Abstract
Background
Migraine is common in women of reproductive age. Migraine’s episodic manifestation and acute and preventive pharmacological treatment options challenge studying drug safety for this condition during pregnancy. To improve such studies, we aimed to develop algorithms to identify and characterize migraines in electronic healthcare registries and to assess the level of care.
Methods
We linked four registries to detect pregnancies from 2009–2018 and used three algorithms for migraine identification: i) diagnostic codes, ii) triptans dispensed, and iii) a combination of both. We assessed migraine severity using dispensed drugs as proxies. ICD-10 diagnostic subcodes of migraine (G43) allowed the allocation of four subtypes: complicated and/or status migrainosus; with aura; without aura; other/unspecified.
Results
We included 535,089 pregnancies in 367,908 women with available one-year lookback. The prevalence of migraines identified was 2.9%–4.3% before, and 0.8%–1.5% during pregnancy, depending on algorithm used. Pregnant women with migraine were mostly managed in primary care.
Conclusions
Primary care data in combination with drug dispensation records were instrumental for identification of migraine in electronic healthcare registries. Data from secondary care and drug dispensations allow better characterization of migraines. Jointly, these algorithms may contribute to improved perinatal pharmacoepidemiological studies in this population by addressing confounding by maternal migraine indication.
Background
One billion people suffer from migraine worldwide, with women being disproportionately affected (1,2). Hormonal fluctuations influence frequency and severity of migraine attacks, with peak prevalence occurring at age 35–39 (3). Usually, pregnancy leads to migraine symptoms’ improvement and temporary remission, but for 4–8% of women the migraine worsens, and for 12–18% it remains the same (4). To date, few studies have been able to explore the prevalence of migraine in pregnant women on a nationwide population level (5).
The pharmacological treatment recommended for migraine includes acute treatments during the attacks (e.g., nonsteroidal anti-inflammatory drugs (NSAIDs), triptans) and preventive drugs for consistent treatment in case of frequent attacks (e.g., beta-blockers, anti-seizure medications, calcitonin gene-related peptide (CGRP) inhibitors) (6). Migraine is often accompanied by further symptoms requiring additional pharmacological treatment (e.g., antiemetics) (6). Research on the reproductive safety of migraine drugs is crucial to safeguard maternal and infant health as clinical trials usually exclude pregnant women (7). Routinely collected electronic healthcare data offers great opportunities to investigate drug safety in pregnancy (8). However, this data is not primarily collected for research purposes and algorithms to identify pregnant women with migraine are needed to inform pharmacoepidemiologic studies on migraine drug use in pregnancy. Identifying the migraine type, and severity is essential to limit confounding by indication (9–11). Migraine increases the risk for pregnancy complications such as hypertensive disorders, stroke, embolism, or preterm birth (10). Migraine drugs during pregnancy are also associated with different risks on maternal-child outcomes (9,12). Additionally, restricting the study to women with migraine at baseline (i.e., at the start of pregnancy) can help emulate the clinical target trial (13).
Yet, registry-based safety studies of migraine drugs in pregnancy face multiple challenges: i) the pattern of use of acute migraine drugs is episodic (14); ii) drugs other than triptans and CGRP inhibitors can also be used for other indications (6,15); and iii) due to concerns about risk of teratogenicity, pregnant women may switch from triptans or long-term preventive treatment and NSAIDs drugs to paracetamol (9,16).
Considering the above challenges, we aimed to: i) identify women with migraines at baseline and during pregnancy, using different algorithms based on diagnostic codes from primary and secondary care registries and drug codes from the prescription registry; ii) characterize migraine type and severity around pregnancy; and iii) describe the level of care where women with migraine around pregnancy were managed (2). An overall goal is to provide methodological suggestions to appraise better the role of confounding by underlying maternal migraine and to inform quantitative bias analysis to address misclassification of migraine indication.
Material and methods
Study design and data sources
This is a retrospective cohort study using four population-based, nationwide Norwegian healthcare registries between 2008 and 2018. The unique personal identification numbers of each resident in Norway were used to deterministically link the Medical Birth Registry of Norway (MBRN), the Norwegian Patient Registry (NPR), the Norway Control and Payment of Health Reimbursement Registry (KUHR), and the Norwegian Prescription Database (NorPD). Reporting in all four registries is mandatory for all health-care services in the whole country.
Since 1967, the MBRN has registered all pregnancies reaching at least 12 gestational weeks. It contains information on parental demographics, maternal pre-existing conditions, pregnancy, and perinatal outcomes (17).
The NPR has registered information on secondary health care encounters, including outpatient specialist consultations and hospital admissions since 2008 (18). It contains primary and secondary diagnoses coded according to the International Classification of Disease Classification, Version 10 (ICD-10).
KUHR has maintained records of all encounters with general practitioners, primary emergency care, and specific specialist outpatient visits for reimbursement since 2006. Diagnostic codes are listed according to ICD-10 and the International Classification of Primary Care, Version 2 (ICPC-2).
NorPD has been collecting information on all prescribed drugs dispensed in public pharmacies since 2004, regardless of their reimbursement status. It does not include over-the-counter (OTC) drugs. The dataset contains each dispensation's date, type, and characteristic, including the number of defined daily doses, categorized by the Anatomical Therapeutic Chemical (ATC) Classification System (19).
Observation periods
We defined distinct observation periods where migraine-related codes were relevant. “At baseline” was defined using a one-year lookback period before pregnancy start. “Pregnancy start” was defined in MBRN by subtracting the gestational length, as assessed by ultrasound in the first trimester, from the date of delivery or end of pregnancy in case of non-live outcome (“pregnancy end”). “During pregnancy” was defined as the period from “pregnancy start” until “pregnancy end,” with the first trimester from pregnancy start to day 97, the second from day 98 to day 195, and the third from day 196 to pregnancy end (20).
Study population
The study population comprises pregnancies lasting at least 12 or more gestational weeks in women aged 15 to 49 registered in MBRN. To allow the data to go back one year from the start of pregnancy (one year lookback) in all registries, we included pregnancies starting between 1 January 2009 and 1 March 2018. Pregnancies with missing or unrealistic gestational lengths (<12 and >45 gestational weeks) were excluded (Online Supplemental Figure 1).
Identification of women with migraine
We employed three algorithms to identify pregnancies in women with migraine; Dx: at least one diagnostic code as registered in primary or secondary care (ICD-10: G43; or ICPC-2: N89); Rx: at least one triptan dispensed in community pharmacies (ATC: N02CC); Dx/Rx: at least one code of either Dx and/or Rx. We defined “new medically diagnosed migraine” if the first migraine diagnosis and/or triptan prescription fill was during pregnancy. We excluded these women from the primary analysis to emulate a clinical trial design (Online Supplemental Figures 1 and 2).
Migraine type
We defined the following groups of migraine types based on the International Classification of Headache Disorders, Version 3, employing diagnostic codes from primary and secondary care (ICD-10, ICPC-2) (21). The groups are mutually exclusive and their hierarchical order is based on presumed severity: i) Status migrainosus (ICD-10: G43.2) or complicated migraine (ICD-10: G43.3); ii) migraine with aura (ICD-10: G43.1;); iii) migraine without aura (ICD-10: G43.0); iv) unspecified or other migraine (ICD-10: G43.8, G43.9). If more than one type was recorded, we allocated the migraine to the higher hierarchical group. Migraine identified only by ICPC-2 or triptan dispensation does not allow for type allocation, so we added two more groups: v) migraine in primary care (ICPC-2: N89); or vi) triptan-users. (ATC: N02CC). We further assessed the number of diagnoses for cluster headaches (ICD-10: G44.0; ICPC-2: N90) among triptan-users, as sumatriptan and zolmitriptan can be prescribed for cluster headaches.
Proxy for migraine severity
Expanding on our previous work (22), we classified migraine severity using drugs as proxies and established the following hierarchical and mutually exclusive categories: i) very mild (a migraine diagnosis, no dispensations); ii) mild (a migraine diagnosis, general analgesics dispensed (i.e., paracetamol, NSAIDs and/or prochlorperazine); iii) moderate (a migraine diagnosis, oral triptans dispensed; and, iv) severe (migraine Dx, preventive drugs and/or triptan injections dispensed). During pregnancy, we added a “very severe” category (preventive drugs) (Online Supplemental Table 1).
Level of care
We determined the level of care based on whether the migraine was identified in primary (KUHR) or secondary care registries (NPR), and on the type of code used for its registration (ICD-10 or ICPC-2).
Population characteristics
We identified the characteristics of the pregnancies by using information on maternal demographics, obstetric history, maternal health, and pregnancy outcomes through the MBRN. Maternal demographics included: age (continuous and in five-year age groups); reproductive history such as parity, previous miscarriages up to 12 gestational weeks, and previous stillbirth (none, one, two or more); maternal health: smoking (never, smoked before pregnancy, smoked during pregnancy), use of folic acid (none, started before pregnancy, started during pregnancy); pregnancy outcomes: delivery mode (cesarean section (cs) yes or no), child sex (male, female, unclear), mean gestational age, preterm birth (<34 weeks, 34–37 weeks, at term), mean birthweight and low birthweight (<2500 g, ≥2500 g).
Statistical analyses
We assessed the number of pregnancies and determined the proportion of pregnancies exposed to migraine using the three algorithms during each observation period (at baseline, during pregnancy, and by trimester). Pregnancy characteristics were described for migraineurs and non-migraineurs according to identification at baseline. We further assessed the proportions of migraine type and severity at baseline, during pregnancy, and by trimester.
We conducted three sensitivity analyses: First, we extended the lookback to five years and included pregnancies starting after 1 January 2013, and before 1 March 2018. (Online Supplemental Figure 2, Online Supplemental Table 2). Second, we restricted the study population to the first recorded pregnancy of a woman registered in MBRN within the study period irrespective of parity. This was done to control for any variations in the number of pregnancies between migraineurs and non-migraineurs (Online Supplemental Table 3). Third, we included the pregnancies of women with a new medically diagnosed migraine during pregnancy as a third category and described their baseline characteristics, as they might differ from those with migraine at baseline (Online Supplemental Tables 4 and 5).
We used STATA version 16.0 for data management and statistical analysis and R version 4.1.1. using the eulerr library (a statistical package in R) to create Figure 1 (23). All analyses were performed on the secure server at the University of Oslo, TSD.
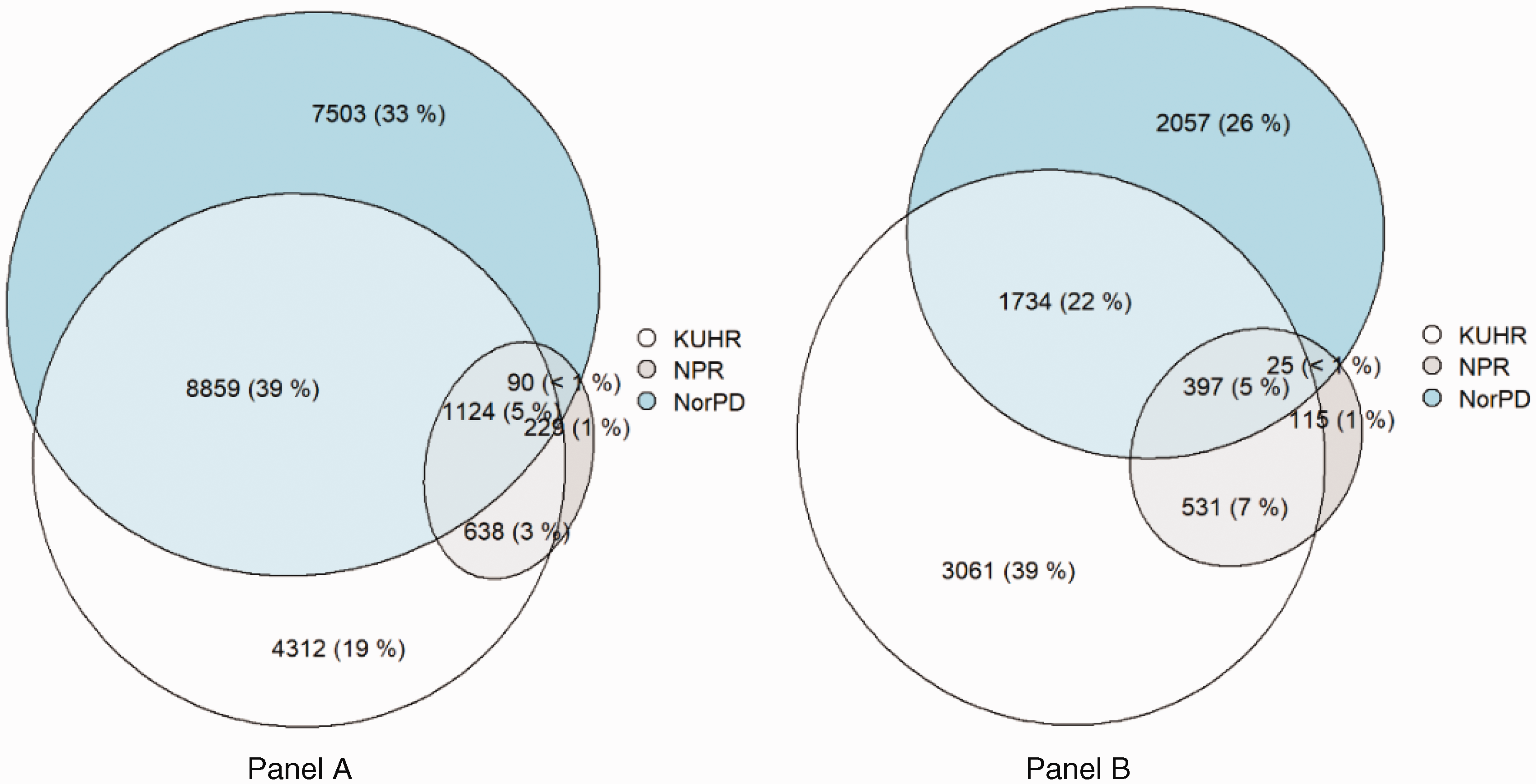
Source of identification/Level of care of migraine at baseline (Panel A) and during pregnancy (Panel B). Circle and field sizes proportionally represent the number of cases identified in the registries or group of registries. (KUHR: Primary care registry, NPR: Secondary care registry, NorPD: Norwegian Prescription Database (based on triptan dispensation)).
Ethics
The study was approved by the Regional Committee for Research Ethics in South-East Norway (approval number 2018/140/REK) and by the Data Protection Officer at the University of Oslo (approval number 58033). Data access was provided by the Norwegian Institute of Public Health (https://www.fhi.no/en/hd/access-to-data/applying-for-access-to-data/). Data were handled and stored in accordance with the General Data Protection Regulation.
Results
We identified 535,089 pregnancies of 367,908 women with a one-year lookback period (Online Supplemental Figure 1). For the five-year lookback, we included 298,354 pregnancies in 241,328 women (Online Supplemental Figure 2).
Identification of pregnancies affected by migraine
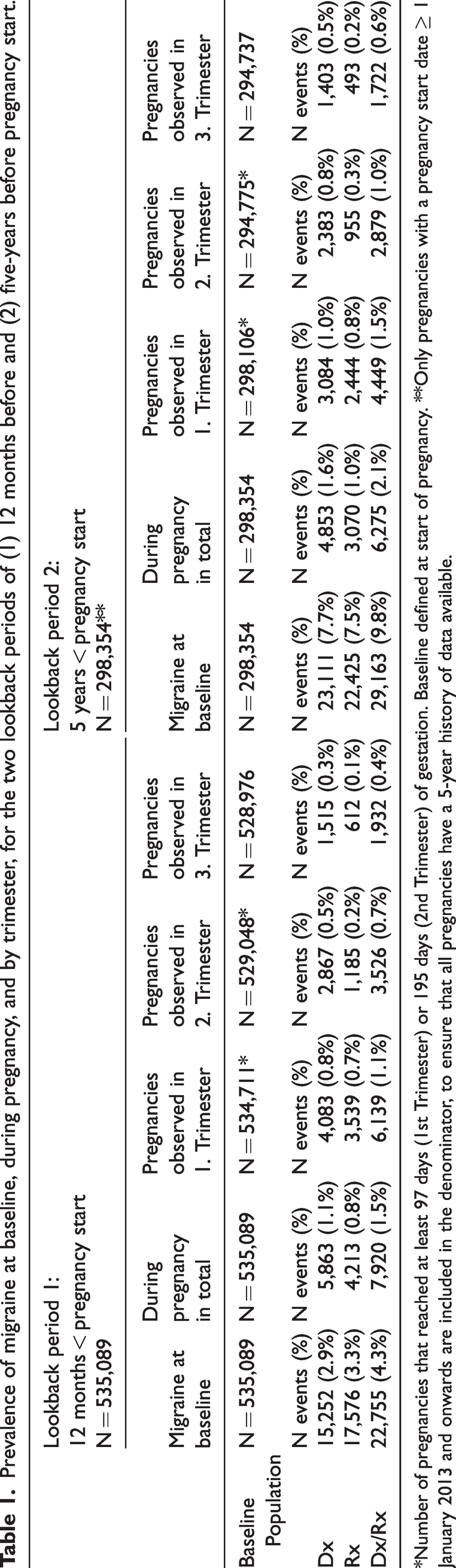
At baseline, we identified a population-based one-year prevalence of migraine of 2.9% using the Dx algorithm, 3.3% using the Rx, and 4.3% with the combined Dx/Rx algorithm (Table 1). From the Dx/Rx algorithm (n = 22,755), the majority (44.3%) had both a diagnostic and a prescription code, followed by solely a prescription code (33.0%), and lastly, only a diagnostic code (22.8%). Of the Dx algorithm (n = 5,179), 81.5% only had the ICPC-2 code (N89) for migraine. Figure 1 depicts the registry where the migraine was identified. Most migraines were identified in primary care registries before (65.6%) and during pregnancy (72.3%).
*Number of pregnancies that reached at least 97 days (1st Trimester) or 195 days (2nd Trimester) of gestation. Baseline defined at start of pregnancy. **Only pregnancies with a pregnancy start date ≥ 1 January 2013 and onwards are included in the denominator, to ensure that all pregnancies have a 5-year history of data available.
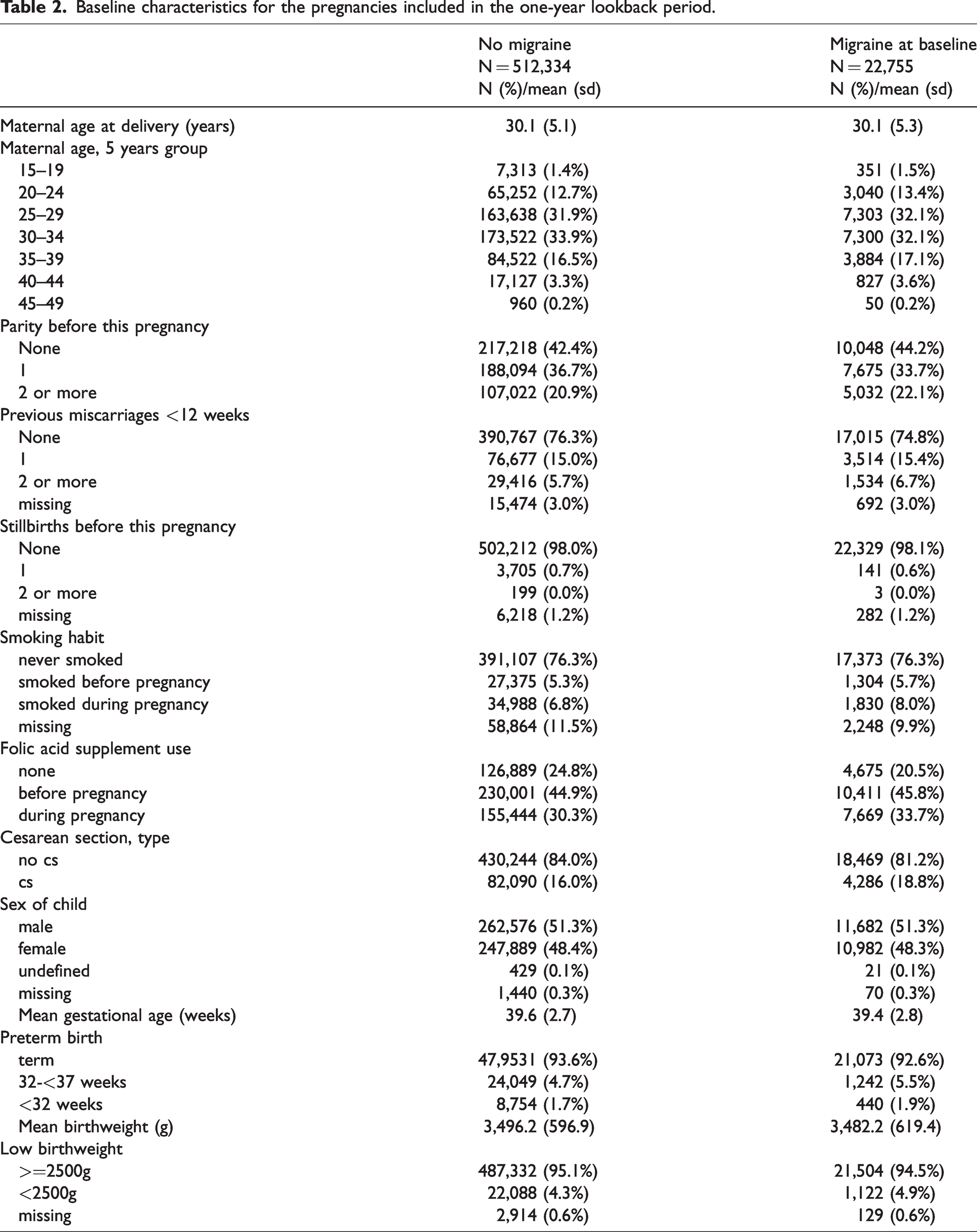
Table 2 displays the pregnancy characteristics by migraine at baseline in the one-year lookback. Migraineurs and non-migraineurs are mostly similar, except for slightly increased proportions of hypertensive conditions (2.3% vs. 1.7%) and of preeclampsia/eclampsia (3.9% vs. 2.8%) in migraineurs.
Baseline characteristics for the pregnancies included in the one-year lookback period.
Migraine type
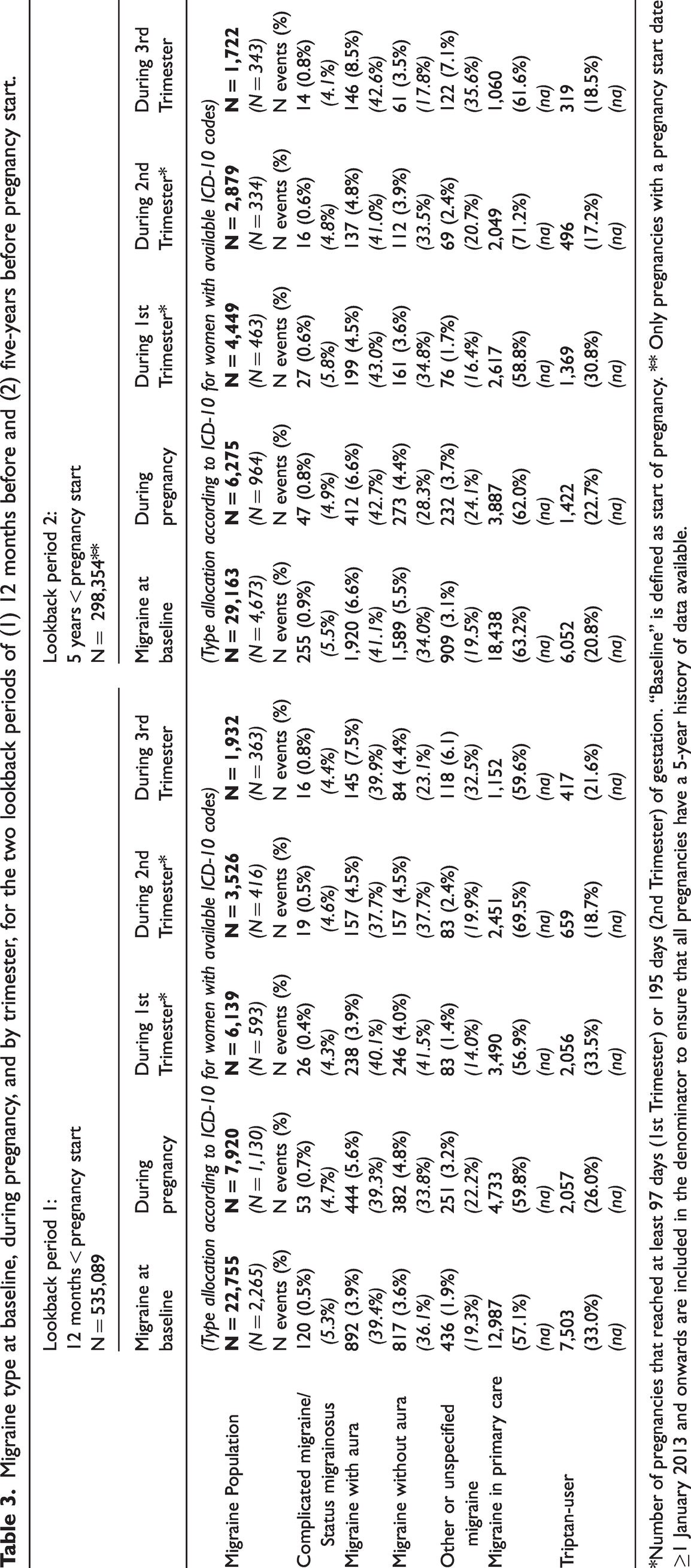
The largest group in all observation periods was “migraine in primary care” (57–70%), followed by “triptan-users” (17–34%). Among triptan-users, less than 1% had a diagnosis of cluster headache in each period (between 0%−0.83%).
Migraine with aura (baseline: 39.4%, during: 39.3%) and migraine without aura (baseline: 36.1%, during: 33.8%) were the most frequent migraine diagnoses when an ICD-10 code was available. The proportion of migraine with aura was highest in the first trimester (Table 3, Figure 2).
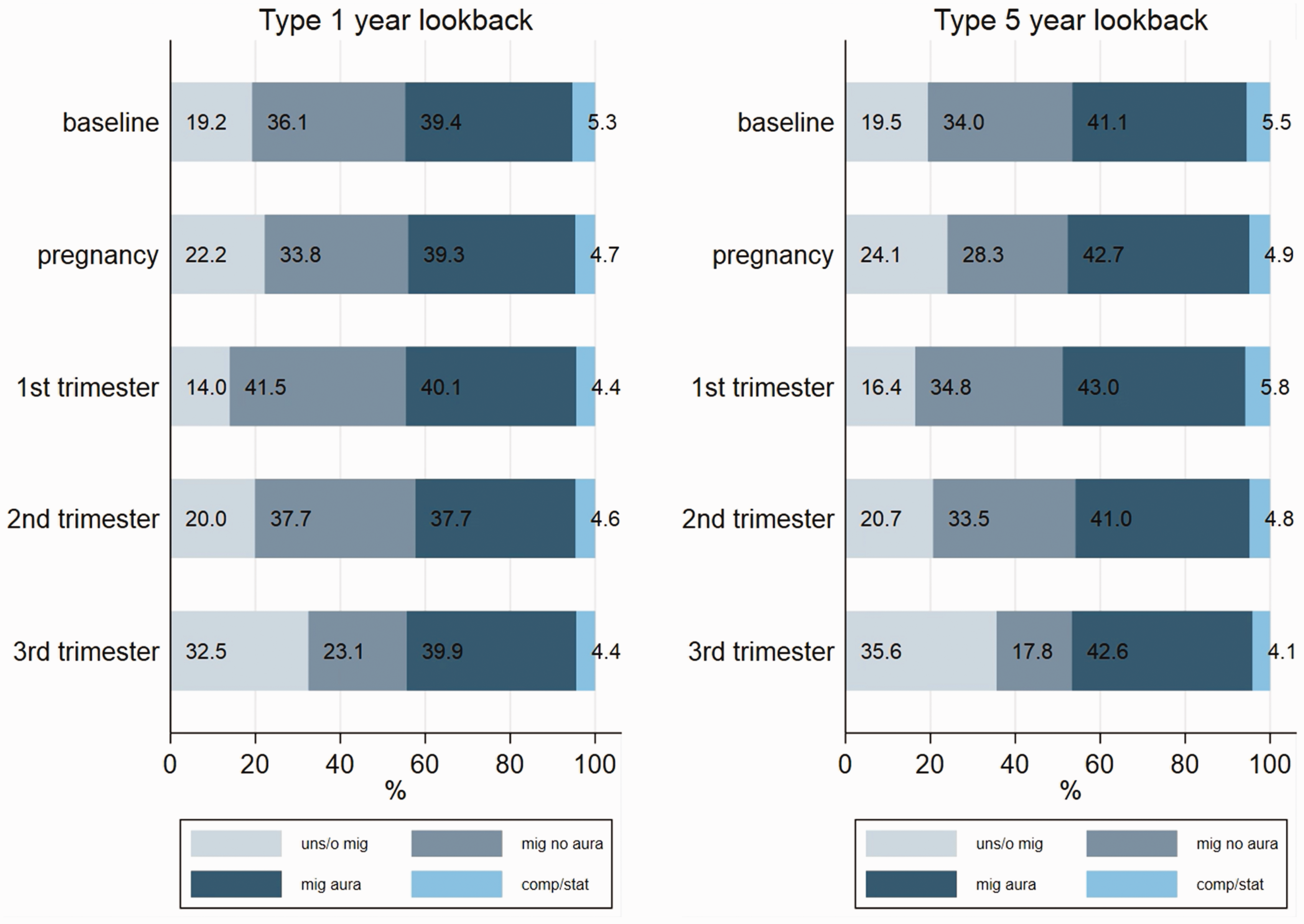
Type of migraine at baseline, during pregnancy, and by trimester, for the two lookback periods of one year before (left) and five years before (right) pregnancy start (excluding migraine in primary care and triptan-users). Abbreviations: uns/o: unspecified/other migraine; mig aura: migraine with aura; mig no aura: migraine without aura; comp/stat: complicated migraine/status migrainous.
Proxy of migraine severity
Figure 3 depicts migraine severity by timing, illustrating the impact of pregnancy: At baseline, we mostly identified moderate migraine (64.3%) followed by severe migraine (15.8%), very mild (12.8%), and mild migraine (7.2%). The proportion of moderate migraine decreased to 46.6% during pregnancy, and very mild migraine increased to 38.2%, reaching 63.3% in the third trimester. Very severe migraine was identified in 9.4% of pregnancies.
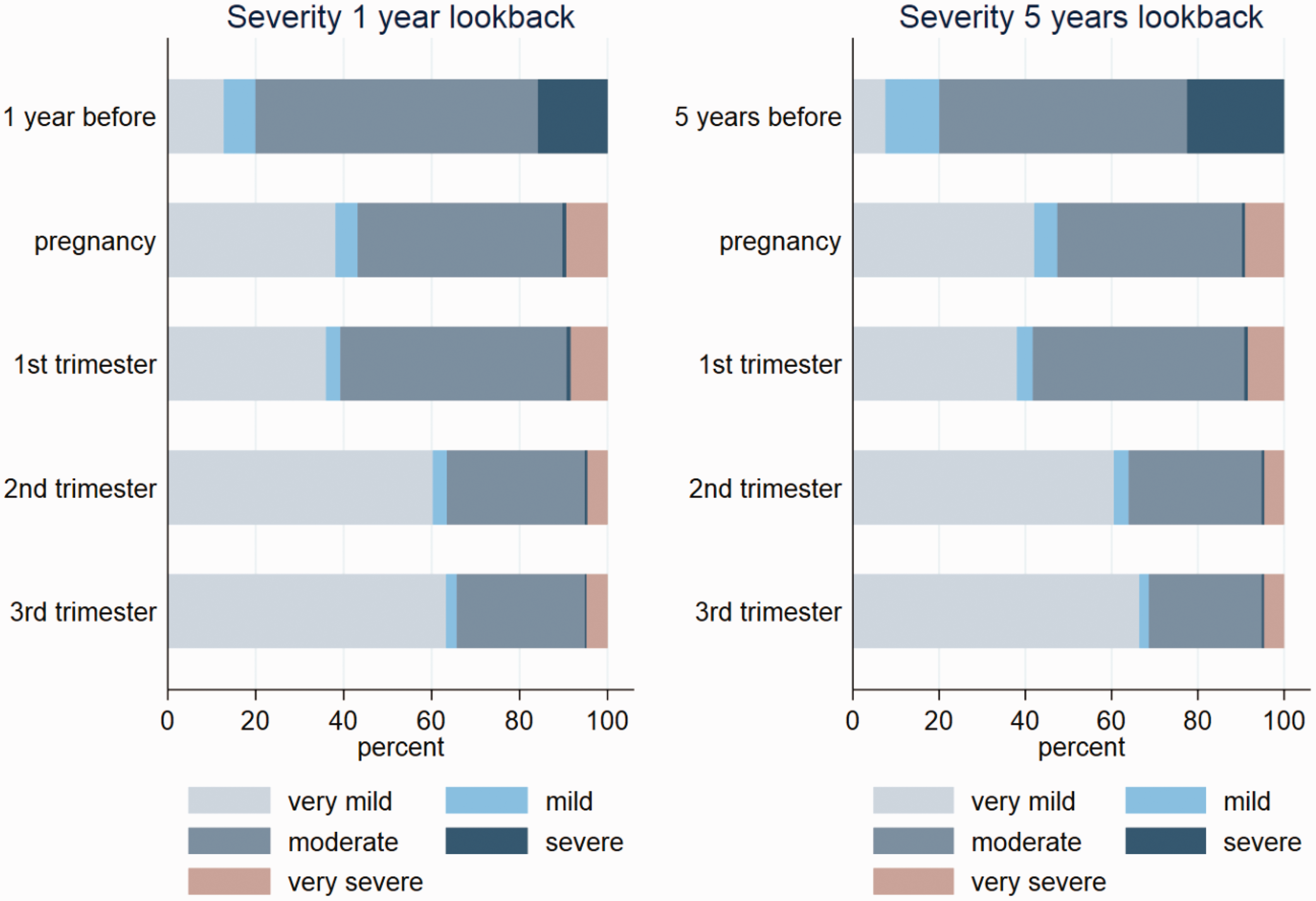
Severity of migraine at baseline, during pregnancy, and by trimester, for the two lookback periods of one year before (left) and five years before (right) pregnancy start.
Sensitivity analyses
Prevalence rates at baseline were considerably higher when using a five-year lookback (Dx: 7.7%, Rx: 7.5%, and Dx/Rx: 9.8%) but had a minor impact on prevalence during pregnancy (Table 1). The pregnancy characteristics between the two lookback periods were similar (Table 2, Online Supplemental Table 2). Extending the lookback resulted in changes to patterns for type and severity at baseline but not during pregnancy (Table 3; Figures 2 and 3).
*Number of pregnancies that reached at least 97 days (1st Trimester) or 195 days (2nd Trimester) of gestation. “Baseline” is defined as start of pregnancy. ** Only pregnancies with a pregnancy start date ≥1 January 2013 and onwards are included in the denominator to ensure that all pregnancies have a 5-year history of data available.
Migraine first medically diagnosed in pregnancy was most often with aura (around 52–58%) in both lookback periods (24). The pregnancy characteristics did not differ compared to migraineurs at baseline (Online Supplemental Tables 4 and 5).
When we included only the first pregnancy of each woman, prevalence estimates were similar to the primary analysis (Online Supplemental Table 3).
Discussion
This Norwegian registry-linkage study provides population-based estimates of migraine prevalence around pregnancy. The identified prevalence estimates vary depending on the algorithm and lookback time used: at baseline, the prevalence ranged from 2.9% to 9.8% while during pregnancy it ranged from 0.8% to 2.1%. The most frequently identified group was “migraine in primary care” (>57%), though in the cases where allocation of subtype was possible, “migraine with aura” was most common. In terms of migraine severity, “moderate” was most frequent (64.3% at baseline, 46.6% in pregnancy), whereas “very mild” became most prevalent in the third trimester (63.3%).
The global one-year prevalence of migraine in women is around 18% (5). In Norway, it was 15% in a questionnaire study (25), and 23% in an interview study (26). Prior Norwegian birth cohort studies found a self-reported prevalence of migraine of 11- 13% before pregnancy and 5% during pregnancy (9,27). Compared to these studies, our identified, population-based prevalence of migraine was lower at baseline (4.3%) and during pregnancy (1.5%). Using a five-year lookback, the identified prevalence (9.8%) is closer to the self-reported findings.
Our study used diagnostic and prescription codes that depend on healthcare contacts and medical follow-up instead of self-reported information. We are therefore unable to capture women with episodic migraine who never contacted their physician or women who manage their migraine nonpharmacologically or using OTC drugs (6,28). In self-reported questionnaire-based studies, it may be challenging for participants to distinguish headache disorders with overlapping features and misclassification may be present (29). Whereas sensitivity for self-reported tension-type headache was high (96%), it was fair to moderate for migraine and subtypes of migraine (35–67%) as compared to a neurologists’ assessment (30). Norway has universal health and social insurance coverage and pregnancy care are free of charge. As our study covers all inhabitants of Norway filling prescriptions or seeking public healthcare, no selection bias is expected. It is essential to consider these methodological differences when comparing prevalence rates between studies (5).
Other explanations for the lower prevalence might lie in the changing nature of the disease. In women, migraine prevalence increases after puberty and peaks around middle age (3). It decreases during the second and third trimesters of pregnancy, during breastfeeding, and after menopause (3). In a population-based cohort of pregnant women, inactive or lower disease activity might lead to reduced healthcare contact due to migraine (31).
In women, migraine is also believed to be underdiagnosed and undertreated (32). The Eurolight study estimated that only 10–18% of individuals with migraine receive an adequate headache assessment, and results from the European My-LIFE anamnesis survey showed that many chronic migraineurs were not properly managed or referred to specialist care (33,34).
Sixty-six percent of women with migraine were seen in primary care before pregnancy, and 73% during pregnancy. At baseline, this reflects international and Norwegian treatment guidelines for common headache disorders (2,35). During pregnancy, the guidelines suggest neurological follow-up of women with migraine using triptans or preventive migraine treatment (6). Surprisingly, less than 8% of women using triptans and less than 30% of women with very severe migraine were seen in specialist care during pregnancy (Online Supplemental Table 6).
According to ICD-10 codes in secondary and specialist care, around 40% were diagnosed with “migraine with aura”, despite “migraine without aura” usually being the most frequent subtype among all patients with migraine (6). Guidelines suggest referring those suffering from migraine with atypical or prolonged aura to a neurologist, which might explain why migraine with aura, as compared to migraine without aura, is more frequently reported in secondary and specialist healthcare data. This is particularly important during pregnancy, where headache and aura can be signs of complications such as preeclampsia or stroke (36).
Migraine in pregnant women itself, mainly migraine with aura, is associated with increased risks of thromboembolic events, cardiac diseases, hypertensive disorders, and stroke (36). Migraine has also been found to be associated with adverse perinatal outcomes such as, preterm birth, and low birth weight (12,37). The increase in migraine with aura and decrease in migraine without aura in the second and third trimester may have two causes: first, the higher hormone dependency of migraine without aura usually induces headache improvement during pregnancy (36). Second, the higher level of care for pregnant women with migraine with aura (3). Our findings regarding the severity of migraine further support the general improvement of migraine during pregnancy (4).
Confounding by disease severity and type (i.e., confounding by indication) is a major issue in perinatal drug safety studies (8,11). Expanding on our previous work, we used drugs as a proxy to characterize migraine severity better (22). The algorithm is based on acute and preventive treatment, with preventive drugs indicating more severe migraine (22,38). It did not take the amount of drug dispensed into account and therefore did not incorporate medication overuse headache that can complicate migraine. This algorithm based on drug type dispensed is not without risks. Women might discontinue drug use during pregnancy due to lower disease activity, or due to fear of teratogenicity (14,16). Finally, co-morbidities and polypharmacy are frequent among women with migraine, and most preventive drugs are also used to treat other diseases (e.g., depression or epilepsy) (15).
The challenges described may lead to underestimation of prevalence and misclassification of migraine cases, migraine type, and severity. Prior studies have shown that relying on diagnostic codes in electronic health care data may be, compared with gold-standard clinical diagnoses, precise and lead to high negative predictive values, but sensitivity is usually poor, and positive predictive values may only be moderate (39). A Norwegian study assessing agreement between self-reported triptan-use and a dispensation code found lower sensitivity (32–63%) and higher specificity (90–99%) (14). The absence of a code makes us less confident in identifying a true non-case (39). In case of an effect, the one by the drug might be overestimated whereas the one by the underlying disease might remain underestimated (11). Quantitative bias analysis may be used to estimate the impact of misclassification on final effects when considering the assumed proportion of misclassified individuals (39).
Medication safety studies that emulate the clinical target trial may generate similar results to those from a clinical trial. To do so, identification of current disease activity at hypothetical enrolment is essential to allocate the patients to the correct, hypothetical study arm. Selecting the optimal length of lookback time is essential to generate reliable results. A more extended lookback period may increase migraine prevalence at baseline but simultaneously increases the risk of capturing individuals with no current disease activity (13).
The main strength of this study is that it covers pregnancies from an entire country and includes prospectively recorded information from both primary and secondary care, as well as drugs dispensed prior to and during pregnancy. This approach enables an efficient and cost-effective assessment of diagnoses and drug use around pregnancy (8).
However, our study has limitations. First, it is essential to note that the shorter time periods of pregnancy and pregnancy trimesters (three to nine months) cannot be directly compared to the one-year lookback. This time difference may partially account for the lower prevalence estimates observed during pregnancy. Second, we could not classify migraine type for >85% of women due to unspecific ICPC-2 codes in primary care or identification solely through triptan dispensation. Third, the hierarchical and mutually exclusive categories for type of migraine could have slightly influenced identified proportions of migraine type. Triptan-users also contain a proportion of cluster headaches, but this group constitutes less than 1% of our cases. Fourth, since the severity algorithm was based on the type of the drugs dispensed, we could not ascertain whether changes in migraine severity were due to improvement of migraine or the discontinuation of treatment. Validation of the proposed algorithms such as through detailed chart reviews or patient interviews, was not feasible due to data de-identification. Future studies should consider clinical validation of registry-based variables. Whereas the algorithms can be transferred to other registry or claims data, the results may be highly dependent on the origin of the health care data, variations in the health care system and the structure and purpose of data collection (39).
Conclusion
When using electronic healthcare registries, we think a one-year lookback period proves to be adequate for identifying women with active, moderate to severe migraine at the start of pregnancy. Primary care registry data is important and adding secondary care data and drug dispensation records allows for a better characterization of migraine around pregnancy. This approach not only improves the ability to address confounding by indication but may help to identify potential misclassification in migraine drug safety studies during pregnancy.
For future studies, employing electronic healthcare data with a one-year lookback, combined with causal inference techniques, or to emulate the target trial, will enable the generation of higher quality evidence on the safety of migraine drugs during pregnancy.
Key findings
We developed and validated algorithms to identify and characterize migraine in electronic healthcare data, to inform future studies on drug safety of migraine treatment during pregnancy to avoid confounding by indication. Migraine is a chronic condition with episodic disease manifestation often associated with hormonal changes around pregnancy. We identified a population-based one-year prevalence of migraine of 2.9–4.3% before pregnancy and 0.8–1.5% during pregnancy. Most migraines were identified in primary care data, meaning they are mostly treated at that level. In combination with dispensed drug data, primary care data seems best to reliably identify migraine in electronic healthcare data. Self-managed milder migraine remains challenging to capture in electronic healthcare data and false negatives are possible. Misclassification analysis is recommended.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024241248846 - Supplemental material for Identification and characterization of migraine in pregnancy: A Norwegian registry-based cohort study
Supplemental material, sj-pdf-1-cep-10.1177_03331024241248846 for Identification and characterization of migraine in pregnancy: A Norwegian registry-based cohort study by Vera R. Mitter, Angela Lupattelli, Marte-Helene Bjørk and Hedvig M.E. Nordeng in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024241248846 - Supplemental material for Identification and characterization of migraine in pregnancy: A Norwegian registry-based cohort study
Supplemental material, sj-pdf-2-cep-10.1177_03331024241248846 for Identification and characterization of migraine in pregnancy: A Norwegian registry-based cohort study by Vera R. Mitter, Angela Lupattelli, Marte-Helene Bjørk and Hedvig M.E. Nordeng in Cephalalgia
Footnotes
Acknowledgments
We performed this work on the TSD (Tjeneste for Sensitive Data) facilities owned by the University of Oslo and operated by the IT department (USIT). We thank Mike Mitter for the support with programming the Venn/Euler diagram in R. We used ChatGPT UiO Version 3.5 and Grammarly v 1.0.54.1209 to support editing of the English language and to improve readability in some parts of the manuscript.
Authors’ contributions
HN, AL, and MHB conceived and wrote the study protocol and the statistical analysis plan. AL and VRM performed the analysis. VRM drafted the manuscript, and AL and HN contributed to the writing. All authors revised the manuscript and critically interpreted the findings. All authors are accountable for the final version submitted.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MHB reports receiving fees paid to her institution by valproate market authorization holders for EMA-mandated contract research; speaking and/or advisory board honoraria from Eisai, Novartis Norway, Jazz Pharmaceuticals, Angelini Pharma, Teva, Lilly, and Lundbeck; the other authors do not declare any conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.