Abstract
Objective
While a substantial body of research describes the disabling impacts of migraine attacks, less research has described the impacts of migraine on physical functioning between migraine attacks. The objective of this study is to describe physical impairment during and between migraine attacks as a dimension of burden experienced by people living with chronic migraine.
Methods
The physical impairment domain of the Migraine Physical Function Impact Diary was recorded in headache diaries from the Medication Overuse Treatment Strategy trial. Days with moderate to severe headache were used to approximate migraine attacks. Factor analysis and regression analysis were used to describe associations between migraine and physical impairment.
Results
77,662 headache diary entries from 720 participants were analyzed, including 25,414 days with moderate to severe headache, 19,149 days with mild headache, and 33,099 days with no headache. Mean physical impairment score was 41.5 (SD = 26.1) on days with moderate to severe headache, 12.8 (SD = 15.0) on days with mild headache, and 5.2 (SD = 13.1) on days with no headache. Physical impairment on days with mild headache and days with no headache was significantly associated with days since last moderate to severe headache, physical impairment with last moderate to severe headache, mild headache (compared to no headache), depression, hypersensitivities and cranial autonomic symptoms.
Conclusions
Physical impairment occurs on migraine and non-migraine days. Study participants with frequent headaches, symptoms of depression, hypersensitivities and cranial autonomic symptoms experience physical impairment at a higher rate on days with no headache and days with mild headache.
Introduction
The World Health Organization ranks migraine as the second leading cause of disability measured in years lived with disability (YLDs), accounting for 41.1 million YLDs in 2019, 4.8% of all YLDs globally (1). Physical impairment during and between migraine attacks constitutes a key dimension of migraine-associated disability (2). While much of the disability resulting from migraine occurs during migraine attacks, disability can persist between migraine attacks. Quantifying disability during migraine attacks without also considering disability between migraine attacks underestimates the total burden experienced by people with migraine. While there have been numerous studies reporting migraine-associated disability during migraine attacks and total migraine-related disability based on answers to questionnaires requiring patient recall over a given period (e.g. MIDAS, HIT-6), there has been a lack of studies quantitatively assessing disability on migraine and non-migraine days using daily headache diary data.
The objectives of this study were to describe physical impairment on migraine days and non-migraine days and to identify factors associated with physical impairment.
Methods
Standard protocol approvals, registrations, and patient consents
The Medication Overuse Treatment Strategy (MOTS) Trial was approved by the Mayo Clinic Institutional Review Board and the IRB of record for each enrolling site, and it was registered at ClinicalTrials.gov (NCT02764320) with predefined primary and secondary outcome measures. All participants underwent an informed consent process and signed IRB-approved consent forms.
Study design and population
The present study analyzed data from 720 chronic migraine patients collected in the MOTS Trial (3). Participants were recruited in the course of routine clinical care at 34 sites in the United States including six primary care clinics, 14 general neurology clinics, and 14 headache specialty clinics. Eligibility rules required participants to be 21 years of age or older and meet International Classification of Headache Disorders 3 beta (ICHD-3β) (4) diagnostic criteria for chronic migraine and for medication overuse as defined within the medication overuse headache diagnostic criteria. ICHD-3β was the most recent ICHD diagnostic criteria available at the time of study initiation. Nine hundred and seventy eight individuals were assessed for eligibility (see Figure 1).
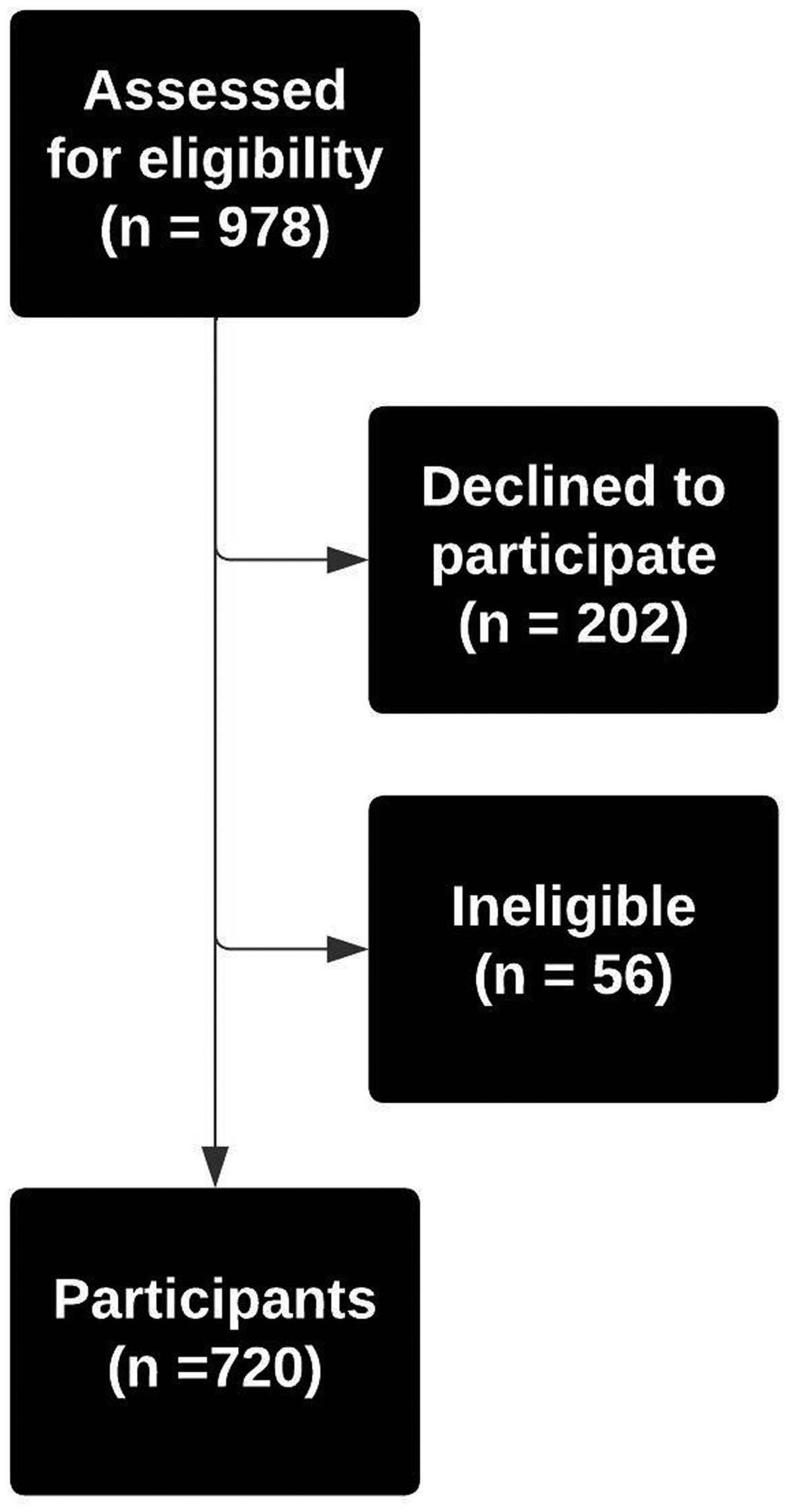
Flow Diagram
Measures
Headache diary
Participants completed daily electronic headache diaries for up to 20 weeks during their involvement in the MOTS Trial. Each day in the headache diary, participants indicated whether they experienced headache, peak headache intensity (mild, moderate, or severe), headache duration, a one-item measure indicating whether they found the headache disabling on a four-point scale from “able to function normally” to “not able to function at all”, migraine medication use, pain medication use, and the Physical Impairment domain of the Migraine Physical Function Impact Diary (MPFID-PI), discussed below. Diary entries were date and time stamped. Compliance with diary keeping was maximized by allowing access through a mobile application and a web-based platform, sending automated e-mail reminders to complete diary entries, and providing remuneration for diary compliance. In total, 77,662 headache diary entries were recorded. The headache diary employed in MOTS was designed to measure the primary outcome, frequency of days with moderate to severe headache (DMoH). The diary did not collect all information required to formally assess whether symptoms met criteria for a migraine attack. In our analysis, self-reported DMoH was used as a proxy for a migraine attack. In the study population (diagnosed chronic migraine with medication overuse, headache days per 28 days mean 22.5 [SD 5.1]), it is likely that the vast majority of DMoH were attributable to migraine attacks (3). Days were considered DMoH when patients reported headache that lasted at least two hours and peaked at moderate or severe intensity. Days were classified as days with mild headache (DMiH) when patient diary data indicated a headache that did not meet criteria for moderate to severe headache and days with no headache (DNH) when the diary indicated no headache. Self-reported DMiH and DNH were used as a proxy for non-migraine days. While these proxy definitions may misclassify some migraine days, we believe they are useful in describing findings.
Migraine Physical Functioning Impact Diary
In each headache diary entry, participants were prompted to complete select questions from the MPFID, a validated instrument measuring migraine impact on physical functioning over the prior 24 hours (5,6). The selected questions constitute the physical impairment (PI) domain of the MPFID, five items, each rated on a five-point scale. Three items ask about severity of difficulty (e.g., “In the past 24 hours, were you able to get out of bed?”) rated on a scale from “without any difficulty” to “unable to do”. Two items ask about duration of difficulty (e.g., “In the past 24 hours, how much of the time did you have difficulty moving your head?”) rated on a scale from “none of the time” to “all of the time”. Sum scores are linearly transformed to take a range of zero to 100 where higher score values indicate greater migraine-related physical impairment.
Covariate measures
Age, sex, race, and ethnicity were collected as demographic measures. Participants completed a headache characteristics questionnaire at baseline, constructed using methodology aligned with the NIH National Institutes of Neurological Disorders and Stroke Common Data Elements for headache (7). The questionnaire included headache frequency, date of chronic migraine onset, medication use, headache pain characteristics, and an inventory of non-headache migraine symptoms. Headache pain characteristics included average headache duration if untreated or inadequately treated, average headache duration if successfully treated, pain location (right, left, front, back, and/or side), whether the pain was typically unilateral, pain quality (pulsating/throbbing, pressure/aching, stabbing, and/or burning), average pain intensity, maximum pain intensity, and whether headache worsened with physical activity. Non-headache symptoms included nausea, vomiting, sensitivity to light, sensitivity to sound, conjunctival injection, tearing, nasal congestion/rhinorrhea, eyelid drooping, visual aura, sensory aura, motor aura, language aura, and other auras.
Participants also completed several validated questionnaires at baseline: Headache Impact Test 6 (HIT-6), Patient Reported Outcomes Measurement Information System (PROMIS) Pain Interference Questionnaire 6b, Generalized Anxiety Disorder 7 (GAD-7), and Patient Health Questionnaire 9 (PHQ-9). HIT-6 is used to assess the impact of headaches on the respondent’s functioning in vocational, social, and home settings (8). HIT-6 scores range from 36 to 78, where 36 to 49 indicates “little or no impact”; 50 to 55, “some impact”; 56–59, “substantial impact”; and 60 to 78, “severe impact”. PROMIS Pain Interference 6b measures the extent to which pain has interfered with social, cognitive, emotional, physical, and recreational activities over the prior seven days (9). GAD-7 is used to screen for symptoms of generalized anxiety disorder (10,11). GAD-7 scores range from 0 to 21, where 0 to 4 indicates “minimal anxiety”; 5 to 9, “mild anxiety”; 10 to 14, “moderate anxiety”; and 15 to 21, “severe anxiety”. PHQ-9 is used to screen for symptoms of depressive mood disorders (12). PHQ-9 scores range from 0 to 27, where 0 to 4 indicates “none-minimal”; 5 to 9, “mild”; 10 to 14, “moderate”; 15 to 19, “moderately severe”; and 20 to 27 “severe” depression.
Statistical analyses
Variables from the questionnaires and daily diaries were analyzed individually by frequency and distribution. MPFID-PI, HIT-6, PROMIS Pain Interference Questionnaire 6b, GAD-7, and PHQ-9 scales were scored according to published guidelines and MPFID-PI was evaluated for internal consistency. In headache diary data, only entries with complete responses to the headache diary and MPFID-PI were analyzed. Exploratory factor analysis and confirmatory factor analysis were used to extract common factors from the inventory of non-headache migraine symptoms contained within the headache characteristics questionnaire. Correlations among headache diary variables, MPFID-PI, and covariate measures were examined to inform regression modeling. An initial linear mixed model of MPFID-PI on DMoH was used to assess the utility of common factors extracted from non-headache symptoms and inform the design of a time series regression model of MPFID-PI on DMiH and DNH.
Results
Headache diary data
Diary data contained 25,414 DMoH and 52,248 combined DMiH and DNH, including 19,149 DMiH and 33,099 DNH. Headache information and MPFID-PI were both recorded in 77,381 of 77,662 diary entries (99.6%). MPFID-PI scores differed by headache status: Mean on DMoH was 41.5 (SD = 26.1). Mean on DMiH and DNH combined was 8.0 (SD = 14.3), including a mean of 12.8 (SD = 15.0, n = 19,149) on DMiH and 5.2 (SD = 13.1, n = 33,099) on DNH (see Table 1). Internal consistency of the scores was excellent (α = 0.96) with overall mean 19.0 (SD = 24.7).
Mean MPFID-PI by Headache Status: Mean MPFID-PI is higher DMoH than DMiH and DNH. Among mild and headache free days, mean MPID-PI is higher on mild headache days than headache free days.
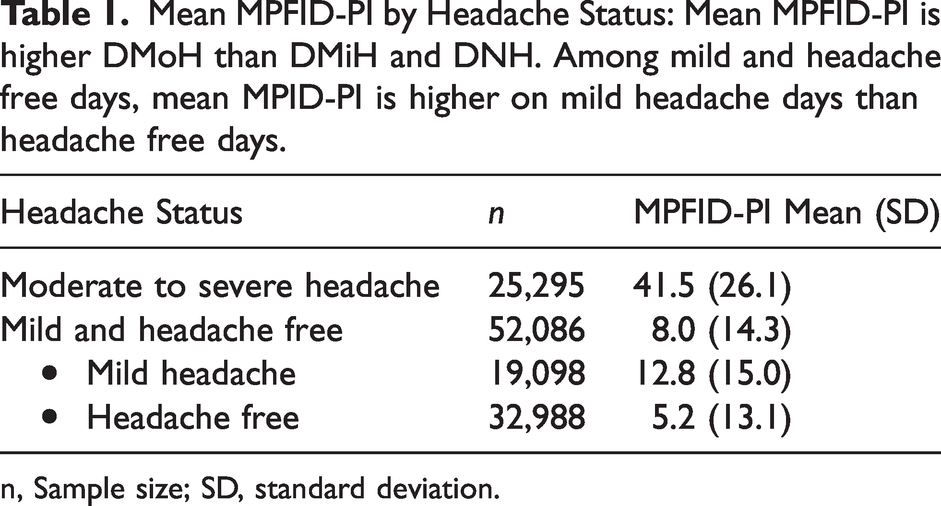
n, Sample size; SD, standard deviation.
MPFID-PI on DMoH was distributed roughly normally (median = 40.0, mean = 41.5, skewness = 0.3, kurtosis = 2.4; see Figure 2) and conducive to modeling as a continuous variable. MPFID-PI on DMiH and DNH was strongly zero-inflated; 63.5% of combined DMiH and DNH entries reported no physical impairment (median = 0, mean = 8.0, skewness = 2.2, kurtosis = 8.2; see Figure 2). MPFID-PI on DMiH and DNH was not conducive to modeling as a continuous variable. However, the relative frequency with which participants reported any physical impairment (MPFID-PI > 0) varied systematically with respect to migraine characteristics and time (see, e.g., rates of physical impairment by day since last DMoH in Table 2). Thus, the likelihood of physical impairment (as defined by MPFID-PI > 0) was conducive to modeling, and logistic regression was used to model the likelihood of physical impairment on DMiH and DNH.
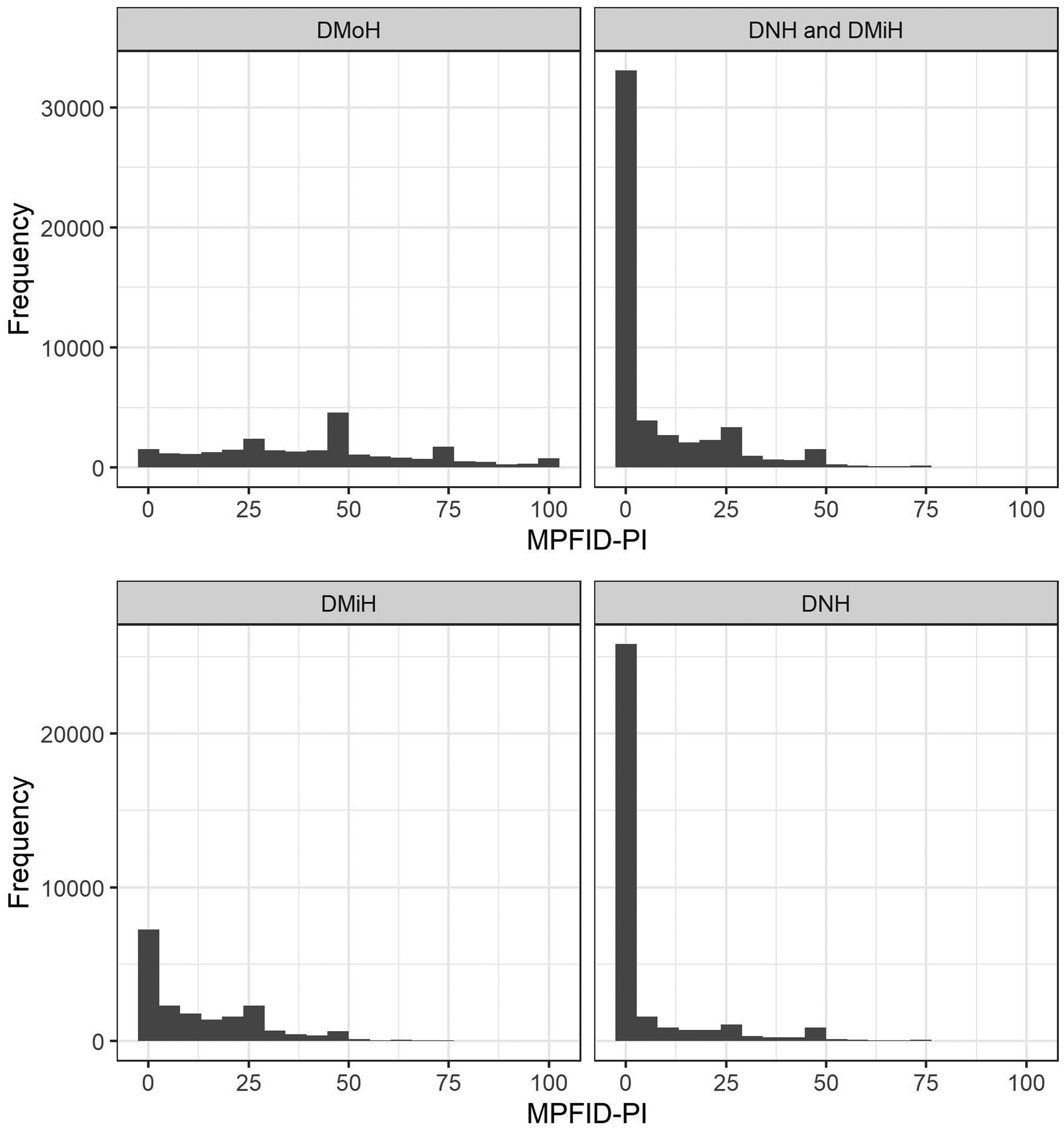
MPFID-PI Distribution by Headache Status
Physical Impairment on Non-Migraine Days: The proportion of individuals reporting any physical impairment on non-migraine days decreased as the number of days since last DMoH increased.
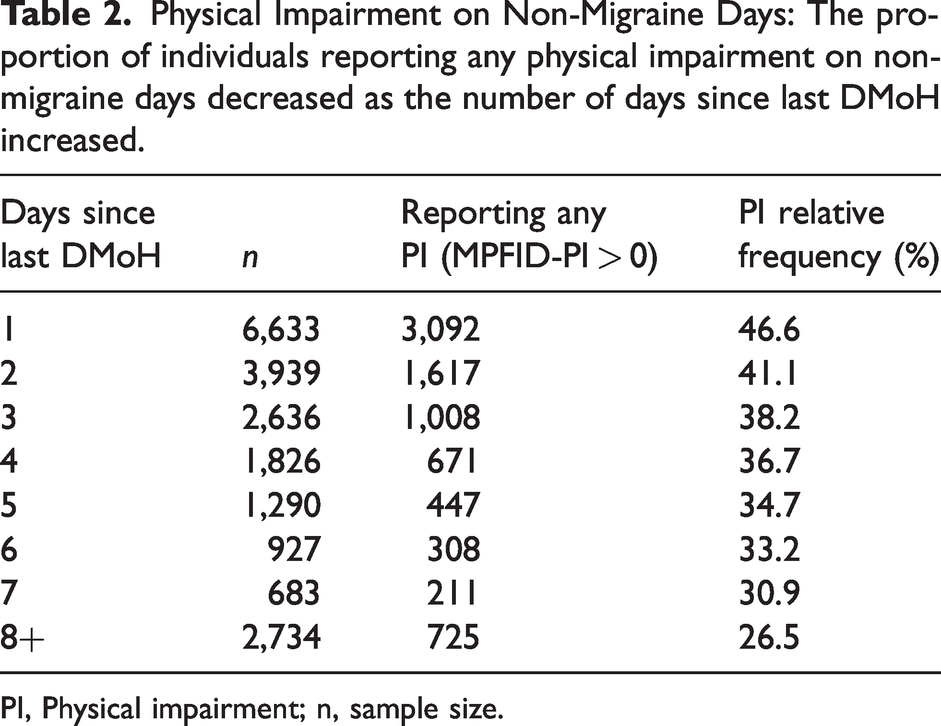
PI, Physical impairment; n, sample size.
Covariate measures
Among 706 participants who responded to both the demographic questionnaire and patient questionnaires: Age ranged from 21 to 83 years with mean 43.9 (SD = 13.0). Six hundred and seventeen participants were assigned female at birth (87.4%), 88 were assigned male (12.5%), and one declined to identify (0.1%). With respect to race, two participants identified as American Indian/Alaska Native (0.3%), nine Asian (1.3%), 44 Black/African American (6.2%), 0 Hawaiian/Other Pacific Islander (0.0%), 574 White (81.3%), and 67 other (9.2%), while 10 declined to identify (1.4%). With respect to ethnicity, 94 participants identified as Hispanic (13.3%), 600 as not Hispanic (85.0%), and 12 declined to identify (1.7%). Statistics describing HIT-6, PROMIS Pain Interference 6b, GAD-7, and PHQ-9 scores are presented in Table 3.
Covariate measures.
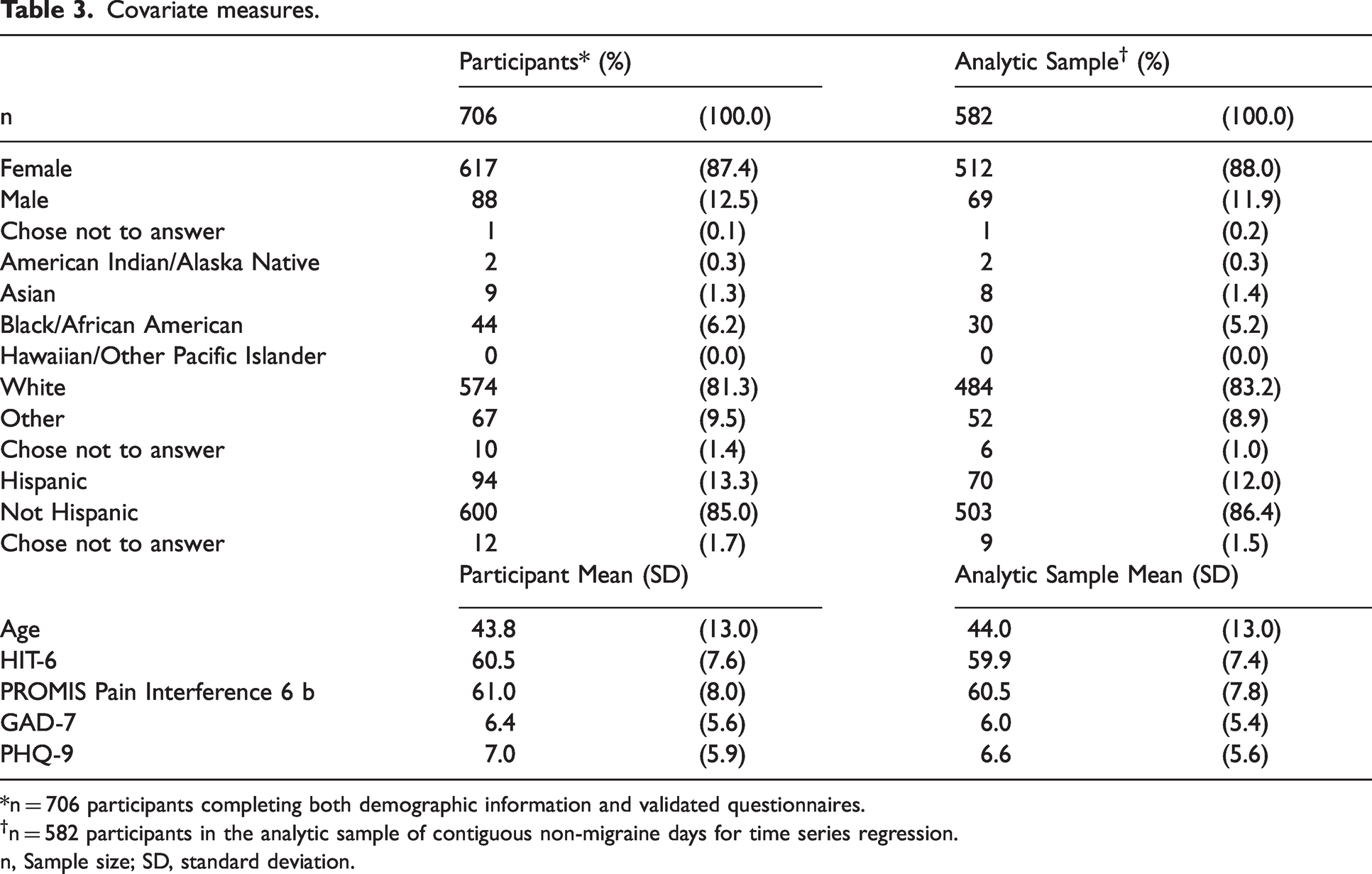
*n = 706 participants completing both demographic information and validated questionnaires.
n = 582 participants in the analytic sample of contiguous non-migraine days for time series regression.
n, Sample size; SD, standard deviation.
Factor analysis of headache characteristics
Among headache characteristics, non-headache symptoms were correlated with MPFID-PI on DMoH (e.g., ρphotophobia, MPFID-PI = 0.24) and with one another (e.g., ρnausea, vomiting = 0.73). This prompted factor analysis of non-headache migraine symptoms to attempt to extract common factors, such that non-headache symptoms could be retained as explanatory variables in regression without introducing several correlated, binary regressors. In exploratory factor analysis, a model with four factors in geomin rotation proved to be the most analytically useful, cumulatively explaining 59% of item variance and organizing symptoms into four common factors:
F1 (Auras): language aura, motor aura, visual aura, and sensory aura; F2 (GI Symptoms): nausea and vomiting; F3 (Hypersensitivities): photophobia and phonophobia; and F4 (Cranial Autonomic Symptoms): tearing, nasal congestion/rhinorrhea, conjunctival injection, and drooping eyelid.
Confirmatory factor analysis (CFA) was used to assess the fit of a model with four factors retaining factor loadings > 0.3 from EFA (see CFA model as specified in Figure 3). The CFA model fit well with Tucker-Lewis fit index (TLI) 0.96.
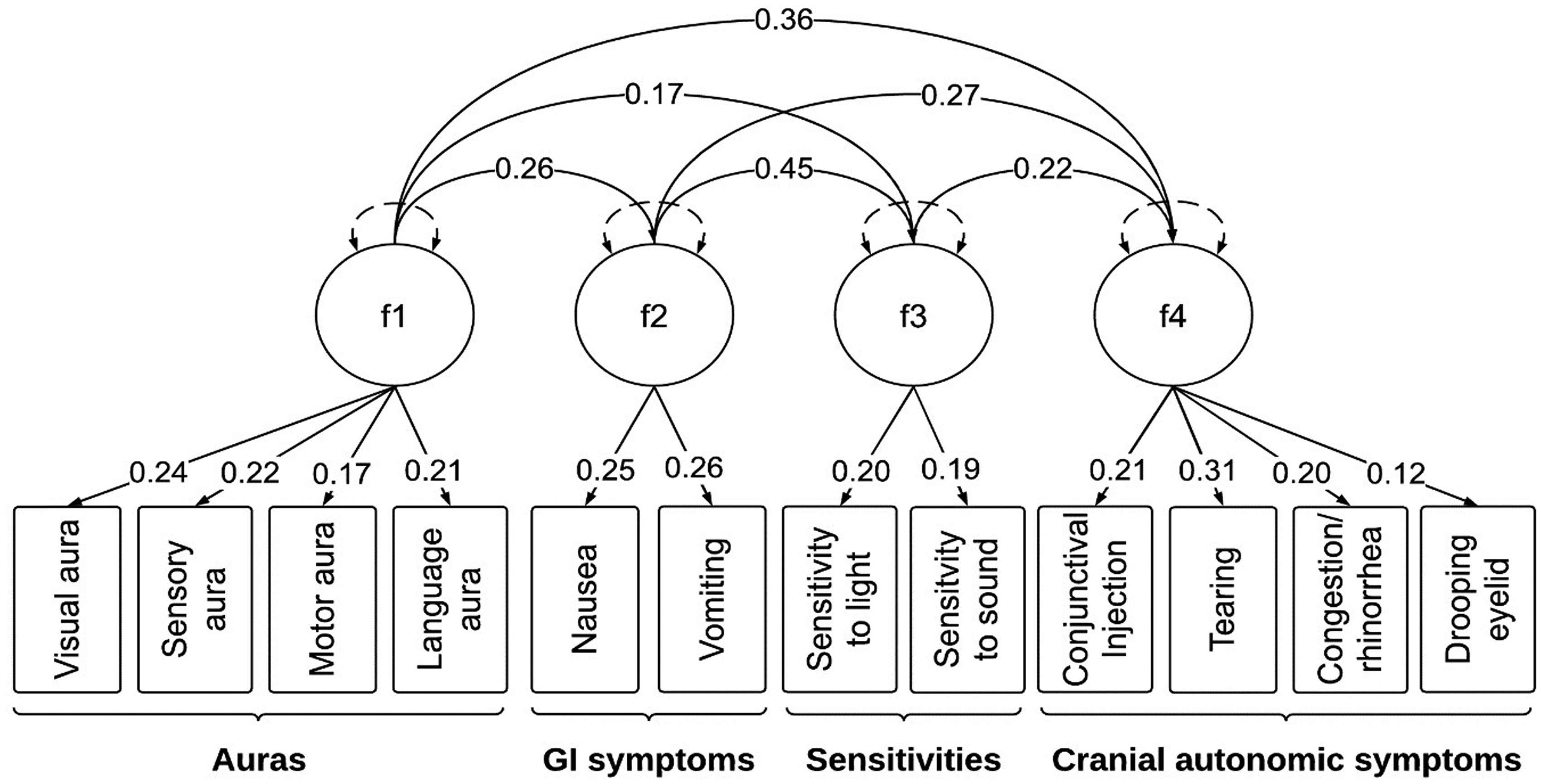
Confirmatory Factor Analysis Model of Non-Headache Symptoms
Regression analysis of physical impairment on DMoH
To test the utility of the common factor scores, severity scores were calculated for each common factor and included in regression analysis of MPFID-PI on DMoH. Scores were calculated by counting the number of symptoms assigned to each factor that were endorsed by the participant. For example, a participant who reported no history of auras would be assigned a score of zero on F1 (Auras), while a participant who reported a history of visual auras and sensory auras would be assigned a score of two. Diary data contained repeated observations of each participant, which raised the possibility that data were overdispersed due to clustering by participant. To test for a clustering effect and correct for overdispersion, linear mixed effects regression was employed. We first constructed a reference model using headache duration and peak pain intensity to predict MPFID-PI on DMoH (n = 25,295). In the reference model, fixed effects of headache duration and peak headache intensity explained 27% of the variation in MPFID-PI (
Regression Analysis of MPFID-PI on DMoH: Fixed effects in the reference model explain 27% of variation in MPFID-PI (
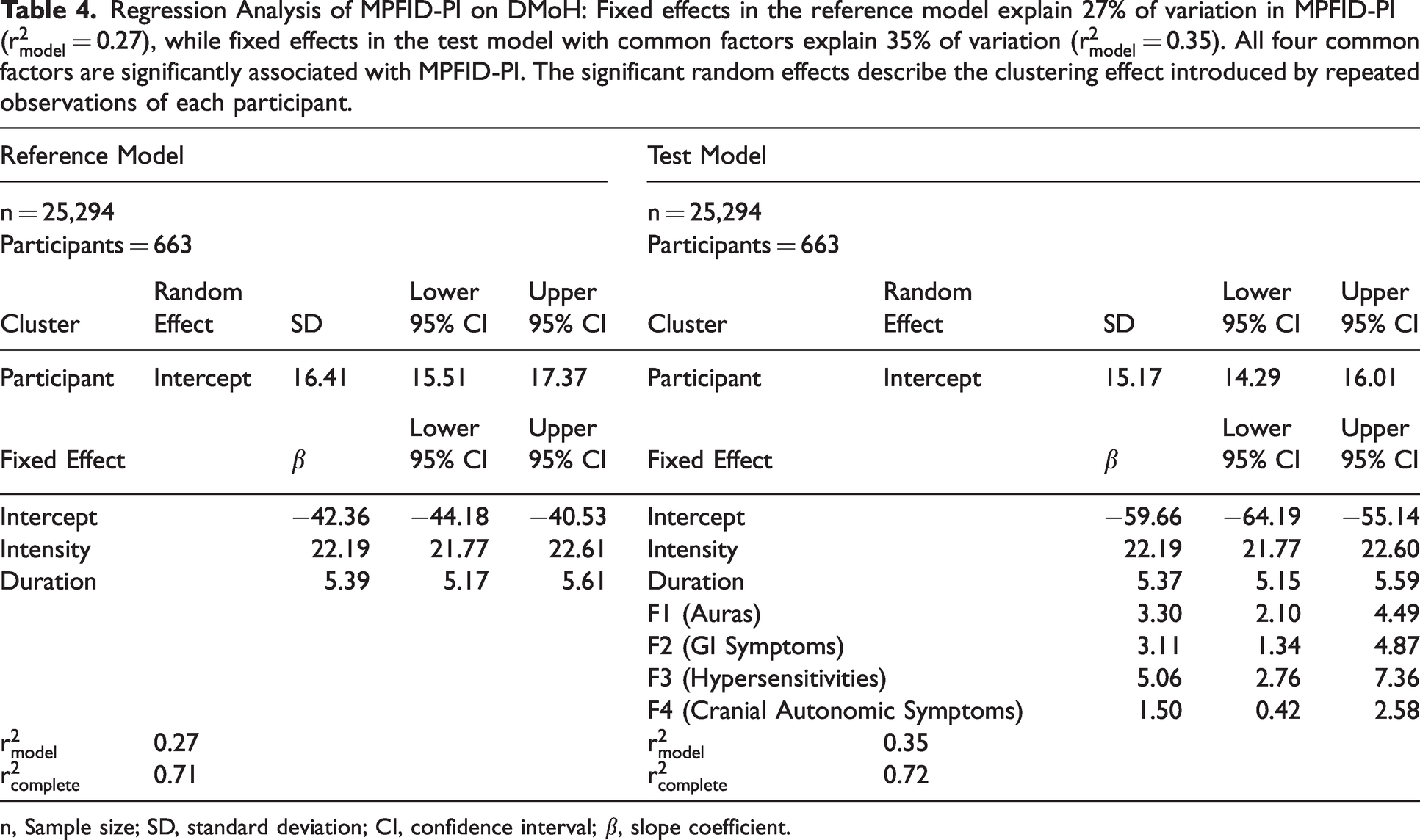
n, Sample size; SD, standard deviation; CI, confidence interval; β, slope coefficient.
Regression analysis of physical impairment on DMiH and DNH
Analyzing physical impairment on DMiH and DNH as a time series required the calculation of days since last migraine and days until next migraine (using DMoH as a proxy). In that calculation, an unlogged DMoH could render an erroneous count of days since last DMoH or days until next DMoH. To rule out that possibility, diary entries were only retained for time series analysis when each day since the last DMoH and until the next DMoH was also logged (n = 21,652). The shortest continuous span of combined DMiH and DNH was one day and the longest was 80 days (mean = 4.6, SD = 6.2). The analytic sample was trimmed at the third standard deviation of days since last DMoH and days until next DMoH, retaining only diary entries within 23 days of the last DMoH and 23 days of the next DMoH, resulting in an analytic sample of 20,658 complete records from 580 participants.
Subsequently, associations between MPFID-PI and each other variable were tested using univariable logistic mixed models. Those variables that were significantly associated in univariable models were retained in multivariable modeling (with certain exceptions described below). The multivariable model included: mild headache on diary day, presence of any physical impairment on last DMoH, F1 (auras), F3 (hypersensitivities), F4 (cranial autonomic symptoms), days since last DMoH, duration of last moderate to severe headache, GAD-7, and PHQ-9. Pain medication and migraine abortive medication use on a diary day were positively associated with physical impairment, which was interpreted as a confounded relationship (i.e., a third variable such as headache intensity may have caused both physical impairment and pain medication use). Pain medication and migraine abortive medication use were excluded from multivariable modeling. Days since last DMoH and days until next DMoH are characterized by a determinative algebraic relationship; days since last DMoH was advanced to multivariable modeling for its stronger correlation to physical impairment (ρDLM, PI = −0.16, ρDNM, PI = −0.14). The presence of physical impairment on the last DMoH expressed as an event (event = 1, non-event = 0) had better explanatory power than MPFID-PI score on last DMoH in univariable models and the event variable was advanced to multivariable modeling.
The final model is presented in Table 5. Mild headache, physical impairment on last DMoH, F3 (hypersensitivities), F4 (cranial autonomic symptoms), days since last DMoH, and PHQ-9 were significantly associated with physical impairment on DNH and DMiH (α = 0.05). F1 (auras), duration of last moderate to severe headache, and GAD-7 were not significantly associated with physical impairment on DNH and DMiH. Mild headache was associated with physical impairment as both a fixed effect, describing the mean effect among all participants, and a random effect, describing differences in the effect size among individual participants. A random intercept term describing individual differences in likelihood of physical impairment was also significant. Using odds ratio estimates to describe the size of fixed effects in the model of physical impairment on non-migraine days:
Logistic Mixed Model of PI on DMiH and DNH: Likelihood of physical impairment on DMiH and DNH was significantly associated with mild headache on that diary day, physical impairment on last DMoH, F3 (hypersensitivities), F4 (cranial autonomic symptoms), days since last DMoH, and PHQ-9.
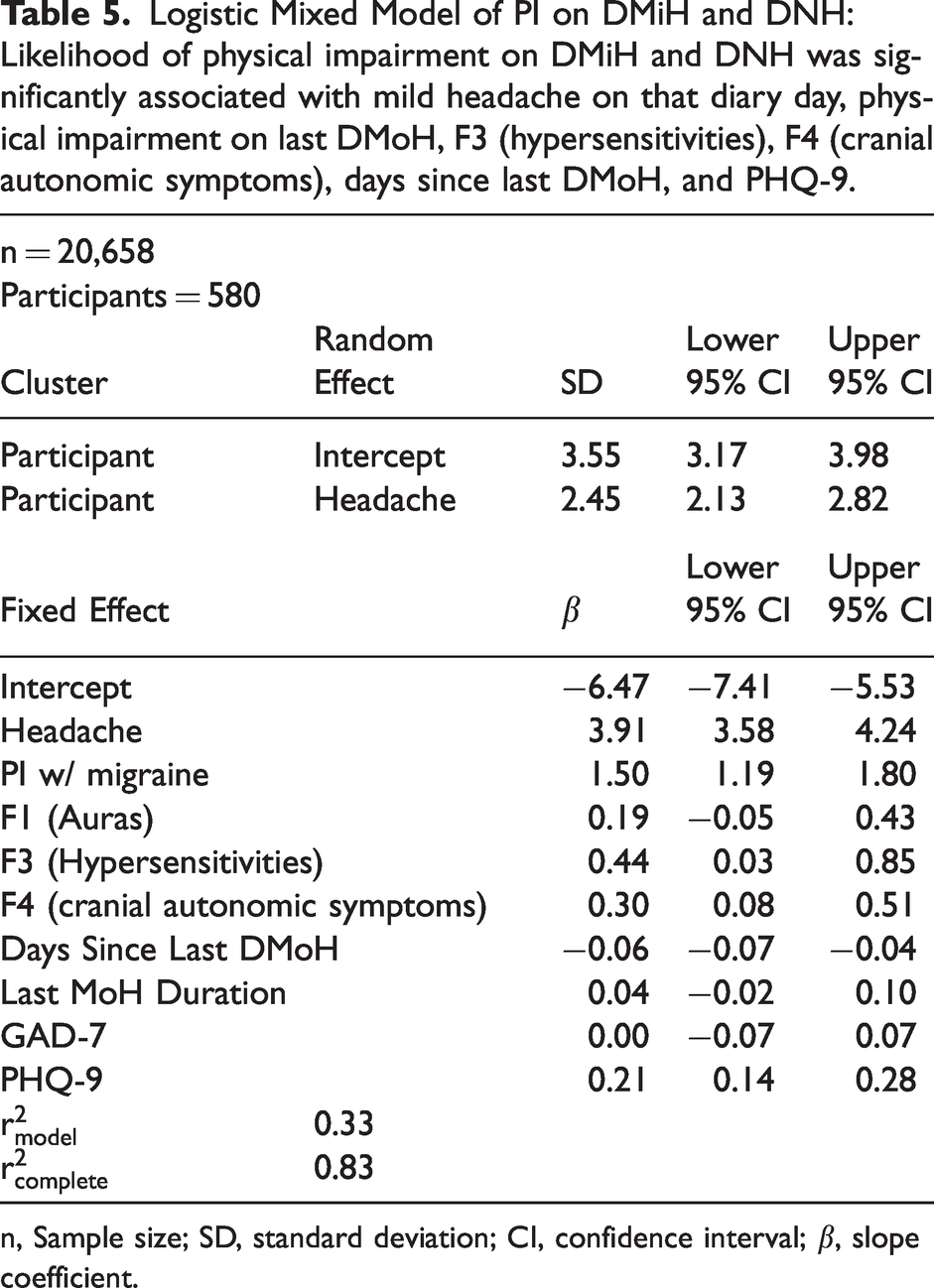
n, Sample size; SD, standard deviation; CI, confidence interval; β, slope coefficient.
On DMiH, participants were 49.9 times more likely to report physical impairment compared to DNH (95% CI 35.9 to 69.4).
Participants were 4.5 times more likely to report physical impairment on DMiH and DNH when they reported physical impairment on their last DMoH (95% CI 3.3 to 6.0).
With an additional day since last DMoH, participants were 5.8% less likely to report physical impairment (95% CI 3.9% to 6.8%).
While fixed effects explained 33% of the variation in likelihood of physical impairment on DMiH and DNH, random effects explained an additional 50% of the variation.
Discussion
Patterns of physical impairment on DMoH, DMiH, and DNH suggest that physical impairment: 1) is greatest on migraine days, but also occurs on non-migraine days; 2) is less likely to be present as one gets further from their last migraine; 3) is higher on DMiH than DNH; 4) is partially explainable by migraine headache intensity, history of non-headache symptoms, days since last migraine, occurrence of mild headache, and depression as measured in PHQ-9; and 5) is characterized by individual variation among participants.
Physical impairment and, more broadly, functional impairment during migraine attacks have been consistently described and thoroughly quantified in the literature. For example, in Brandes’s study (13) of migraine in the United Kingdom, France, Germany, Italy, and the United States, 52.3% of the 516 adult migraine patients who were interviewed required bed rest to manage migraine attacks. In Buse and colleagues’ systematic review (14), they found that impairment related to migraine incurred enormous indirect costs through missed work, school absenteeism, and adverse impacts to family relationships.
While functional impairment on non-migraine days has been described in the literature, a smaller body of work has attempted to quantify this dimension of disease burden. Buse and colleagues’ review (14) further describes the nature of burden between migraine attacks, citing worry, poorer sleep, and lower energy levels between attacks, with fewer than half of migraine patients surveyed reporting that they recover fully between attacks. Dahlöf and Dimenäs (15) compared self-report measures from 101 healthy control participants to those of 103 migraine patients surveyed between migraine attacks and found migraine patients reported poorer contentment, vitality, and sleep. Findings from Mannix and colleagues’ (2) systematic review and subsequent concept elicitation interviews with 32 participants elaborated the role of physical impairment in migraine-associated disability during and between migraine attacks. Twenty-eight of 32 participants (88%) mentioned physical function without prompting, citing impairments such as needing to lie down and difficulty moving their heads, bending over, getting out of bed, or performing activities that required physical effort both during and between migraine attacks. Ultimately, Mannix and colleagues (2) found physical function to be the primary concept of interest in elicitation from patients, present in ictal and interictal periods in both episodic migraine and chronic migraine.
Our analysis is unique since it: 1) measured physical impairment daily using a headache diary, yielding more detailed information than the use of questionnaires with weekly, 30 day, or 90 day recall timeframes; 2) measured physical impairment on DMoH, DMiH, and DNH; 3) investigated the impact of time since last migraine on physical impairment; and 4) identified other factors associated with physical impairment, such as symptoms that occur during migraine attacks.
The following limitations of the MOTS Trial design and our analysis should be considered. 1) These findings may not generalize to populations with migraine diagnoses other than chronic migraine with medication overuse. 2) While history of non-headache symptoms was found to be significantly associated with physical impairment, the occurrence of non-headache symptoms was not included in the daily diary. It is unclear whether the occurrence of these symptoms directly caused physical impairment. 3) The measurement point in time for anxiety and depression—years into chronic migraine for some participants—was not conducive to investigating direction of causation between depression and physical impairment. 4) Our analysis did not account for the use of acute migraine medications. The use of acute migraine medications may partially explain variation in physical impairment on subsequent days through either the therapeutic effect or the side effects of those medications. 5) Since data about non-headache symptoms such as sensitivities and nausea were not collected in the MOTS Trial headache diaries, we used DMoH as a proxy to analyze general trends surrounding migraine attacks. Although we expect the vast majority of moderate to severe headaches lasting at least two hours would meet criteria for a migraine day in this population, it is possible that some would not. It is also possible that participants with mild headache may still fulfill criteria for a migraine or probable migraine attack on a given day.
Future analyses should describe physical impairment in diverse populations and populations with diagnoses other than chronic migraine with medication overuse to investigate the extent to which these findings are generalizable. Non-headache symptoms should be included in daily diary entries to analyze the nature of the relationship between these symptoms and physical impairment. Prospectively collecting repeated measures of anxiety and depression would yield data more conducive to meaningful interpretation of the significant associations among anxiety, depression, and physical impairment that we observed. Finally, future analyses should investigate the possible relationship between acute migraine medication use and physical impairment between migraine attacks.
Conclusions
Our analysis offers further evidence that physical impairment on non-migraine days constitutes a substantive dimension of disease burden among people living with migraine and advances the quantitative description of the topic. Physical impairment on DMoH is associated with headache duration, headache intensity, and history of aura, GI symptoms, hypersensitivities, and cranial autonomic symptoms. Physical impairment on DMiH and DNH is partially explainable by mild headache, days since last DMoH, physical impairment on last DMoH, depression, history of hypersensitivities, and individual variation among participants. Study participants with more frequent headaches, more symptoms of depression, and history of hypersensitivities and cranial autonomic symptoms experience physical impairment more often on DMiH and DNH.
Key findings
People living with chronic migraine experience physical impairment on days with headache and days without headache. Physical impairment on days with mild or no headache is associated with days since last moderate to severe headache, physical impairment on the last day with moderate to severe headache, currently having a mild headache (rather than no headache), depression, hypersensitivities, and cranial autonomic symptoms.
Footnotes
Acknowledgement
The authors acknowledge and thank the patient partners who helped plan and run the The Medication Overuse Treatment Strategy (MOTS) trial. They also acknowledge the MOTS investigators, MOTS Steering Committee, and Data Safety Monitoring Board.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DJW reports nothing to disclose. GMD reports grants from the American Brain Foundation/American Academy of Neurology and Amgen. JGH reports grants from the Patient Centered Outcomes Research Institute (PCORI). DWD reports personal fees from Amgen, Atria, CapiThera Ltd., Cerecin, Ceruvia Lifesciences LLC, CoolTech, Ctrl M, Allergan, AbbVie, Biohaven, GlaxoSmithKline, Lundbeck, Eli Lilly, Novartis, Impel, Satsuma, Theranica, WL Gore, Genentech, Nocira, Perfood, Praxis, AYYA Biosciences, Revance, and Pfizer. He has received speaking fees from from Teva, Amgen, Eli Lilly, Lundbeck, and Pfizer and honoraria from American Academy of Neurology, Headache Cooperative of the Pacific, Canadian Headache Society, MF Med Ed Research, Biopharm Communications, CEA Group Holding Company (Clinical Education Alliance LLC), Vector Psychometric Group, Clinical Care Solutions, CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Academy for Continued Healthcare Learning, Majallin LLC, Medlogix Communications, Medica Communications LLC, MJH Lifesciences, Miller Medical Communications, WebMD Health/Medscape, Wolters Kluwer Health, Oxford University Press, Cambridge University. He has stock or stock options in Ctrl M, Aural Analytics, Axon Therapeutics, ExSano, Palion, Man and Science, Healint, Theranica, Second Opinion/Mobile Health, Epien, Nocira, Matterhorn, Ontologics, King-Devick Technologies, Precon Health, Ayya Biosciences, Cephalgia Group, Atria Health. He sits on the board of directors for Epien, Matterhorn, Ontologics, King-Devick Technologies, Precon Health, Axon Therapeutics, and Cephalgia Group. He has received research support from United States Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation, and Patient Centered Outcomes Research Institute (PCORI). He holds Patent 17189376.1-1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis without fee and has submitted a patent application for Synaquell® (Precon Health). TJS reports a grant from PCORI during the conduct of the MOTS Trial; grants from Amgen, American Migraine Foundation, United States Department of Defense, National Institutes of Health, American Heart Association, Spark Neuro, and Henry Jackson Foundation, personal fees from Abbvie, Allergan, Amgen, Axsome, Biodelivery Science, Biohaven, Click Therapeutics, Collegium, Eli Lilly, Ipsen, Linpharma, Lundbeck, Novartis, Satsuma, Theranica, Tonix, and Scilex. He has submitted a patent application for predicting speech outcomes in post-traumatic headache and migraine. He reports stock options from Aural Analytics and Nocira and royalties from Up To Date.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this article was funded through a Patient Centered Outcomes Research Institute (PCORI) award (PCS-1504-30133). The views in this publication are solely the responsibility of the authors and do not necessarily represent the view of the PCORI, its Board of Governors, or its Methodology Committee. Amgen granted free use of the Migraine Physical Function Impact Diary for the Medication Overuse Treatment Strategy (MOTS) Trial under a license agreement with Mayo Clinic.
