason; ineffectiveness-only cohort is a part of the discontinuation cohort that withdrew from treatment for ineffectiveness.
As anticipated, the ≥50% response rate (in MHDs) compared to baseline was achieved by a low proportion of patients. A reduction ≥50% in MHDs was achieved by 21.4%, 30.5%, 34.6%, and 40.0% of patients at month 1, 3, 6, and 12 of treatment, respectively (Figure 3). In the last month of treatment, the percentage of patients achieving a 50% response was 16.9% (136/136) (Online Supplementary Table S4).
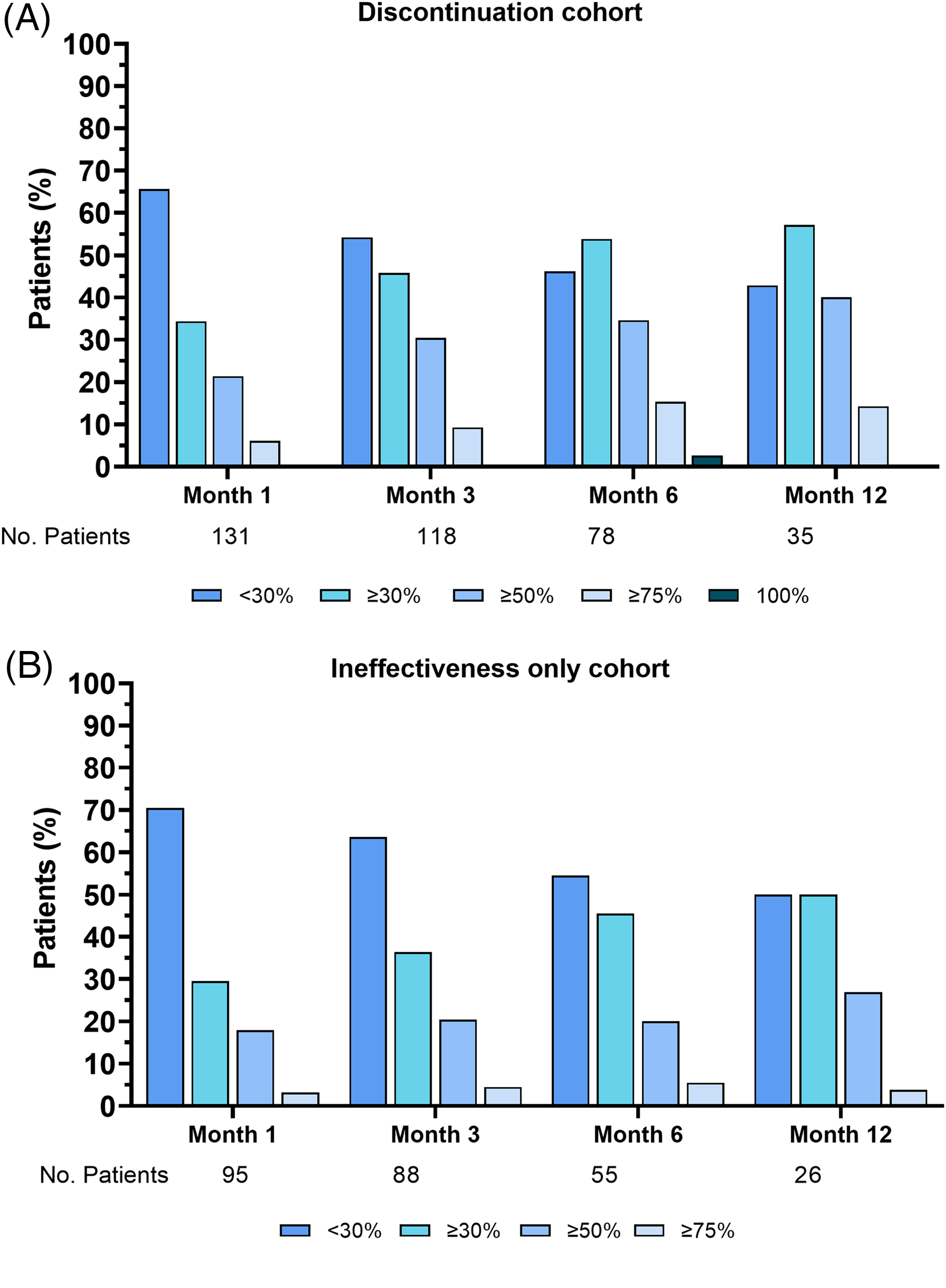
Response rates in the discontinuation (A) and ineffectiveness-only cohorts (B). Percentages are calculated on the total number of patients per follow-up reported below. Discontinuation cohort includes all patients who withdrew from treatment regardless of reason; ineffectiveness-only cohort is a part of the discontinuation cohort that withdrew from treatment for ineffectiveness.
Patients with MO were reduced compared to baseline from 84.8% to 68.2% at month 3, and 60.3% at month 6 (p < 0.001) (but not at month 12, 73.5%, p > 0.05) (Figure 4), and in the last month of treatment (72.0%, p = 0.005). Notably, the percentage of patients with CM who had MO did not decrease during treatment. Disability questionnaire (MIDAS and HIT-6) scores were reduced (for both p < 0.001) compared to baseline at all follow-up time points, as reported in Figure 5 and Online Supplementary Table S2. AMDs and AMNs were significantly reduced at months 1, 3, 6, and 12 of treatment compared to baseline (Figure 2 and Online Supplementary Table S2) as well as the last month of treatment (p ≤ 0.0001) (Online Supplementary Table S2).
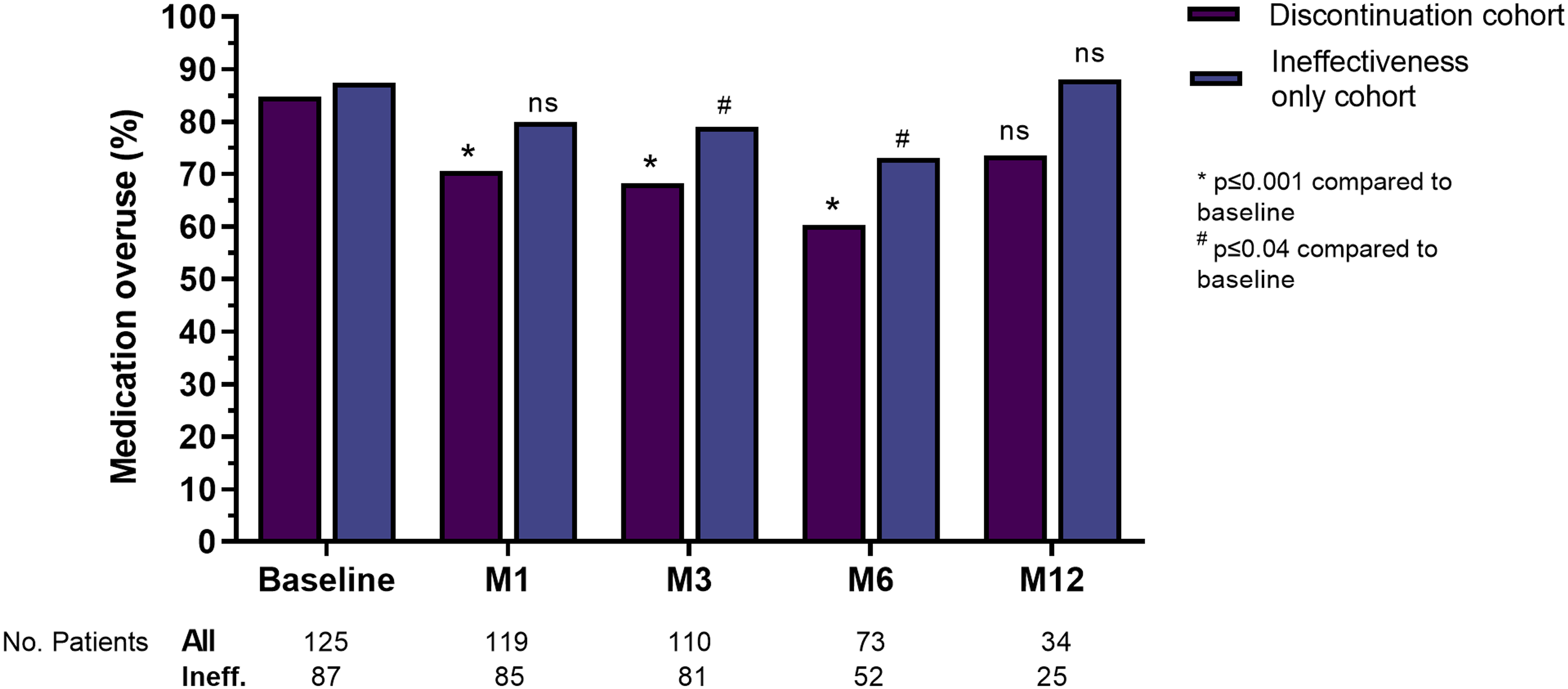
Medication overuse in patients with chronic migraine in the discontinuation and ineffectiveness-only cohorts. Percentages are calculated on the total number of patients per follow-up reported below per each group. Discontinuation cohort includes all patients who withdrew from treatment, regardless of reason; ineffectiveness-only cohort is a part of the discontinuation cohort that withdrew from treatment for ineffectiveness. Ns, not significative.
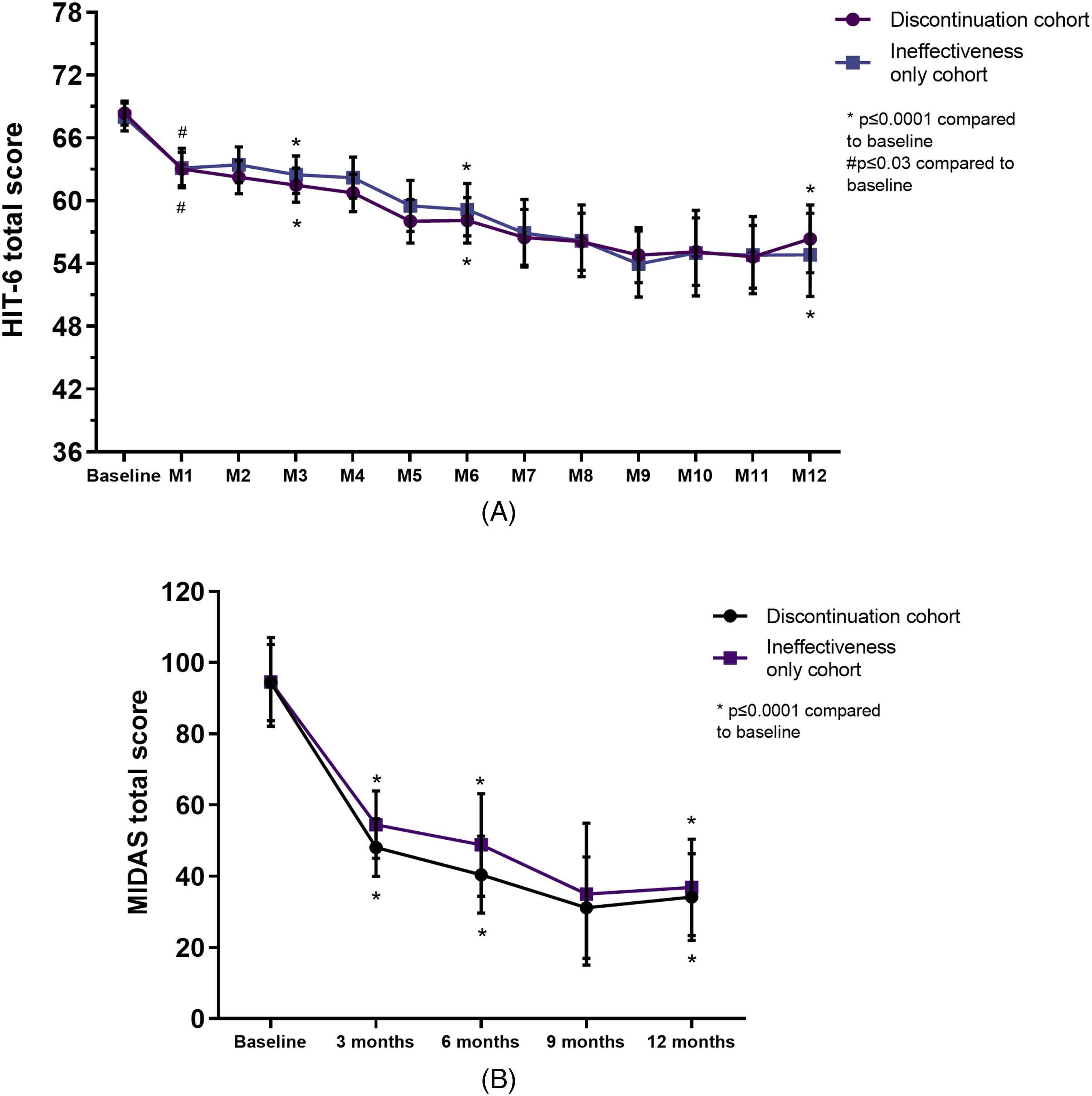
HIT-6 (A) and MIDAS (B) total score questionnaire. HIT-6, Headache Impact Test 6; M, month; MIDAS, Migraine Disability Assessment. Discontinuation cohort including all patients who withdrew from treatment regardless of reason; ineffectiveness-only cohort is a part of the discontinuation cohort that withdrew from treatment for ineffectiveness.
Ineffectiveness-only cohort
In the cohort that included only patients who withdrew treatment for ineffectiveness (96/136, subgroup of patients from the overall cohort), MHDs were significantly reduced at month 1, 3, 6, and 12 compared to baseline (Figure 2 and Online Supplementary Table S2), as well in the last month of treatment, regardless of follow-up (p ≤ 0.002). A reduction in MHDs compared to baseline ranged from −2.0 [IQR 7.0] to −7.0 [IQR 8.0], with a significant reduction reported also in the last month of treatment (−2.0 [IQR 7.0], p ≤ 0.0001) (Figure 2 and Online Supplementary Table S3). A ≥ 50% reduction in MHDs was achieved by 17.9%, 20.4%, 20.0%, and 26.9% of patients at month 1, 3, 6, and 12 of treatment, respectively (Figure 3). In the last month of treatment, the patients achieving 50% response were 4.1% (Online Supplementary Table S4).
As reported for the overall cohort (i.e., all patients that withdrew from mAbs for any reason), the percentage of patients with MO remained constantly high during treatment also in this subgroup (i.e., patients that withdrew from mAbs only for ineffectiveness). Patients with MO were significantly reduced compared to baseline from 87.4% to 79.0% at month 3 and 73.1% at month 6 (p ≤ 0.001), but not at month 1 (80.0%) and month 12 (88.0%), (p > 0.05) (Figure 4), as well as in the last month of treatment (83.9%, p = 0.54). Disability questionnaire (MIDAS and HIT-6) scores were reduced (p ≤ 0.002 and p ≤ 0.03, respectively) compared to baseline at all follow-up time points, as reported in Figure 5 and Online Supplementary Table S3. AMDs and AMNs were significantly reduced at months 1, 3, 6, and 12 of treatment (Figure 2 and Online Supplementary Table S3) and last month of treatment (p ≤ 0.0001) (Online Supplementary Table S3) compared to baseline.
Safety and tolerability during anti-CGRP treatment
Two severe, not lethal, adverse events (i.e., acute myocardial infarctions, AMI) that led to treatment discontinuation were reported during the study. These AEs were classified as probably not related to the drug (erenumab and galcanezumab). The patients had no known cardiovascular risk factors or relevant comorbidities. Ten patients (7.3%) discontinued treatment due to adverse events (Figure 1). These AEs were: asthenia (four patients), AMIs (two patients), stypsis (one patient), generalized pruritus (one patient), de novo onset of right leg paresthesia (one patient), arterial hypertension (one patient, with erenumab). Overall, 20 patients (14.7%) reported at least one mild and transient AE, with 24 total AEs detailed in Online Supplementary Table S5. The most commonly reported AEs were fatigue (eight, 5.8%) and constipation (five, 3.6%).
Discussion
In this real-world study, we described a large cohort of patients who withdrew from treatment with anti-CGRP/R mAbs, and the subsequent management strategies. We reported an overall withdrawal rate of 28.8%, in line with observational studies (2) and RCTs (11), but expanding these findings with patient's characterization and reporting the treatments (or lack of) after withdrawal. However, the number of patients that withdrew from anti-CGRP mAbs is far lower than previous preventive treatments (5,6).
Evidence-based recommendations regarding further management are still lacking in case of non-response to one anti-CGRP/R mAb in resistant/refractory migraine (12,13). In this cohort of migraine patients, therapeutic options are often limited or absent. In principle, patients could switch to another anti-CGRP/R drug, start a monotherapy with an oral preventative or BTX-A (regardless if naïve or with prior failures), add oral preventive or BTX-A on the anti-CGRP/R treatment, or add/start non-pharmacological treatments. However, in our cohort, we showed that a large number of patients were lost to follow up or decided to not start a new treatment probably due to their perceived refractoriness to treatment and loss of trust in therapies/medical support. Furthermore, limited data, costs, and specific restrictions by national agencies still preclude most drug alternatives. For instance, in Italy, BTX-A and anti-CGRP/R mAbs co-administration (14) as well as switching to another anti-CGRP/R (at healthcare system expenses), are not permitted (15). Considering these limitations and uncertainties, drugs with unclear effectiveness in migraine are often used, as reported in our cohort.
The treatment course of patients withdrawing from anti-CGRP/R mAbs has been reported in one previous retrospective study that primarily assessed the difference between super-responders and non-responders to anti-CGRP/R mAbs. The non-responders were defined as those with ≤25% reduction in MHDs after three months of treatment with an anti-CGRP/R mAb targeting the ligand and subsequent three months of treatment with an anti-CGRP mAb targeting the receptor, or vice versa (3). In non-responders, therapy was switched to the third and last available anti-CGRP/R mAb in 31% of cases (eptinezumab was still not available in EU), and the most recent anti-CGRP/R mAb was continued in 19% of patients due to subjectively reported headache improvement. In the other cases, non-responders switched back to prior not-effective oral preventatives or BTX-A (3). Our findings are in line with and expand these results, with a similar pattern of treatment after anti-CGRP/R mAbs discontinuation. However, no patients in our cohort switched back or tried more than two anti-CGRP/R mAbs.
The effectiveness of anti-CGRP/R mAbs is well established, providing several advantages compared to standard prophylaxis treatments (16), including high adherence (5–7) and lower and mild side effects (17). Despite these favorable data, in RCTs and real-world studies, different percentages of non-responders have been reported, ranging from 5% to 30% (11,18,19) according to different effectiveness criteria (2,15).
The definition of responder to anti-CGRP/R mAbs treatment and whether the same cutoff should be applied in RCTs and observational studies is a matter of debate (20,21). Even a 50% MMD reduction can be associated with a residual clinically relevant burden (22), but, on the other hand, a 30% reduction can have clinical relevance (1,21) and for the most severely impacted patients (e.g., daily migraine attacks) even a small reduction could be significant. The current definition of responders and non-responders might not accurately reflect clinical practice results. Including multiple alternative measures of effectiveness to evaluate treatment, as MIDAS, HIT6, analgesic use as well as patient's self-reported impression of wellness (e.g., patients’ global impression of change, PGIC) should be always considered.
Indeed, in our study, the overall 50% response rate was very low considering other observational studies, as expected (9). However, several parameters considered (i.e., MHD, AMN, AMD, MIDAS, and HIT-6) show statistically significant reductions in both cohorts, suggesting at least some effect of treatment, although not sufficient to univocally discriminate patients who discontinued treatment. Therefore, although some variables have been classified as more or less useful to define a clinical response, an isolated parameter can hardly reflect the multifaced action of anti-CGRP/R mAbs (20,23) and the complex symptomatology of migraine other than pain. Assessments based on inclusion of patient-reported outcomes, which evaluate the impact on quality of life, disability, improved efficacy of analgesics, and patient preference should be, in principle, superior to one single variable (20,24).
Non-responders often have medication overuse, which is linked to poor response to acute treatments and contributes to chronicity and treatment resistance, making it a poor prognostic factor (25,26). In our cohort, there is a high percentage of patients with MO and a high median number of acute medications used per month. Nevertheless, several studies have shown the efficacy of anti-CGRP/R mAbs in patients with MO, leading to MO remission (9,27). Therefore, MO alone may not be the only factor for therapy non-responsiveness, but rather a combination of various clinical features (3). Finally, the complete or partial ineffectiveness of anti-CGRP/R monoclonal antibodies could be related to the involvement of other neurotransmitters in the pathophysiology of migraine such as vasoactive intestinal peptide (VIP), pituitary adenylate cyclase-activating polypeptide (PACAP), or prostaglandins (28,29). The definition of refractory migraine should be carefully used and the definition itself is changing due to introduction of novel treatments (13,30). Several patients with reported refractory migraine responded to anti-CGRP mAbs. Also, in the future, non-responders to anti-CGRP drugs could be responders to different not yet available target therapies.
Several strengths characterize our study. This is the first study reporting a large group of multi-assessed non-responders to anti-CGRP/R mAbs in clinical practice, evaluating a long treatment course and treatment outcomes. Further strengths include the use of different analyses of acute medication use (including days and total number of acute medications use) and disability questionnaires. There are, however, some limitations, including the use of only MHDs instead of distinguishing MHDs from MMDs (not available as separated) and the single center design, therefore reporting judgments and practice only of physicians involved in our center. We were also unable to record the number of patients with MO who underwent detoxification procedures. Finally, being an open label study, we cannot rule out the cyclical exacerbations of migraine over time or the regression toward the mean for effectiveness variables.
Conclusions
Undoubtedly, anti-CGRP/R mAbs have led to a paradigm shift in migraine management. However, a significant percentage of patients remain unresponsive, prompting the need for alternative therapeutic strategies and approaches. Further research is required to identify specific signatures of non-responders, address unmet needs in migraine treatment, and determine the possible benefits of treatment switches and combinations in selected patients.
Better characterization of patients that withdrawn anti-CGRP mAbs and understanding of their unmet needs is crucial in order to optimize their clinical management. Evidence-based recommendations regarding further management are still lacking in case of non-response to one anti-CGRP/R mAb in resistant/refractory migraine. Some patients remain unresponsive and require additional therapeutic support.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241273968 - Supplemental material for Outcomes, unmet needs, and challenges in the management of patients who withdraw from anti-CGRP monoclonal antibodies: A prospective cohort study
Supplemental material, sj-docx-1-cep-10.1177_03331024241273968 for Outcomes, unmet needs, and challenges in the management of patients who withdraw from anti-CGRP monoclonal antibodies: A prospective cohort study by Andrea Burgalassi, Marina Romozzi, Giulia Vigani, Roberto De Icco, Bianca Raffaelli, Alberto Boccalini, Francesco De Cesaris, Paolo Calabresi, Pierangelo Geppetti, Alberto Chiarugi and Luigi Francesco Iannone in Cephalalgia
Footnotes
Acknowledgment
Authors’ contributions
All authors critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P.G.: received personal fees from Allergan, Eli Lilly, Novartis, Amgen, TEVA; Grants from Amgen, TEVA, Eli-Lilly, Allergan, Chiesi; Scientific Advisory Board, Endosome Therapeutics; Founding scientist of FloNext srl, Spinoff of the University of Florence.
F.D.C. received personal fees from TEVA, Eli Lilly, Novartis, Abbvie.
B.R. reports research grants from Novartis, and personal fees from Abbvie/Allergan, Hormosan, Lilly, Novartis, and Teva.
R.D.I. received honoraria for scientific presentations from Eli-Lilly and Teva.
L.F.I. received personal fees from TEVA, Eli Lilly, Pfizer, Abbvie, Lundbeck.
Other authors have no conflicting interests.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
