Abstract
Objective
To compare plasma adrenomedullin (AM) levels between adults with migraine and healthy controls (HCs), assessing subgroup differences and clinical associations.
Methods
This cross-sectional, observational single-center study was carried out from September 2020 to June 2022. Adults with migraine and HCs were enrolled and underwent blood sampling. Migraine subgroups included episodic migraine, chronic migraine, migraine without aura and migraine with aura. Plasma AM concentrations were quantified using a validated immunoluminometric assay by trained personnel who were blinded to group status.
Results
In total, 667 participants with migraine and 147 HCs provided data eligible for analysis. Plasma AM concentrations did not differ significantly between the migraine and HC group (18.2 ± 9.1 pg/ml vs. 18.5 ± 8.4 pg/ml; p = 0.08). However, participants with episodic migraine exhibited lower AM levels than HCs (16.9 ± 7.7 pg/ml vs. 18.5 ± 8.4 pg/ml; p = 0.04). Multivariate regression models showed that plasma AM concentrations are positively associated with body mass index (4.2% increase in AM per kg/m2; 3.6 to 4.8%; p < 0.001) and monthly migraine days (0.6% increase in AM per MMD; 0.2 to 1.0%; p = 0.006).
Conclusions
Plasma AM concentrations were comparable between participants with migraine and HCs; however, those with episodic migraine exhibited slightly lower levels. Future studies should investigate other candidate blood-based biomarkers for migraine.
This is a visual representation of the abstract.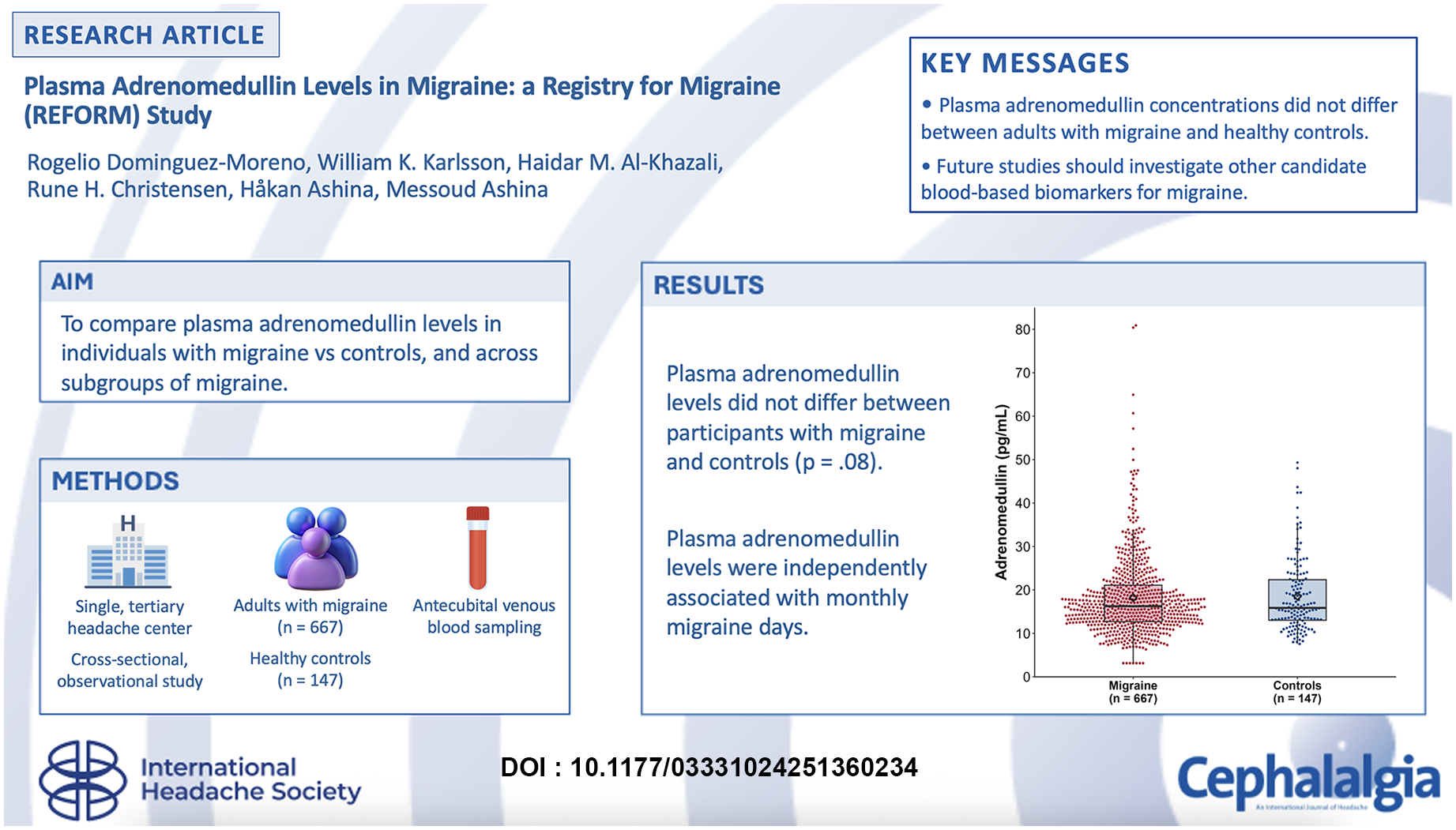
Introduction
Adrenomedullin (AM) is a neuropeptide that belongs to the same family of peptides as calcitonin gene-related peptide (CGRP).1–3 It exerts its effects by binding to G protein-coupled receptors, including the canonical CGRP receptor and distinct AM1 and AM2 receptors, located on cell membranes. 4 Preclinical data have revealed that AM and its receptors are expressed in trigeminal afferent neurons,5–8 which are widely accepted to be involved in migraine pathogenesis.9,10 In rodents, AM administration elicits hyperalgesia, whereas AM receptor antagonism produces anti-nociceptive effects. 11 Complementing these findings, a human experimental study demonstrated that intravenous administration of AM induced migraine attacks in 55% of individuals with migraine, whereas only 15% experienced attacks following placebo, underscoring its potential clinical relevance. 12 These observations position AM as a promising therapeutic target for migraine management, yet its broader implications remain underexplored.13,14
Despite its biological plausibility and emerging clinical significance, it remains uncertain whether circulating AM levels differ between individuals with migraine and healthy controls (HCs), or whether AM levels reflect clinically meaningful variation within the migraine population. To date, only two small studies have assessed plasma AM concentrations in individuals with migraine, yielding conflicting results.15,16 These discrepancies might reflect methodological limitations. Most notably, both studies included fewer than 30 participants with migraine and controls,15,16 considerably limiting statistical power and the ability to detect subtle differences.
To address current gaps in understanding, we enrolled a large sample of adults diagnosed with migraine and HCs, evaluating plasma AM concentrations across both groups and within migraine subgroups. In addition, we investigated the associations between plasma AM levels and selected demographic and clinical factors.
Methods
The data reported here originate from the parental Registry for Migraine (REFORM) study. 17 The REFORM protocol received approval from the relevant ethics committee, and all participants provided their written informed consent before enrollment. The research adhered to the principles of the Declaration of Helsinki, including subsequent amendments. This manuscript was drafted in accordance with the STROBE checklist. 18
Design and participants
This single-center study applied a cross-sectional, observational design to investigate adults with migraine and HCs. A detailed description of the recruitment process and eligibility criteria for both groups has been published elsewhere. 17
Eligible participants were adults aged ≥18 years with a diagnosis of migraine without aura, migraine with aura or chronic migraine according to the International Classification of Headache Disorders, 3rd edition (ICHD-3). 19 Migraine with aura was defined in accordance with ICHD-3 criteria. This classification was applied regardless of whether the individual had episodic or chronic migraine, thereby allowing comparisons across the full migraine spectrum. 19 Participants were also required to report an average of at least 4 monthly migraine days (MMDs) over the 3 months preceding enrollment. A migraine day was defined using ICHD-3 criteria as any day with definite migraine or probable migraine.
Other primary headache disorders were allowed in the migraine group, with the exception of hemiplegic migraine and cluster headache. Participants were required to reliably distinguish their migraine attacks from other headache types. Individuals with a history of secondary headache disorders – except medication-overuse headache – were excluded, as were those with clinically significant medical or psychiatric comorbidities. Participants had not been previously treated with erenumab, and any other anti-CGRP monoclonal antibody was discontinued at least 3 months before enrollment. Only one participant used a gepant (rimegepant) for acute treatment during the study period. Participants receiving preventive migraine medications had been on stable regimens for at least three months prior to blood sampling, reducing the likelihood of acute treatment effects on AM levels (see supplementary material, Table S1).
The HC group comprised adults with no personal history of any headache disorder, except for infrequent episodic tension-type headache. Potential HCs were also excluded if they reported first-degree relatives with a primary headache disorder, any clinically significant medical conditions, or regular use of medications.
Procedures
Participants with migraine underwent a semi-structured interview and physical examination conducted by trained personnel. This process systematically captured sociodemographic data, clinical characteristics, and details of current and prior migraine treatments, ensuring a thorough assessment of each individual's profile.
At the time of sampling, site investigators documented whether participants with migraine were experiencing an active migraine attack (ictal phase), a non-migraine headache or were headache-free (interictal phase). The ictal phase was defined as the presence of migraine meeting ICHD-3 criteria for a migraine attack without aura at the time of blood sampling. 19 Participants were also asked whether they had used a triptan or non-steroidal anti-inflammatory drug (NSAID) within 72 hours of blood sampling. These variables were included in the exploratory analyses.
Blood samples were collected from all participants via venipuncture performed in the cubital fossa, using 9-ml K2EDTA tubes. Following collection, samples were centrifuged at 1851 g for 10 minutes at 4°C to separate plasma from cellular components. The resulting plasma was then aliquoted into cryovials and stored at −80°C until subsequent analysis, maintaining stability of the target analytes. To minimize potential bias, laboratory personnel who were blinded to group status processed all samples in a randomized sequence.
Plasma AM concentrations were measured using the Sphingotest® bio-ADM® immunoluminometric assay (SphingoTec GmbH, Hennigsdorf, Germany; ASKA Biotech GmbH, Hennigsdorf, Germany). With a detection range of 10.8–1094.0 pg/ml, this assay reliably captured the full spectrum of plasma AM values observed across all participants with migraine and HCs.
Outcomes and measures
The primary outcome was the difference in plasma AM levels between participants with migraine and HCs. This comparison aimed to elucidate migraine-specific alterations in plasma AM concentrations. Secondary analyses included comparisons of plasma AM concentrations between clinically relevant subgroups: episodic vs. chronic migraine, migraine with vs. without aura, ictal vs. interictal sampling and preventive medication users vs. non-users. Each subgroup was also compared with HCs to contextualize findings. The patients were considered to have migraine with aura if they reported two or more aura episodes during their lifetime in accordance with the ICHD-3 diagnostic criteria. 19
The exploratory outcomes investigated associations between plasma AM concentrations and key variables in participants with migraine. These variables encompassed age, sex, body mass index (BMI), monthly headache days (MHD), MMD, and monthly number of days with acute headache medication use. MHD, MMD and monthly number of days with acute medication use were assessed for the previous 30 days.
Statistical analysis
The sample size was pragmatically determined based on the recruitment capacity of the REFORM study and the objective of enabling adequately powered subgroup comparisons. The target was to enroll at least 600 participants with migraine and approximately 150 HCs. Continuous data are presented as means ± standard deviations (SDs) or medians with interquartile ranges (IQR), as appropriate. The data distribution was assessed visually using histograms and QQ plots. Categorical data were summarized as frequency counts with percentages.
Plasma AM concentrations were modeled using linear regression, with pairwise comparisons of the estimated marginal means conducted via the emmeans package in R (R Foundation, Vienna, Austria). These comparisons included: (1) all participants with migraine vs. HCs; (2) each migraine subgroup vs. HCs; and (3) specific migraine subgroup pairs (e.g. episodic migraine vs. chronic migraine). Age, sex and BMI were included as covariates to control for potential confounding effects. As a sensitivity analysis, a BMI × diagnostic group interaction term was included in the regression model. As an additional sensitivity analysis, we stratified participants by number of migraine days (≥15 vs. <15) and headache days (≥15 vs. <15) within the last month. Each of these models was tested both with and without an interaction term between BMI and group. For the exploratory analyses, we performed univariate linear regression on the data of participants with migraine data to investigate associations between plasma AM concentrations (as the dependent variable) and MMD, MHD and acute headache medication use. All variables with p < 0.05 were then incorporated into a multivariable linear regression. Models assumptions were evaluated by assessing residuals to ensure normality and homoscedasticity. Logarithmic transformation was applied for plasma AM concentrations to address skewed residuals. Multicollinearity was examined by computing the variance inflation factor, and values <5 were considered acceptable. Missing covariate data were minimal (<5%) for the main analyses. Visual inspection identified extreme AM outlier values, which were excluded to maintain data integrity. p < 0.05 was considered statistically significant, with Bonferroni correction applied for multiple comparisons. All analyses were performed using R, version 4.3.3 software (R Foundation ).
Results
Between September 2020 and June 2022, 751 participants with migraine were enrolled in the REFORM study. A random sample of 669 participants with migraine was selected for plasma AM analysis, based on a predefined target of at least 600 cases. Two participants were excluded due to extreme outlier values (>29 SDs above the mean), resulting in a final analytic sample of 667 participants with migraine. Data from 147 HCs were included for comparison (see supplementary material, Figure S1).
Participants with migraine had a mean ± SD age of 43.9 ± 1.2 years, were mostly female (n = 595; 89.2%) and had a mean ± SD BMI of 25.2 ± 5.1 kg/m2. The migraine and HC groups were well-balanced in terms of sex and BMI (both p > 0.05). Table 1 outlines demographic and clinical information for both groups.
Demographics and baseline clinical characteristics of participants.

Abbreviations: BMI = body mass index; MHD = monthly headache days; MMD = monthly migraine days; NSAID = non-steroidal anti-inflammatory drug.
The migraine group had a mean ± SD of 19.7 ± 7.8 MHDs, 14.4 ± 7.1 MMDs and 12.0 ± 6.7 days per month with acute headache medication use. Furthermore, 406 (60.9%) participants had chronic migraine, 197 (29.5%) had migraine with aura and 198 (33.6%) were post-menopausal. Current use of preventive migraine medication(s) was reported by 338 (50.7%) participants with migraine. At the time of blood sampling, 303 (45.4%) participants reported experiencing a migraine attack and were classified as ictal. In terms of recent use of acute medications, 174 (28.4%) of participants with migraine had used a NSAID within 72 hours of blood sampling, whereas 217 (35.4%) had used a triptan. Gepants were used by only one participant in the REFORM study, and exclusively for acute treatment, because these agents were not approved or available for clinical use in Denmark during the study period.
Comparisons of plasma AM levels
Plasma AM levels in participants with migraine and HCs are presented in Figure 1 and Table 2. Linear regression, adjusted for age, sex and BMI, revealed no significant difference in plasma AM levels between all participants with migraine (18.2 ± 9.1 pg/ml) and HCs (18.5 ± 8.4 pg/ml; p = 0.08). Among subgroups, participants with episodic migraine had lower plasma AM concentrations (16.9 ± 7.7 pg/ml) compared to HCs (p = 0.04). By contrast, levels in those with chronic migraine did not differ from either HCs (p = 0.95) or the episodic migraine subgroup (p = 0.13). Characteristics of the chronic and episodic migraine subgroups are available in the supplementary material (Table S2).

Plasma adrenomedullin (AM) concentrations in migraine populations and healthy controls. Plasma AM levels (pg/ml) in participants with migraine compared to healthy controls and across various migraine subgroups, categorized by chronicity (episodic migraine [EM] vs. chronic migraine [CM]), aura status (migraine with aura [MA] vs. migraine without aura [MO]) and headache status at the time of blood sampling (migraine headache vs. no headache). The controls are depicted in blue. The box in each plot represents the interquartile range (Q1–Q3), the central horizontal line indicates the median, the diamond denotes the mean and individual dots correspond to measured values. Significant differences based on pairwise comparisons in linear regression, adjusted for age, sex and body mass index, are indicated by blue brackets with the corresponding p-values.
Plasma adrenomedullin (AM) levels (pg/ml) in participants with migraine (n = 667) and healthy controls (HCs) (n = 147).

Abbreviations: BMI = body mass index; CM = chronic migraine; EM = episodic migraine.
*Linear regression model adjusted for age, BMI and sex, with p-values adjusted using the Bonferroni method.
No significant difference was found in plasma AM concentrations between participants with migraine with aura (18.8 ± 9.5 pg/ml) and those with migraine without aura (18.0 ± 8.9 pg/ml; p = 0.50). These subgroups also did not differ from HCs (both p > 0.10). Furthermore, no differences were observed between ictal (18.3 ± 9.6 pg/ml) and interictal (17.7 ± 8.0 pg/ml) participants or between either subgroup and HCs (both p > 0.10). Likewise, plasma AM levels did not differ between participants using preventive migraine medication (18.5 ± 9.0 pg/ml) and those not using it (17.9 ± 9.2 pg/ml; p = 0.49). Both of these subgroups did not differ from HCs (both p > 0.05). Including an interaction term between BMI and migraine status did not alter the main findings, supporting the robustness of the observed associations (see supplementary material, Table S3). Stratifying participants by number of migraine days (≥15 vs. <15) and headache days (≥15 vs. <15) within the last month also yielded comparable results to our main analysis (see supplementary material, Table S4).
Exploratory multivariable regression analysis
The exploratory multivariable regression analysis revealed that plasma AM concentrations were positively associated with BMI (exponentiated β = 1.042; 95% confidence interval = 1.036–1.048; p < 0.001) (Table 3). This corresponds to a 4.2% increase in plasma AM levels for each one-unit increase in BMI. Likewise, plasma AM concentrations were positively associated with MMD (exponentiated β = 1.006; 95% confidence interval = 1.002–1.010; p = 0.006) (Table 3), indicating a 0.6% increase in plasma AM levels for each additional MMD. No significant association was found between plasma AM levels and age, sex, postmenopausal status, MHDs, monthly use of acute medications, or recent use of triptans or NSAIDs.
Linear regression model for am levels and clinical variables.

Abbreviations: BMI = body mass index; CI = confidence interval; MHD = monthly headache day; MMD = monthly migraine days; NSAID = non-steroidal anti-inflammatory drug.
Discussion
In this cross-sectional study, we investigated whether plasma AM concentrations differ between adults with migraine and sex- and BMI-matched HCs, using a validated assay. Our results showed no significant difference in plasma AM levels between the overall migraine population and HCs. However, participants with episodic migraine had lower plasma AM concentrations compared to HCs. Moreover, we observed positive associations between plasma AM concentrations and both BMI and MMD. These associations support the hypothesis that circulating AM is regulated by metabolic factors and disease burden. Nonetheless, the magnitude of these effects was small. For MMD, each additional MMD was associated with only a 0.6% increase in plasma AM levels. Although statistically significant, this incremental change lacks clinical relevance.
Existing evidence and added value
Preclinical reports have shown that AM and its responsive G protein-coupled receptors are expressed in trigeminal afferent neurons.5–8 In rodent pain models, AM administration induces hyperalgesia, whereas AM receptor antagonists produce anti-nociceptive effects. 11 These findings suggest that AM might modulate migraine-relevant pathways within the trigeminovascular system.
Previous data on circulating AM levels in people with migraine have been scarce and inconclusive. Two small studies reported conflicting results regarding circulating AM levels in migraine: one of them included 26 participants showing lower levels of AM during migraine attacks compared to HCs, whereas the other study with 30 participants did not find any difference between participants with migraine and HCs during migraine attacks.15,16 These incongruent findings raise questions about the pathophysiological role of AM in migraine.
Controlled experimental studies have demonstrated that intravenous AM administration provokes migraine attacks in a substantial proportion of affected individuals, further supporting its relevance as a pathophysiological target. 12 However, our results suggest that endogenous plasma AM levels do not reflect this pathogenic potential.
The modest reduction in plasma AM concentrations observed in episodic migraine is nonetheless somewhat intriguing. It might represent a compensatory downregulation in response to intermittent trigeminal activation. The absence of a similar pattern in chronic migraine is less easily explained but highlights the need to distinguish between migraine phenotypes when evaluating blood-based biomarkers. We acknowledge that the episodic migraine group represents a heterogeneous population, with individuals ranging from low- to high-frequency attack patterns. To provide greater granularity, we report detailed characteristics of the episodic and chronic migraine subgroups in the supplementary material (Table S2). Although further stratifying EM into low-, moderate- and high-frequency categories would have clinical relevance, our sample size was insufficient to support adequately powered subgroup comparisons. We therefore retained the ICHD-3 framework as our primary classification approach.
Although we identified positive associations with BMI and MMD, the effects were modest and do not have clinical implications on their own. These findings, however, point to an important interaction between metabolic status and neuropeptide signaling. Because obesity is a risk factor for migraine chronification,20,21 AM might be one of several mediators connecting these conditions.22–26
Taken together, our data suggest that blood-based AM measurements are unlikely to hold value as a clinically actionable biomarker for migraine. The lack of robust group-level differences, modest subgroup effects and small effect sizes with clinical variables all argue against utility in migraine care. Nonetheless, circulating AM might still provide some pathophysiological insight, particularly in the context of metabolic regulation and migraine frequency.
Future directions
Future investigations must confirm or refute our findings and contextualize it with longitudinal analyses. Repeated measurements over time, including pre-ictal, ictal and post-ictal phases, would clarify whether plasma AM undergoes cyclical variations. Furthermore, exploring central (e.g. cerebrospinal fluid) vs. peripheral AM levels could elucidate whether peripheral measurement fails to capture relevant neural processes. Although our findings suggest that preventive medication use does not significantly influence circulating AM levels, future studies with larger samples may be better positioned to evaluate class-specific pharmacologic effects. Combining AM with other well-studied neuropeptides, such as CGRP or pituitary adenylate cyclase-activating peptide, could help generate a multi-biomarker panel with the potential to refine our disease understanding. While we found no association between AM levels and menopausal status, our analysis was limited by the absence of menstrual phase tracking, which may be relevant for neuropeptide fluctuations. This limitation highlights an area for future investigation.
Strengths and limitations
This study has several strengths. First, the relatively large sample size allowed for stratified subgroup analyses. Despite the relatively large sample size allowing for subgroup analyses, the absence of a formal power calculation introduces the possibility that the study might have been underpowered to detect smaller but relevant differences. Second, we applied randomized sample processing and blinded measurement of plasma AM concentrations to minimize the risk of bias. Third, plasma AM levels were quantified using a validated immunoluminometric assay. 27 Although bio-ADM is less stable than MR-proADM, its functional relevance to vasodilatory and nociceptive processes made it a more appropriate biomarker for the study's mechanistic focus.28,29 Strict pre-analytical handling procedures were followed to preserve sample integrity and minimize degradation. Lastly, our investigation benefited from detailed collection of demographic and clinical information, enabling adjustment for potential confounders.
Nonetheless, certain limitations warrant mention. The cross-sectional design precluded longitudinal assessment of plasma AM concentrations, limiting our ability to capture dynamic alterations across migraine phases. Moreover, we did not systematically classify participants according to prodromal or postdromal states, limiting our ability to assess AM concentrations during these distinct migraine phases. Furthermore, participants with migraine were primarily recruited from the outpatient clinic of a national referral hospital. As a result, our migraine population might over-represent people with more severe, frequent and difficult-to-treat migraine, reducing generalizability to community-based populations or primary care settings. Information on menstrual cycle phase at the time of blood sampling was not collected, limiting our ability to assess short-term hormonal influences on AM levels beyond menopausal status.
Conclusions
Among adults with migraine, plasma AM concentrations did not differ from those in HCs, although individuals with episodic migraine had slightly lower levels than HCs. These results, combined with a positive association between plasma AM levels and MMD, suggest that AM might be a pathogenic contributor to migraine. Future investigations should explore whether preventive medications targeting the CGRP receptor, which is known to interact with AM, modulate plasma AM concentrations, potentially providing novel scientific insights.
Key findings
Plasma AM concentrations did not differ between adults with migraine and HCs.
Participants with episodic migraine had slightly lower plasma adrenomedullin levels than HCs.
Higher plasma AM concentrations were positively associated with BMI and MMDs.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251360234 - Supplemental material for Plasma adrenomedullin levels in migraine: A registry for migraine (REFORM) study
Supplemental material, sj-docx-1-cep-10.1177_03331024251360234 for Plasma adrenomedullin levels in migraine: A registry for migraine (REFORM) study by Rogelio Dominguez-Moreno, William K. Karlsson, Haidar M. Al-Khazali, Rune H. Christensen, Håkan Ashina and Messoud Ashina in Cephalalgia
Footnotes
Author contributions
Concept and design: MA and HA. Acquisition, analysis or interpretation of data: RDM, WKK, HMA, RHC, HA and MA. Drafting of the manuscript: RDM, WKK, HMA and RHC. Critical revision of the manuscript for important intellectual content: RDM, WKK, HMA, RHC, HA and MA. Statistical analysis: RDM and WKK. Administrative, technical or material support: MA. Supervision: MA and HA.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
RDM has received personal fees from Novartis and Pfizer, outside of the submitted work. RDM serves as a Visual Abstract Editor of Cephalalgia Reports. RHC has received personal fees from Pfizer and Teva, outside of the submitted work. WKK has received personal fees from Lundbeck and Pfizer, outside of the submitted work. HMA has received personal fees from Lundbeck and Pfizer, outside of the submitted work. HA has received personal fees from AbbVie, Lundbeck, Pfizer and Teva, outside of the submitted work. HA serves as an Editorial Board Member of The Journal of Headache and Pain. MA has received personal fees from AbbVie, AstraZeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva, outside of the submitted work. MA serves as an Associate Editor of Brain and The Journal of Headache and Pain.
Ethical statement
The REFORM protocol received approval from the relevant ethics committee, and all participants provided written informed consent before enrollment. The research adhered to the principles of the Declaration of Helsinki, including subsequent amendments.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from the Lundbeck Foundation (R310-2018–3711 to MA).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
