Abstract
Introduction
In this drug utilization study, we aimed at assessing the pattern of triptan use in Italy by means of the drug prescription databases of two local health authorities, accounting for approximately 1 million citizens.
Methods
The study population included all residents aged 18 to 84 years in the Vercelli province (about 175,000 inhabitants) and in the Umbria region (about 885,000 inhabitants), who had at least one dispensation for triptans in 2012. A frequent user, who might be at risk of medication-overuse headache (MOH), was defined as a patient being dispensed at least 10 defined daily doses (DDD) of triptans every month for at least three consecutive months.
Results
Triptans were used by 0.7%–1% of the population. While most patients were dispensed fewer than 60 DDDs per year, about 10% of all triptan users were classified as frequent users. In both areas, patients below the age of 29 were less likely to be frequent users while the 40- to 49-year-old population was the most affected, with no sex difference. About two-thirds of frequent users persisted in this behavior for an additional three-month period in the following six months.
Conclusions
Our data indicate that approximately 10% of all triptan users in the Italian population are potentially at risk for MOH. An approach based on drug prescription databases could be useful to identify patients at risk for MOH.
Introduction
According to the International Classification of Headache Disorders (ICHD-III edition beta version), medication-overuse headache (MOH) is a chronic headache occurring as a consequence of regular overuse of acute or symptomatic headache medications on at least 10 days per month (15 days for simple analgesics and nonsteroidal anti-inflammatory drugs (NSAIDs)) for at least three consecutive months (1). While the previous definition of the International Headache Society (IHS) included the resolution or improvement of the headache after withdrawal of symptomatic treatment (2), the latest criteria have excluded this parameter although it is mentioned that chronic headache usually resolves after the overuse is stopped (1). In brief, therefore, the cause of MOH is the headache treatment itself. Even though every new agent that arrived on the market raised the hope that it would not lead to MOH, it is now accepted that all symptomatic treatments for migraine can trigger chronic headache. Indeed, ergotamine, triptans, NSAIDs, opioids and combination treatments are all listed in the ICHD-III beta as causative factors (1).
The therapy of MOH entails the discontinuation of the abused drugs, which, according to the severity of MOH and to different clinical practices, can be achieved with a variety of means, ranging from simple advice (3) to hospitalization (4). Nonetheless, MOH is a costly disease in terms of social burden as well as from a pharmacoeconomic perspective (e.g. increased medicine prescriptions, increased costs for headache clinics and hospitalization, etc.) (5,6). Furthermore, while the condition is well known to neurologists and to experts in headache clinics, it is probably less known to general practitioners and pharmacists. Epidemiological data indicate that MOH affects approximately 1% of the general population, with a similar prevalence in Europe, North America and Asian countries (7). However, given that most analgesics are over-the-counter drugs, it is also difficult to have accurate figures because patients overusing medications not reimbursed by the Italian National Health Service (NHS) may not be caught in the statistics. On the other hand, in a number of countries, including Italy, triptans, which are considered by many experts as the gold-standard therapy for acute treatment of migraine, are prescribed by general practitioners and are reimbursed by the NHS. In principle, therefore, it is possible to identify individual patients’ drug intake to estimate the prevalence of triptan use. In a study conducted in the Netherlands, prevalence of triptan use in the Dutch population was 1.3%, and 10.4% of these patients could be considered overusers following the IHS criteria (8). In Italy, data collected in selected areas suggested a slightly lower use of triptans (0.6%–0.8% of the population) (9–12) but a higher proportion of patients at risk of MOH using the IHS criteria (14.3%) (12). These data, nonetheless, run back in time and it would be important to have an updated snapshot of triptan use in Italy.
According to sales statistics published by the Italian medicines agency (AIFA), almost the entire amount of triptan use in the 2001–2011 decade (around 95%) was covered by the NHS (13). Thus, when individual prescriptions are recorded, it is possible to identify almost all triptan users. To quantify the prevalence of MOH related to triptan use and to assess the role of potential risk factors, we carried out a drug utilization study in two Italian areas: the Province of Vercelli (about 175,000 inhabitants) and the Umbria region (about 885,000 inhabitants). The use of two separate databases allowed us to investigate which clinical and demographic factors were consistent across populations.
Methods
Triptans are reimbursed by the NHS and data relevant to each prescription are recorded for administrative purposes (the reimbursement of retail pharmacies). The following data were used in this study: drug code, number of dispensed packages, encrypted patient identification number and date of prescription. From the drug code we were able to identify the information on substances, expected duration of the prescription (on the basis of the defined daily doses (DDD) contained in each package), and anatomical, therapeutic and chemical (ATC) classification.
Prescribing data were derived from the drug dispensation monitoring system of the Local Health Authority of Vercelli and of the Umbria region. A descriptive drug utilization study was conducted on subjects living in Vercelli (exploratory cohort) and in the Umbria region (validation cohort). All participants aged 18 to 84 years who received at least one triptan (ATC N02CC) prescription in the period from January 1, 2012 to December 31, 2012 were included in the study. Patient demographic data were obtained by means of automated record-linkage with the regional health office database in which all individuals assisted by the NHS are listed. The record linkage procedure is based on the NHS patient identification code, which does not contain any sensitive data related to patient identity, thus ensuring confidential treatment of personal data.
Triptans are reimbursed by the NHS and related prescription details are entirely recorded in regional health authorities’ databases. All triptans available in 2012 on the Italian market (almotriptan, rizatriptan, frovatriptan, sumatriptan, zolmitriptan and eletriptan) were included in the analysis. A “
We also identified a subgroup of patients,
Triptan users in the study populations were described in terms of age, sex, administered triptan and months of treatment. Drug consumption was evaluated in terms of: prevalence of use (by dividing the number of drug users by the overall resident population) and DDDs per 1000 users/inhabitants per day (the mean number of doses consumed every day by 1000 patients included in the study/resident population). The risk of frequent use was estimated for each triptan found in the observation period.
Concomitant drug prescriptions were also retrieved for frequent users to identify comorbidities and consumption of migraine-prophylactic medications. Online Supplementary Table S2 illustrates the categories of drugs that were considered as indicators of comorbidities. With regard to migraine-prophylactic medications, given that no information on the indication is recorded in the administrative databases, it was impossible to establish whether these drugs were prescribed for migraine or for other indications. However, we assumed that the following drugs might be prescribed as prophylactic medicines: metoprolol (ATC C07AB02), propranolol (C07AA05), topiramate (N03AX11), valproic acid (N03AG01), timolol (C07AA06), atenolol (C07AB03), amitriptyline (N06AA09), venlafaxine (N06AX16), and pizotifen (N02CX01).
Statistical analysis
Clinical characteristics of frequent users and regular users and the difference for continuous variables are presented with 95% confidence intervals (CIs). The two groups were compared using the Mann-Whitney
Results
Triptan use in Italy
First, we referred to a comprehensive database on drugs bought by the NHS, updated to 2012 (13). The prescription of triptans has steadily increased in the last decade in Italy (online Supplementary Figure S1(a)). In 2011, 0.76 DDDs/1000 inhabitants per day were prescribed/dispensed. The prescription pattern differs significantly from region to region, ranging from 40% below (e.g. Umbria, Campania) to 40% (e.g. Sardegna, Valle d’Aosta) above the national average (online Supplementary Figure S1(b)). Unlike many other drug classes (13), there is no North-to-South trend in the number of prescriptions and there is little variability in the regional expenditure per DDD since prices of various triptans are fairly similar.
Triptan use in the two Italian areas in 2012
Characteristics of the triptan users in the two areas.
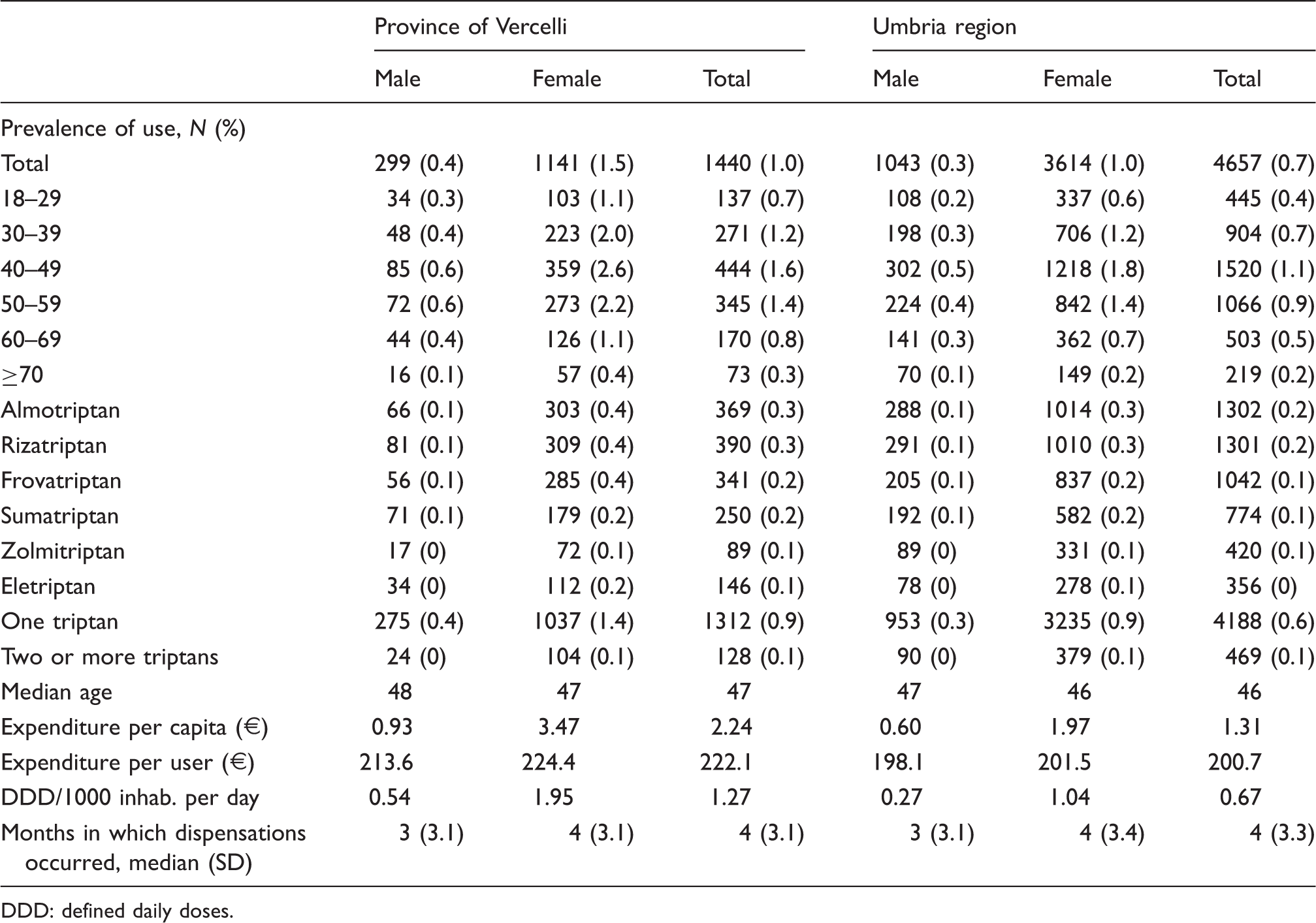
DDD: defined daily doses.
Number of defined daily doses (DDDs) per user in 12 months.

Characteristics of frequent triptan users compared with regular users.
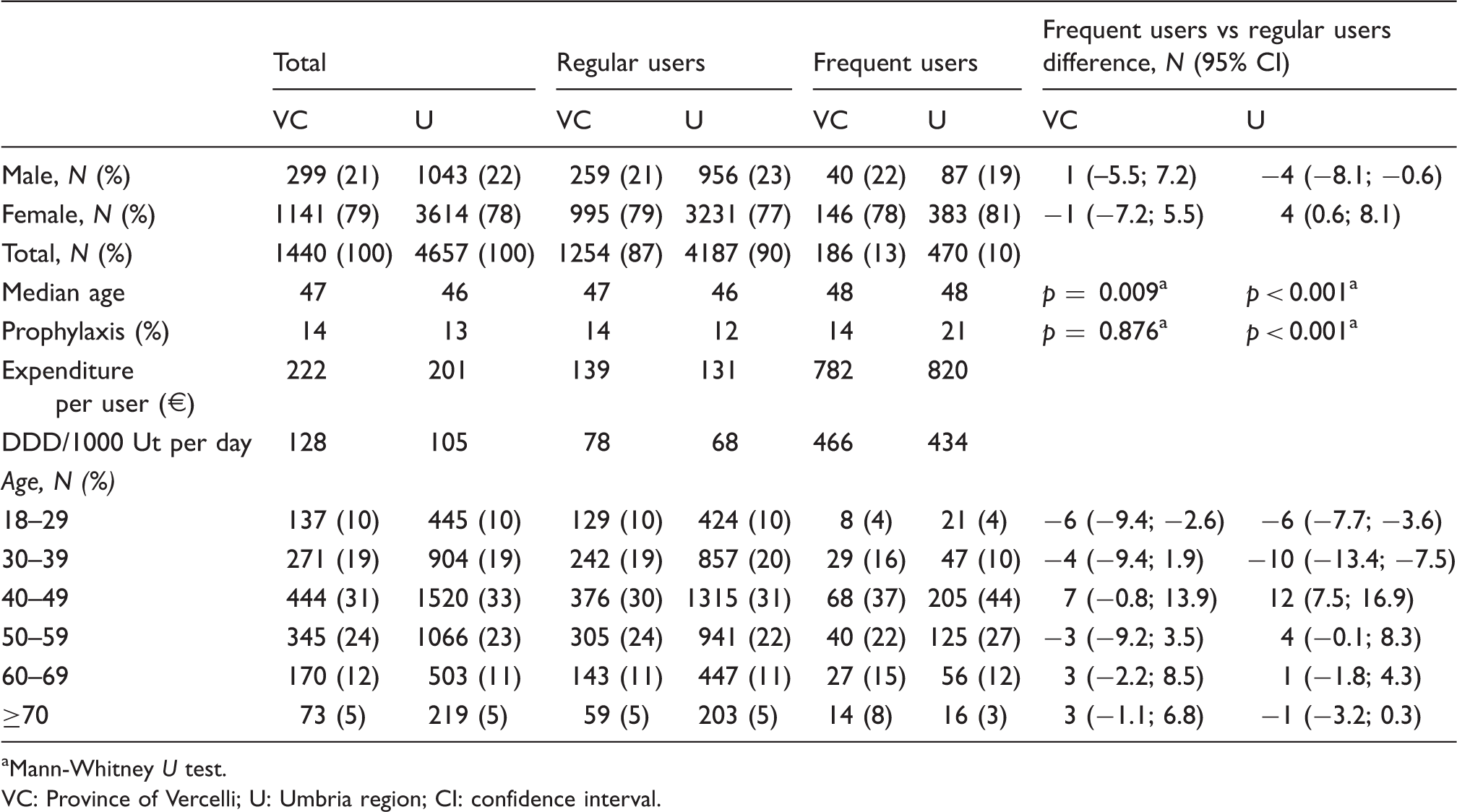
Mann-Whitney
Risk of overuse for each triptan in the two areas.

Adjusted by age, gender and prophylaxis. VC: Vercelli; U: Umbria region; CI: confidence interval. In the analysis were considered users of only triptan in the period.
Consistently frequent users in the two areas by age and sex.

VC: province of Vercelli; U: Umbria region.
Discussion
Our analysis suggests that about 10% of triptan users are prescribed and dispensed a number of monthly doses that, if taken on separate days, would lead them to meet the IHS criteria for MOH. These frequent users are responsible for 40% of the total cost of triptans. Focusing medical attention on this relatively small proportion of migraineurs might save and optimize a significant part of NHS resources. With respect to our study design, by using two separate populations, we were able to use the two databases as both exploratory and confirmatory and to find common determinants of this pattern of use. While a number of factors (e.g. type of triptan) showed a consistent trend in both populations, only age correlated significantly with frequent use. The finding that relatively older patients (40–49 age category) appeared at higher risk is consistent with the data reported in a meta-analysis of 29 studies comprising a total of 2612 patients with MOH, where patients had a mean duration of primary headache of around 20.4 years (14).
With respect to the prescription of prophylactic medications, we found conflicting results in the two areas, with a higher level of use in the Umbria population. It must be acknowledged that migraine prophylaxis is difficult to monitor using NHS databases, as most medications are not solely prescribed for migraine and many are not dispensed by the NHS. For example, the prescription of flunarizine, which in a recent study was found to be the most used preventive medication in two provinces (Novara and Pavia) (15) bordering with the Vercelli province, could not be monitored as flunarizine is not covered by the NHS. In a broader study, involving 10 headache centers from all over Italy, flunarizine was the third used preventive medication (16). Nonetheless, our study suggests that only 14%–21% of frequent users of triptans are prescribed a prophylactic medication that can be monitored and these data are in line with a recent Dutch study on migraine-preventive medications (17). It would therefore be important to design a study to assess the use of preventive medication in Italy.
The main strength of our study is represented by the fact that triptans in Italy are all reimbursed via the NHS and therefore our census covers the entirety of triptan use in the population of two geographic areas. The very limited amount of triptans that are purchased out of pocket (around 5% of total prescriptions) cannot modify our estimates.
The study also faced several limitations: (i) we focused solely on triptan use and were unable to monitor over-the-counter drugs or other prescription drugs that are not dispensed by the NHS. It is likely that including these drugs would increase significantly the number of patients who might meet IHS criteria for MOH; (ii) we defined as frequent user a patient who is dispensed more than 10 DDDs per month for three consecutive months. It must be acknowledged that the relation “one DDD one attack” might not hold, as guidelines suggest that patients might decide to take a second dose for the same attack in the same day. It is possible, therefore, that triptan overusers are less than frequent users, as defined in this study; (iii) as in all pharmacoepidemiological studies, this report is unable to discriminate between dispensation and use, and some factors (e.g. sharing of the same prescription among family members, accumulating the drug for future use) might contribute to overestimating the phenomenon.
Prevalence of frequent triptan users in population studies.

Criteria to define frequent triptan users: a30–59 DDDs/month. b≥60 DDDs/month. cUsers of 120 DDDs or more per year. dUsers of 216 DDDs or more per year. e>244 DDD/year. f≥15 doses/month. g15–29 DDD/month among new users of triptans (
Clinical implications
In the present study, a descriptive drug utilization study was conducted by means of the drug prescriptions databases of two local health authorities in Italy to evaluate triptan use. About 10% of identified triptan users are dispensed at least 10 defined daily doses (DDDs) of triptans every month for at least three consecutive months. If these doses were actually taken on different days, these patients would meet the IHS criteria for medication-overuse headache (MOH). An approach based on drug prescription databases could be useful for early identification of patients at higher risk of MOH. Such an approach might also reduce the prevalence and duration of MOH as well as decrease its social and economic burden.
Footnotes
Funding
This work was partly supported by a Ministry of Health (RC2011) grant to the C. Mondino National Neurological Institute. S.C. holds a PhD fellowship supported by the Compagnia di San Paolo.
Conflict of interest
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
