Abstract
Data from the Prolonged Migraine Prevention (PROMPT) with Topiramate trial were evaluated post hoc to determine whether topiramate could prevent migraine auras, and whether its efficacy in preventing migraine headaches was similar in patients with (MA; n = 269) and without (MoA; n = 542) aura. Migraines and auras were recorded during prospective baseline, 6-month open-label (OL) topiramate and 6-month double-blind (DB), placebo-controlled phases. In the last 28 OL days, migraines without aura and migraine auras decreased by 43.1% and 54.1%, respectively, in MA patients. MoA patients experienced a 44.3% reduction in migraines. In the DB phase, increases in migraines with placebo vs. topiramate were similar to the full study, but were generally not statistically significant, probably due to lack of power in the subgroup analysis. Similarly, there were no statistically significant changes in number of auras between groups. Thus, topiramate appears to reduce migraine auras in parallel with headache reductions, which are similar in patients with and without aura.
Introduction
Migraine is a common disorder, affecting up to 6% of men and 18% of women annually (1). A throbbing headache is the most bothersome feature, which explains why migraine therapies have focused on reducing the severity, duration and occurrence of these headaches. As a complete neurological disorder, however, migraine is not limited to headache but also involves hypersensitivity to light and sound, as well as gastrointestinal disturbances. The typical migraine attack can also—but not necessarily—be preceded by visual, auditory and other sensory neurological symptoms, collectively known as ‘aura’. Importantly, aura is not just a silent precursor of the upcoming migraine headache, but can be disabling by bringing about temporary speech disturbances, loss of single-sided vision and hemiparesis. Migraine auras usually develop over 15–20 min, last up to 60 min (2) and occur during some or all migraine attacks in 30–40% of migraine sufferers (3–6).
Migraine headaches can be treated acutely with the help of non-steroidal anti-inflammatory drugs or triptans, but these agents are not effective against auras (7). Some substances not used for acute treatment of migraine headaches may, however, be beneficial for the treatment of prolonged aura. In a small uncontrolled trial and single case observations, the use of intravenous verapamil (8), intravenous furosemide (9) and intranasal ketamine (10) showed some success. However, the modes of application of these agents, and the occurrence of adverse events, mean that they are not useful in daily practice.
Another approach to reducing migraine-related suffering is aimed at preventing the occurrence of migraines. Various agents—belonging to pharmacological classes as different as β-blockers, calcium antagonists and antiepileptic drugs (AEDs)—have been reported to reduce or prevent migraine headaches, but whether these prophylactic agents can also prevent migraine auras is largely unknown. Topiramate, a drug first developed for use in epilepsy, is at present the most investigated drug in migraine prevention. Its efficacy has been demonstrated in at least three randomized controlled trials of 6 months' duration (11–13) and is licensed for migraine preventive treatment.
In a recent double-blind, placebo-controlled withdrawal study—the Prolonged Migraine Prevention with Topiramate (PROMPT) trial—patients were randomized to continue or discontinue topiramate after 6 months of open-label (OL) treatment. Patients receiving placebo for 6 months experienced an increase from baseline of 1.19 migraine days per 4-week period (P < 0.0001 vs. baseline), compared with no significant change in patients who continued topiramate (0.10 migraine days per 4-week period; P = 0.0011 vs. change with placebo) (14). In the present analysis of the PROMPT data, we assessed whether topiramate treatment could also prevent migraine auras, and whether topiramate treatment reduced migraines to a similar extent in patients with auras compared with those without.
Methods
This was a post hoc analysis of the PROMPT trial (protocol TOPMAT-MIG-303; EudraCT number 2005-000321-29), details of which have been published previously (14). In brief, adult patients with established migraine at an average frequency of four or more migraine days per month first entered a 4–8-week prospective baseline (PB) phase during which they received no preventive study medication. Patients recorded the occurrence of auras and migraines, the latter with start and end time, as well as the use of acute medication for migraine headaches, in a diary throughout the study. During the second, OL phase of the study, all patients were treated with topiramate for 26 weeks (initially titrated to 100 mg, with further adjustments to 50–200 mg/day permitted). Patients completing the OL phase entered the third phase, a 26-week double-blind (DB) treatment phase in which they were randomly assigned to continue receiving their final topiramate dose or switch to placebo. All patients provided written informed consent; the trial was done in accordance with the international guidelines on good clinical practice, and was approved by local ethics committees.
Definitions
For the purposes of the study, a ‘migraine’ was defined as a continuous period of migraine headache, and a ‘migraine day’ was defined as a calendar day during which the patient experienced migraine headache at any time (14). Auras could be registered once per day and were classified—depending on the presence or absence of migraine headache—as either ‘migraine with aura’ or ‘aura only’. Each migraine and, by extension, each migraine day was classified as being with or without aura. Patients were categorized as ‘patient with aura’ (MA) if they experienced aura at least once during the study, otherwise they were categorized as ‘patient without aura’ (MoA). Over the course of the study, MA patients had migraines both with and without aura.
Analyses
The primary end-point in PROMPT was the difference in number of migraine days during the last 4 weeks of the DB phase compared with the last 4 weeks of the OL phase (14). The post hoc analyses presented here were performed in quite a similar manner. Monthly (28-day) frequencies of auras, migraines and migraine days were assessed per subgroup for the indicated periods of each of the three phases (PB, OL, DB). For the observed-case analyses, changes in frequency during the OL phase were calculated vs. the last 4 weeks of the PB phase (defined as OL baseline). Changes in frequencies during the DB phase were calculated vs. the last 4 weeks of the OL phase (defined as DB baseline). End-point analyses used the last observation carried forward method.
Within-group differences for continuous data were assessed using a Wilcoxon signed rank test. Between-group comparisons were made using a Wilcoxon two-sample test. Fisher's exact test and McNemar's test were used for between-group and within-group comparisons of categorical data, respectively. All tests were two-tailed and used an α-level of 5%. No corrections for multiple comparisons were made. Percentage changes from baseline were analysed as symmetrized percentage change (SPC): 100 × (post-baseline − baseline)/(post-baseline + baseline). The values presented in this study are the SPCs transferred back to percentage changes.
Results
In total, 818 patients entered the OL phase, with 514 patients entering the DB phase (topiramate, n = 253; placebo, n = 257) (Fig. 1). The DB phase was completed by 83.0% of patients receiving topiramate (n = 210/253) and 80.5% (n = 207/257) receiving placebo. Baseline demographics and disease characteristics were well balanced between the treatment arms (Table 1). Most patients were female (708/818; 87%), and the average age of the patient population was almost 40 years. Active concomitant diseases were present in 401/818 patients (49%) at baseline, of which the most common were endocrine (n = 96; 11.7%), cardiovascular (n = 89; 10.9%) and psychiatric disorders (n = 89; 10.9%).
Patient flow in the study. MA, patients who experienced aura at least once during the study; MoA, patients who did not experience aura. Baseline demographics and disease characteristics

Number and frequency of auras
In the last 4 weeks of the PB phase, 216/811 patients (26%) experienced an aura at least once. Of the 847 auras that occurred during the PB phase, six were auras without headache (0.7% of the total number of auras). In the entire study (all phases taken together), 269/811 patients (33%) experienced an aura at least once. In total, 4699 auras were reported, of which 4659 (99.1%) were auras with migraine and 40 (0.9%) were auras without migraine.
During the 6-month OL topiramate treatment phase, the percentage of patients experiencing an aura decreased gradually in the consecutive 4-weekly periods, to 15% in weeks 21–24 (P < 0.001 vs. PB end; Fig. 2a). In the last 28 OL days, 131 patients (16%) experienced an aura. In the DB phase, the percentage of patients experiencing an aura rose initially in both treatment arms (P < 0.05 for weeks 26–30 vs. OL end-point), but returned in weeks 47–50 to the lower level that was obtained by the end of the OL phase (topiramate, P = 0.8084; placebo, P = 0.4328 vs. OL end-point; Fig. 2b). In the last 28 DB days, 82/510 patients (16.1%) overall experienced an aura.
Percentage of patients with aura in the 4-week period preceding the end of the indicated week in (a) the open-label (OL) phase and (b) the double-blind (DB) phase.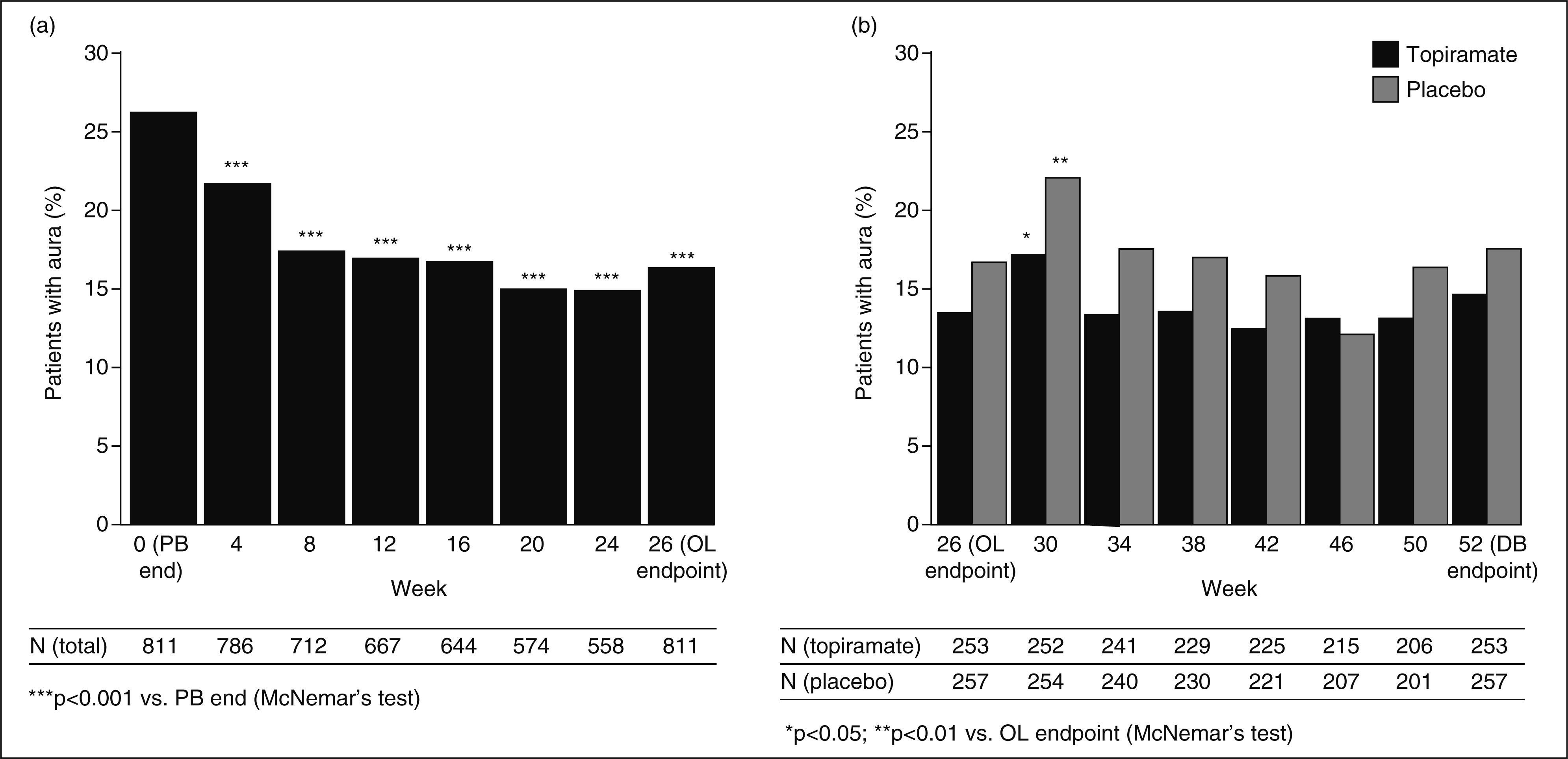
MA patients
Of the 811 patients who qualified for the OL analysis, 269 (33.2%) were identified as having migraine with aura at least once during the study (MA patients). Of the 510 patients who qualified for the DB analysis, 184 (36.1%) were MA patients. On average, about one-third of the migraines of these MA patients was preceded by aura [during the PB phase, 36.3% of their migraines were with aura; during the OL phase, 32.8% of migraines were with aura (n = 269)]. The percentage of migraines with aura was not evenly distributed among MA patients: > 50% of MA patients experienced an aura in < 25% of their migraines, whereas 25% of MA patients experienced an aura in > 50% of their migraines (results not shown).
Migraines in MA patients
In the MA group (n = 269), the number of all migraines (with or without aura) per 4 weeks decreased gradually throughout the OL phase from an average of 8.5 in the PB phase to 4.2 in weeks 17–26 (Fig. 3a). In the last 4 weeks of the OL phase, the change in migraines (all) vs. PB was −3.08 per 4 weeks (P < 0.001; end-point analysis) (Table 2).
Mean (± Monthly frequency of migraines with aura, migraines without aura, and all migraines (either with or without aura) during the prospective baseline (PB) phase (28-day rate) and last 4 weeks of the open-label (OL) phase in the migraine with aura (MA) and migraine without aura (MoA) subgroups **P < 0.001. Symmetrized percentage change transferred back to percentage change values.

Number of migraines without aura per 4-week period at the end of the open-label (OL) phase and change during the double-blind (DB) phase

***P < 0.001; **P < 0.01; *P < 0.05 vs. open-label end-point (Wilcoxon signed-rank test, two-tailed).
P-value for between-treatment group difference (Wilcoxon two-sample test, two-tailed).
The number of migraine auras (with or without headache) per 4 weeks decreased gradually from an average of 2.8 in the PB phase to 1.2 in weeks 17–26 (change vs. PB, −1.32 per 4 weeks; P < 0.001). The mean decrease in number of migraine auras (Table 2) and migraine days with aura in the last 28 OL days was 54.1% in both cases. The number of migraines with aura, assessed as a percentage of the total number of migraines, decreased from 36.3% in the PB phase to 31.1% in weeks 17–26. Thus, the reduction in auras during topiramate treatment tended to be somewhat more pronounced than the reduction in migraine headaches. No significant changes in the number of auras during the DB phase were found in the topiramate arm [mean (S.E.) +0.10 (0.36); P = 0.7677], nor in the placebo arm [+0.39 (0.524); P = 0.2360], nor between the two arms (P = 0.2675).
Migraines in MoA patients
About two-thirds of all migraine patients (542/811) did not report aura at any time during the study. In these MoA patients, the number of migraines per 4 weeks decreased gradually from an average of 7.7 in the PB phase to 4.3 in weeks 17–26 (Fig. 3b). In the last 4 weeks of the OL phase, the change in migraines vs. PB was −2.55 migraines per 4 weeks (P < 0.001; end-point analysis) (Table 2). When migraines in MA and MoA patients in the last 4 weeks of the OL phase were compared, the mean percentage reductions in migraines were 46.9 and 44.3%, respectively (P = 0.36) (Table 2).
During the DB phase, the number of migraines increased significantly in the placebo arm (P < 0.001 at end-point) but not in the topiramate arm, and the difference between the treatment groups was statistically significant (P < 0.01 at end-point; Table 3).
Adverse events in MA vs. MoA patients
Treatment-emergent adverse events reported during 6 months of treatment with topiramate (open-label phase) by ≥ 4% of patients in either subgroup: migraine without aura (MoA) or migraine with aura (MA)
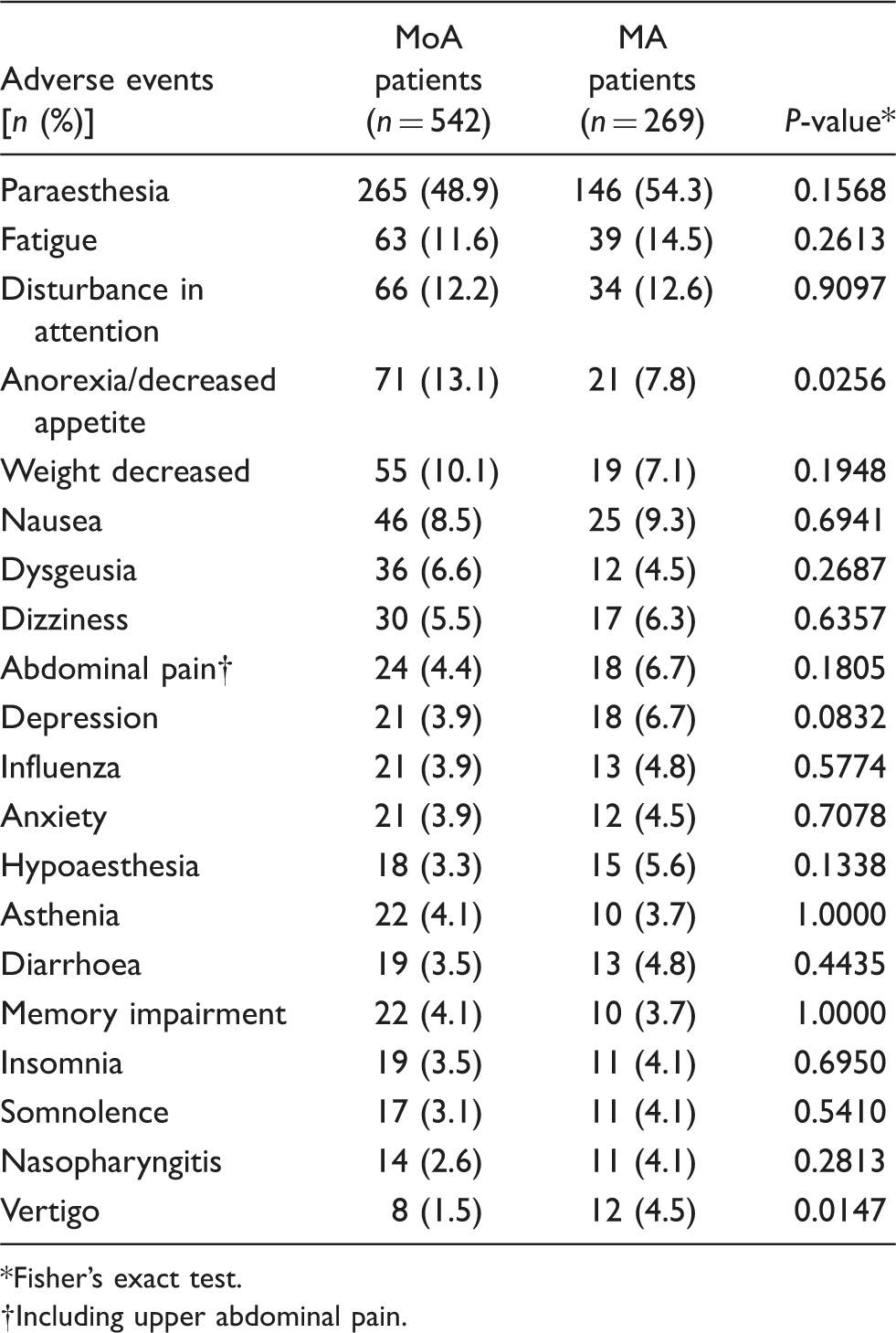
Fisher's exact test.
Including upper abdominal pain.
In patients who received placebo during the DB phase, paraesthesia (29.5% vs. 16.7%; P = 0.0184) and depression (9.5% vs. 2.5%; P = 0.0182) appeared to occur more frequently in MA than in MoA patients. There was also a tendency for memory impairment to be more frequent (4.2% vs. 0.6%; P = 0.064) and disturbance in attention to be less frequent (1.1% vs. 6.8%; P = 0.061) in MA compared with MoA patients. In patients maintained on topiramate during the DB phase, no differences between MA and MoA patients were seen except for a tendency to less frequent memory impairment in MA compared with MoA patients (3.7% vs. 0%; P = 0.093).
Discussion
The present post hoc analysis has demonstrated for the first time that topiramate, in addition to reducing migraine headaches, significantly reduced the number of migraine auras. Moreover, topiramate treatment reduced migraine headaches in a similar manner in patients with and without aura. Comparing the change in the number of migraines without aura between MA and MoA patients, we found a similar percentage reduction in MA compared with MoA patients (43% vs. 44%). The reduction in the number of auras with or without migraine headache (about 1% were auras only) was even more pronounced (54%). Furthermore, topiramate was effective in MA patients in preventing migraines with and without aura, reducing the frequency of migraines overall by 47%.
Earlier reported findings from the present study had shown that topiramate continued to provide sustained benefit after discontinuation, but that migraine frequency increased slightly in patients who discontinued topiramate treatment after 6 months (14). The results from the present analysis indicate that the increase in migraines after discontinuation occurs to a similar extent in MA and MoA patients, while the benefits of continued topiramate are also similar in the two groups. Immediately after discontinuation there was a rise in the number of auras, which was not maintained during the course of the DB phase. An initial rise in aura frequency, although somewhat less pronounced, was also seen in patients continuing topiramate treatment. The increases in frequency of migraine auras during the first month of the DB phase thus closely resemble the increases in migraine headaches that were found earlier for both treatment groups in the full study population (14). Changes in the frequency of migraine auras in the OL and DB phases of this study therefore appear to mirror changes in the frequency of migraine headaches in these phases. Absolute numbers of migraines with aura were, as expected, lower than the total number of migraine headaches, which may explain why the results often failed to reach statistical significance.
The tolerability profile of topiramate in the PROMPT study was consistent with the known clinical profile of the drug (14), and the present analysis shows that it did not differ markedly between patients with and without auras.
AEDs are a heterogeneous class that act via a number of different mechanisms to prevent epileptic seizures by reducing neuronal excitation in the central nervous system. This variation in pharmacological mechanisms of action is likely to be responsible for the observation that, although some AEDs are effective for migraine prevention, others are not. Of the ‘classical’ AEDs, for example, evidence has been presented that valproate is effective in migraine prevention (15), whereas carbamazepine (16) and oxcarbazepine (17) are probably not. It is possible that the migraine-preventive effects of drugs such as topiramate and valproate are the result of increased inhibitory GABAergic neurotransmission, a property not possessed by other AEDs such as carbamazepine, oxcarbazepine and lamotrigine (18). Although the neurological basis of migraine auras has not yet been established, increasing evidence indicates that they are a clinical manifestation of a cortical spreading depression (CSD)-like phenomenon (19). In animal models, topiramate has been shown to inhibit CSD (20,21). Although this has not been demonstrated in humans, recently published case studies have shown that topiramate treatment can lead to resolution of aura symptoms, even in the presence of continued migraine headaches (22). Data suggest that 3–4 weeks' treatment may be necessary to achieve therapeutic efficacy, with potential for increased efficacy with longer treatment (21). Indeed, chronic administration of the drug is essential for the suppression of CSD (23–25).
Trials of migraine prophylactic drugs generally focus on reduction in the number of migraine days as the key efficacy parameter, and little attention has been paid to the influence of these drugs on the occurrence of auras. Indeed, a literature search revealed no evidence for an effect of β-blockers on migraine auras, and only limited information on some AEDs. Valproic acid has been reported as being useful in three patients with persistent migraine aura (27,28), and open-label trials of lamotrigine suggest effectiveness in migraine aura prevention (29–31). For example, a reduction in frequency of migraine auras was observed in a 3-year OL study with lamotrigine that enrolled 59 patients who had experienced migraine with aura or migraine aura without headache for ≥ 1 year (31). A recent Cochrane meta-analysis, however, concluded overall that lamotrigine was not significantly more effective than placebo in the prevention of migraine headache (32). The effects of lamotrigine on migraine auras remain to be confirmed.
In addition to the negative results for non-GABAergic AEDs, a similar lack of efficacy in migraine and aura prevention has been reported for triptans and other analgesics. For example, triptans given during the aura phase neither shorten the aura nor prevent the development of headache (7). Again, these clinical results are in line with experimental findings that triptans do not suppress CSD. CSD leads to an increase in cyclic guanosine monophosphate levels in the cortex and brainstem that is inhibited by tonabersat but not by sumatriptan (26).
As discussed previously, the major strength of this analysis is the large number of patients in each cohort and the long (12-month) duration of the study (14). A weakness of the study is its considerable drop-out rate; almost one-third of patients (32%) did not complete the first 6 months' treatment. This number does not, however, compare unfavourably with three earlier studies in migraine prevention, which used topiramate in fixed doses for 6 months and found drop-out rates between 37 and 47% (11–13). Unlike these studies, PROMPT used flexible dosing for topiramate (50–200 mg/day) in the OL phase, which may have reduced the frequency of early discontinuation related to poor tolerability at higher doses. By applying flexible dosing, PROMPT was designed to mimic daily prescribing practices, thereby raising the applicability of its results both for prescribers and recipients. Even though the PROMPT study was not primarily designed to answer questions about migraine auras, the mandatory registration of such auras in the patients' diaries and the large amount of data obtained in the study enabled us to estimate the occurrence of auras with sufficient accuracy. The incidence of auras in the study population (36% overall) falls well within the range of values published in the literature (30–40%) (3–6).
In conclusion, topiramate was shown to reduce the number of migraine auras in parallel with its reduction in migraine headaches. As popular acute treatments such as triptans are ineffective against auras, preventive treatment with topiramate can be a useful addition to therapy, particularly when the auras are disabling or extremely bothersome. Finally, topiramate appeared to reduce migraine headaches in a similar manner in patients with and without aura.
Footnotes
Acknowledgements
The authors thank all the investigators who participated in PROMPT. The study was funded by Janssen-Cilag EMEA. Editorial assistance with drafting and completion of the manuscript was provided by D. Booth (Bioscript Stirling Ltd, London, UK).
